Abstract
Early-onset sepsis (EOS) is one of the leading causes of neonatal death and morbidity worldwide and timely initiation of antibiotic therapy is, therefore, of paramount importance. This study aimed to evaluate the predictive effect of lactate and base excess (BE) values in the cord arterial blood gas and the 6th hour of life venous blood gas analysis on clinical sepsis in newborns. This is a cohort case–control study. In this study, 104 cases were divided into clinical and suspected sepsis groups according to the evaluation at the 24th hour after delivery. Lactate and BE values were evaluated in the cord arterial blood gas analysis (ABGA) and at the postnatal 6th-hour venous blood gas. The cord ABGA and postnatal 6th-hour results were compared in the clinical and suspected sepsis groups. Clinical sepsis was found to be associated with a lactate value above 2 mMol/L at postnatal 6th-hour venous blood gas (p = 0.041). This association was the highest when the clinical sepsis group's postnatal 6th-hour lactate cut-off value was determined as 3.38 mMol/L (sensitivity 57.9% and specificity 68.5%) (p = 0.032). However, no association was found between clinical sepsis diagnosis and venous BE's value in cord ABGA at the postnatal 6th hour. We found that a venous lactate value above 3.38 mMol/L at the postnatal 6th hour was the cut-off value that could indicate early-onset clinical sepsis. However, none of the biomarkers used in diagnosing EOS can accurately show all cases.
Similar content being viewed by others
Introduction
Neonatal sepsis is the leading cause of global death in children under 5 years1. Proven neonatal early-onset sepsis (EOS) has mortality rates of up to 30% in high-income and 60% in low-income countries2. EOS is defined as the onset of sepsis within 72 h after birth. EOS risk factors were reported as group B streptococcal colonization, chorioamnionitis, prelabor rupture of membranes (at least 18 h before the onset of delivery), maternal fever, and gestational week3,4.
Rapid diagnosis and treatment of neonatal EOS are crucial to preventing morbidity and mortality. Clinical findings are often vague and non-specific, and commonly used biomarkers have low predictive values for early sepsis5. Although many biomarkers have been tried to predict sepsis, no definitive diagnostic marker has yet been found6. Another method to increase the accuracy of EOS diagnosis is the European Medicines Agency (EMA) scoring, which has been developed using a combination of clinical and laboratory findings and is frequently applied in neonatal units. This scoring system criteria include lactate and BE values7. pH indicates metabolic acidosis and results from the balance between lactate, which tends to lower pH and BE, which stabilizes it8. Mild metabolic acidosis detected in cord blood gas of term newborns is associated with morbidity9. In addition, recent studies show a relationship between lactate level and neonatal sepsis and morbidity10,11.
This study aimed to evaluate the predictive effect of lactate and BE analysis in cord artery blood gas (ABGA) and 6th-hour venous blood gas on clinical sepsis and suspected sepsis.
Materials and methods
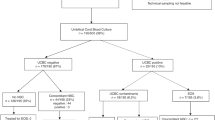
The study was conducted Cerrahpasa Faculty of Medicine, Istanbul University and Kanuni Sultan Suleiman Training and Research Hospital in Istanbul between January 1, 2019, and December 31, 2020, with newborn infants who were admitted to the neonatal unit in the first six hours after birth with suspicion of early-onset sepsis. The data from 104 cases that met the inclusion criteria were analysed in Fig. 1. Newborns with fetal distress in antenatal follow-up, with cord ABGA values below 7.0, a 5-min Apgar score below 5, cases for whom informed consent could not be obtained from the parents, and those with cardiac or metabolic disease were excluded from the study.
Ethics committee approval was obtained from Ethics Committee of the Cerrahpasa Faculty of Medicine, Istanbul University-Cerrahpasa (date: 05/12/21, approval no; 186107). Written informed consent from the parents was obtained for all participants.
Blood culture evaluation was performed in all cases before starting antibiotic treatment; following the guidelines of the Turkish neonatology society, lumbar puncture and chest X-ray were performed in necessary cases, and blood samples were taken into heparinized injectors and measured by ABL Flex (Radiometer) within 20 min12. Lactate and base excess (BE) in the cord ABGA and at the postnatal 6th-hour venous blood gas were registered. In addition, C-reactive protein (CRP) and procalcitonin (PCT) values of all cases were evaluated at the postnatal 24th hour. An algorithm was not determined for initiating antibiotics for the cases with EOS, and the decision to start antibiotic treatment in all cases was left to the consultant specialist neonatologist.
