Abstract
Low birth weight predisposes to the development of hypertension in middle- and high-income countries. We examined the relation of early life length-for-age score (Z-score) on cardiovascular function in young adults in Malawi, a low-income country. Capture of supine, seated, and standing brachial pulse waveforms (Mobil-O-Graph) were performed in 223 females and 152 males (mean age 21 years), and analyzed according to the length-for-age Z-score tertiles during the first month of life. Plasma LDL cholesterol in young adulthood was slightly lower in the lowest versus highest tertile. Otherwise, blood hemoglobin and plasma chemistry were similar in all tertiles. Irrespective of posture, blood pressure, forward and backward wave amplitudes, and pulse wave velocity were corresponding in all tertiles. In the three postures, the lowest tertile presented with 4.5% lower systemic vascular resistance than the highest tertile (p = 0.005), and 4.4% and 5.5% higher cardiac output than the middle and highest tertiles, respectively (p < 0.01). Left cardiac work was 6.8% and 6.9% higher in the lowest tertile than in the middle and highest tertiles, respectively (p < 0.01). To conclude, in a low-income environment, low length-for-age Z-score after birth predicted hyperdynamic circulation at 21 years of age without changes in blood pressure and metabolic variables.
Similar content being viewed by others
Introduction
People living in low-income countries have on average better overall cardiovascular risk profile than people in high-income countries, but the incidence and mortality from cardiovascular diseases are higher in low-income countries1. This may be attributed to nutritional factors, higher burden of infections, lower level of education, and suboptimal health care services1,2. Underweight and growth stunting are common problems among children in low-income African countries2. According to the United Nations Children’s Fund (UNICEF) data from Malawi in 2019–2020, 13% of the newborn babies had low birth weight3.
Fetal undernutrition can have a permanent influence on tissue structure and physiology4. Early life undernutrition may induce changes in gut microbiota that predispose to growth faltering, as the transfer of microbiota from undernourished Malawian children into germ-free mice resulted in transmission of the impaired growth phenotype to the experimental animals5. Infancy is a critical developmental period, during which severe malnutrition may influence metabolic capacity in the long term and confer elevated susceptibility to non-communicable diseases6. Growth restriction can affect children’s cognitive and motor development7, but has also potential influences on the cardiovascular system4,8. In Malawi, 36% of all children under 5 years of age present with growth stunting3, while the prevalence of wasting (low weight-for-height) is 3%3, which is still within the acceptable range (< 5%) according to the WHO criteria2.
According to the Barker hypothesis, early life undernutrition predisposes to disturbances of glucose and lipid metabolism, obesity, coronary heart disease, and high blood pressure (BP) that emerge in adulthood8. Such pathophysiological changes can take place despite adequate catch-up growth during later childhood when energy-rich nutrition is available8. In 272 Malawian hospitalized children aged 0.5–5 years, malnutrition was associated with lower BP and systemic vascular resistance, whilst increased cardiac output was observed in the most severely wasted children9.
Currently, oscillometric recordings of BP and pulse waveforms from the brachial artery allow for the derivation of various hemodynamic variables10,11,12,13,14,15,16. These variables encompass aortic BP10,17, forward and backward wave amplitudes10,12, cardiac output, and total peripheral resistance14,18,19. Moreover, devices like the Mobil-O-Graph can estimate pulse wave velocity (PWV) by comparing the time delay between the pulse waveforms evaluated at the aortic and brachial sites15,20. Assessment of cardiac output18,19 and wave reflections11,12 using this method have provided values of acceptable accuracy. Overall, this approach provides a reasonable compromise between accuracy and ease of use.
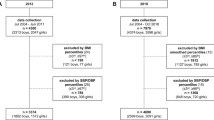
Limited information exists from low-income countries regarding the impact of early-life growth on cardiovascular function in adulthood. From 1995 to 1996, a total of 795 pregnant women attending the Lungwena health center antenatal clinic in Malawi were enrolled in a longitudinal study to track their descendants21. Previous reports have been published on the follow-up22,23,24,25,26, and it is still ongoing. In this cohort, the prevalence of underweight, growth stunting, and wasting reached its highest point between 3 and 21 months of age within the first three years of life23,27. We conducted a study on 375 individuals from this cohort, recording their brachial BPs and oscillometric pulse waveforms, to investigate whether early-life body size influences their hemodynamic variables at the age of 21 years.
