Abstract
Adolescents are at increased risk to develop substance use disorders and suffer from relapse throughout life. Targeted weakening of drug-associated memories has been shown to reduce relapse-like behavior in adult rats, however this process has been understudied in adolescents. We aimed to examine whether adolescent-formed, cocaine-associated memories could be manipulated via reconsolidation mechanisms. To accomplish this objective, we used an abbreviated operant cocaine self-administration paradigm (ABRV Coc-SA). Adult and adolescent rats received jugular catheterization surgery followed by ABRV Coc-SA in a distinct context for 2 h, 2×/day over 5 days. Extinction training (EXT) occurred in a second context for 2 h, 2×/day over 4 days. To retrieve cocaine-context memories, rats were exposed to the cocaine-paired context for 15 min, followed by subcutaneous injection of vehicle or the protein synthesis inhibitor cycloheximide (2.5 mg/kg). Two additional EXT sessions were conducted before a 2 h reinstatement test in the cocaine-paired context to assess cocaine-seeking behavior. We find that both adult and adolescent cocaine-exposed rats show similar levels of cocaine-seeking behavior regardless of post-reactivation treatment. Our results suggest that systemic treatment with the protein synthesis inhibitor cycloheximide does not impair reconsolidation of cocaine-context memories and subsequent relapse during adulthood or adolescence.
Similar content being viewed by others
Introduction
Substance-use disorders (SUDs) are a chronic disease characterized by cycles of repeated use, abstinence, and relapse. Patients that suffer from SUDs often report starting drug use during adolescence1,2, with recent reports showing that the severity of SUD diagnosis during adolescence increases the risk for problematic drug use in adulthood3,4. Cocaine use disorders affect up to 1.5 million people in the US, with reports of increased cocaine use in adolescents and young adults from 2005 to 2018, highlighting the importance of studying relapse during this developmental period5,6.
The cues and contextual environments linked with drug use are major triggers for relapse7,8,9. Exposure to cocaine-associated contexts can elicit craving and relapse in part through the retrieval of cocaine-associated memories10,11,12. Normally, when memories are retrieved, they enter a temporary labile state and can be reconsolidated back into long-term storage13,14,15. Each drug-taking or relapse episode can serve to strengthen cocaine-associated memories. Targeted weakening of maladaptive drug-related memories, via reconsolidation mechanisms, has been proposed as a potential therapeutic method to reduce the risk to relapse16,17,18. The process of reconsolidation is dependent on new protein synthesis, with reduced cocaine conditioned place preference (CPP), cue- and context-elicited cocaine seeking observed in adult rats when protein synthesis is inhibited during the labile window10,19,20,21.
There is evidence that adolescent fear-associated and social CPP memories are sensitive to reconsolidation-based manipulations22,23,24,25. However, much less is known about the potential lability of drug-associated memories formed during adolescence, despite the critical role of this developmental period in initiation of drug use18. We therefore examined whether protein synthesis inhibition would impair the reconsolidation of adult or adolescent-formed cocaine-context-memories and subsequent relapse-like behavior. We used a published, abbreviated cocaine self-administration model (ABRV Coc-SA) that allows for self-administration training and reinstatement testing in rats to occur during the adolescent window, between postnatal days 35–6326,27. We modified the ABRV Coc-SA paradigm by incorporating a memory reactivation session to elicit retrieval of cocaine-associated contextual memories in adult and adolescent rats, followed by systemic protein synthesis inhibition before a test of cocaine-seeking behavior.
Materials and methods
Animals
For all experiments, male Sprague Dawley rats (Envigo Inc., Haslett, MI) arrived on postnatal day 25 (P25, average of 75 g, n = 14), or P56 (average of 257 g, n = 16). All rats were pair housed upon arrival with ad libitum access to food and water. The vivarium housing was humidity and temperature controlled under reversed light conditions (8 am off, 8 pm on) with all behavioral experiments conducted during the rats’ active cycle. Rats were handled 6–7 days with jugular catheterization surgeries performed on P33 for adolescent or P66 for adult rats.
Previous reports did not find behavioral differences associated with single- vs pair-housed conditions using ABRV Coc-SA, therefore after a surgery and recovery period (2 days before behavioral start) all rats were single housed with adolescents maintained on 15 g and adults on 18 g of chow, which allowed for weight gain throughout the experiment27,28. All protocols were approved by the Institutional Animal Care and Use Committee (IACUC) at Michigan State University (MSU) and followed the National Research Council’s Guide for the Care and Use of Laboratory Rats.
Surgery
Intravenous (IV) catheters were constructed in house, with a 10 cm length for adults and 9.7 cm length for adolescents. A silicone ball was placed 3.25 or 3.0 cm from the inserted end of the catheter for adults or adolescents27,28,29. On the surgery day, rats were fully anesthetized via intraperitoneal (IP) injection of ketamine + xylazine (80–100 mg/kg + 5–10 mg/kg, respectively; Covetrus, Cats:11695-6840, 33198). Catheters were implanted into the right jugular vein, exiting subcutaneously at the back below the shoulder blade27,30,31. Rats were administered oral meloxidyl 2 days pre- and post surgery (Covetrus, Cat:50401), topical lidocaine and gentamycin applied to suture areas, and catheters flushed with 0.1 mL of cefazolin (0.1 mg/mL, Covetrus, Cat:54846) dissolved in heparinized saline (Hep-Sal; 70 U/mL; Covetrus, Cat:49130), followed by 0.1 mL of 10 U/mL Hep-Sal. Catheters were flushed before Coc-SA with 70U Hep-Sal, and after with 10U Hep-Sal. Cefazolin was administered after the last Coc-SA session of the day. When catheter patency was uncertain, 0.1 mL propofol was administered IV and rats monitored for brief loss of movement (Covetrus, Cat:54899).
