Abstract
Cystic fibrosis transmembrane conductance regulator modulators have revolutionized cystic fibrosis (CF) care in the past decade. This study explores the CF-related mortality trends in the US from 1999 to 2020. We extracted CF-related mortality data from the CDC WONDER database. CF age-standardized mortality rates (ASMRs) were identified by ICD-10 code E84 and were stratified by demographic and geographical variables. Temporal trends were analyzed using Joinpoint modeling. CF-related ASMRs decreased from 1.9 to 1.04 per million population (p = 0.013), with a greater reduction in recent years. This trend was replicated in both sexes. The median age of death increased from 24 to 37 years. CF mortality rates decreased across sex, white race, non-Hispanic ethnicity, census regions, and urbanization status. Incongruent trends were reported in non-white races and Hispanic ethnicity. A lower median age of death was observed in women, non-white races, and Hispanic ethnicity. SARS-CoV-2 infection was the primary cause of death in 1.7% of CF decedents in 2020. The national CF-related mortality rates declined and the median age of death among CF decedents increased significantly indicating better survival in the recent years. The changes were relatively slow during the earlier period of the study, followed by a greater decline lately. We observed patterns of sex, ethnic, racial, and geographical disparities associated with the worsening of the gap between ethnicities, narrowing of the gap between races and rural vs. urban counties, and closing of the gap between sexes over the study period.
Similar content being viewed by others
Introduction
Cystic fibrosis (CF) is an inherited multi-system disease with a shortened life expectancy, affecting approximately 40,000 people in the United States (US) and 105,000 people worldwide1. Although CF is most common in Caucasians, affecting 1 in 2500 live births, it is being increasingly recognized in other races and ethnicities including 1 in 10,500 Native Americans, 1 in 13,500 Hispanic-Americans, 1 in 15,000 African-Americans, and 1 in 35,000 Asian-Americans2,3. Cystic fibrosis is an autosomal recessive disease caused by mutations in the cystic fibrosis transmembrane conductance regulator protein (CFTR), resulting in defective chloride and bicarbonate secretion in epithelial cells, with greater than 2000 CFTR mutations identified to date4. While CFTR dysfunction is genetically influenced by the mutation class, often leading to life-threatening complications5, the F508del mutation accounts for the majority of mutated alleles in North American and northern European populations.
Cystic fibrosis care has evolved steadily over the last 70-plus years, most dramatically in the past decade, with advances in diagnostic and therapeutic options including nationwide newborn screening to identify CF at birth, aggressive infection prevention, targeted nutritional support, augmentation of pancreatic function, improved mucociliary clearance, and the implementation of highly effective CFTR modulators6. Several recent studies have reported encouraging survival trends7,8,9,10. To date, the available studies from the US utilized data from the Cystic Fibrosis Foundation Patient Registry (CFFPR), composed of patients cared for at specialized CF centers. The US CFFPR is a comprehensive registry that records 81–84% of CF patients with a 90% retention rate, capturing close to 72–75% of CF patients for clinical research11. However, no recent US study has reported CF-related mortality trends incorporating the decedents who did not participate in CFFPR. The Centers for Disease Control and Prevention (CDC) Wide-ranging Online Data for Epidemiologic Research (WONDER) Multiple Cause of Death (MCOD) data files, which cover the death records of the whole US population, provide a unique opportunity to analyze the evolution of CF mortality independent of the decedents' participation in the CFFPR.
In this study, we aimed to investigate temporal trends of CF-related mortality in the US. We also explored disparities by demographic characteristics and geographical regions.
Methods
Study design
This is a retrospective observational study performed using CDC WONDER MCOD datafiles from 1999 to 2020. MCOD data files are compiled by the Division of Vital Statistics, the National Center for Health Statistics (NCHS), and the United States Department of Health and Human Services. The death certificate of US residents is the primary source of data. The method of translation of death certificate data into MCOD files has been reported previously12. MCOD files include an underlying cause of death (UCOD), up to 20 additional contributing causes of death, and demographic information about the decedents. The UCOD is defined as “the disease or injury which initiated the train of events leading directly to the death". Deidentified data was publicly available for researchers and an institutional review board endorsement was not required.
Study definitions
CF-related deaths were identified by the International Classification of Diseases-10 (ICD-10) code E84. Age-standardized mortality rates (ASMRs) were stratified by sex, race, ethnicity, census region, urbanization, and age. For race-specific estimates, we reported four race categories: White, African American, American Indians/Alaska Natives, and Asian/Pacific Islanders. For urbanization-specific data, the analysis was reported based on counties’ population sizes > 250 K, and < 250 K. In addition, we calculated the median age of death in the years 1999, 2005, 2010, 2015, and 2020 for the CF population and at the start and end of the study period for each sex, race, and ethnicity to make our analysis comparable to previously published literature.