Methods
The present cohort has been followed prospectively since their birth in Lungwena, a rural area in Southern Malawi. The original participants were 795 pregnant mothers visiting the Lungwena health center between June 1995 and August 1996. The distance to the nearest town, Mangochi, was 30–40 km, and no commercially manufactured food products were available at local shops. The livelihoods of the participants were dependent on rain-fed small-scale agriculture, fishing, and petty trading. Based on 24-h dietary recall, maize provided two-thirds (63%) of the dietary energy in Lungwena, while other energy sources were roots and tubers (11%), fish (5%), fruit (4%), legumes (4%) and vegetables (3%)22. The number of children born alive was 76726, and they were recruited to the follow-up. By the age of 15 years, 179 children from the cohort had deceased25.
When the subjects were ~ 21 years of age, they were invited to participate in brachial pulse waveform analyses. Successful recordings were performed on 419 from in total 429 participants27. Information about weight and length at birth and at one month was available from 375 participants: 223 females (age range 20.4–22.3 years) and 152 males (age range 20.4–22.2 years). The average values of body size during these two measurements were used in the analyses. The recruitment, data collection, and follow-up have been described previously26. Length and height values were converted to Z-scores using the WHO Multi-Center Growth Reference Study for children data28.
Ethical approval for the original Lungwena Child Survival Study (LCSS) was obtained from the National Health Science Research Committee in Malawi (HSRC 93⁄94), while approval for the present follow-up study was obtained from the College of Medicine Research and Ethics Committee (COMREC) on the 31st of October 2016. The study complies with the Declaration of Helsinki. Informed consent was acquired verbally from all caregivers of the children in the beginning of the study21. Later, informed consent was obtained again from the guardians and the children before the study visits until the age of 15 years25,26, and from the present participants when they were ~ 21 years of age. The consents were acquired by a person living in Lungwena and speaking the local dialect, and the participants either signed the consent or confirmed the document with their fingerprint image.
Laboratory analyses
Blood hemoglobin concentration was determined using HemoCue (Angelholm, Sweden). Malaria was screened using SD Bioline Malaria Ag Pf test (Standard Diagnostic Inc., South Korea) specific to Plasmodium falciparum (sensitivity 99.7%, specificity 99.5%). Plasma was separated and stored at − 70 °C until cold-shipped to Finland. The laboratory analyses were performed at the Medical Research Unit, Seinäjoki Central Hospital, Seinäjoki, Finland (SFS-EN ISO/IEC 15189:2013 accredited unit). Plasma C-reactive protein, glucose; total, high-density lipoprotein (HDL), low-density lipoprotein (LDL) cholesterol; triglycerides, sodium, potassium, and creatinine were determined using Cobas c702 or Cobas c702 ISE photometric assay tests (F. Hoffmann-Laroche Ltd, Basel; Switzerland). Glomerular filtration rate (eGFR) was estimated using a creatinine-based formula29.
Blood pressure and analyses of brachial pulse waveforms
An automated oscillometric device (Mobil-O-Graph, I.E.M., Stolberg, Germany) was applied to measure brachial BP and perform waveform-derived evaluation of aortic BP, wave reflections, PWV, heart rate, stroke volume, cardiac output, and systemic vascular resistance27. The last three values were normalized to body surface area and expressed as stroke index, cardiac index, and systemic vascular resistance index (SVRI). The formula 0.01439 × (mean aortic pressure − pulmonary artery occlusion pressure) × cardiac index was applied to calculate left cardiac work index (LCWI)30,31,32. Normal pulmonary artery occlusion pressure was assumed (6 mmHg), while 0.0143 is the conversion factor of pressure from mmHg to centimeters of water, volume to density of blood (in kg/L), and centimeters to meters. The brachial cuffs were chosen according to the upper arm circumference. The Mobil-O-Graph has been validated for measurement of peripheral and central BP16,33 and 24-h ambulatory BP17,34. Moreover, the evaluated PWV has been compared with tonometrically and invasively acquired values with acceptable accuracy35. The recordings were performed by the Lungwena research team that had ~ 20 years of experience in child health-related research.