The threshold values of 15 mg/L for CRP, 2 ng/ml for PCT, − 10 mEq/L for blood gas BE were accepted according to EMA sepsis scoring, previous studies, and the limit values used by our hospital4,7. Threshold values of 2 and 4 mMol/L for lactate measures were determined relying on previous studies8,9,10.
The diagnosis of clinical sepsis was defined as having any relevant clinical findings and positive results for either CRP (> 15 mg/L) or PCT (2 ng/ml) at the postnatal 24th hour. Suspected sepsis was defined as whether there is a clinical symptom or not the presence of sepsis risk factors (chorioamnionitis, premature rupture of membranes above 18 h or above, maternal fever) in the newborns or clinical findings suggesting sepsis in follow-up without positive CRP or PCT results at the postnatal 24th hour's blood measures11,12. Laboratory findings are considered positive when CRP is above 15 mg/dl and/or PCT above 2 ng/ml. Clinical findings are irregular body temperature (> 38.5 °C or < 36 °C), cardiovascular instability (bradycardia or tachycardia and/or rhythm irregularity, oliguria (< 1 ml/kg/hour), hypotension, peripheral perfusion), skin and subcutaneous lesions (petechiae, sclerema), respiratory instability (apnea or tachypnea or increased oxygen demand or increased need for ventilation support), gastrointestinal (nutritional intolerance, insufficient breastfeeding, abdominal distention), and non-specific signs (irritability, lethargy, hypotonia) based on EMA score7.
Based on the postnatal 24th-hour evaluation, newborns were divided into clinical and suspected sepsis groups. First, the lactate and BE values in cord ABGA of the clinical sepsis group were compared to that of the suspected sepsis group. Then, the groups were compared regarding their lactate and BE values at the postnatal 6th hour shown in Fig. 1.
Fetal distress was accepted as abnormal cardiotocography detection in the regular antenatal follow-up of specialist obstetricians. Prelobor rupture of membranes (PROM) is defined as the rupture of membranes at least 18 h before the onset of labor13. Fever ≥ 39.0 °C once or 38.0–38.9 °C in two or more measurements 30 min apart with no other apparent source and one or more of the following: Except for accelerations, decelerations, and periods of significant variability, baseline fetal heart rate > 160 beats/min for ≥ 10 min, maternal white blood cell count > 15,000/mm3, in the absence of corticosteroids and ideally shifting to the left, purulent-appearing fluid from the cervical os seen by speculum examination14. Chorioamnionitis is defined as an acute inflammation of the membranes and chorion of the placenta, typically due to ascending polymicrobial bacterial infection in the setting of membrane rupture. Fetal growth restriction (FGR) is defined as the failure of the fetus to achieve its genetically determined growth potential15.
All statistical analyses were performed with R software (version 4.0.5) (R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, Available online: http/www.r-project.org/). Categorical variables were given as frequencies (percentage), and comparisons between groups were made by Pearson's Chi-squared test and Fisher's exact test. Continuous variables were analyzed with the Kolmogorov–Smirnov test, Shapiro–Wilk test, Q-Q plots, and histograms to check the normality assumption. Normally distributed variables were expressed as means ± standard deviations and compared with independent samples t-test. Non-normally distributed variables were shown as median (25–75th percentiles) and compared with the Mann–Whitney U test. pROC package was used to plot ROC curves (Receiver Operating Characteristic) and calculate sensitivity/specificity16. Optimal cut-off values were estimated according to Youden Index. p < 0.05 was considered significant.
Ethics approval and consent to participate
An informed consent document with information about the study and in accordance with the requirements established by the “Ethics Committee of the Cerrahpasa Faculty of Medicine, Istanbul University-Cerrahpasa” was offered to the parents. Informed consent was obtained from parents. Ethics approval was obtained from the Ethics Committee of the Cerrahpasa Faculty of Medicine, Istanbul University-Cerrahpasa (date: 05/12/21, approval no; 186107) conducted in accordance with the Declaration of Helsinki.
Consent to publish
Written informed consent was obtained from the parents.