Statistical analysis
CF-related ASMRs with a 95% confidence interval (CI) were extracted. The use of age standardization is common in epidemiological studies to provide information regarding changes in mortality trends over time as well as for making comparisons between demographic variables. For this analysis, we used the 2000 US standard population as provided by the CDC, and mortality rates were computed as deaths per million population. The extracted ASMRs were analyzed using Joinpoint regression modeling (Command Line Version 4.5.0.1), a statistical software developed by the US National Cancer Institute Surveillance Research Program to identify any changes in mortality trends and time points with significant inflections13,14. The average annual percentage changes (AAPCs) with CI were reported. Additionally, the model computes an estimated annual percentage changes (EAPCs) for each trend segment by fitting a regression line to the natural logarithm of ASMRs. Null hypothesis testing of whether EAPCs or AAPCs were non-zero was performed. Per CDC protocol to ensure confidentiality, individual mortality trends for each non-white race were not reported due to < 10 deaths per annum and automated suppression of MCOD datafiles. We re-analyzed the CF mortality trends by using UCOD as the case definition to record any disparities based on the alternate case definition. UCOD CF mortality rates were stratified similarly to the primary analysis, but Joinpoint analysis was performed on overall national-level data only.
Sensitivity analysis
After completing the primary analysis, we planned two separate post hoc sensitivity analyses to assess the potential influence of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic on the observed trends. First, we extended the observation period to include provisional mortality statistics for the year 2021 which are also provided by the CDC. Briefly, the provisional mortality data reports all the variables similar to the final MCOD files. The only difference is provisional data might not include all the deaths. However, this entry process lags by just a few weeks making 2021 data equally reliable. We graphically inspected the mortality trends and repeated the Joinpoint analysis including the additional year of data. Second, if the observed trends in CF mortality were due to changes that came with the SARS-CoV-2 pandemic, it is possible that mortality from other chronic respiratory diseases would follow a similar trend after the onset of the pandemic. Therefore, we analyzed mortality data of other chronic respiratory diseases (ICD-10 codes: J40-J47; J84).
Results
From 1999 to 2020, 56,806,341 individuals died in the US, and CF accounted for 11,068 deaths (0.02%) including 5489 (49.6%) men, and 5579 (50.4%) women. The corresponding average CF-related ASMR was 1.65 per million population (95% CI 1.61–1.68). Individually in men and women, average CF-related ASMRs were 1.67 (CI 1.62–1.71) and 1.68 (CI 1.64–1.72) respectively. The highest number of deaths (n = 10479, [95%]) were recorded in Whites with ASMR of 2.01 (CI 1.97–2.05), followed by 461 (4%) in African American (ASMR, 0.48 [CI 0.43–0.52)]), 68 (0.5%) in American Indians/Alaska Native (ASMR, 0.75(CI 0.57–0.96]), and 60 (0.5%) in Asian/Pacific Islanders with ASMR of 0.17 (CI 0.13–0.23). By ethnicity, the majority of deaths [n = 10333, (93%)] occurred in non-Hispanic ethnicity with ASMR of 1.87 (CI 1.84–1.91) in comparison to Hispanic (n = 722 [7%]; ASMR, 0.64 [CI 0.59–0.69]) (Table 1).
The CF mortality rates varied across the census regions with the highest average ASMR in the Midwest (1.82 per million population [CI 1.75–1.89]) and lowest in the West (1.44, CI [1.38–1.5]). CF mortality burden was lower in counties with a population > 250 K (1.53 [CI 1.50–1.56]) versus counties < 250 K (2.13 [CI 2.06–2.20]) (Table 1).
Table 2 and Fig. S1 report the CF mortality rates analyzed by age group and sex (Fig. 1). Women CF patients reached higher and earlier peak mortality (15–24 age group) as compared to men (25–34 age group). The highest number of deaths was identified in the 5–24 age group (n = 3212 [29%]), and the lowest number of deaths was estimated in the 85 + age group (n = 105 [0.9%]). The corresponding ASMR was highest in the 15–24 age group (3.43 per million population [CI 3.31–3.55]), and lowest in the 1–4 years age group (0.31 [CI 0.25–0.37]).
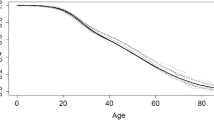
The median age of death was 24 years (interquartile range [IQR] 18–33) in 1999, and 37 years (IQR 25–55) in 2020 (Fig. 2). In 1999, a higher median age of death was observed in men compared to women, but the difference resolved over the study period (Table 3). The difference in the median age of death between Whites and other races slightly improved over the study period, but the gap widened between non-Hispanic and Hispanic ethnicities over time (Table 3).
Temporal joinpoint trends
In 1999, 539 individuals died from CF, and the annual number of deaths decreased to 342 in 2020. The corresponding CF ASMRs decreased from 1.9 per million population (CI 1.74–2.06) to 1.04 (CI 0.93–1.16) (AAPC, − 2.6% [CI − 4.7 to − 0.6]; p = 0.013). Two Joinpoint trends indicated a steady decline in mortality from 1999 to 2018 (EAPC, − 0.7% [CI, − 1.4 to 0]; p = 0.037), followed by a trend toward significant reduction from 2018 to 2020 (EAPC, − 19.1% [CI: − 35.7 to 1.8]; p = 0.068). Similar trends were observed individually in each sex (Tables 4, S1; Figs. 1, 3).
