Abstract
This study aimed to assess recurrence-free survival (RFS) rates and recurrence-related factors of patients who received adjuvant therapy (AT) with radioactive iodine (RAI) for differentiated thyroid cancer (DTC) following thyroidectomy. We evaluated 284 patients who underwent AT between January 2011 and July 2020 at our hospital. Recurrence was defined as visible recurrent lesions on image analysis or need for repeat surgery with pathologically confirmed recurrent lesions. RFS rate and prognostic factors were statistically evaluated. The median observation period was 30.2 months (range, 5.7–294 months). Overall, 192 patients were female and 92 were male, and the median age was 54 years (range, 9–85 years). Initial assessment revealed 39 recurrence cases. The 3-year RFS rate was 85.8% (95% confidence interval: 81.1–90.9%). Univariate analysis revealed that histology (except for papillary carcinoma), Tg level > 4 ng/dL before AT, and AT result significantly exacerbated the RFS rate. In multivariate analysis, histology and AT result were also important contributors to the worsening RFS rate. Results of AT can be determined relatively early and are important in predicting future recurrence in patients with DTC. Increasing the success rate of AT may lead to an improved prognosis.
Similar content being viewed by others
Introduction
Thyroid cancer is the most frequent cancer of the endocrine system, and its incidence is constantly rising worldwide. This is mainly because of advances in examination modalities that have made it possible to detect smaller cancers at an early stage. Owing to advancements in screening techniques and treatment methods, the number of patients who die from thyroid cancer is small compared to the incidence. Women are three times more likely to develop thyroid cancer than men1. The most common type of thyroid cancer is differentiated thyroid cancer (DTC), which includes papillary and follicular carcinomas. In Japan, the prevalence of DTC is reported to be more than 90%2,3.
The use of radioactive iodine (RAI) as part of radioisotope therapy for DTC following thyroidectomy, including “remnant ablation,” “adjuvant therapy (AT),” and “cancer treatment,” is well-accepted. AT is recommended, because of its therapeutic impact on micro-invasions or micro-metastases, to reduce the likelihood of recurrence in patients who do not have metastases but have a high risk of recurrence according to the Japanese guidelines and American Thyroid Association guidelines4,5. However, such situations cannot be easily identified. Ruel et al. reported that AT could prolong the survival of patients with intermediate-risk DTC6. Additionally, a meta-analysis has shown that AT is effective in reducing recurrence in some studies but not in others7, and the effect of AT on recurrence prevention, especially in high-risk DTC, remains unclear. One study has reported the death-suppressing effect of RAI administration in a high-risk patient group, but this report includes cases with metastasis and a high risk of recurrence, for which AT is not used8. In addition, it is not known whether the full benefits of AT can be obtained in individual patients or clinical settings.
Although there are reliable reports discussing the relationship between the success or failure of remnant ablation9,10, recurrence after AT is rarely reported and the number of cases is small11,12. In addition, no study has so far discussed the relationship between the success or failure of AT or remnant ablation and recurrence or death. No conclusions about the indicated doses for remnant ablation and AT have been drawn, with some studies showing no difference in success rates between high and low doses and others showing better results at higher doses13,14,15. The recommended dose for AT is stated in the guidelines but the statement is evaluated as “Weak recommendation, Low-quality evidence”4,5. Therefore, the aim of this study was to assess the recurrence-free survival (RFS) rate and recurrence-related factors, especially the relationship between RFS and AT dose, in patients who received AT with RAI after thyroidectomy.
Results
The median observation period was 30.2 months (range, 5.7–294 months). The median age of the patients was 54 years (range, 9–85 years). The study included 192 female patients and 92 male patients. Doses of 1110, 1850, and 3700 MBq were administered to 111, 16, and 157 patients, respectively. Detailed characteristics are shown in Table 1.
Overall, 39 cases of recurrence were observed in the initial assessment. The following were the locations of the first recurrences: thyroid bed in two patients, cervical lymph nodes (LNs) in 16, mediastinal LNs in three, other LN sites in two, and lungs in 16 (including overlap). The 3-year RFS rate was 85.8% (95% confidence interval (CI): 81.1–90.9%; Fig. 1). Twenty-nine cases of recurrence were observed within 3 years.