Results
All patients' mean gestational age, birth weight, and 5-min Apgar score were 34.1 ± 3.8 weeks, 2.368 ± 896 g, and 7.65 ± 1.30, respectively. Other patient characteristics are presented in Table 1. Bacterial growth was detected in 3 blood cultures, one in the suspected sepsis group (coagulase-negative staphylococcus) and two in the clinical sepsis group (Escherichia coli).
The characteristics of the patients classified as clinical sepsis and suspected sepsis at the 24th hour are summarized in Table 2. No difference was observed between the two groups regarding gestational age, birth weight, type of delivery, gender, FGR, chorioamnionitis, urinary tract infection, PROM (18 h or longer before birth), and maternal antibiotic treatment. The 5-min Apgar score was lower in the clinical sepsis group (7 vs. 8, p = 0.0352).
When the groups with the diagnosis of clinical sepsis and suspected sepsis were compared, clinical sepsis was more common in the cases with a lactate value above 2 mMol/L at the postnatal 6th-hour evaluation (p = 0.041). No association was found between the cord ABGA lactate, BE value, and the value of venous BE at the postnatal 6th hour and clinical sepsis in Table 3. The optimal 6th-hour lactate cut-off value, which identifies the cases diagnosed with clinical sepsis, was determined to be 3.38 mMol/L (sensitivity 57%, specificity 68%, p = 0.032) shown in Table 4, Fig. 2.
The discrimination power between suspected and clinical early-onset neonatal sepsis in the postnatal 6th-hour CRP and PCT values was statistically insignificant (CRP; AUC = 0.567, p = 0.259 and PCT; AUC = 0.531, p = 0.599, respectively).
Discussion
This study compared the cord ABGA and postnatal 6th-hour venous lactate and BE values of clinical and suspected sepsis cases. It was found that lactate value over 3.38 mMol/l (sensitivity 57%, specificity 68%, p = 0.032) measured at the postnatal 6th hour had a discriminative power on early-onset clinical sepsis. Lactate values are closely associated with morbidity and mortality since it is essential for final tissue oxygenation17,18. Therefore, when interpreting the acuity of lactate elevations in patients, it is crucial to determine the underlying cause of lacticemia and the associated prognosis. It is essential to exclude secondary causes of lacticemia (such as hypoxic-ischemic encephalopathy, necrotizing enterocolitis, and cyanotic congenital heart disease) in newborns with high lactate values19. Clinicians aim to detect and treat sepsis as early as possible, but no single or combined biomarker can provide this with 100 percent sensitivity and specificity20. Although a specificity higher than 85% is ideal for biomarkers, some authors find a specificity higher than 50% acceptable21,22,23. In our study, a lactate value above 3.38 mMol/L measured at the postnatal 6th hour was 68% specific for diagnosing clinical sepsis.
Rapid diagnosis and treatment of neonatal EOS are crucial to preventing morbidity and mortality. Clinical findings are often vague and non-specific, and commonly used biomarkers have low predictive values for early sepsis5. Although CRP is widely used to indicate bacterial sepsis in neonates and children, it may have several disadvantages, like a late response. CRP can generally be detected 12 h after the onset of clinical symptoms and reaches a plateau after 20–72 h6. It has been shown that the CRP level is high in cases diagnosed wit hypoxic ischemic encephalopathy and undergoing hypothermia treatment24. However, cases with suspected hypoxia were not included in our study, and the umbilical cord blood gas pH value and 5-min Apgar score of our cases show that there is no diagnosis neonatal asphyxia. Another frequently used parameter is PCT, which may increase earlier than CRP and reaches a peak in 6–8 h, remaining high for 24 h6,25. The measurement of lactate in blood gas results is faster, easier, and cheaper than these two frequently used parameters. In this study, lactate measurements at the 6th hour of life might be used to differentiate clinical sepsis from suspected sepsis.