CF ASMRs decreased in the white population from 2.28 per million population (CI 2.09–2.48) to 1.23 (CI: 1.09–1.37) (AAPC: − 2.6% [− 4.5 to − 0.6]; P = 0.013). However, a flat trend was observed in all other races combined (AAPC, 0% [CI − 1.5 to 1.5]; p = 0.98) (Table 4; Fig. 3). In the non-Hispanics, ASMR decreased from 2.16 (CI 1.97–2.34) to 1.14 (CI 1–1.3) [AAPC, − 2.6% (CI − 4.6 to − 0.5); p = 0.016]. In the Hispanic cohort, ASMR increased from 0.5 (CI 0.31–0.78) in 2000 to 0.63 (CI 0.44–0.89) in 2020 (AAPC 2.1% [CI 0.6–3.6]; p = 0.008) (Table 4; Fig. 4). The CF mortality decreased in all the census regions (p < 0.05) except West (p = 0.096) (Table 4). CF mortality rates decreased irrespective of urbanization status (p < 0.05) (Table 4).
In 1999, the highest CF mortality was seen in the 15–24 years age group [5.09 per million population (CI 4.38–5.80)] and the lowest mortality was observed in > 55-years age group [0.34 (CI 0.21–0.53)]. The CF mortality improved in < 35 years age groups (p < 0.05). There was no change in mortality in the 35–44 years age group (p = 0.379). Conversely, an increase in mortality was recorded in > 45-year age groups (p < 0.001) (Table 3).
The CF-related mortality was reanalyzed using UCOD as the case definition. The ASMR decreased from 1.71 per million population (CI 1.56–1.87) in 1999 to 0.82 (CI 0.72–0.92) in 2020 (AAPC, − 3.2% [CI − 5.2 to − 1.3%]; p < 0.001). Again, two Joinpoint trends were observed (Tables 4, S1 and S2). Notably, out of 342 CF-related deaths in 2020, only 6 (1.7%) had SARS-CoV-2 as the UCOD.
Sensitivity analysis
The first post hoc sensitivity analyses with provisional data from 2021 demonstrated a decrease in deaths from 342 in 2020 to 314 in 2021. The corresponding ASMRs decreased from 1.04 per million population (CI 0.93–1.16) to 0.95 (CI 0.85–1.06) (AAPC, − 3.2% [CI − 5.2 to − 1.3]; p < 0.01). Similar to the primary analysis, the Joinpoint analysis showed two trends; 1999–2018 (EAPC, − 0.8% [CI − 1.5 to − 0.2]; p = 0.019) and 2018–2021 (EAPC, − 16.2% [CI − 25.2 to − 6.1); p = 0.004], suggesting the durability of the significant decrease in CF-related mortality following 2018 and beyond 2020 (Fig. S2). In the second post hoc analysis, the mortality trends of chronic respiratory diseases showed an increase from 84.13 (CI 83.85–84.41) in 2019 to 92.22 (CI 91.93–92.51) in 2020 which was incongruent with CF-related ASMRs in the same period.
Discussion
There has been a significant decline in CF-related mortality in the US. While there were only gradual improvements in the earlier part of the past two decades, there has been a rapid decline in recent years. The median age of death has increased by 13 years, and the mortality rates decreased consistently across sex, white race, non-Hispanic ethnicity, census regions, and urbanization status. Conversely, the mortality rates worsened in Hispanic ethnicity and persisted without change in non-white races. In parallel, significantly lower median age of death was observed in Hispanic ethnicity and non-white races, associated with a widening gap with their respective counterparts over time. It is also worth noting that men and women had similar overall ASMRs, but women have higher mortality rates from 1 to 24 years of age and a lower median age of death, indicating a relatively poor prognosis in younger females.
The temporal trends of CF-related mortality from 1979 through 1991 were reported using the US CDC data files12, and our work builds upon this previous study. Halliburton et al.12 reported a 21% decrease in CF-ASMR and an increase in the median age of death among CF decedents from 15 to 23 years. Our analysis demonstrated a 45% decrease in CF-ASMR and an increase in the median age of death from 24 to 37 years. Consistent with our findings, the CFFPR annual report documented an improvement in the mortality rate from 1999 to 2020, although the magnitude of change was relatively higher at 62%8. Stephenson and colleagues also reported improved survival in the US and Canada; however, there was a notably lower median age of survival in the US CF population, with an absolute difference of 10 years compared to their Canadian counterparts10. Of note, our analysis identified 1107 CF-related deaths from 2019 to 2021, whereas the CFFPR annual report8 reported a relatively lower number of deaths (n = 861), indicating some degree of selection bias in registry-based mortality studies and underscoring the inclusive nature of the current analysis.