General Coc-SA training
As shown in Fig. 1, Coc-SA occurred in an operant conditioning chamber with distinct odor, tactile, auditory and visual stimuli, as previously described27. Operant chambers (29.5 × 24 × 28 cm; Med Associates Inc., St. Albans, NY) were configured with the following contextual stimuli: Context A contained a continuous red house light (0.4 fc brightness), intermittent pure tone (80 dB, 1 kHz; 2 s on, 2 s off), pine-scented air freshener (Car Freshener Corp., Watertown, NY), and wire mesh flooring (26 cm × 27 cm). Context B contained an intermittent white stimulus light over the inactive lever (1.2 fc brightness; 2 s on, 2 s off), continuous pure tone (75 dB, 2.5 kHz), vanilla-scented air freshener (Car Freshener Corp., Watertown, NY), and a slanted black acrylic panel bisecting the bar flooring (19 cm × 27 cm). Rats were counter-balanced to start Coc-SA in context A or B and importantly, background stimuli were not paired with cocaine infusion8,27,32.
Schematic and timeline of behavioral experiments to examine reconsolidation of cocaine-context memories in adult and adolescent cocaine-exposed rats. All experiments included abbreviated cocaine self-administration training (Coc-SA, 2 h sessions, 2×/day over 5 days, minimum of 10 sessions) in a distinct context, followed by extinction training (EXT, 2 h sessions, 2×/day over 4 days, minimum of 8 sessions). On average, adolescent rats started Coc-SA at P38 and adults at P70. The average age at the start of EXT was P45 for adolescent rats and P76 for adult rats, due to differences in the number of sessions needed to acquire Coc-SA criteria. To retrieve cocaine-associated contextual memories, rats were returned to the previous cocaine-paired context for memory reactivation (MR, 15 min session), followed by a subcutaneous injection of vehicle or 2.5 mg/kg cycloheximide (VEH, CHX) immediately after the MR session. Two additional sessions of EXT were given 1 day after MR (post-MR EXT), followed by a reinstatement test in the previous cocaine-paired context (Test, 2 h).
Coc-SA consisted of 2 h sessions, 2×/day over 5 days, for a minimum of 10 sessions (criteria = at least 10 infusions). Active lever responses resulted in an IV infusion of 0.05 mL of Cocaine-HCl (0.5 mg/kg per each infusion) on a Fixed-Ratio 1 (FR1) schedule of reinforcement, whereas inactive lever responses resulted in no infusions. Cocaine hydrochloride (Cocaine-HCl; NIDA Drug Supply System, Research Triangle Park, NC) was dissolved in sterile saline. Drug delivery was controlled by an infusion pump (Med Associates Inc; Model PHM-107) and each infusion lasted for 2 s with a 20 s time-out period. Weight was recorded daily and cocaine concentration in syringes adjusted for every 50 g increase in weight27,33. As shown in Fig. 1, Coc-SA began on P38 (adolescent) or P70 (adult), and importantly at a post-pubertal period for adolescents26. Rats were returned to their home-cage environments for a break period of 2 h between training sessions.
Extinction training, memory reactivation and reinstatement tests
As shown in Fig. 1, extinction training (EXT) consisted of 2 h sessions, 2×/day over 4 days, for a minimum of 8 sessions (criteria = less than 25 responses on last 2 sessions). During EXT training, responses on the active and inactive lever had no programmed consequences. Rats received a 15 min memory reactivation session (MR) in the cocaine-paired context. The MR session functions to elicit brief memory reactivation, without extensive extinction11,34. Immediately after MR, rats were given a subcutaneous (SC) injection of vehicle (VEH) or cycloheximide (CHX, 2.5 mg/kg) and returned to their home cage. We used a 2.5 mg/kg dose based on previous studies which showed that 2.2 mg/kg given immediately after retrieval of a cocaine-cue memories diminished subsequent relapse21. To assess whether CHX affected responding in the EXT context, two additional 2 h EXT sessions were administered (post-MR EXT). After the final post-MR EXT, rats were returned to the original cocaine-paired context for a 2 h reinstatement test (Test). During the Test, both active and inactive lever presses resulted in no programmed consequences; therefore, active lever responses served as an index of drug-seeking behavior. On average, MR and Tests occurred on P49 and P51 for adolescent rats and P80 and P82 for adult rats.
Statistical analysis
Separate analyses of variance (ANOVAs) or independent t-tests were conducted to examine for pre-existing differences between VEH and CHX groups for: lever responses during Coc-SA (Mean ± SEM, final 3 sessions), last EXT session, 15 min MR session, final post-MR EXT session, and number of sessions to reach acquisition criteria for Coc-SA and EXT.
To assess behavioral differences by age, data from adult and adolescent VEH and CHX groups pre-treatment were collapsed. Interaction effects for within subject factors. Coc-SA, EXT, MR and the between subject factor of Age were assessed (Fig. 2). For all behavioral phases (Coc-SA, EXT, MR, final post-MR EXT and Test), no Age × Context × Treatment interaction effects were found and therefore subsequent ANOVAs were analyzed separately for adult or adolescent groups.