Experimental protocol
As posture can individually influence BP, and especially diastolic BP increases when changing from the supine to the seated or standing positions27,36,37,38, the present Mobil-O-Graph recordings were performed in supine, seated, and standing postures in a quiet room. (1) The brachial BP cuff was attached, and after ~ 5 min in the seated position, two measurements were taken. (2) The subjects rested supine for ~ 5 min before the third recording was performed. (3) The subjects stood up and the fourth and the fifth measurements were performed after ~ 5 min of standing. We have previously found that the hemodynamic variables remain quite stable after about two minutes in either supine or upright posture31,32,36. The supine-to-upright change in body position complied with the protocol to examine orthostatic BP responses39. The figures present the results in the order supine-seated-standing.
Statistics
The participants were divided to sex-specific tertiles according to the length-for-age Z-score during the first month of life. The characteristics between the tertiles were analyzed using analysis of variance (ANOVA) followed by t-test for normally distributed variables, and Kruskal–Wallis test followed by Mann–Whitney U-test for variables with skewed distribution. Tertile proportions were compared using Chi-square test. The homogeneity of variances was tested with the Levene’s test, the Bonferroni correction was applied for all post-hoc analyses.
Hemodynamic differences between the tertiles in the supine, seated and standing postures were examined using generalized estimating equations (GEE) to examine the influences of tertile membership, posture, and their interaction on the variable of interest. The linear scale response was selected, and the autoregressive option was chosen, as the serial measures of hemodynamic variables are autocorrelated. As the tertiles presented with differences in gestation length and plasma concentration of LDL cholesterol (Table 1), the analyses were adjusted for these differences by including the variables as covariates. The PWV analyses were additionally adjusted for mean aortic pressure40,41.
The results were presented as mean and standard deviation (SD) or as median [25th–75th percentile] in the text and tables, and as mean and standard error of the mean (SEM) in the figures, and p < 0.05 was considered statistically significant. SPSS version 26.0 (IBM SPSS Statistics, Armonk, NY, USA) was used.
Results
Study group
The demographic and anthropometric data according to the tertiles of the length-for-age Z-score are presented in Table 1. During the first month of life body length (p < 0.001) and weight-for-age Z-score (p < 0.001) were lowest in the lowest tertile, and lower in the middle tertile than in the highest tertile (p < 0.001 for both). The proportion of females ranged from 57.6 to 62.1% without significant difference between the tertiles (p = 0.989). At 21 years of age, mean seated brachial BP was similar in all tertiles: 121, 119, and 121 mmHg for systolic BP (p = 0.456), and 76, 74, and 75 mmHg for diastolic BP (p = 0.298), in the lowest, middle, and highest tertiles, respectively. However, small differences in height and weight persisted (p < 0.05 for all between-tertile comparisons), while body mass indexes were not statistically significantly different (p = 0.054) in the tertiles.
Laboratory data
Hemoglobin, prevalence of Plasmodium falciparum positivity in the rapid test, and estimated glomerular filtration did not differ between the tertiles (p > 0.435 for all) (Table 2). Plasma creatinine, sodium, potassium, urid acid, glucose, C-reactive protein, alanine aminotransferase, alkaline phosphatase, total and HDL cholesterol, and triglyceride concentrations did not differ between the tertiles, either (p > 0.053 for all). LDL cholesterol was 0.18 mmol/l higher in the highest versus the lowest tertile (p = 0.034) (Table 2).
Hemodynamic results
Oscillometric brachial pulse waveforms were recorded in supine, seated, and standing postures (Figs. 1, 2). No differences between the tertiles were observed in brachial and aortic systolic and diastolic BP (Fig. 1A–D), forward wave amplitude (Fig. 1E), backward wave amplitude (Fig. 1F), and augmentation index (p = 0.442, data not shown). All these variables changed significantly in response to change in posture, while no significant interactions between the tertiles and posture were found (Fig. 1A–F).