Due to the observational nature of this data, we are unable to make causal statements about the exact causes of the significant decrease in CF-related mortality recently. However, a potential contributor to the decreasing CF mortality could be the introduction of CFTR modulator therapy. Ivacaftor, the first CFTR modulator, received approval in 2012 and treated only 4–6% of the CF population15. Subsequently, the introduction of Lumacaftor-ivacaftor in July 2015 expanded the coverage to 25–30% of the CF population15. With the introduction of tezacaftor-ivacaftor in February 2018, the coverage further increased to 45% of CF patients. In October 2019, triple modulator therapy was approved for those with the F508del mutation, covering 90% of the CF population8,15. The introduction of the early access program for triple modulator therapy in the summer of 2019 for those with the most advanced disease led to the fast uptake of triple modulator therapy in the US. Out of all the eligible patients for any modulator therapy, 25% were on triple therapy in 2019, 63% in 2020, and 76% in 20218,16. Our analysis revealed a parallel trend between the stepwise approval of CFTR modulators and an increase in the median age of death. Specifically, from 2011 to 2015, there was a 4-year increase, and from 2016 to 2020, there was a 7-year increase, compared to 2 years in the previous decade. Additionally, the introduction of triple modulator therapy correlated with a fast decrease of 26% in CF-ASMR in 2020. The provisional CDC MCOD data files showed a continuous but relatively slower drop of 9% in 2021. These findings align with the mortality trends reported by the CFFPR in their annual reports8,16, although the magnitude of mortality rate reduction recorded by the CFFPR annual reports was higher at 33.3% and 13% in 2020 and 2021, respectively. Notably, the decrease in mortality burden documented in the CFFPR reports was predominantly related to the non-lung transplant CF population8,16. Another possible explanation could be the onset of the SARS-CoV-2 pandemic which coincided with the beginning of the fast decrease in CF mortality since 2019. We conducted a post hoc sensitivity analysis of national mortality trends in other common chronic respiratory diseases, which may mimic the CF population but did not observe similar trends in those populations. Interestingly, we found that SARS-CoV-2 infection was identified as a contributing cause of death in only 8 (2.3%) CF decedents in 2020.
Historically, studies have reported a lower median age of death or median survival age in CF females compared to males. The current analysis found a similar disparity at the beginning of the study but a complete closure of the gap in the year 2020, with the same median age of death between genders, which is a novel finding. Previous studies from the 1990s indicated lower survival age in CF females17,18. In a study by Rosenfield et al. in 199719, the median survival age was reported as 25.3 years in females and 28.4 years in males, using the CFFPR database. The exact reasons for this disparity are not yet fully understood. Demko et al.20 observed that CF females tend to acquire Pseudomonas aeruginosa infection at an earlier age and experience a faster decline in forced expiratory volume in 1 s (FEV1). Estrogen has been implicated in modulating ion channels in the airway epithelium, leading to increased mucus viscosity and promoting the transition of non-mucoid Pseudomonas aeruginosa infection to the mucoid type, which is a drug-resistant subtype associated with a poorer prognosis21,22. In 2014, Harness-Brumley et al.23 conducted a large cohort analysis using the CFFPR database, involving 32,766 patients over a 13-year period, and reported persistent differences in life expectancy between males and females. The authors also found that females become colonized with various microorganisms such as Pseudomonas aeruginosa, Staphylococcus aureus, Hemophilus influenzae, Aspergillus, and non-tuberculous mycobacterium at earlier ages23. Possible explanations for the closing of the mortality gender gap are the modern aggressive treatment of CF, including the use of newer antipseudomonal antimicrobials, DNase therapy to improve mucus clearance, and most recently, the highly effective CFTR modulators. Furthermore, there has been increased standardization of care for CF across the country, with specialized CF care centers playing a crucial role.
To our knowledge, this is the first nationwide US study that explores the temporal trends of CF-related mortality by ethnicity over a period of two decades. In 1999, the median age of death among Hispanic CF decedents was 17 years, compared to 24 years in non-Hispanic individuals, indicating poorer survival in the former. Over the study period, the gap between the two ethnicities widened from 7 to 12 years, and the median age of death increased to 26 years in Hispanics and 38 years in non-Hispanics in 2020. The Joinpoint analysis also revealed divergent mortality trends between the two ethnic groups, indicating a worsening of inequalities over time. A study by Rho et al.24 documented a lower mean age of death among Hispanic CF decedents from 2010 to 2014, but the gap between the ethnicities was relatively smaller. Two earlier studies conducted from 1982 to 1998 and 1991 to 2010 reported up to three times the risk of death among Hispanic CF individuals, even after adjusting for socioeconomic factors25,26. The exact reasons for the relatively poor outcomes in the Hispanic CF population could not be determined from our analysis. Possible explanations, extrapolated from other diseases, include lower health literacy, lower medical insurance coverage27, language barriers, treatment non-adherence28,29, higher exposure to tobacco or air pollution30,31,32,33, cultural acculturation34, and higher rates of malnutrition35.