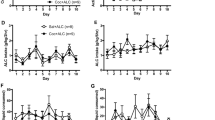
Effect of age on behavioral phases. Mean ± SEM of active and inactive lever responses for adult and adolescent rats during (A) cocaine self-administration (Coc-SA, 2 h sessions, 2×/day over 5 days), (B) extinction (EXT, 2 h sessions, 2×/day over 4 days), (C) memory reactivation (MR, 15 min session) in the previous cocaine-paired context to retrieve cocaine-associated memories. Rats received subcutaneous injection of vehicle or 2.5 mg/kg cycloheximide (VEH, CHX) immediately after the MR session. (D) Two additional sessions of EXT were given 1 day after MR (post-MR EXT), followed by a reinstatement test in the previous cocaine-paired context (Test, 2 h). (E) Active and inactive lever responses during 20 min Bins of the 2 h Test. Symbols indicate significant within-subject differences revealed by Tukey’s test (B) #p < 0.01: EXT session 1 > 8, (D) final post-MR EXT session < Test, (E) 20 min Test Bin 1 > 6. Groups denoted by: Blue = adult (n = 16), Orange = adolescent (n = 14), Light blue and Light orange individual points = VEH, Black individual points = CHX.
To assess behavioral differences for adults (Fig. 3) or adolescents (Fig. 4), interaction effects for within-subjects factors: Coc-SA, EXT, MR, Context (final post-MR EXT session vs Test), 20 min Test Bins, 5 min MR or 5 min Test Bins and between subjects factor of Treatment (VEH vs CHX) were conducted. Significant effects, when appropriate, were followed by analysis with Tukey’s HSD post-hoc tests, with alpha set at 0.05. Rats that did not acquire Coc-SA or did not pass catheter patency tests were excluded from analysis.
Effect of cycloheximide on reconsolidation of cocaine-context memories in adult cocaine-exposed rats. Mean ± SEM of active and inactive lever responses for adult rats during (A) cocaine self-administration (Coc-SA, 2 h sessions, 2×/day over 5 days), (B) extinction (EXT, 2 h sessions, 2×/day over 4 days), (C) memory reactivation (MR, 15 min session) in the previous cocaine-paired context to retrieve cocaine-associated memories. Adult rats received subcutaneous injection of vehicle or 2.5 mg/kg cycloheximide (VEH, CHX) immediately after the MR session. (D) Two additional sessions of EXT were given 1 day after MR (post-MR EXT), followed by a reinstatement test in the previous cocaine-paired context (Test, 2 h). (E) Active and inactive lever responses during 20 min Bins of the 2 h Test. Symbols indicate significant within-subject differences revealed by Tukey’s test (B) #p < 0.01: EXT session 1 > 8, (D) final post-MR EXT session < Test, (E) 20 min Test Bin 1 > 6. Groups denoted by: Light blue = adult VEH (n = 8), Black = adult CHX (n = 8).
Effect of cycloheximide on reconsolidation of cocaine-context memories in adolescent cocaine-exposed rats. Mean ± SEM of active and inactive lever responses for adolescent rats during (A) cocaine self-administration (Coc-SA, 2 h sessions, 2×/day over 5 days), (B) extinction (EXT, 2 h sessions, 2×/day over 4 days), (C) memory reactivation (MR, 15 min session) in the previous cocaine-paired context to retrieve cocaine-associated memories. Adolescent rats received subcutaneous injection of vehicle or 2.5 mg/kg cycloheximide (VEH, CHX) immediately after the MR session. (D) Two additional sessions of EXT were given 1 day after MR (post-MR EXT), followed by a reinstatement test in the previous cocaine-paired context (Test, 2 h). (E) Active and inactive lever responses during 20 min Bins of the 2 h Test. Symbols indicate significant within-subject differences revealed by Tukey’s test (B) #p < 0.01: EXT session 1 > 8, (D) #p < 0.01, active; #p < 0.05, inactive: final post-MR EXT session < Test (E) #p < 0.01, active; #p < 0.05, inactive: 20 min Test Bin 1 > 6. Groups denoted by: Light orange = adolescent VEH (n = 6), Black = adolescent CHX (n = 8).
ARRIVE guidelines
The current study is reported in accordance with ARRIVE guidelines.
Results
Adolescent and adult behavior during Coc-SA, EXT, MR, and tests
To examine potential behavioral differences between adult and adolescent rats, lever response data for all phases before VEH or CHX administration (Coc-SA, EXT, MR) were combined by age. Overall, there were no pre-existing differences between adult and adolescent rats in the number of sessions to acquire Coc-SA, lever responses, or cocaine infusions during the last three sessions of Coc-SA (Fig. 2A). The 2 × 10 ANOVAs during Coc-SA did not reveal significant Coc-SA session × Age or Coc-SA infusion × Age interaction effects.
During EXT, there were no pre-existing differences between adult and adolescent rats in the number of sessions needed to complete EXT training, or lever responses on the last EXT session (Fig. 2B). The 2 × 8 ANOVA of lever responses revealed a significant EXT session main effect (active: F7, 28 = 44.395, p < 0.001; inactive: F7, 28 = 28.558, p < 0.001), but no EXT session × Age interaction or Age main effects. Tukey’s post-hoc comparisons revealed that both adult and adolescent groups decreased lever responses by the final EXT session (active and inactive: EXT S1 > S8, #p < 0.01).
Adult and adolescent rats had similar lever responses during the 15 min MR session before systemic VEH or CHX administration (Fig. 2C). The 2 × 3 ANOVA of lever responses during 5 min Bins of the 15 min MR session revealed a significant MR Bin main effect (active: F2, 28 = 22.226, p < 0.001; inactive: F2, 28 = 3.646, p = 0.032) with no MR Bin × Age interaction or Age main effects. Tukey’s post-hoc comparisons revealed that both adult and adolescent groups displayed reduced responses on the active lever by the final 5 min of MR (active: Bin 1 > 3, #p < 0.05) (Suppl Fig. 2A). Tukey’s post-hoc comparisons did not reveal significant differences in inactive lever responses during 5 min MR Bins (Suppl Fig. 2A).
