Abstract
COVID-19 sequelae are varied, and whether they are temporary or permanent is still unknown. Identifying these sequelae may guide therapeutic strategies to improve these individuals' recovery. This prospective cohort aimed to assess body composition, cardiopulmonary fitness, and long-term symptoms of overweight individuals affected by COVID-19. Participants (n = 90) were divided into three groups according to the severity of acute COVID-19: mild (no hospitalization), moderate (hospitalization, without oxygen support), and severe/critical cases (hospitalized in Intensive Care Unit). We assessed body composition with a tetrapolar multifrequency bioimpedance, hemodynamic variables (heart rate, blood pressure, and peripheral oxygen saturation-SpO2) at rest, and the Bruce test with direct gas exchange. Two assessments with a one-year interval were performed. The most prevalent long-term symptoms were memory deficit (66.7%), lack of concentration (51.7%), fatigue (65.6%), and dyspnea (40%). Bruce test presented a time effect with an increase in the distance walked after 1 year just for severe/critical group (p < 0.05). SpO2 was significantly lower in the severe/critical group up to 5 min after the Bruce test when compared to the mild group, and diastolic blood pressure at the end of the Bruce test was significantly higher in the severe/critical group when compared to mild group (p < 0.05; for all comparisons). A time effect was observed for body composition, with increased lean mass, skeletal muscle mass, fat-free mass, and lean mass just for the severe/critical group after 1 year (p < 0.05). Cardiopulmonary fitness parameters did not differ among the groups, except for respiratory quotient with higher values for the severe/critical group when compared to itself after 1 year. All COVID-19 patients might present long-term sequelae, regardless of the acute disease severity. Reassessing and identifying the most prevalent long-term sequelae are essential to perform more precise health promotion interventions.
Similar content being viewed by others
Introduction
Studies have emphasized that COVID-19 is a respiratory and multisystem syndrome1,2. The persistent symptoms of COVID-19 at the end of the acute infectious response, like other viral disorders, are characterized as chronic symptomatic disorders that mainly affect the respiratory, cardiovascular, neurocognitive, digestive, and muscular systems3. These symptoms, characterized in the literature as "long COVID", are present in many of the population, ranging from 10 to 35% of those who contracted the disease4. The long COVID can present in different forms according to each individual's environmental, physiological, and lifestyle characteristics, such as age, sex, ethnicity, functional activity, social exposure, hospitalization factors, and chronic diseases5.
The persistent symptoms of long COVID have been classified into two categories1. The first classification encompasses long-term tissue damage mainly affecting the heart, lung, and neurological tissues6. The second classification focuses on chronic inflammation leading to viral persistence, lymphopenia, intestinal dysbiosis, and autoimmunity1,5. In addition, the effects of long COVID can be social, physical, and mental, generally reducing the quality of life and health of those infected by SARS-CoV-2. Fatigue, difficulty breathing, reduced activities of daily living, and memory problems are frequent sequelae reported in patients after acute infection4,7.
Studies have shown that the sequelae of long COVID are a public health problem that implies the indispensability of developing strategies for controlling and caring for these patients8,9,10. In addition, patients with severe acute COVID-19 who require admission to the intensive care unit (ICU) generally showed a higher fat mass, lower cardiorespiratory fitness, myopathies, and neuropathies6,9. Given the multifactorial effects, monitoring the clinical evolution of patients has become essential in unraveling the evolution of long COVID3,10. This follow-up is indispensable given that physical fitness is vital to performing daily living activities and obtaining good working conditions since the deleterious effects of long COVID are unknown in different organ systems until the present moment6,7,8,9,10.
Preventing and treating the physical domains impaired by the syndrome is essential9. Among them, monitoring cardiopulmonary and neuromuscular systems could promote aspects that help health professionals during rehabilitation9,11. At the best of our knowledge, few studies have assessed the recovery of COVID-19 sequelae after a first diagnostic assessment. Given this, the present study aimed (1) to evaluate body composition, cardiopulmonary fitness, and long symptoms of individuals affected by COVID-19; (2) to compare the features according to the severity of acute disease; and (3) to assess cardiopulmonary and hemodynamic recovery post stress-test after 1 year. We hypothesize that individuals who presented with more severe acute disease have a slower recovery of cardiopulmonary fitness and long-term symptoms.