Cord ABGA lactate was shown not to affect the differentiation of clinical sepsis and suspected sepsis in the diagnosis of EOS. Early-onset sepsis may be secondary pathogens from the mother's gastrointestinal or urinary flora by ascending the uterine compartment and infecting the amniotic fluid. Less commonly, it is caused by vertical transmission of bacteria from the mother's lower genital tract during vaginal delivery or by hematogenous spread through the placenta26. Sepsis is a systemic inflammatory response syndrome that develops secondary to an infection and is accompanied by circulatory dysfunction27. The following features of the placenta are well defined; It provides oxygen and nutrients to the fetus while removing carbon dioxide and other waste products, metabolizes several substances, and releases metabolic products into the maternal and/or fetal circulation, helping to protect the fetus against specific xenobiotic molecules, infections, and maternal diseases28. In addition, the protective effect of the placenta against cytokine release has been demonstrated in Covid 19 positive maternal placentas29. In our study, the absence of a correlation between lactate value in cord blood gas and clinical sepsis cases might be related to the protective effect of the placenta against the inflammatory process.
Cord ABGA and the postnatal 6th-hour BE measurements were shown not to affect the differentiation of clinical sepsis from suspected sepsis in the diagnosis of EOS. BE is primarily used to differentiate the metabolic or respiratory component of acidosis and affects the outcome mainly from the changes in blood bicarbonate level30. Base excess is not considered a sepsis biomarker by itself; it is mainly included in the EMA sepsis scoring system and is used to diagnose sepsis by demonstrating the presence of acidosis7. Similarly, our study showed that BE measurements do not differentiate between clinical and suspected sepsis.
The Apgar score quickly assesses the newborn's clinical status 1 and 5 min after birth. The Apgar score is obtained by evaluating five parameters (skin color, heart rate, reflexes, muscle tone, and respiratory activity). Apgar score was lower in the clinical sepsis group than in the suspected sepsis group in our study. In particular, a 5-min Apgar score below 7 indicates an increased risk of neonatal asphyxia31. Other objective indicators of antenatal hypoxia are cord blood gas pH, BE, and lactate levels19,32,33. In this study, no difference was observed between the two groups regarding cord blood gas lactate and BE values. This result is likely related to excluding cases with fetal distress from this study. It has been shown that a low Apgar score increases the risk of early-onset sepsis12,34. Similar to previous studies, a lower 5-min Apgar score was associated with clinical sepsis in our study.
The gold standard in diagnosing EOS is the presence of growth in culture from sterile fields (blood, urine, cerebrospinal fluid, pleura, peritoneum, and joint fluid)4,23,35. Our study observed a blood culture growth in two cases (1.9%) in the clinical sepsis group. Coagulase-negative staphylococci culture growth in the blood culture in the suspected sepsis group was considered more in favor of contamination. Initial clinical signs and biomarkers are not specific in EOS, which causes intensive use of antibiotics in suspected patients. The number of babies that need to be treated for 1 proven case of EOS in term and late preterm babies are reported in the literature to be between 40 and 10036. Studies have reported blood culture growth rates ranging from 0.1 to 40% in EOS37. This range is usually ascribed to an insufficient blood sample, maternal antibiotic use, and low value of bacteremia6,20. In a recent prospective multicenter study, a CRP value above 16 mg/L at 36 h and a procalcitonin value above 2.8 ng/L could distinguish it from culture-positive sepsis at a high sensitivity rate38. In addition, over time, EOS, which is caused by group B streptococci, decreased with the increase in the use of antenatal antibiotics, and gram-negative enteric bacteria (mainly Escherichia coli) became the leading factor in preterm infants39,40. Similarly, the number of proven sepsis was found to be relatively low in our study, and Escherichia coli was shown in the blood cultures of only two cases diagnosed with clinical sepsis. Therefore, using the threshold values of 15 mg/L for CRP and 2 ng/L for procalcitonin value is acceptable to differentiate clinical and suspected sepsis at the postnatal 24th hour in our study.
The main limitation of this study is the low number of culture-positive sepsis cases. The study's strength is the number of cases, the number of markers evaluated, and the fact that it was conducted with a homogeneous patient group with close laboratory follow-up.
In conclusion, the diagnosis of EOS and the duration of treatment remain controversial for non-culture-positive cases. Clinicians can use lactate levels as an easily measured laboratory test in the neonatal intensive care unit. Our study showed that the patients with high lactate value in the postnatal 6th-hour venous lactate value were associated with an early-onset clinical sepsis diagnosis. Therefore, clinicians should closely monitor cases with high lactate levels in the postnatal 6th hour for early diagnosis of neonatal sepsis.