Line graphs show brachial systolic (A) and diastolic (B), and aortic systolic (C) and diastolic (D) blood pressure, and forward wave amplitude (E) and backward wave amplitude (F) according to the tertiles of the length-for-age Z-score during first month of life during supine, seated and standing positions; lowest tertile n = 125 when supine and seated, n = 123 when standing; middle tertile n = 124 when supine and seated, n = 122 when standing; highest tertile n = 126 when supine and seated, n = 125 when standing; mean ± standard error of the mean. Statistics by generalized estimating equations with adjustments for plasma concentration of LDL cholesterol; p values denote differences between tertiles.
Line graphs show pulse wave velocity (A), heart rate (B), stroke index (C), cardiac index (D), left cardiac work index (E), and systemic vascular resistance index (SVRI) (F), division to tertiles and statistics as in Figure, pulse wave velocity was additionally adjusted for mean aortic blood pressure.
PWV (Fig. 2A) and heart rate (Fig. 2B) did not differ between the tertiles, while both variables changed in response to posture without significant interactions between the tertiles. Stroke index progressively decreased from supine to seated, and from seated to standing postures, while no significant tertile-posture interactions were observed. However, the lowest tertile presented with higher stroke index than the middle tertile, the average difference though postures being 2.7 ml/min/m2 (p = 0.014) (Fig. 2C). Cardiac index and LCWI also decreased from supine to seated, and from seated to standing positions in all tertiles without significant tertile-posture interactions (Fig. 2D,E). On average, cardiac index was 0.15 l/min/m2 higher in the lowest than in the middle tertile (p = 0.002), and 0.20 l/min/m2 higher in the lowest than in the highest tertile (p < 0.001). Mean LCWI was 0.26 kg × m/m2 higher in the lowest than in the middle tertile (p = 0.003), and 0.29 kg × m/m2 higher in the lowest than in the highest tertile (p = 0.001). SVRI gradually increased from the supine to the seated and standing postures in all tertiles without significant tertile-posture interactions. Average SVRI was 0.10 s × mmHg/ml/m2 lower (p = 0.002) in the lowest tertile than in the highest tertile (Fig. 2F).
To eliminate the influence of differences in body size on the cardiac variables and vascular resistance, the above results were adjusted for body surface area. The following comparison emphasizes the significance of this adjustment: The present young women were smaller than men, and the respective body surface areas were 1.50 (0.12) m2 vs. 1.63 (0.11) m2 (p < 0.001). Estimated cardiac output was lower in women than in men 4.69 (0.45) vs. 5.12 (0.55) l/min (p < 0.001). However, when related to body surface area the cardiac index values were identical between women and men: 3.14 (0.35) vs. 3.14 (0.34) l/min/m2, respectively.
Discussion
To examine the influence of early life size on cardiovascular function in young adults from a low-income country, the present participants from rural Malawi were divided to tertiles according to the length-for-age Z-score during their first month of life. Small early-life body size was reflected in the hemodynamics of these subjects at 21 years of age, as in the lowest tertile cardiac index and cardiac workload irrespective of posture, as evaluated by LCWI, were higher than in the other two tertiles. There were no differences in brachial or central BP between the tertiles, probably due to the lower SVRI in the lowest tertile that reached statistical significance when compared with the highest tertile.
Previously, low birth weight (n = 20) was associated with higher risk of elevated BP among 77 Swedish men who were examined at the age of 28 years (odds ratio 3.63), and the authors concluded that being born small for gestational age may be a predictor for raised BP in adult life42. In 4–18-year-old children and adolescents in Spain (n = 630), the participants with lowest birth weights (n = 35) had highest 24-h ambulatory BP values in the absence of differences in heart rate43. However, contradictory findings have also been reported, showing no significant differences in laboratory BP in children and teenagers aged 7–18 years (n = 219), despite differences in birthweight44. In the present study we did not observe differences in brachial or aortic BP between the tertiles of the length-for-age Z-score in the supine, seated or standing postures. BP assessment in three postures was included in the measurements, as exaggerated systolic BP response to standing was recently found to be an independent predictor of adverse cardiovascular events45. Moreover, we included aortic BP in the analyses, as central BP is considered to be a more reliable predictor of cardiovascular risk than brachial BP46,47.