Materials and methods
Study design
According to the recommendations of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, this was an observational, longitudinal, and prospective study12. We assessed body composition, cardiorespiratory fitness, and persistent symptoms of COVID-19 at two time points with a one-year interval. Participants were allocated into three groups according to disease severity: mild group (no hospitalization), moderate group (hospitalization inward care only), and severely/critically ill group (hospitalized in ICU). The evaluations were conducted at the Exercise Physiology Laboratory of the Higher Education Institution between August and December 2021 and between August and December 2022. In this period, no interventions were performed. Both assessments followed the same procedures. The participants fulfilled a detailed anamnesis with personal information, previous medical history, continuous use of medication, persistent symptoms after COVID-19, and if applied, time of hospitalization and respiratory support modalities required during hospital care.
Participants
One hundred and seventy-one patients (52 with severe/critical cases of COVID-19, 58 moderate cases, and 61 mild cases) were evaluated in 2021 (baseline). After 1 year, 90 patients were re-evaluated: 29 severe/critical cases, 32 moderate cases, and 29 mild cases. Eighty-one patients did not return for reevaluations. Figure 1 shows the flowchart of the present study. COVID-19 symptoms classified patients according to the Independent Oversight Advisory Interim Report on Wealth Health Organization (WHO's) response to COVID-1913. Inclusion criteria were: (i) all patients with a positive diagnosis of COVID-19 by reverse transcription-polymerase chain reaction testing (RT‒PCR) between January and July 2021; (ii) aged between 18 to 65 years; (iii) who were overweight or obese classified by body mass index (≥ 25.00 kg/m2)14; (iv) who received at least one dose of the vaccine against COVID-19; and (v) who received medical clearance to perform the stress test. Exclusion criteria were (i) individuals with disabling neurological disease and reduced mobility or (ii) another physical condition that could affect the performance of the Bruce test.
The sample size was calculated in the G*Power software version 3.1 using the repeated measures analysis of variance (ANOVA) test, with an estimate of the effect based on the values of peak oxygen consumption (VO2 peak) found in a previous study with long COVID-19 patients9, considering a significance level of α = 0.05 and a correlation between repeated measures of 0.5; a sample size of 90 individuals was estimated for β = 80%. All participants were informed about the study's aims and possible risks and benefits. They were guaranteed confidentiality and invited to sign the free and informed consent form. The Local Research Ethics Committee approved the study under number 4.546.726/2021, according to resolution 466/2012. The study was conducted according to the provisions of the Declaration of Helsinki.
Baseline and follow-up assessments
All assessments were performed over two days, with a 24-h interval. A medical clearance, anthropometry, and body composition were performed on the first day. Twenty-four hours later, if the patients were eligible, the Bruce test with direct gas analyzer was performed with monitoring of systolic and diastolic blood pressure (SBP and DBP, mmHg), heart rate (HR, bpm) and peripheral oxygen saturation (SpO2, %) up to 15 min after the test according to a previous study9. All assessments (baseline and after 1 year) were conducted by the same technical team (with four researchers), which presented an intraclass coefficient correlation of 0.96 to 0.99.
All anamnesis data were self-reported by the patients. The following information was collected: (i) sociodemographic data; (ii) previous morbid history and continuous use of medication; (iii) characteristics of SARS-CoV-2 infection (symptoms, hospital length of stay, and respiratory support modalities during hospitalization); (iv) persistent symptoms after COVID-19 infection; (v) lifestyle habits; and (vi) self-reported physical activity routine.
Anthropometry and body composition
Height was measured using a Sanny stadiometer (model ES 2030, São Bernardo do Campo, São Paulo, Brazil), measuring 2.20 m with an accuracy of 0.1 cm. Body mass was measured using a Welmy mechanical scale (Model 104A, Santa Bárbara d'Oeste, São Paulo, Brazil), with a capacity of 300 kg and accuracy of 100 g. Body composition was analyzed by the bioelectrical impedance analysis used with an INBODY 570 device (Biospace Co. Ltd., Seoul, Korea), which has eight tactile points, 250 kg capacity, and 100 g precision.
The variables analyzed were body mass (kg), lean mass (LM, in kg), skeletal muscle mass (SMM, in kg), fat mass (FM, in kg), fat-free mass (FFM, in kg), and body fat (BF). The following equation was used to calculate the body mass index (BMI): body mass (kg)/height (m)2. The participants were previously instructed to fast for 4 h, not to ingest (caffeine and water), to stop drinking alcohol for at least two days before, not to practice physical exercise for at least 24 h earlier, to urinate 30 min before, and not to wear metal accessories on the body during assessments15.