To examine for potential behavioral differences between adult and adolescent rats during the reinstatement Test after VEH or CHX administration, we compared lever response data for (1) VEH groups only or (2) VEH and CHX groups combined by age and observed similar results. Therefore we presented data for all groups. Adult and adolescent groups displayed increased cocaine-seeking behavior when exposed to the cocaine-paired context (Fig. 2D). The 2 × 2 ANOVAs of active lever responses during the final post-MR EXT vs Test session revealed a significant Context main (active: F1, 14 = 56.826, p < 0.001), but no Context × Treatment interaction or Treatment main effects. Tukey’s post-hoc comparisons revealed that lever responses in the VEH and CHX groups were higher during the Test compared to the final post-MR EXT session (active: #p < 0.01) therefore, both VEH and CHX-treated groups exhibited cocaine-seeking behavior. The 2 × 2 ANOVAs of inactive lever responses during the final post-MR EXT vs Test session did not reveal significant Context × Treatment interaction, Context or Treatment main effects.
The 2 × 6 ANOVA of lever responses during 20 min Test Bins of the 2 h Test revealed significant Test Bin main (active: F5, 28 = 42.136, p < 0.001; inactive: F5, 28 = 13.150, p < 0.001), with no Test Bin × Age interaction or Age main effects (Fig. 2E). Tukey’s post-hoc comparisons revealed that both adult and adolescent groups decreased active lever responses by the final 20 min of the Test (active: Bin 1 > 6, #p < 0.01). Tukey’s post-hoc comparisons did not reveal significant differences in inactive lever responses during 20 min Test Bins.
For all behavioral phases, no Context × Age × Treatment interaction effects were found, therefore adult and adolescent groups were further independently analyzed.
Adult behavior during Coc-SA, EXT, MR and tests: effect of CHX on reconsolidation of cocaine context memories
Adult rats in the VEH (n = 8) and CHX (n = 8) groups displayed similar responses on both levers during Coc-SA, with no pre-existing differences observed in the number of sessions to acquire Coc-SA, lever responses or cocaine infusions during the last three sessions of Coc-SA (Fig. 3A). The 2 × 10 ANOVAs during Coc-SA did not reveal significant Coc-SA session × Treatment or Coc-SA infusion × Treatment interaction effects.
Adult rats in the VEH and CHX groups had similar lever responses during EXT training (Fig. 3B). The 2 × 8 ANOVAs of lever responses revealed a significant EXT session main effect (active: F7, 14 = 26.439, p < 0.001; inactive: F7, 14 = 13.301, p < 0.001), with no EXT session × Treatment interaction or Treatment main effects. Tukey’s post-hoc comparisons revealed that both groups decreased lever responses by the final EXT session (active and inactive: EXT S1 > S8, #p < 0.01).
Adult rats in the VEH and CHX groups had similar lever responses during the MR session (Fig. 3C). The 2 × 3 ANOVA of lever responses during 5 min bins of the 15 min MR session revealed a significant MR Bin main effect (active: F2, 14 = 14.646, p < 0.001) with no MR Bin × Treatment interaction or Treatment main effects. Tukey’s post-hoc comparisons revealed that both VEH and CHX groups displayed reduced active lever responses by the final 5 min of MR (active: Bin 1 > 3, #p < 0.01). Tukey’s post-hoc comparisons did not reveal significant differences in inactive lever responses during 5 min MR Bins (Suppl Fig. 3A).
Adult rats in VEH and CHX groups displayed increased cocaine-seeking behavior when exposed to the cocaine-paired context (Fig. 3D). The 2 × 2 ANOVAs of active lever responses during the final post-MR EXT vs Test session revealed a significant Context main (active: F1, 14 = 56.826, p < 0.001), but no Context × Treatment interaction or Treatment main effects. Tukey’s post-hoc comparisons revealed that lever responses in the VEH and CHX groups were higher during the Test compared to the final post-MR EXT session (active: #p < 0.01) therefore, both VEH and CHX-treated groups exhibited cocaine-seeking behavior. The 2 × 2 ANOVAs of inactive lever responses during the final post-MR EXT vs Test session did not reveal significant Context × Treatment interaction, Context or Treatment main effects.
The 2 × 6 ANOVA of lever responses during 20 min Test Bins of the 2 h Test revealed significant Test Bin main effect (active: F5, 14 = 25.197, p < 0.001; inactive: F5, 14 = 8.955, #p < 0.001) with no Test Bin × Treatment or Treatment main effects (Fig. 3E). Tukey’s post-hoc comparisons revealed that responses for both VEH and CHX groups decreased by the final 20 min of the Test (active and inactive: Bin 1 > 6, #p < 0.01). Further analysis of lever responses during 5 min Bins of the 2 h Test did not reveal Bin × Treatment interaction, Bin or Treatment main effects at the active or inactive levers (Suppl Fig. 3B).
Adolescent behavior during Coc-SA, EXT, MR and tests: effect of CHX on reconsolidation of cocaine context memories
Adolescent rats in the VEH (n = 6) and CHX (n = 8) groups displayed similar responses on both levers with no pre-existing differences observed in the number of sessions to acquire Coc-SA, lever responses, or cocaine infusions during the last three sessions of Coc-SA (Fig. 4A). The 2 × 10 ANOVAs during Coc-SA did not reveal significant Coc-SA session × Treatment or Coc-SA infusion × Treatment interaction effects.