Other factors that have been identified include older age at diagnosis33 and differential access to lung transplantation36,37. Hispanic patients tend to acquire Pseudomonas infection more frequently and at an earlier age, have lower FEV1, and are underrepresented in clinical trials34,37,38,39,40. The differences in mortality may also be influenced by variations in the identification of genetic mutations, with 39% of CFTR variants in Hispanic CF individuals remaining unclassified33,41,42. Moreover, the heterogeneity in CFTR genotype distribution results in 25% of Hispanic CF patients currently being ineligible for available CFTR modulator therapy based on their genotypes, compared to 10% in non-Hispanic whites26. This relative ineligibility for CFTR modulator therapy may further contribute to the survival differences43.
There is a wealth of literature reporting higher morbidity and mortality in rural areas, termed "rural penalty mortality"44,45. The rural–urban divide for chronic lower respiratory diseases such as COPD and asthma has been reported, with worsening inequalities over time46. We believe that this is the first study exploring CF-related mortality by urbanization status. We found a higher CF-ASMR in the rural population; however, the rural–urban divide has narrowed in recent years due to improvements in rural population mortality. Potential reasons for these inequalities may include fewer specialized CF care centers in smaller counties, different smoking rates, and socioeconomic factors. Future studies focused on exploring the key factors responsible for these disparities are required to identify areas for intervention.
Cystic fibrosis patients frequently die young from the complications of the disease, while common geriatric diseases contribute less8,9,47. Recent advances in CF management have led to a step-change in survival. Studies on the impact of recent advances in CF therapies predict a survival age of 56 years and 66 years for individuals born in 2010 and 2021, respectively8,48. With the increasing survival, the number of adult CF patients is estimated to increase by 75% from 2015 through 202549. The rapidly changing epidemiology of CF presents new challenges, requiring different and more collaborative approaches to healthcare delivery to meet future needs adequately. The increased risk of gastrointestinal malignancies50 and complications associated with long-standing CF-related diabetes underscore the importance of surveillance and interventional care in the coming decades. This will necessitate more active partnerships with primary care physicians, as well as cardiovascular, endocrine, and gastrointestinal specialists51,52.
Our study is observational, and there are several inherent limitations to consider. Although the CDC WONDER database provides a comprehensive assessment of mortality statistics, it relies on information from death certificates, which may be prone to reporting errors or misclassification of the cause of death53,54. Centralized data sources have been reported to underestimate mortality55,56. Additionally, not all individuals with CF may have CF documented as a contributing cause of death on their death certificates. It is important to note that the mortality estimates analyzed by the CDC do not account for changes in the incidence or prevalence of the disease. To address this limitation, we reported the median age of death as an additional surrogate marker of survival to assess temporal trends. However, it is pertinent to note that the median age of death reflects the age distribution of the decedents in a given year and is not analogous to the survival age of the entire CF population. Finally, due to the observational nature of our study and the unavailability of CFTR modulator treatment data, the results cannot be used to establish causality.
Conclusions
The national CF-related mortality rates have declined, and the median age of death among CF decedents has significantly increased, indicating improved survival. These changes were relatively slow during an earlier period of the study, followed by a rapid decline more recently. We observed patterns of disparities based on sex, ethnicity, race, and geographical factors. These disparities were associated with a widening gap between ethnicities, a narrowing gap between races and rural vs. urban counties, and a complete closer of the gap between sexes over the study period.
Data availability
The datasets generated and/or analyzed during the current study are available in the CDC WONDER repository, [https://wonder.cdc.gov/controller/datarequest/D77].
Abbreviations
- AAPC:
-
Average annual percentage change
- ASMR:
-
Age-standardized mortality rate
- CDC WONDER:
-
Centre for Disease Control and Prevention Wide-ranging Online Data for Epidemiologic Research
- CF:
-
Cystic fibrosis
- CFFPR:
-
Cystic Fibrosis Foundation Patient Registry
- CFTR:
-
Cystic fibrosis transmembrane conductance regulator
- CI:
-
Confidence interval
- EAPC:
-
Estimated annual percentage change
- ICD-10:
-
International Classification of Diseases-10
- IQR:
-
Interquartile range
- MCOD:
-
Multiple cause of death
- NCHS:
-
National Center for Health Statistics
- PC:
-
Percentage change
- SARS-CoV2:
-
Severe acute respiratory syndrome coronavirus 2
- UCOD:
-
Underlying cause of death
- US:
-
United States
References
Guo, J., Garratt, A. & Hill, A. Worldwide rates of diagnosis and effective treatment for cystic fibrosis. J. Cyst. Fibros. 21(3), 456–462. https://doi.org/10.1016/j.jcf.2022.01.009 (2022).
Grody, W. W., Cutting, G. R. & Watson, M. S. The cystic fibrosis mutation “arms race”: When less is more. Genet. Med. 9(11), 739–744. https://doi.org/10.1097/gim.0b013e318159a331 (2007).
Grody, W. W. et al. Laboratory standards and guidelines for population-based cystic fibrosis carrier screening. Genet. Med. 3(2), 149–54. https://doi.org/10.1097/00125817-200103000-00010 (2001).