Bruce’s protocol with vital signs monitoring
The stress test was performed using an INBRAMED treadmill (model ATL 24, Porto Alegre, Rio Grande do Sul) according to the Bruce protocol16. First, the vital signs at rest were measured: HR (H10 model, Polar, Kempele, Finland), SpO2 (ALFAMED, sense 10 model, Lagoa Santa, Minas Gerais, Brazil) positioned on the index finger, and blood pressure with a stethoscope and sphygmomanometer (PREMIUM brand, standard model, São Paulo, Brazil). All vital signs were measured according to the recommendations of the VIII Brazilian Guideline on Arterial Hypertension17.
The tests were performed with complete monitoring of a multidisciplinary team (physicians, nurses, and physical education professionals with specialization in exercise physiology), continuous monitoring of HR, SpO2, and measurement of the patient's rating of perceived exertion (RPE) using the Borg scale (6–20 points)18. All participants were previously instructed to answer the RPE on the Borg scale. The distance walked in meters (m) during the test was also recorded since the test time could not be used because, in 2021, at least 20% of the patients performed the Adapted Bruce test. The exercise test was terminated in the following cases: (i) voluntary withdrawal of the participant; (ii) RPE ≥ 19; (iii) respiratory quotient (RQ) > 1.15; (iv) signs of exercise intolerance such as lower limb fatigue; and (v) physical inability to maintain intensity during the test.
Gas exchange was analyzed using a VO2000 device (MEDGRAPHICS Corp., Saint Paul, United States of America), with measurement of VO2 (L/min) and VCO2 (L/min), minute-ventilation (VE, L/min), oxygen pulse (VO2/HR), RQ and HR, via a Polar H10 heart rate monitor (POLAR, Kempele, Finland). The equipment was calibrated at each ergo spirometric analysis according to the standardization recommendation9. The peak relative values for VO2 and VCO2 were used for the statistical analyses. Peak values were determined by the highest values obtained during the stress test. The laboratory room temperature was standardized at 24 °C.
Monitoring of vital signs
At the end of the Bruce test, the patients were placed in a quiet environment. HR and SpO2 were monitored minute by minute for 15 min. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured immediately after the exercise and every 5 min for the next 15 min by the same evaluators at both measurement times, i.e., 2021 and 20229.
Statistical analysis
The distributions of the continuous variables were analyzed using skewness, kurtosis tests, and visual inspection of the histograms. Data with a normal distribution are described as the mean ± standard deviation. Data with a non-normal distribution is described as the median and percentiles (25–75%). Categorical data were expressed as absolute (n) and relative frequency (%). A significance of 5% was established for all statistical tests (p < 0.05). To analyze possible differences between the characteristics of the three groups, we used the one-way ANOVA for the continuous variables and the chi-square test (χ2) for the categorical variables. A repeated two-way ANOVA was used for the stress test performance and body composition variables with time (baseline vs 1 year), group (mild, moderate, and severe/critical group), and time-group interaction effects. An ANOVA was also used to analyze the delta variability in post-Bruce test vital signs (from 1 to 15 min after the end of the test). Post-hoc Bonferroni correction was used for all ANOVA analyses when a significant difference was found. A paired t-test (before vs after 1 year) was applied when a time difference was identified to verify potential statistical significance in intra-group conditions, and the confidence interval (CI) was also calculated19. The partial effect size eta square (ƞ2p) was classified according to Richardson20: 0.0099 (small effect), 0.0588 (moderate effect), and 0.1379 (large effect). Cohen's d was calculated and classified following the classification: 0.2 (small effect), 0.5 (moderate effect), and 0.8 (large effect)21. Statistical analyses were performed using JAMOVI software, version 1.6.23.
Ethical approval
This study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Scientific Ethics Committee of University Cesumar (approval number: No. 4.546.726/2021). Informed consent was obtained from all subjects involved in the study.
Results
Sample characteristics
Table 1 presents the participants' characteristics according to each group. No significant differences existed between the groups for any aspects (p > 0.05), except for the length of hospitalization (p < 0.05), with more days of hospitalization for the severe group when compared to the other groups.
Long-term symptoms
Table 2 presents self-reported persistent long COVID symptoms in the second time point. The most prevalent symptoms were memory deficit (66.7%), fatigue (65.6%), difficulty concentrating (51.7%), and dyspnea (40.0%). No significant differences were found in the groups (p > 0.05).
Body composition
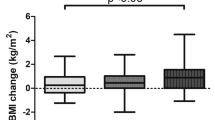
Table 3 presents the participants' body mass, BMI, and body composition.