Univariate analysis revealed that histology (except for papillary carcinoma), Tg levels > 4 ng/dL before AT, and AT outcomes were significant factors that worsened the RFS rate. In Cox regression analysis, histology and AT findings were also important contributors to the worsening RFS rate (Table 2, Fig. 2). Two patients relapsed before the AT results were determined and were treated as failures for convenience in the aforementioned analysis. However, even when these two patients were excluded from the analysis, the AT result was a significant predictor of RFS in both univariate and multivariate analyses (p < 0.01). The median observation period of the AT success group was 28.6 months and that of the failure group was 33.2 months (p = 0.41). Although the low-dose group (1110 MBq) tended to have a better prognosis than the high-dose group (2960 and 3700 MBq), the difference was not significant (p = 0.07 in both univariate and multivariate analyses).
Five patients died during observation: four died from the primary disease and one died from other causes. The estimated OS was 97.8% (95% CI 95.6–100%) at 3 years (Fig. 3). The median observation period of the high-dose group was 31.0 months and that of the low-dose group was 27.6 months (p = 0.63). All the patients who died were from the high-dose group, and the estimated OS was 96.2% (95% CI 90.9–98.4%). In univariate analysis, histology (except for papillary carcinoma) (p < 0.01) and AT outcomes (p = 0.04) were significant factors that worsened the OS rate. In Cox regression analysis, histology (p < 0.01) and AT results (p = 0.05) were also statistically significant predictors of a worsening OS rate.
Discussion
We retrospectively evaluated RFS rates and recurrence patterns after AT in patients with DTC. The 3-year RFS rate was 85.8% (95% CI 81.1–90.9%) and the most common recurrence site was cervical LNs.
Histology (except for papillary carcinoma, N = 15) was a significant poor prognostic factor for RFS. The breakdown of this group was as follows: follicular carcinoma, 7; poorly differentiated carcinoma, 7; and mixed papillary and follicular pattern, 1. A possible explanation for these observed results could be that poorly differentiated carcinoma has a poor prognosis16,17. AT success/failure is associated with Tg levels, and cases with a high pre-Tg level also have more recurrences. The pre-Tg level can imply the presence of residual lesions at the thyroid bed or distant metastases elsewhere after surgery, which may contribute to future recurrences. Patients whose metastases were identified by 131I scintigraphy during AT (excluded from this study) had higher Tg levels than the overall population (mean, 99.1; range, 0.25–429.6 ng/mL).
AT failure is significantly associated with recurrence. We analysed the prognostic factors for AT in a previous study and found that cases with Pre-Tg levels greater than 4 ng/mL had significantly more AT failures18. AT results can be evaluated within 1 year at most and may be useful as an early predictor of recurrence9,10. To date, there have been no reports of methods or favourable populations that increase the success rate of AT; therefore, careful follow-up is desirable for patients with failed AT. This is the first report to discuss the relationship between the success and failure status of AT and DTC recurrence. Criteria for successful AT have not been definitively established, and they tend to vary across different reports. For example, they are based on serum Tg concentration with thyroid-stimulating hormone (TSH) stimulation or non-stimulation, imaging studies (131I scintigraphy or ultrasonography), or a combination of these. Indeed, the definition of AT success in the present study is not always used, but we use the same criteria as in previous studies in order to be consistent in our discussion18: “no accumulation of 131I in the thyroid bed on scintigraphy and a serum Tg concentration < 2.0 ng/mL without TSH stimulation”. Radiation dose and positive surgical margins or extranodal invasion are sometimes said to be associated with recurrence19,20; however, in this study, there was no significant effect on RFS. Rather, RFS tended to be better in the low-dose group, which may have been influenced by the tendency to use high doses in high-risk cases. There are no studies on the appropriate doses in AT yet, but guidelines recommend doses up to 5550 MBq4,5. Reports of AT at even higher doses are awaited.