Veit, G. et al. From CFTR biology toward combinatorial pharmacotherapy: Expanded classification of cystic fibrosis mutations. Mol. Biol. Cell. 27(3), 424–433. https://doi.org/10.1091/mbc.E14-04-0935 (2016).
Strug, L. J., Stephenson, A. L., Panjwani, N. & Harris, A. Recent advances in developing therapeutics for cystic fibrosis. Hum. Mol. Genet. 27(R2), R173–R186. https://doi.org/10.1093/hmg/ddy188 (2018).
Shteinberg, M., Haq, I. J., Polineni, D. & Davies, J. C. Cystic fibrosis. Lancet 397(10290), 2195–2211. https://doi.org/10.1016/S0140-6736(20)32542-3 (2021).
Quintana-Gallego, E. et al. Mortality from cystic fibrosis in Europe: 1994–2010. Pediatr. Pulmonol. 51(2), 133–142. https://doi.org/10.1002/ppul.23337 (2016).
Cystic Fibrosis Foundation Cystic Fibrosis Foundation Patient Registry 2020 Annual Data Report. Cystic Fibrosis Foundation, 2020.
Cystic Fibrosis Canada. (2020). The Canadian Cystic Fibrosis Registry 2019 Annual Data Report. Cystic Fibrosis Canada.
Stephenson, A. L. et al. Survival comparison of patients with cystic fibrosis in Canada and the United States: A population-based cohort study. Ann. Intern. Med. 166(8), 537–546. https://doi.org/10.7326/M16-0858 (2017).
Knapp, E. A. et al. The cystic fibrosis foundation patient registry. Design and methods of a National Observational Disease Registry. Ann. Am. Thorac. Soc. 13(7), 1173–9. https://doi.org/10.1513/AnnalsATS.201511-781OC (2016).
Halliburton, C. S., Mannino, D. M. & Olney, R. S. Cystic fibrosis deaths in the United States from 1979 through 1991. An analysis using multiple-cause mortality data. Arch. Pediatr. Adolesc. Med. 150(11), 1181–5. https://doi.org/10.1001/archpedi.1996.02170360071012 (1996).
Joinpoint Regression Program. 4.9.0.0 ed: statistical methodology and applications branch, surveillance research program, National Cancer Institute; 2021.
Clegg, L. X., Hankey, B. F., Tiwari, R., Feuer, E. J. & Edwards, B. K. Estimating average annual per cent change in trend analysis. Stat. Med. 28(29), 3670–3682. https://doi.org/10.1002/sim.3733 (2009).
Savant, A. P. & McColley, S. A. Cystic fibrosis year in review 2019: Section 1. Pediatr. Pulmonol. 55(12), 3236–3242. https://doi.org/10.1002/ppul.25039 (2020).
Cystic Fibrosis Foundation Cystic Fibrosis Foundation Patient Registry 2021 Annual Data Report. Cystic Fibrosis Foundation, 2021
Corey, M. & Farewell, V. Determinants of mortality from cystic fibrosis in Canada, 1970–1989. Am. J. Epidemiol. 143(10), 1007–1017. https://doi.org/10.1093/oxfordjournals.aje.a008664 (1996).
Liou, T. G. et al. Predictive 5-year survivorship model of cystic fibrosis. Am. J. Epidemiol. 153(4), 345–352. https://doi.org/10.1093/aje/153.4.345 (2001).
Rosenfeld, M., Davis, R., FitzSimmons, S., Pepe, M. & Ramsey, B. Gender gap in cystic fibrosis mortality. Am. J. Epidemiol. 145(9), 794–803. https://doi.org/10.1093/oxfordjournals.aje.a009172 (1997).
Demko, C. A., Byard, P. J. & Davis, P. B. Gender differences in cystic fibrosis: Pseudomonas aeruginosa infection. J. Clin. Epidemiol. 48(8), 1041–1049. https://doi.org/10.1016/0895-4356(94)00230-n (1995).
Nick, J. A. et al. Effects of gender and age at diagnosis on disease progression in long-term survivors of cystic fibrosis. Am. J. Respir. Crit. Care Med. 182(5), 614–26. https://doi.org/10.1164/rccm.201001-0092OC (2010).
Assael, B. M. et al. Epidemiology and survival analysis of cystic fibrosis in an area of intense neonatal screening over 30 years. Am. J. Epidemiol. 156(5), 397–401. https://doi.org/10.1093/aje/kwf064 (2002).
Harness-Brumley, C. L., Elliott, A. C., Rosenbluth, D. B., Raghavan, D. & Jain, R. Gender differences in outcomes of patients with cystic fibrosis. J. Womens Health (Larchmt). 23(12), 1012–1020. https://doi.org/10.1089/jwh.2014.4985 (2014).
Rho, J. et al. Disparities in mortality of hispanic patients with cystic fibrosis in the United States. A National and Regional Cohort Study. Am. J. Respir. Crit. Care Med. 198(8), 1055–1063. https://doi.org/10.1164/rccm.201711-2357OC (2018).