A time effect was observed for all groups, with a significant increase in LM (p = 0.003; ƞ2p = 0.10—moderate), SMM (p = 0.002; ƞ2p = 0.12—moderate), and FFM (p < 0.001; ƞ2p = 0.19—large); and a significant reduction in BF (p = 0.014; ƞ2p = 0.07—small).
A time-group interaction was observed for LM (p = 0.025; ƞ2p = 0.09—moderate), with higher values for the severe/critical group after 1 year (p = 0.003); and FFM (p = 0.046; ƞ2p = 0.07—moderate), with higher values in the severe/critical group after 1 year (p = 0.001). After time effect analysis, a t-test was performed to identify possible intra-group differences, with the severe/critical group showing a significant increase in LM (p = 0.017; d = 0.47, 95% CI 0.08 to 0.85—small), SMM (p < 0.001; d = 0.92, 95% CI 0.47 to 1.37—large) and FFM (p < 0.001; d = 0.79, 95% CI 0.35 to 1.22—moderate), but no significant differences were observed in BF (p > 0.05). The moderate group significantly decreased BF (p < 0.003; d = 0.68, 95% CI − 1.08 to − 0.22—moderate). No significant differences were found in LM, SMM, BF, and FFM for mild and moderate groups (p > 0.05).
There was a group effect for FM (p = 0.043; ƞ2p = 0.08—moderate), with higher values in the severe/critical group compared to the mild group (p = 0.037). There were no group, time, or interaction effects for body mass and BMI (p > 0.05).
Bruce test
Table 4 shows the parameters from the Bruce test.
A time effect was observed for all patients about the distance walked (p = 0.037; ƞ2p = 0.05—small), with an increase after 1 year. However, the t-test showed a significant difference in the severe/critical group (p = 0.017; d = 0.47, 95% CI 0.08 to 0.85—small). Mild and moderate groups did not present a significant difference for distance walked (p > 0.05). A group effect was observed for SpO2 (p = 0.043; ƞ2p = 0.08—moderate), with lower values in the severe/critical group compared to mild (p = 0.002) and moderate (p < 0.001) groups. A group effect was observed for DBP (p = 0.005; ƞ2p = 0.12—moderate), with lower values in the mild group compared to the moderate (p = 0.008) and severe/critical (p = 0.018) groups. A time-group interaction was observed for RQ (p < 0.001; ƞ2p = 0.31—large), with significantly higher values in the severe/critical group (p = 0.003) after 1 year. However, no time, group, or interaction effects were observed for peak VO2 peak, HR peak, final SBP, or RPE (p > 0.05).
Monitoring vital responses after the Bruce test
Figure 2 shows the SpO2, HR, SBP, and DBP monitoring from 1 to 15 min after the stress test. For the post-stress test, SpO2, we observed a time-group interaction (p < 0.001; ƞ2p = 0.16—large) from 1 to 6 min after the test, with lower values in the severe/critical group compared to mild (p < 0.001) and moderate (p < 0.001) groups.
Peripheral oxygen saturation, heart rate, systolic and diastolic blood pressure after Bruce testing at different times in the post-COVID-19 survivor groups. Panel (A) = SPO2 (peripheral oxygen saturation for 15 min after the test); Panel (B) = HR (heart rate for 15 min after the test); Panel (C) = SBP (systolic blood pressure, 5 min after the test, 10 min after the test and 15 min after the test); Panel (D) = DBP (diastolic blood pressure, 5 min after the test, 10 min after the test and 15 min after the test); † = group difference (p < 0.05, severe/critical group vs moderate and mild groups); # = Time difference with higher values 5 min after test when compared to 15 min after test (p < 0.05).
Also, a group effect for SpO2 was detected (p < 0.001; ƞ2p = 0.20—large), with lower values in the severe/critical group compared to mild (p < 0.001) and moderate (p < 0.001) groups.
For the post-stress test SBP, a group effect was observed (p = 0.024; ƞ2p = 0.08—moderate), with lower values in the mild group than in the moderate group (p = 0.02). Post-stress test HR and DBP had no group, time, or interaction effects (p > 0.05).
Discussion
The present study aimed to analyze the self-reported sequelae after COVID-19 and possible changes in body composition and cardiorespiratory fitness after 1 year. The main outcomes observed were (i) the most recurrent persistent symptoms were related to memory deficit, fatigue, difficulty concentrating, dyspnea, and capillary loss, although there were no differences between the severity of disease; and (ii) increasing LM, SMM, and FFM in the severe/critical group after 1-year; (iii) a group effect with lower values of BF in the FM mild group when compared to the severe/critical group; (iv) increase in the distance walked in the Bruce test just for the severe/critical group after 1 year; (vi) a group effect for DBP post-Bruce test with lower values for mild group when compared to moderate and severe/critical groups. No significant differences were observed for the anthropometric and other ergospirometric and hemodynamic variables.