There are few reports on clinical outcomes after AT with RAI, particularly in patients at intermediate or high risk of recurrent DTC. Watanabe et al. reported the RFS rate after AT with microscopically positive tumour margins11. They evaluated 48 patients who underwent AT with low-dose (1110 MBq, N = 17) or high-dose (3700 MBq, N = 31) RAI after total thyroidectomy and reported nine cases (52.9%) of recurrence in the low-dose group and seven (22.5%) in the high-dose group. The 3-year RFS rates were approximately 85% in the low-dose group and 94% in the high-dose group. RFS in the low-dose group was significantly shorter than that in the high-dose group. Lymph node metastases were the most common type of recurrence. Jeong et al. reported the RFS rate in 253 patients with T4 or N1b DTC who underwent AT with different 131I doses (3700 MBq vs 5550 MBq). Of the 253 patients, 22 (8.7%) experienced recurrence after AT, and the high-dose and low-dose groups did not differ significantly from one another. The 3-year RFS rate was approximately 93%, with the most common type of recurrence being cervical LN metastasis. Our results were worse than those of the Korean study, and the cause of this is unclear. The authors stated that these patients were operated on by only two experienced surgeons at a high-volume tertiary care centre. This finding suggests that skilfully done surgery reduces the risk of recurrence. In addition, recurrence was histologically or cytologically proven, and it is possible that lung metastasis, which can be diagnosed only by imaging, had been overlooked. The primary endpoint of the study by Jeong et al. was the success or failure of AT, and the relationship between this and RFS was not described12.
The definition of recurrence is controversial, and the clinical diagnosis of recurrence is difficult. Lesions that take up iodine can be diagnosed with high sensitivity by 131I scintigraphy but those that do not take up iodine change a little over time and are often difficult to diagnose with imaging. 18F-Fluorodeoxyglucose positron emission tomography may be used in patients with negative 131I scintigraphy or anti-Tg antibodies21,22. Lymph node metastases may be pathologically diagnosed after neck dissection in patients with a high Tg level, but small pulmonary nodules may be indistinguishable from inflammatory changes. In this study, lung lesions were diagnosed using imaging, which was less accurate. An accurate diagnosis of non-iodine-uptake lesions is also needed.
This study had a few limitations. Firstly, it was an observational retrospective study conducted at a single institution. However, the treatment policy and dose were consistent, and the selection bias was small. Secondly, the observation period was short, and the sample size was small. Lastly, we observed only five cases of death, and we could not evaluate whether AT prolonged the OS. To assess the relationship between AT and OS rates, a long-term comparison study with a larger sample size is required.
In patients who received AT with RAI, the estimated 3-year RFS rate was 85.8%. Histological and AT results significantly influenced the RFS rate. The AT result can be determined relatively early and is an important index for predicting future recurrence. Thus, increasing the success rate of AT may lead to an improved prognosis.
Materials and methods
Study design
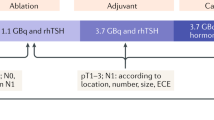
This retrospective evaluation was performed on patients who received AT in our hospital for DTC after surgical resection without macroscopic residual or metastatic lesions (patients with gross residual lesions at the tumour bed or at the site of lymph node resection who were considered to be receiving “cancer treatment” were not included in this study). A total of 343 patients underwent AT between January 2011 and July 2020. Of these, 284 patients were evaluated after excluding those whose metastases were confirmed during AT and those whose AT results were unknown (Fig. 4).
Procedure for AT
Preparations for AT were as follows: iodine intake restriction for 2–3 weeks strictly and thyroid hormone restriction or use of recombinant human thyroid-stimulating hormone (rhTSH) to elevate the serum TSH level prior to 131I administration.
131I was administered in doses of 1110 MBq (low-dose group) or 2960–3700 MBq (high-dose group). Radiation oncologists recommend these doses based on pathological findings, ATA risk stratification, patient preference, and the environment of the patient’s residence. None of the patients were included in the ATA low-risk group, and all patients were treated with 131I as AT. In Japan, as only a dose below 1110 MBq of 131I is permitted for use in AT when administered in outpatient settings, outpatients who underwent AT were included in the low-dose group and inpatients were classified into the high-dose group. Three days after 131I administration, whole-body 131I scintigraphy was performed using a gamma camera with high-energy collimators (Infinia Hawkeye4, GE Healthcare, Milwaukee, WI, USA). Serum thyroglobulin (Tg) concentration was also determined. Three to 12 months after AT, a diagnostic 131I scintigraphy (370 MBq) was performed to determine whether AT was successful. According to our study and earlier investigations, initial AT success was defined as the absence of 131I accumulation in the thyroid bed on 131I scintigraphy and a serum Tg concentration of < 2.0 ng/mL without TSH stimulation. We used the same criteria as in our previous study18, determined with reference to previous reports9,10. Patients who tested positive for anti-Tg antibodies were determined based on their 131I scintigraphy results, whereas those who did not undergo 131I scintigraphy were evaluated based on serum Tg concentration. Eastern Cooperative Oncology Group performance status (ECOG-PS) was determined through a medical interview and physical examination at the first visit by the doctor in charge.