Adolescent rats in the VEH and CHX groups had similar lever responses during EXT training (Fig. 4B). The 2 × 8 ANOVA of lever responses revealed a significant EXT session main effect (active: F7, 12 = 19.713, p < 0.001; inactive: F7, 12 = 14.304, p < 0.001), with no EXT session × Treatment interaction or Treatment main effects. Tukey’s post-hoc comparisons revealed that responding decreased in both VEH and CHX groups by the final EXT session (active and inactive: EXT S1 > S8, #p < 0.01).
Adolescent rats in the VEH and CHX groups had similar lever responses during the MR session (Fig. 4C). The 2 × 3 ANOVA of lever responses during 5 min bins of the 15 min MR session revealed significant MR Bin main effect (active: F2, 12 = 8.311, p = 0.002) with no MR Bin × Treatment interaction or Treatment main effects. Tukey’s post-hoc comparisons revealed that only VEH groups displayed reduced active lever responses by the final 5 min of MR (VEH: Bin 1 > 3, #p < 0.01; CHX: Bin 1 ≠ 3, 2 ≠ 3). Tukey’s post-hoc comparisons did not reveal significant differences in inactive lever responses during 5 min MR Bins (Suppl Fig. 4A).
Adolescent rats in the VEH and CHX groups displayed increased cocaine-seeking behavior when exposed to the cocaine-paired context (Fig. 4D). The 2 × 2 ANOVA of active lever responses during the final post-MR EXT vs Test sessions revealed a significant Context main (active: F1, 12 = 31.633, p < 0.001; inactive: F1, 12 = 7.010, p = 0.021) with no Context × Treatment interaction or Treatment main effects. Tukey’s post-hoc comparisons revealed that lever responses in the VEH and CHX groups were higher during the Test compared to the final post-MR EXT session (active: #p < 0.01; inactive: #p < 0.05) therefore, both VEH and CHX-treated adolescent groups exhibited cocaine-seeking behavior.
The 2 × 6 ANOVA of lever responses during 20 min Test Bins of the 2 h Test revealed significant Test Bin main effect (active: F5, 12 = 18.524, p < 0.001; inactive: F5, 12 = 6.149, p < 0.001) with no Test Bin × Treatment or Treatment main effects (Fig. 4E). Tukey’s post-hoc comparisons revealed that active lever responses for both VEH and CHX groups, and inactive for the CHX group decreased by the final 20 min of the Test (Bin 1 > 6, active: #p < 0.01; inactive: #p < 0.05). Further analysis of lever responses during 5 min Bins of the 2 h Test revealed Bin main effect (active: F2, 12 = 8.618, p = 0.002; inactive: F2, 12 = 4.441, p = 0.023), and Bin × Treatment interaction for active lever (F2, 24 = 3.972, p = 0.032); however Tukey’s post-hoc comparisons revealed no significant effects at Test Bins within or between groups (Suppl Fig. 4B).
Discussion
Overall, systemic protein synthesis inhibition after retrieval of cocaine-associated contextual memories did not reduce relapse-like behavior in adult or adolescent cocaine-exposed groups. The current results did not reveal age-dependent differences during operant phases of behavioral training (Fig. 2). Adult and adolescent rats had similar lever responses during Coc-SA and EXT training phases and these data are in line with previous reports in which self-administration of cocaine occurred in the presence of diffuse environmental stimuli or paired with explicit cues27,28,29,35,36.
There is evidence that adolescent rats have increased resistance to extinguish fear and cocaine-related behaviors24,29,37. A previous study determined that adolescent rats were less responsive to a rodent version of cue-exposure therapy (CET). In this study, investigators combined a Coc-SA paradigm with passive presentation of a cocaine-associated cue to mimic human CET and observed that adult and adolescent rats displayed similar levels of EXT training behavior, as observed in our study, but were resistant to passive cue extinction compared to adults29. We have also noted a potential resistance to extinguish lever responses in adolescent rats within the original cocaine-paired context, but not within the EXT context28. Similar to our previous reports, adolescent rats had higher total inactive lever presses during reinstatement tests; however both age groups displayed increased inactive presses during the first 20 min bin of the reinstatement test. It is possible that across the entire test session, adolescent cocaine-exposed rats had increased motivation or response generalization to both levers in the drug-paired context38,39.
We modified our ABRV Coc-SA paradigm to incorporate standard procedures to test whether protein synthesis was required for the reconsolidation of adult- and adolescent-formed cocaine-context memories. Importantly, lever responses were similar between adult and adolescent cocaine-exposed rats during a reactivation session used to retrieve cocaine-context memories. We chose 15 min of exposure to the previous cocaine-paired context since this timeframe is sufficient to elicit retrieval in adult rats without engaging extensive extinction processes11,12,40. We examined 5 min intervals of the 15 min MR session but did not observe age-dependent differences in the curves of lever responses (Suppl Fig. 2A) or in the latency for the first lever press during MR; therefore, the MR session likely served to retrieve cocaine-associated memories to a similar extent for both age groups. Overall, operant lever response behavior was similar between adult and adolescent rats during Coc-SA and EXT training and MR, which suggests that an age-dependent difference in the strength of drug-context associations did not contribute to the lack of effect of CHX on reconsolidation and subsequent relapse.