Long COVID symptoms
The persistent symptoms of COVID-19 require early intervention to minimize possible sequelae in different severity symptoms, i.e., mild, moderate, and severe/critical patients with follow-up. Asymptomatic patients also require follow-up, especially those with some vulnerable condition or associated comorbidities, combined with multidisciplinary actions to promote better outcomes given the reported sequelae22,23.
Body composition responses in long COVID patients
Patients with greater loss of LM 6 months after contracting COVID-19 could not recover muscle health24, and unplanned hospitalizations tend to promote a reduction in upper limb muscle strength, LM, limb muscle strength, maximum isometric handgrip strength, and of one-repetition maximum in the extensor chair25,26. Long COVID-19 patients also have a decreased LM compared to a control group (without a diagnosis of COVID-19)23. However, the present study showed increased LM, SMM, and FFM for severe/critical patients after 1 year. Discrepancies in the measurement time between the present study and the others may justify this finding. In this regard, there was a need for medical clearance of the present study participants to perform submaximal exercise tests, and the recovery time will depend on each patient and possible limitations sequels.
The convalescent period after infection is susceptible to different complications, especially with distinct immunological signatures that open an "immunological window," allowing the development of complications such as acute myocardial infarction and myocarditis, among other clinical manifestations27,28. Therefore, the morphophysiological parameters were collected after recovery from the acute sequelae recorded within the subdivision of symptoms (mild, moderate, and severe/critical). Another study indicates that long COVID-19 patients showed significant improvement in muscle strength, mobility, and cardiorespiratory fitness after 12 months of in-person physical therapy rehabilitation29.
Previous studies found that BF was higher in long COVID patients compared to a control group (i.e., without a diagnosis of COVID-19), and outpatients had a lower FM compared to patients hospitalized for COVID-19 with the same BMI9,26. Considering the difference in FM between groups persisted after 1 year (mild with lower FM vs severe/critical cases), actions to provide improvement in body composition, with reduction of FM, BF, and increase in LM, remain substantial for improving the body composition of COVID-19 survivors. Concurrent training may be a relevant strategy for improving health-related physical fitness by increasing muscle strength, cardiorespiratory fitness, and LM, in addition to reducing FM30. Rehabilitation sessions via aerobic and resistance exercise should control volume, intensity, density, frequency, and progression based on each clinical case, as well as on the physical fitness indicators that were most affected by the disease20,30.
Cardiorespiratory responses in long COVID patients
VO2 peak, HR peak, and RPE did not differ between groups, and there was no difference between the times (at baseline vs after 1 year), suggesting that the intensity was similar. Significant differences between outpatients and severe/critical patients were found previously9. The absence of differences between the groups in the present study may be related to a reduction in endothelial damage during convalescence and a subsequent return to activities of daily living for severe/critical patients9,31. However, the self-reported level of physical activity of patients with different symptoms did not differ.
The distance walked in the Bruce test increased the severe/critical group after 1 year, indicating an improvement in physical fitness and a possible reduction in sequels provoked by COVID-19 survivors. Similar responses were identified in another study, with a significant increase in the distance walked in the 6-min walk test 12 months after hospital discharge30. In the present study, less than 50% of the patients (mild, moderate, and severe/critical) reported being physically active, i.e., > 150 min of physical activity/week. However, the improvement in patients' cardiorespiratory fitness is also associated with the physical reconditioning of individuals who returned to their respective activities of daily living, in addition to a possible reduction in residual inflammation and organic damage (this condition was not analyzed in the present study)9,31.
Considering the increased distance walked during the Bruce test, an interaction was also observed with higher values for the RQ of severe/critical patients after 1 year, suggesting an improvement in high exercise tolerance in long duration, justified by the increased intolerance of exercise intensity29. Final SpO2 post-Bruce test showed a group effect, with significantly lower values for severe/critical patients that may be related to chronic hypoxemia after physical effort or even to vascular and pulmonary changes and a decrease in pulmonary function10,31,32.
The main signals that affect respiratory control are derived from the response of peripheral chemoreceptors and mechanoreceptors, in addition to an abnormal muscular effort of thoracic muscles and a reduction in lung compliance, accentuating dyspnea that affects performance in the effort33. Cardiorespiratory rehabilitation of these patients requires monitoring of SpO2, blood pressure, and cardiac function in addition to the application of the principles of interdependence between volume and intensity, increasing loads, biological individuality, and periodic assessments for evaluation of outcomes and mitigation of possible sequelae of COVID-1934.