Data availability
The datasets generated and/or analysed during the current study are not publicly available due to our hospital policy but are available from the corresponding author on reasonable request.
References
GBD 2015 Child Mortality Collaborators. Global, regional, national, and selected subnational levels of stillbirths, neonatal, infant, and under-5 mortality, 1980–2015: A systematic analysis for the Global Burden of Disease Study. Lancet 388(10053), 1725–1774 (2016).
Thaver, D. & Zaidi, A. K. Burden of neonatal infections in developing countries: A review of evidence from community-based studies. Pediatr. Infect. Dis. J. 28(1 Suppl), S3–S9 (2009).
Shane, A. L. & Stoll, B. J. Neonatal sepsis: Progress towards improved outcomes. J. Infect. 68(Suppl 1), S24–S32 (2014).
Flannery, D. D. & Puopolo, K. M. Neonatal early-onset sepsis. NeoReviews 23(11), 756–770 (2022).
Weiss, S. L. et al. Delayed antimicrobial therapy increases mortality and organ dysfunction duration in pediatric sepsis. Crit. Care Med. 42(11), 2409–2417 (2014).
Hedegaard, S. S., Wisborg, K. & Hvas, A. M. Diagnostic utility of biomarkers for neonatal sepsis–A systematic review. Infect. Dis. (Lond.) 47(3), 117–124 (2015).
Tuzun, F. et al. Is European Medicines Agency (EMA) sepsis criteria accurate for neonatal sepsis diagnosis or do we need new criteria?. PLoS One 14(6), e0218002 (2019).
Sood, P., Paul, G. & Puri, S. Interpretation of arterial blood gas. Indian J. Crit. Care Med. 14(2), 57–64 (2010).
Bailey, E. J. et al. Mild neonatal acidemia is associated with neonatal morbidity at term. Am. J. Perinatol. 38(S01), e155–e161 (2021).
Sun, Y. S. & Yu, J. L. Clinical value of blood lactate in predicting the prognosis of neonatal sepsis. Zhongguo Dang Dai Er Ke Za Zhi 21(7), 629–634 (2019).
Jia, Y., Wang, Y. & Yu, X. Relationship between blood lactic acid, blood procalcitonin, C-reactive protein and neonatal sepsis and corresponding prognostic significance in sick children. Exp. Ther. Med. 14(3), 2189–2193 (2017).
Satar, M., Arısoy, A. E. & Çelik, İH. Turkish Neonatal Society guideline on neonatal infections-diagnosis and treatment. Turk. Arch. Pediatr. 53(Suppl 1), S88–S100. https://doi.org/10.5152/TurkPediatriArs.2018.01809 (2018).
Polin, R. A., Committee on Fetus and Newborn. Management of neonates with suspected or proven early-onset bacterial sepsis. Pediatrics 129(5), 1006–1015 (2012).
Tita, A. T. & Andrews, W. W. Diagnosis and management of clinical chorioamnionitis. Clin. Perinatol. 37(2), 339–354 (2010).
Burton, G. J. & Jauniaux, E. Pathophysiology of placental-derived fetal growth restriction. Am. J. Obstet. Gynecol. 218(2S), S745–S761 (2018).
Robin, X. et al. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 12, 77 (2011).
Bou Chebl, R. et al. Serum lactate is an independent predictor of hospital mortality in critically ill patients in the emergency department: A retrospective study. Scand. J. Trauma Resusc. Emerg. Med. 25(1), 69 (2017).
Filho, R. R. et al. Blood lactate levels cutoff and mortality prediction in sepsis-time for a reappraisal? A retrospective cohort study. Shock 46(5), 480–485 (2016).
Ganetzky, R. D. & Cuddapah, S. R. Neonatal lactic acidosis: A diagnostic and therapeutic approach. NeoReviews 18(4), e217–e227 (2017).
Deleon, C., Shattuck, K. & Jain, S. K. Biomarkers of neonatal sepsis. NeoReviews 16(5), e297–e308 (2015).
Mehr, S. & Doyle, L. W. Cytokines as markers of bacterial sepsis in newborn infants: A review. Pediatr. Infect. Dis. J. 19(9), 879–887 (2000).
Ng, P. C. Diagnostic markers of infection in neonates. Arch. Dis. Child. Fetal Neonatal. Ed. 89(3), F229–F235 (2004).