Follow-up and outcome measures
Patients were examined every 2–6 months after AT, serum Tg concentration was measured every 3–6 months, and neck-to-chest computed tomography (CT) or neck ultrasonography was performed if the physician thought the examination was needed, until the end of follow-up. The last day of follow-up was the date of death or when survival was confirmed (mainly the date of the outpatient visit). Patients with visible recurrent lesions on image analysis or those who underwent repeat surgery with pathologically confirmed recurrent lesions were defined as having recurrent lesions. RFS was defined as the absence of recurrence or mortality from any cause, whereas OS was defined as mortality from any cause.
Statistical analysis
R Software version 4.0.2 (R Foundation for Statistical Computing, Vienna, Austria) was used to perform all statistical analyses23. The Kaplan–Meier method was used to estimate RFS and OS rates, and the log-rank test was used to determine the contribution of each factor to RFS. Age, sex, histology, performance status, ATA risk classification, and thyroglobulin concentration all affected the therapy dose; hence, Cox regression analysis was performed to adjust the effect of these confounders. Because it is presumed that some cases may involve latent lesions that do not show the effect of AT at the time of AT and only become evident later, an additional multivariate analysis was performed that did not take the time factor into account. Statistical significance was set at p < 0.05.
Ethics approval
The 1964 Declaration of Helsinki and all amendments thereto, as well as the recommendations of the Ethical Guidelines for Medical and Health Research Involving Human Subjects, served as the foundation for this retrospective observational study. The study was approved, and due to its retrospective nature, the need for informed consent was waived by Kyoto University Graduate School and Faculty of Medicine and Kyoto University Hospital Ethics Committee (R1087; April 19, 2017). The study was carried out utilising the opt-out procedure described on our hospital website.
Consent to participate
Due to the retrospective nature of the study, the need for informed consent was waived by Kyoto University Graduate School and Faculty of Medicine and Kyoto University Hospital Ethics Committee. This study was carried out utilising the opt-out procedure described on our hospital website.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
References
Vigneri, R., Malandrino, P. & Vigneri, P. The changing epidemiology of thyroid cancer: Why is incidence increasing?. Curr. Opin. Oncol. 27, 1–7. https://doi.org/10.1097/CCO.0000000000000148 (2015).
National Cancer Center Cancer Information Service. https://ganjoho.jp/public/index.html (2019).
Hori, M. et al. Cancer incidence and incidence rates in Japan in 2009: A study of 32 population-based cancer registries for the Monitoring of Cancer Incidence in Japan (MCIJ) project. Jpn. J. Clin. Oncol. 45, 884–891. https://doi.org/10.1093/jjco/hyv088 (2015).
Sawka, A. M. et al. American Thyroid Association guidelines and statements: Past, present, and future. Thyroid 28, 692–706. https://doi.org/10.1089/thy.2018.0070 (2018).
The Japanese Society of Thyroid Surgery/The Japan Association of Endocrine Surgeons. Guidelines for the management of thyroid tumors. J. Jpn. Assoc. Endocr. Surg. Jpn. Soc. Thyroid Surg. 35, Suppl 3 (2018).
Ruel, E. et al. Adjuvant radioactive iodine therapy is associated with improved survival for patients with intermediate-risk papillary thyroid cancer. J. Clin. Endocrinol. Metab. 100, 1529–1536. https://doi.org/10.1210/jc.2014-4332 (2015).
Lamartina, L., Durante, C., Filetti, S. & Cooper, D. S. Low-risk differentiated thyroid cancer and radioiodine remnant ablation: A systematic review of the literature. J. Clin. Endocrinol. Metab. 100, 1748–1761. https://doi.org/10.1210/jc.2014-3882 (2015).
Jonklaas, J. et al. Outcomes of patients with differentiated thyroid carcinoma following initial therapy. Thyroid 16, 1229–1242. https://doi.org/10.1089/thy.2006.16.1229 (2016).
Mallick, U. et al. Ablation with low-dose radioiodine and thyrotropin alfa in thyroid cancer. N. Engl. J. Med. 366, 1674–1685. https://doi.org/10.1056/NEJMoa1109589 (2012).
Schlumberger, M. et al. Strategies of radioiodine ablation in patients with low-risk thyroid cancer. N. Engl. J. Med. 366, 1663–1673. https://doi.org/10.1056/NEJMoa1108586 (2012).