The current results revealed that adult and adolescent cocaine-exposed rats had similar magnitudes of context-induced cocaine-seeking behavior regardless of post-retrieval treatment with VEH or CHX (Figs. 3, 4). Surprisingly, CHX treatment failed to reduce cocaine-seeking behavior in adult rats which contrasts with reports of attenuated cocaine reward and cue-induced reinstatement following systemic CHX administration21,41. A subcutaneous dose of 1.0 mg/kg CHX has been shown to reduce protein synthesis in the brain by up to 57%42. A range of CHX doses, from 1.0 to 15 mg/kg, has been used for cocaine CPP and operant self-administration studies. A systemic dose of 2.2 mg/kg CHX reduced cue-induced cocaine-seeking behavior; whereas a 1.0 mg/kg dose did not reduce seeking behavior21. Higher doses of systemic CHX (15 mg/kg) resulted in reduced cocaine CPP41,43. We therefore chose the lowest dose of CHX that was within the range expected to observe behavioral effects, to avoid potential negative effects related to toxicity with protein synthesis inhibitors in general44,45. Weight loss has been noted after SC administration of CHX at a dose of 1.5 mg/kg; therefore, we examined weight at similar intervals—2 days before and after VEH or CHX administration45. Adult and adolescent groups both gained weight at similar magnitudes, with no reductions in weight observed in CHX-treated groups (Suppl Fig. 1). The most parsimonious explanation would suggest unsuccessful CHX delivery, however differences in experimental procedures and food delivery could also account for these results. Rats in the current study received restricted, rather than ad libitum feed. Both age groups gained weight on restricted feed, but in combination with a history of cocaine intake, our experimental procedures may have obscured the ability to detect subtle differences in weight loss due to CHX administration.
The current study used comparable lengths of memory reactivation and doses of CHX, but a major difference from previous reports was the type of stimuli used to elicit retrieval. Previous studies that reported CHX-induced impairment in reconsolidation and drug-seeking behavior used a cocaine-associated explicit cue to elicit retrieval, while the current study exposed rats to a previous cocaine-paired context. It is known that different brain regions and circuits support cue vs contextual cocaine-seeking behavior7,32,46. Furthermore, signaling cascades that are important for memory reconsolidation are shown to be differentially involved in cue vs contextual retrieval and subsequent relapse behavior. For example, post-MR inhibition of protein kinase A activity in the BLA impairs both cue- and context-elicited reconsolidation and subsequent cocaine-seeking; whereas post-MR inhibition of CAMKII in the BLA only impairs cue-elicited drug-seeking behavior12,18,40. Another difference from previous studies is that reinstatement Tests in the current study were conducted 2 days after MR and CHX treatment, whereas previous reports conducted Tests 3 days after MR + CHX treatment. The lack of a behavioral effect on relapse from adult systemic CHX manipulation likely is specific to retrieval of contextual vs cue memories, as has also been observed in the fear conditioning literature47.
While beyond the scope of the current work, future studies could focus on use of the unconditioned stimuli (US) of cocaine to retrieve and destabilize cocaine-associated memories. Previous reports that used the US to retrieve and destabilize both drug CPP and operant drug-associated memories observed reduced reward and cue-induced cocaine-seeking in adult rats48,49,50,51. Use of the US to retrieve cocaine-linked memories could be a particularly relevant approach in adolescent rats, given that this age group displays higher stress and cocaine-primed reinstatement compared to their adult counterparts, suggesting potential age differences in reactivity to external vs interoceptive cues33,52. In line with modifying the stimuli used to retrieve cocaine-context memories, future studies could present a CS-US pairing that differs in duration from the original CS-US used during training to provide for a larger prediction error during the memory retrieval session53.
Lastly, it is also possible that systemic administration of CHX may have exerted effects in several brain regions involved in reconsolidation and motivated behavior, thereby obscuring potential reductions in cocaine-seeking behavior. A large body of literature has demonstrated brain-specific reconsolidation mechanisms contribute to relapse-like behavior11,12,20,34,54. Delivery of protein synthesis inhibitors to specific brain regions to investigate adult vs adolescent differences in drug memory reconsolidation may be a more promising avenue for future experiments.
Conclusions and future directions
The current results suggest that both adult and adolescent-formed cocaine-context associations were not sensitive to, or weakened by, reconsolidation manipulations via systemic protein synthesis inhibition. A large body of literature demonstrates that brain-specific reconsolidation mechanisms contribute to cocaine-associated memories and relapse. Therefore, future directions should investigate the brain region specific mechanisms that strengthen cocaine-context memories in adult and adolescent rats. We recently demonstrated that adolescent cocaine-exposed rats display a time-dependent increase in context-induced cocaine-seeking behavior after 15 days of abstinence from cocaine intake, whereas adult cocaine-exposed counterparts do not28. Future studies could aim to use similar procedures of memory reactivation in a cocaine-paired context after extinction training and then examine whether reduced context-induced incubation of craving is observed in adolescent cocaine-exposed rats.
Data availability
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
References
Ramo, D. E. & Brown, S. A. Classes of substance abuse relapse situations: A comparison of adolescents and adults. Psychol. Addict. Behav. 22, 372–379 (2008).
Winters, K. C., Tanner-Smith, E. E., Bresani, E. & Meyers, K. Current advances in the treatment of adolescent drug use. Adolesc. Health. Med. Ther. 5, 199–210 (2014).
McCabe, S. E., Schulenberg, J. E., Schepis, T. S., McCabe, V. V. & Veliz, P. T. Longitudinal analysis of substance use disorder symptom severity at age 18 years and substance use disorder in adulthood. JAMA Netw. Open 5, e225324 (2022).
Volkow, N. D. & Wargo, E. M. Association of severity of adolescent substance use disorders and long-term outcomes. JAMA Netw. Open 5, e225656 (2022).