Blood pressure responses in long COVID patients
Post-Bruce test DBP test also differed among long COVID-19 patients, with higher values for the moderate and severe/critical group than for the mild group. DBP response during physical exertion is related to comorbidities (prevalence of obesity, systemic arterial hypertension, diabetes mellitus, and tobacco use) but is not independently associated with a higher risk of death from cardiovascular diseases35. A systematic review with meta-analysis identified that SBP ≥ 210 mmHg for males and ≥ 190 mmHg for females in moderate effort intensity can be considered an independent risk factor for cardiovascular events and increased mortality36. The physiology and pathophysiology of DBP after physical effort have not yet been fully elucidated, and the increase in DBP may be concatenated to greater peripheral arteriolar resistance, increased afterload, and even arterial stiffness or dysfunction, and early signs of atherosclerotic vascular disease35.
A high DBP response to physical effort is a predictive factor for increased systemic arterial hypertension. Because systemic arterial hypertension can often be asymptomatic, patients can progress to structural and/or functional changes in target organs and endothelial dysfunction, with an imbalance between vasodilating and vasoconstrictor substances affecting vascular function, with reduced blood pressure compliance capacity of the great arteries, impairing pressure homeostasis37,38. A study observed a significant increase in SBP and DBP in males and females during the pandemic period (between 2019 and 2020)39, probably associated with increased alcohol consumption, weight gain, lower level of physical activity, emotional stress, and less continuous medical care (with reduced medication adherence), although the parameters above were not observed and/or measured in the present study, since the collections were performed strictly during the pandemic period. However, the increase in SBP during physical exercise is a normal response related to the intensity of effort40.
Limitations, strengths, and future directions
This study has some limitations. First, the lack of follow-up during the acute infection of the patients is justified by the intolerance to exercise. Second, there was no follow-up after the 1 year between the evaluations, and behavior changes (e.g., physical activity and nutrition habits) and other features not accessed in the study might be associated with improving body composition and cardiopulmonary fitness in the hospitalized groups. Third, the loss of follow-up in the second evaluation may have impacted the results; however, loss rates were similar in all groups. Unfortunately, these patients opted not to return for re-assessment, and this was related to (i) lack of time; (ii) not understanding the necessity to perform a re-assessment; (iii) patients believe they are completely recovered from COVID; and (iv) lack of financial resources to travel to university and not work part-time. Considering the future perspectives for research, Patients may be evaluated over months and even years to understand pathophysiological responses. Furthermore, actions that seek to assess, intervene, and re-evaluate long COVID patients associated with a control group (without the disease) may guide more assertive rehabilitation actions.
Clinical applications
Given clinical relevance, some points can be highlighted: (i) hospitalized patients need to be monitored periodically on body composition, cardiorespiratory fitness, and vital signs; (ii) all COVID-19 survivors independently of the disease severity can be monitored about fatigue, dyspnea, muscle pain, joint pain, dizziness, tinnitus, sensation of hearing loss, otalgia, ageusia, anosmia, memory deficit, difficulty concentrating and capillary loss and (iii) earlier interventions with health professionals can reduce the possible impacts (sequels) of COVID-19.
Conclusions
Cardiopulmonary fitness parameters did not differ among the groups, but severe/critical cases maintained worse hemodynamic responses to exercise. Fat mass showed higher values in severe/critical cases than in mild cases in which excess adiposity is related to low-grade inflammation. Considering these responses, regular physical activity and healthy nutrition programs are fundamental for all long COVID patients (biological individuality, symptoms, possible limitations, among other aspects). The prevalence of long-term symptoms among the groups was not different either. Neurocognitive dysfunction, fatigue, and dyspnea are the most prevalent long-term symptoms. Regardless of the severity of COVID-19, it is essential to reassess and identify the most prevalent long-term sequelae so that more precise health promotion interventions can be performed.
Data availability
The datasets generated during and/or analyzed during the current research are available from the corresponding author upon reasonable request.