Ng, P. C. & Lam, H. S. Diagnostic markers for neonatal sepsis. Curr. Opin. Pediatr. 18(2), 125–131 (2006).
Ruhfus, M. et al. Association of routinely measured proinflammatory biomarkers with abnormal MRI findings in asphyxiated neonates undergoing therapeutic hypothermia. Front. Pediatr. 9, 624652. https://doi.org/10.3389/fped.2021.624652 (2021).
Mukhopadhyay, S. & Puopolo, K. M. Risk assessment in neonatal early onset sepsis. Semin. Perinatol. 36(6), 408–415 (2012).
Puopolo, K. M., Benitz, W. E., Zaoutis, T. E., Committee on Fetus and Newborn; Committee on Infectious Diseases. Management of neonates born at ≥35 0/7 weeks’ gestation with suspected or proven early-onset bacterial sepsis. Pediatrics 142(6), e20182894 (2018).
Yuki, K. & Murakami, N. Sepsis pathophysiology and anesthetic consideration. Cardiovasc. Hematol. Disord. Drug Targets 15(1), 57–69 (2015).
Gude, N. M., Roberts, C. T., Kalionis, B. & King, R. G. Growth and function of the normal human placenta. Thromb. Res. 114(5–6), 397–407 (2004).
Rad, H. S. et al. The effects of COVID-19 on the placenta during pregnancy. Front. Immunol. 12, 743022 (2021).
Fischbach, F. T. & Dunning, M. B. A manual of laboratory and diagnostic tests (Lippincott Williams & Wilkins, 2009).
American Academy of Pediatrics Committee on Fetus and Newborn, American College of Obstetricians and Gynecologists Committee on Obstetric Practice. The apgar score. Pediatrics 136(4), 819–822 (2015).
Lie, K. K., Grøholt, E. K. & Eskild, A. Association of cerebral palsy with Apgar score in low and normal birthweight infants: Population based cohort study. BMJ 341, c4990 (2010).
Ahmadpour-Kacho, M. et al. Short term outcome of neonates born with abnormal umbilical cord arterial blood gases. Iran. J. Pediatr. 25(3), e174 (2015).
Polcwiartek, L. B. et al. Early-onset sepsis in term infants admitted to neonatal intensive care units (2011–2016). J. Perinatol. 41(1), 157–163 (2021).
Bethou, A. & Bhat, B. V. Neonatal Sepsis-Newer insights. Indian J. Pediatr. 89(3), 267–273 (2022).
van Herk, W., Stocker, M. & van Rossum, A. M. Recognising early onset neonatal sepsis: An essential step in appropriate antimicrobial use. J. Infect. 72(Suppl), S77–S82 (2016).
Lukacs, S. L. & Schrag, S. J. Clinical sepsis in neonates and young infants, United States, 1988–2006. J. Pediatr. 160(6), 960–5.e1 (2012).
Stocker, M. et al. Procalcitonin-guided decision making for duration of antibiotic therapy in neonates with suspected early-onset sepsis: A multicentre, randomised controlled trial (NeoPIns). Lancet 390(10097), 871–881 (2017).
Cantey, J. B. & Lee, J. H. Biomarkers for the diagnosis of neonatal sepsis. Clin. Perinatol. 48(2), 215–227 (2021).
Vatne, A., Klingenberg, C., Rettedal, S. & Øymar, K. Early-onset sepsis in neonates—A population-based study in South-West Norway From 1996 to 2018. Front. Pediatr. 9, 634798 (2021).
Author information
Authors and Affiliations
Contributions
Conception—A.Y., M.V.; Design—A.Y., M.V.; Supervision—Y.P., M.V.; Materials—A.Y., N.K., I.G., M.V.; Data Collection and/or Processing—A.Y., N.K., I.G., M.V; Analysis and/or Interpretation—A.Y., A.U; Literature Review—A.Y.; Writing—A.Y., A.U.,M.V.; Critical Review—A.Y.,Y.P., M.V.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yilmaz, A., Kaya, N., Gonen, I. et al. Evaluating of neonatal early onset sepsis through lactate and base excess monitoring. Sci Rep 13, 14837 (2023). https://doi.org/10.1038/s41598-023-41776-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-41776-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.