Ryan, S. A. Cocaine use in adolescents and young adults. Pediatr. Clin. N. Am. 66, 1135–1147 (2019).
Guerin, A. A. & Kim, J. H. Age of onset and its related factors in cocaine or methamphetamine use in adults from the United States: Results from NHANES 2005–2018. Int. J. Environ. Res. Public Health 18, 12259 (2021).
Perry, C. J., Zbukvic, I., Kim, J. H. & Lawrence, A. J. Role of cues and contexts on drug-seeking behaviour. Br. J. Pharmacol. 171, 4636–4672 (2014).
Khoo, S. Y. S., Gibson, G. D., Prasad, A. A. & McNally, G. P. How contexts promote and prevent relapse to drug seeking. Genes Brain Behav 16, 195–204 (2017).
Feltenstein, M. W., See, R. E. & Fuchs, R. A. Neural substrates and circuits of drug addiction. Cold Spring Harbor Persp. Med. 11, a039628 (2021).
Miller, C. A. & Marshall, J. F. Molecular substrates for retrieval and reconsolidation of cocaine-associated contextual memory. Neuron 47, 873–884 (2005).
Ramirez, D. R. et al. Dorsal hippocampal regulation of memory reconsolidation processes that facilitate drug context-induced cocaine-seeking behavior in rats. Eur. J. Neurosci. 30, 901–912 (2009).
Sanchez, H., Quinn, J. J., Torregrossa, M. M. & Taylor, J. R. Reconsolidation of a cocaine-associated stimulus requires amygdalar protein kinase A. J. Neurosci. 30, 4401–4407 (2010).
Sara, S. J. Retrieval and reconsolidation: Toward a neurobiology of remembering. Learn. Mem. 7, 73–84 (2000).
Alberini, C. M., Milekic, M. H. & Tronel, S. Mechanisms of memory stabilization and de-stabilization. Cell. Mol. Life Sci. 63, 999–1008 (2006).
Nader, K. & Einarsson, E. Ö. Memory reconsolidation: An update. Ann. N. Y. Acad. Sci. 1191, 27–41 (2010).
Sorg, B. A. Reconsolidation of drug memories. Neurosci. Biobehav. Rev. 36, 1400–1417 (2012).
Tronson, N. C. & Taylor, J. R. Addiction: A drug-induced disorder of memory reconsolidation. Curr. Opin. Neurobiol. 23, 573–580 (2013).
Bender, B. N. & Torregrossa, M. M. Molecular and circuit mechanisms regulating cocaine memory. Cell. Mol. Life Sci. 77, 3745–3768 (2020).
Lee, J. L. C., Milton, A. L. & Everitt, B. J. Cue-induced cocaine seeking and relapse are reduced by disruption of drug memory reconsolidation. J. Neurosci. 26, 5881–5887 (2006).
Wells, A. M. et al. Interaction between the basolateral amygdala and dorsal hippocampus is critical for cocaine memory reconsolidation and subsequent drug context-induced cocaine-seeking behavior in rats. Learn. Mem. 18, 693–702 (2011).
Dunbar, A. B. & Taylor, J. R. Inhibition of protein synthesis but not β-adrenergic receptors blocks reconsolidation of a cocaine-associated cue memory. Learn. Mem. 23, 391–398 (2016).
Achterberg, E. J. M., Trezza, V. & Vanderschuren, L. J. M. J. β-Adrenoreceptor stimulation mediates reconsolidation of social reward-related memories. PLoS ONE 7, e39639 (2012).
Baker, K. D., McNally, G. P. & Richardson, R. Memory retrieval before or after extinction reduces recovery of fear in adolescent rats. Learn. Mem. 20, 467–473 (2013).
Baker, K. D., Bisby, M. A. & Richardson, R. Impaired fear extinction in adolescent rodents: Behavioural and neural analyses. Neurosci. Biobehav. Rev. 70, 59–73 (2016).
Johnson, D. C. & Casey, B. J. Extinction during memory reconsolidation blocks recovery of fear in adolescents. Sci. Rep. 5, 8863 (2015).
McCutcheon, J. E. & Marinelli, M. Age matters. Eur. J. Neurosci. 29, 997–1014 (2009).
Cho, B. R. et al. Role of adolescent-formed, context-drug-associations on reinstatement of drug-seeking behavior in rats. Psychopharmacology 237, 2823–2833 (2020).
Olekanma, D. I. et al. Context-drug-associations and reinstatement of drug-seeking behavior in male rats: Adolescent and adult time-dependent effects. Neurobiol. Learn. Mem. 199, 107722 (2023).
Zbukvic, I. C. et al. Role of dopamine 2 receptor in impaired drug-cue extinction in adolescent rats. Cereb. Cortex 26, 2895–2904 (2016).
Fuchs, R. A. et al. The role of the dorsomedial prefrontal cortex, basolateral amygdala, and dorsal hippocampus in contextual reinstatement of cocaine seeking in rats. Neuropsychopharmacology 30, 296–309 (2005).
Bal, A., Gerena, J., Olekanma, D. I. & Arguello, A. A. Neuronal activation in orbitofrontal cortex subregions: Cfos expression following cue-induced reinstatement of cocaine-seeking behavior. Behav. Neurosci. 135, 489–495 (2019).
Fuchs, R. A., Lasseter, H. C., Ramirez, D. R. & Xie, X. Relapse to drug seeking following prolonged abstinence: The role of environmental stimuli. Drug Discov. Today Dis. Model 5, 251–258 (2008).