Watanabe, K., Igarashi, T., Uchiyama, M. & Ojiri, H. Relapse-free survival after adjuvant radioactive iodine therapy in patients with differentiated thyroid carcinoma with a microscopically positive tumor margin. Ann. Nucl. Med. 34, 920–925. https://doi.org/10.1007/s12149-020-01523-1 (2020).
Jeong, S. Y. et al. Clinical outcomes of patients with T4 or N1b well-differentiated thyroid cancer after different strategies of adjuvant radioiodine therapy. Sci. Rep. 9, 5570. https://doi.org/10.1038/s41598-019-42083-3 (2019).
Zaman, M. et al. A randomized clinical trial comparing 50mCi and 100mCi of iodine-131 for ablation of differentiated thyroid cancers. J. Pak. Med. Assoc. 56, 353–356 (2006).
Fallahi, B. et al. Low versus high radioiodine dose in postoperative ablation of residual thyroid tissue in patients with differentiated thyroid carcinoma: A large randomized clinical trial. Nucl. Med. Commun. 33, 275–282. https://doi.org/10.1097/mnm.0b013e32834e306a (2012).
Pilli, T. et al. A comparison of 1850 (50 mCi) and 3700MBq (100 mCi) 131-iodine administered doses for recombinant thyrotropin-stimulated postoperative thyroid remnant ablation in differentiated thyroid cancer. J. Clin. Endocrinol. Metab. 92, 3542–3546. https://doi.org/10.1210/jc.2007-0225 (2007).
Sanders, E. M., LiVolsi, V. A., Brierley, J., Shin, J. & Randolph, G. W. An evidence-based review of poorly differentiated thyroid cancer. World J. Surg. 31, 934–945. https://doi.org/10.1007/s00268-007-9033-3 (2007).
Sakamoto, A. Definition of poorly differentiated carcinoma of the thyroid: The Japanese experience. Endocr. Pathol. 15, 307–311. https://doi.org/10.1385/ep:15:4:307 (2004).
Iizuka, Y., Katagiri, T., Ogura, K. & Mizowaki, T. Comparison between the different doses of radioactive iodine ablation prescribed in patients with intermediate-to-high-risk differentiated thyroid cancer. Ann. Nucl. Med. 33, 495–501. https://doi.org/10.1007/s12149-019-01357-6 (2019).
Lee, S. H. et al. Risk factors for recurrence after treatment of N1b papillary thyroid carcinoma. Ann. Surg. 269, 966–971. https://doi.org/10.1097/SLA.0000000000002710 (2019).
Sanabria, A., Rojas, A., Arevalo, J., Kowalski, L. P. & Nixon, I. Microscopically positive surgical margins and local recurrence in thyroid cancer. A meta-analysis. Eur. J. Surg. Oncol. 45, 1310–1316. https://doi.org/10.1016/j.ejso.2019.02.007 (2019).
Esteva, D. et al. Clinical and pathological factors related to 18F-FDG-PET positivity in the diagnosis of recurrence and/or metastasis in patients with differentiated thyroid cancer. Ann. Surg. Oncol. 16, 2006–2013. https://doi.org/10.1245/s10434-009-0483-8 (2009).
Seo, J. H., Lee, S. W., Ahn, B. C. & Lee, J. Recurrence detection in differentiated thyroid cancer patients with elevated serum level of antithyroglobulin antibody: Special emphasis on using (18)F-FDG PET/CT. Clin. Endocrinol. (Oxf.) 72, 558–563. https://doi.org/10.1111/j.1365-2265.2009.03693.x (2010).
Team RDC. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2012).
Funding
This study was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI, Grant Number 21K15763.
Author information
Authors and Affiliations
Contributions
Y.I. and T.K. contributed to study conception and design. Clinical data collection and analysis were performed by Y.I., T.K., K.O., M.I., R.N., and K.N. T.M. supervised this study. The first draft of this manuscript was written by Y.I. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
RN and TM received research funding and TM received a scholarship from PDRadiopharma Inc. The other authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Iizuka, Y., Katagiri, T., Ogura, K. et al. Recurrence-free survival and prognosis after adjuvant therapy with radioactive iodine-131 in patients with differentiated thyroid carcinoma. Sci Rep 13, 10795 (2023). https://doi.org/10.1038/s41598-023-37899-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37899-z
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.