O’Connor, G. T. et al. Case-mix adjustment for evaluation of mortality in cystic fibrosis. Pediatr. Pulmonol. 33(2), 99–105. https://doi.org/10.1002/ppul.10042 (2002).
Buu, M. C., Sanders, L. M., Mayo, J. A., Milla, C. E. & Wise, P. H. Assessing differences in mortality rates and risk factors between hispanic and non-hispanic patients with cystic fibrosis in California. Chest 149(2), 380–389. https://doi.org/10.1378/chest.14-2189 (2016).
Berk, M. L., Schur, C. L., Chavez, L. R. & Frankel, M. Health care use among undocumented Latino immigrants. Health Aff. (Millwood) 19(4), 51–64. https://doi.org/10.1377/hlthaff.19.4.51 (2000).
Fernandez, A. et al. Language barriers, physician-patient language concordance, and glycemic control among insured Latinos with diabetes: The Diabetes Study of Northern California (DISTANCE). J. Gen. Intern. Med. 26(2), 170–6. https://doi.org/10.1007/s11606-010-1507-6 (2011).
LeSon, S. & Gershwin, M. E. Risk factors for asthmatic patients requiring intubation. I. Observations in children. J. Asthma 32(4), 285–294. https://doi.org/10.3109/02770909509044836 (1995).
Homa, D. M. et al. Vital signs: Disparities in nonsmokers’ exposure to secondhand smoke–United States, 1999–2012. MMWR Morb. Mortal. Wkly. Rep. 64(4), 103–8 (2015).
Goss, C. H., Newsom, S. A., Schildcrout, J. S., Sheppard, L. & Kaufman, J. D. Effect of ambient air pollution on pulmonary exacerbations and lung function in cystic fibrosis. Am. J. Respir. Crit. Care Med. 169(7), 816–821. https://doi.org/10.1164/rccm.200306-779OC (2004).
Nishimura, K. K. et al. Early-life air pollution and asthma risk in minority children. The GALA II and SAGE II studies. Am. J. Respir. Crit. Care Med. 188(3), 309–18. https://doi.org/10.1164/rccm.201302-0264OC (2013).
McGarry, M. E., Neuhaus, J. M., Nielson, D. W. & Ly, N. P. Regional variations in longitudinal pulmonary function: A comparison of Hispanic and non-Hispanic subjects with cystic fibrosis in the United States. Pediatr. Pulmonol. 54(9), 1382–1390. https://doi.org/10.1002/ppul.24377 (2019).
Rosser, F. J., Forno, E., Cooper, P. J. & Celedón, J. C. Asthma in hispanics. An 8-year update. Am. J. Respir. Crit. Care Med. 189(11), 1316–27. https://doi.org/10.1164/rccm.201401-0186PP (2014).
Iriart, C., Boursaw, B., Rodrigues, G. P. & Handal, A. J. Obesity and malnutrition among Hispanic children in the United States: Double burden on health inequities. Rev. Panam. Salud Publica 34(4), 235–243 (2013).
Quon, B. S. et al. Disparities in access to lung transplantation for patients with cystic fibrosis by socioeconomic status. Am. J. Respir. Crit. Care Med. 186(10), 1008–13. https://doi.org/10.1164/rccm.201205-0949OC (2012).
Watts, K. D., Seshadri, R., Sullivan, C. & McColley, S. A. Increased prevalence of risk factors for morbidity and mortality in the US Hispanic CF population. Pediatr. Pulmonol. 44(6), 594–601. https://doi.org/10.1002/ppul.21037 (2009).
Kosorok, M. R. et al. Acceleration of lung disease in children with cystic fibrosis after Pseudomonas aeruginosa acquisition. Pediatr. Pulmonol. 32(4), 277–287. https://doi.org/10.1002/ppul.2009.abs (2001).
McGarry, M. E., Neuhaus, J. M., Nielson, D. W., Burchard, E. & Ly, N. P. Pulmonary function disparities exist and persist in Hispanic patients with cystic fibrosis: A longitudinal analysis. Pediatr. Pulmonol. 52(12), 1550–1557. https://doi.org/10.1002/ppul.23884 (2017).
McGarry, M. E. & McColley, S. A. Minorities are underrepresented in clinical trials of pharmaceutical agents for cystic fibrosis. Ann. Am. Thorac. Soc. 13(10), 1721–1725. https://doi.org/10.1513/AnnalsATS.201603-192BC (2016).
Januska, M. N., Marx, L., Walker, P. A., Berdella, M. N. & Langfelder-Schwind, E. The CFTR variant profile of Hispanic patients with cystic fibrosis: Impact on access to effective screening, diagnosis, and personalized medicine. J. Genet. Couns. 29(4), 607–615. https://doi.org/10.1002/jgc4.1271 (2020).
Fanous, H. K., Delgado-Villata, S., Kovacs, R., Shalaby-Rana, E. & Sami-Zakahri, I. Phenotypic characterization of the c.1679+1643G>T (1811+1643G>T) mutation in hispanic cystic fibrosis patients. Children (Basel) 5(7), 91. https://doi.org/10.3390/children5070091 (2018).