Anker, J. J. & Carroll, M. E. Reinstatement of cocaine seeking induced by drugs, cues, and stress in adolescent and adult rats. Psychopharmacology 208, 211–222 (2010).
Arguello, A. A. et al. Role of the agranular insular cortex in contextual control over cocaine-seeking behavior in rats. Psychopharmacology 234, 2431–2441 (2017).
Guerin, A. A. et al. Extinction and drug-induced reinstatement of cocaine seeking following self-administration or conditioned place preference in adolescent and adult rats. Dev. Psychobiol. 63, 125–137 (2021).
Madsen, H. B., Zbukvic, I. C., Luikinga, S. J., Lawrence, A. J. & Kim, J. H. Extinction of conditioned cues attenuates incubation of cocaine craving in adolescent and adult rats. Neurobiol. Learn. Mem. 143, 88–93 (2017).
Brenhouse, H. C. & Andersen, S. L. Delayed extinction and stronger reinstatement of cocaine conditioned place preference in adolescent rats, compared to adults. Behav. Neurosci. 122, 460–465 (2008).
Lu, L., Grimm, J. W., Dempsey, J. & Shaham, Y. Cocaine seeking over extended withdrawal periods in rats: Different time courses of responding induced by cocaine cues versus cocaine priming over the first 6 months. Psychopharmacology 176, 101–108 (2004).
Carroll, M. E., Dougen, B., Zlebnik, N. E., Fess, L. & Smethells, J. Reducing short- and long-term cocaine craving with voluntary exercise in male rats. Psychopharmacology 239, 3819–3831 (2022).
Arguello, A. A. et al. Involvement of amygdalar protein kinase A, but not calcium/calmodulin-dependent protein kinase II, in the reconsolidation of cocaine-related contextual memories in rats. Psychopharmacology 231, 55–65 (2014).
Fan, H. Y. et al. Systemic treatment with protein synthesis inhibitors attenuates the expression of cocaine memory. Behav. Brain Res. 208, 522–527 (2010).
Serova, O. N., Solov’ea, N. A., Lagutina, L. V. & Obukhova, M. F. Formation of taste aversion and preference in protein synthesis inhibition in rats. Neurosci. Behav. Physiol. 26, 477–481 (1996).
Kuo, Y. M. et al. Cocaine-but not methamphetamine-associated memory requires de novo protein synthesis. Neurobiol. Learn. Mem. 87, 93–100 (2007).
Exton-McGuinness, M. T. J. & Milton, A. L. Reconsolidation blockade for the treatment of addiction: Challenges, new targets, and opportunities. Learn. Mem. 25, 492–500 (2018).
Luyten, L., Schnell, A. E., Schroyens, N. & Beckers, T. Lack of drug-induced post-retrieval amnesia for auditory fear memories in rats. BMC Biol. 19, 1–15 (2021).
Bossert, J. M., Marchant, N. J., Calu, D. J. & Shaham, Y. The reinstatement model of drug relapse: Recent neurobiological findings, emerging research topics, and translational research. Psychopharmacology 229, 453–476 (2013).
Biedenkapp, J. C. & Rudy, J. W. Context memories and reactivation: Constraints on the reconsolidation hypothesis. Behav. Neurosci. 118, 956–964 (2004).
Sorg, B. A., Todd, R. P., Slaker, M. & Churchill, L. Anisomycin in the medial prefrontal cortex reduces reconsolidation of cocaine-associated memories in the rat self-administration model. Neuropharmacology 92, 25 (2015).
Xue, Y. X. et al. Effect of selective inhibition of reactivated nicotine-associated memories with propranolol on nicotine craving. JAMA Psychiatry 74, 224–232 (2017).
Dunbar, A. B. & Taylor, J. R. Garcinol blocks the reconsolidation of multiple cocaine-paired cues after a single cocaine-reactivation session. Neuropsychopharmacology 42, 1884–1892 (2017).
Briggs, S. B., Hafenbreidel, M., Young, E. J., Rumbaugh, G. & Miller, C. A. The role of nonmuscle myosin II in polydrug memories and memory reconsolidation. Learn. Mem. 25, 391–398 (2018).
Wong, W. C. & Marinelli, M. Adolescent-onset of cocaine use is associated with heightened stress-induced reinstatement of cocaine seeking. Addict. Biol. 21, 634–645 (2016).
Tallot, L. et al. Updating of aversive memories after temporal error detection is differentially modulated by mTOR across development. Learn. Mem. 24, 115–122 (2017).
Fuchs, R. A., Bell, G. H., Ramirez, D. R., Eaddy, J. L. & Su, Z. Basolateral amygdala involvement in memory reconsolidation processes that facilitate drug context-induced cocaine seeking. Eur. J. Neurosci. 30, 889–900 (2009).
Acknowledgements
This work was supported by NIDA Grant R00 DA037271, MSU Grant GE100332.
Author information
Authors and Affiliations
Contributions
A.N.H.C.: Data Acquisition & Analysis, Figure Preparation, Manuscript Text. D.I.O.: Data Acquisition. C.T.V.: Data Analysis, Manuscript Text. C.A.R.: Data Acquisition. B.R.C.: Data Acquisition. A.A.A.: Conceptualization of Projection, Data Acquisition & Analysis, Figure Preparation, Manuscript Text.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Charpentier, A.N.H., Olekanma, D.I., Valade, C.T. et al. Influence of reconsolidation in maintenance of cocaine-associated contextual memories formed during adolescence or adulthood. Sci Rep 13, 13936 (2023). https://doi.org/10.1038/s41598-023-39949-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-39949-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.