Abbreviations
- ANOVA:
-
Analysis of variance
- BF:
-
Body fat
- BM:
-
Body mass
- BMI:
-
Body mass index
- DBP:
-
Diastolic blood pressure
- FFM:
-
Fat-free mass
- FM:
-
Fat mass
- HR:
-
Heart rate
- ICU:
-
Intensive care unit
- LM:
-
Lean mass
- RPE:
-
Rating of perceived exertion
- RQ:
-
Respiratory coefficient
- SBP:
-
Systolic blood pressure
- SMM:
-
Skeletal muscle mass
- SpO2 :
-
Peripheral oxygen saturation
- VCO2 :
-
Carbon dioxide production
- VE:
-
Minute-ventilation
- VO2 :
-
Oxygen consumption
References
Yong, S. J. Long COVID or post-COVID-19 syndrome: Putative pathophysiology, risk factors, and treatments. Infect. Dis. 53, 737–754 (2021).
Naeije, R. & Caravita, S. Phenotyping long COVID. Eur. Respir. J. 58, 2101763 (2021).
Astin, R. et al. Long COVID: Mechanisms, risk factors and recovery. Exp. Physiol. 108, 12–27 (2023).
Van Kessel, S. A. M., Olde Hartman, T. C., Lucassen, P. L. B. J. & van Jaarsveld, C. H. M. Post-acute and long-COVID-19 symptoms in patients with mild diseases: a systematic review. Fam. Pract. 39, 159–167 (2022).
Raman, B., Bluemke, D. A., Lüscher, T. F. & Neubauer, S. Long COVID: post-acute sequelae of COVID-19 with a cardiovascular focus. Eur. Heart J. 43, 1157–1172 (2022).
Aiyegbusi, O. L. et al. Symptoms, complications and management of long COVID: A review. J. R. Soc. Med. 114, 428–442 (2021).
Cau, R. et al. Long-COVID diagnosis: From diagnostic to advanced AI-driven models. Eur. J. Radiol. 148, 110164 (2022).
Chen, C. et al. Global prevalence of post-coronavirus disease 2019 (COVID-19) condition or long COVID: A meta-analysis and systematic review. J. Infect. Dis. 226, 1593–1607 (2022).
Lemos, M. M. et al. Body composition and cardiorespiratory fitness in overweight or obese people post COVID-19: A comparative study. Front Physiol. 13, (2022).
Caruso, D. et al. Post-acute sequelae of COVID-19 pneumonia: Six-month chest CT follow-up. Radiology. 301, E396–E405 (2021).
Wang, F., Kream, R. M. & Stefano, G. B. Long-term respiratory and neurological sequelae of COVID-19. Med. Sci. Monit. 26, e928996 (2020).
Vandenbroucke, J. P. et al. Strengthening the reporting of observational studies in epidemiology (STROBE): Explanation and elaboration. Epidemiology. 18, 805 (2007).
World Health Organization. COVID-19 clinical management: living guidance, 25 January 2021. https://apps.who.int/iris/handle/10665/338882 (2021).
Obesity: Preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser 894, i–xii, 1–253 (2000).
Branco, B. H. M. et al. Proposal of a normative table for body fat percentages of Brazilian young adults through bioimpedanciometry. J. Exerc. Rehabil. 14, 974–979 (2018).
Bruce, R. A., Kusumi, F. & Hosmer, D. Maximal oxygen intake and nomographic assessment of functional aerobic impairment in cardiovascular disease. Am. Heart J. 85, 546–562 (1973).
Barroso, W. K. S. et al. Diretrizes Brasileiras de Hipertensão Arterial—2020. Arq. Bras. Cardiol. 116, 516–658 (2021).
Borg, G. A. V. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 14, 377 (1982).
Sordi, A. F. Effects of a multi-professional intervention on body composition, physical fitness and biochemical markers in overweight COVID-19 survivors: A clinical trial. Front. Physiol. 14, (2023).
Richardson, J. T. E. Eta squared and partial eta squared as measures of effect size in educational research. Educ. Res. Rev. 6, 135–147 (2011).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences. (Routledge, 2013).
Davis, H. E., McCorkell, L., Vogel, J. M. & Topol, E. J. Long COVID: Major findings, mechanisms and recommendations. Nat. Rev. Microbiol. 21, 133–146 (2023).
Peramo-Álvarez, F. P., López-Zúñiga, M. Á. & López-Ruz, M. Á. Medical sequels of COVID-19. Med. Clin. 157, 388–394 (2021).
Gil, S. et al. Acute muscle mass loss predicts long-term fatigue, myalgia, and health care costs in COVID-19 survivors. Am. Med. Dir. Assoc. 24, 10–16 (2023).
Hartley, P. et al. Change in skeletal muscle associated with unplanned hospital admissions in adult patients: A systematic review and meta-analysis. PLoS ONE. 14, e0210186 (2019).
Ramírez-Vélez, R. et al. Reduced muscle strength in patients with long-COVID-19 syndrome is mediated by limb muscle mass. J. Appl. Physiol. 1985(134), 50–58 (2023).