McGarry, M. E. & McColley, S. A. Cystic fibrosis patients of minority race and ethnicity less likely eligible for CFTR modulators based on CFTR genotype. Pediatr. Pulmonol. 56(6), 1496–1503. https://doi.org/10.1002/ppul.25285 (2021).
Miller, C. E. & Vasan, R. S. The southern rural health and mortality penalty: A review of regional health inequities in the United States. Soc. Sci. Med. 268, 113443. https://doi.org/10.1016/j.socscimed.2020.113443 (2021).
Singh, G. K. & Siahpush, M. Widening rural-urban disparities in life expectancy, U.S., 1969–2009. Am. J. Prev. Med. 46(2), e19-29. https://doi.org/10.1016/j.amepre.2013.10.017 (2014).
Reyfman, P. A. et al. Urban-rural mortality disparities from chronic lower respiratory diseases in the United States, 1999–2019. Am. J. Respir. Crit. Care Med. 203(11), 1435–1437. https://doi.org/10.1164/rccm.202011-4150LE (2021).
ECFSPR Annual Report 2020, Orenti A, Zolin A, Jung A, van Rens J et al, 2022.
MacKenzie, T. et al. Longevity of patients with cystic fibrosis in 2000 to 2010 and beyond: Survival analysis of the Cystic Fibrosis Foundation patient registry. Ann. Intern. Med. 161(4), 233–241. https://doi.org/10.7326/M13-0636 (2014).
Burgel, P. R. et al. ERS/ECFS task force on provision of care for adults with cystic fibrosis in Europe. Future trends in cystic fibrosis demography in 34 European countries. Eur. Respir. J. 46(1), 133–41. https://doi.org/10.1183/09031936.00196314 (2015).
Yamada, A. et al. Risk of gastrointestinal cancers in patients with cystic fibrosis: A systematic review and meta-analysis. Lancet Oncol. 19(6), 758–767. https://doi.org/10.1016/S1470-2045(18)30188-8 (2018).
Toledano, M. B. et al. The emerging burden of liver disease in cystic fibrosis patients: A UK nationwide study. PLoS One 14(4), e0212779. https://doi.org/10.1371/journal.pone.0212779 (2019).
Elborn, J. S. et al. Report of the European Respiratory Society/European Cystic Fibrosis Society task force on the care of adults with cystic fibrosis. Eur. Respir. J. 47(2), 420–428. https://doi.org/10.1183/13993003.00592-2015 (2016).
Lakkireddy, D. R., Gowda, M. S., Murray, C. W., Basarakodu, K. R. & Vacek, J. L. Death certificate completion: How well are physicians trained and are cardiovascular causes overstated?. Am. J. Med. 117(7), 492–498. https://doi.org/10.1016/j.amjmed.2004.04.018 (2004).
Smith Sehdev, A. E. & Hutchins, G. M. Problems with proper completion and accuracy of the cause-of-death statement. Arch. Intern. Med. 161(2), 277–284. https://doi.org/10.1001/archinte.161.2.277 (2001).
Navar, A. M. et al. Evaluation of mortality data from the social security administration death master file for clinical research. JAMA Cardiol. 4(4), 375–379. https://doi.org/10.1001/jamacardio.2019.0198 (2019).
Buchanich, J. M., Dolan, D. G., Marsh, G. M. & Madrigano, J. Underascertainment of deaths using social security records: A recommended solution to a little-known problem. Am. J. Epidemiol. 162(2), 193–194. https://doi.org/10.1093/aje/kwi178 (2005).
Schrijver, I. et al. Diagnostic testing by CFTR gene mutation analysis in a large group of Hispanics: novel mutations and assessment of a population-specific mutation spectrum. J. Mol, Diagn. 7(2), 289–299. https://doi.org/10.1016/S1525-1578(10)60557-0 (2005).
Author information
Authors and Affiliations
Contributions
Conception and design: Drs. H.S., C.J., D.C.M., and J.D.S. made significant contributions to the concept and design of this study. Administrative support: Drs. H.S., C.J., D.C.M., R.F., and J.D.S. provided administrative support. Provision of study materials: Drs. H.S., C.J., D.C.M., and J.D.S. Collection and assembly of data: Drs. H.S., C.J., P.B., D.C.M., and J.D.S. Data analysis and interpretation: Drs. H.S., C.J., D.C.M., P.B., R.F., R.N., J.S., D.C.M., and J.D.S. Manuscript writing: All authors (Drs. H.S., C.J., D.C.M., R.F., P.B., S.P., J.S., J.S.K., R.N., A.Z.U., and J.D.S.). Final approval of manuscript: (Drs. H.S., C.J., D.C.M., R.F., P.B., S.P., J.S., J.S.K., R.N., A.Z.U., and J.D.S.). All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Singh, H., Jani, C., Marshall, D.C. et al. Cystic fibrosis-related mortality in the United States from 1999 to 2020: an observational analysis of time trends and disparities. Sci Rep 13, 15030 (2023). https://doi.org/10.1038/s41598-023-41868-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-41868-x
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.