Tu, T. M. et al. Acute ischemic stroke during the convalescent phase of asymptomatic COVID-2019 infection in men. JAMA Netw. Open. 4, e217498 (2021).
Chu, C.-F. et al. Convalescent COVID-19 patients without comorbidities display similar immunophenotypes over time despite divergent disease severities. Front. Immunol. 12, 601080 (2021).
Berentschot, J. C. et al. Physical recovery across care pathways up to 12 months after hospitalization for COVID-19: A multicenter prospective cohort study (CO-FLOW). Lancet Reg. Health Eur. 22, 100485 (2022).
Jimeno-Almazán, A. et al. Effects of a concurrent training, respiratory muscle exercise, and self-management recommendations on recovery from post-COVID-19 conditions: The RECOVE trial. J. Appl. Physiol 1985(134), 95–104 (2023).
Carfì, A., Bernabei, R., Landi, F., & for the Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent symptoms in patients after acute COVID-19. JAMA. 324, 603–605 (2020).
Ueto-Robledo, G. et al. Severe pulmonary hypertension: An important sequel after severe post-acute COVID-19 pneumonia. Curr. Probl. Cardiol. 47, 101004 (2022).
Dhont, S., Derom, E., Van Braeckel, E., Depuydt, P. & Lambrecht, B. N. The pathophysiology of “happy” hypoxemia in COVID-19. Respir. 21, 198 (2020).
Deng, M. C. An exercise immune fitness test to unravel mechanisms of Post-Acute Sequelae of COVID-19. Expert Rev. Clin. Immunol. 0, 1–5 (2023).
Sydó, N. et al. Significance of an increase in diastolic blood pressure during a stress test in terms of comorbidities and long-term total and CV mortality. Am. J. Hypertens. 31, 976–980 (2018).
Schultz, M. G. et al. Exercise-induced hypertension, cardiovascular events, and mortality in patients undergoing exercise stress testing: A systematic review and meta-analysis. Am. J. Hypertens. 26, 357–366 (2013).
Singh, J. P. et al. Blood pressure response during treadmill testing as a risk factor for new-onset hypertension. Circ. 99, 1831–1836 (1999).
Gallo, G., Volpe, M. & Savoia, C. Endothelial Dysfunction in Hypertension: Current Concepts and Clinical Implications. Front. Med. (Lausanne). 8, 798958 (2021).
Laffin, L. J. et al. Rise in blood pressure observed among US adults during the COVID-19 pandemic. Circ. 145, 235–237 (2022).
Wielemborek-Musial, K., Szmigielska, K., Leszczynska, J. & Jegier, A. Blood pressure response to submaximal exercise test in adults. Biomed. Res. Int. 2016, 5607507 (2016).
Acknowledgements
The authors thank all the patients who participated in the present study.
Funding
This research received funding from the PPSUS—Araucaria Foundation Program in partnership with the National Council for Scientific and Technological Development (CNPq), CP-19: Young P.hD´s of Araucaria Foundation, and Cesumar Institute of Science, Technology and Innovation (ICETI). The funding sources had no role in the study design, conduction, or manuscript writing.
Author information
Authors and Affiliations
Contributions
V.A.S.P. contributed to the study design, coordination, data collection, data analysis, manuscript draft, writing, and review. A.F.S. contributed to the study design, coordination, data collection, data analysis, manuscript writing, and review. M.M.L. contributed to study design, coordination, and manuscript draft and review. J.S.A.F. contributed to data collection, manuscript writing, and review. V.B.N.C. contributed to data collection, manuscript writing, and review. J.S.A.F. contributed to data collection, analysis, and manuscript writing and review. V.B.N.C. contributed to data collection, analysis, and manuscript writing and review. B.F.S. contributed to data collection, analysis, and manuscript writing and review. S.P.R. contributed to study design, supervision, and manuscript draft and review. P.V.-B. contributed to the study design and manuscript draft and review. J.M. contributed to the study design, supervision, and manuscript draft and review. B.H.M.B. coordinated the study and contributed to its design, supervision, manuscript draft, writing, and review. All authors have read and agreed to the final version of the manuscript and agreed with the authors' presentation order.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Perli, V.A.S., Sordi, A.F., Lemos, M.M. et al. Body composition and cardiorespiratory fitness of overweight COVID-19 survivors in different severity degrees: a cohort study. Sci Rep 13, 17615 (2023). https://doi.org/10.1038/s41598-023-44738-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-44738-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.