In the absence of differences in BP, the lowest tertile in this study presented with lower SVRI than the highest tertile, and higher cardiac index than the two other tertiles, while the laboratory analyses did not show evidence of unfavorable harmful changes in the metabolic profiles. Thus, young Malawian adults with small stature at birth had hyperdynamic circulation at the age of 21. This was also translated to higher LCWI, which cannot be regarded as a favorable hemodynamic finding. The long-term influences of increased cardiac workload during normotension remain rather unknown. Hyperdynamic circulation may be an early feature of essential hypertension, which predisposes to the development of established hypertension and progression of vascular disease48. In the Bogalusa Heart Study, hyperdynamic circulation, as assessed using pulse pressure and heart rate measurements, was associated with adverse cardiovascular risk profile among children aged 8 to 17 years49. Low systemic vascular resistance and high cardiac output are also detected in patients with high-output cardiac failure, who have high metabolic rate, while the chronic increases in cardiac output in these patients eventually lead to a progressive decline in cardiac function50. Of note, higher left cardiac work in the upright posture, but not in the supine position, has been previously reported in both Malawian and Finnish men when compared with women27,32.
Backward wave amplitude and augmentation index reflect the influence of wave reflection on central pulse pressure51. Wave reflection is influenced by arterial stiffness, heart rate, stroke volume, and systemic vascular resistance51,52. Arterial stiffness increases with ageing, leading to an earlier return of the reflected waves which increases the central augmentation of pulse pressure51. The PWV values evaluated by the Mobil-O-Graph did not differ between the study tertiles and the values were also within the normal limits of a large European reference population53. The amplitude of the reflected wave is associated with hypertensive end organ damage and it may also predict future cardiovascular events54. Among 7–18 year-old Spanish children and teenagers (n = 219) highest augmentation index values were recorded from subjects born with low birth weight44. However, in the present study backward wave amplitude and augmentation index at 21 years did not differ between the tertiles of the length-for-age Z-score during the first month of life.
Malnutrition, inflammation, infections, low-income, and low socio-economic status are factors that may influence growth and cardiovascular function1,55,56. Babies in Malawi have been reported to be born about 2.5 cm shorter with about 500 g lower body weight than babies in a high-income reference population23,24. Among the pregnant mothers of the present study subjects the seroprevalence of human immunodeficiency virus (HIV) infection was high (18%)57. Moreover, impaired linear growth, high incidence of growth stunting (~ 70%) and underweight were observed among the study cohort that peaked at 3–21 months of age23,24. Morbidity in infancy, small size after birth, maternal HIV infection, and home delivery were the strongest predictors of severe underweight in these subjects23. By the age of 15 years, 179 subjects of the present cohort had already been deceased25. The Mangochi area is characterized by high malaria transmission due to high temperatures and frequent rainfalls from October to April58. Reflecting this endemic situation, 17–18% of the present young men and women were tested positive for Falciparum malaria58. However, the mean results of the laboratory tests reflecting inflammation, electrolyte balance, glucose metabolism, renal function, and lipid profiles were well within the normal range in all study tertiles. Also, low length-for-age and weight-for-age Z-scores during the first month of life were not related with adverse metabolic changes at young adulthood. Altogether, the present results suggest that in the absence of energy-rich nutrition, the Barker hypothesis may not apply to young adults in a low-income country.
We evaluated cardiovascular function by the use of an oscillometric device that has been validated for the measurement of peripheral and central BP16,34. Previously, successful comparisons between the evaluated PWV values and tonometrically and invasively obtained values have been reported16,34,35. However, criticism concerning the reliability of the PWV measurement by Mobil-O-Graph has been presented59, and therefore the results must be interpreted with caution. A further limitation is that the cardiac output measurements in 24 hospitalized patients using Mobil-O-Graph presented with slightly lower values than the thermodilution method18. However, the values were reproducible and the accuracy of the device was considered to be acceptable18. The cardiac output measurement also corresponded well to values measured using transthoracic echocardiography14. In the present analyses comparing the study tertiles, cardiac output, left cardiac work, and systemic vascular resistance were adjusted for body surface area. This approach is a strength of our study, as it eliminates the deviations caused by differences in body size among the participants.
In conclusion, young Malawian adults with low length-for-age and weight-for-age Z-scores after birth presented with hyperdynamic circulation and increased left ventricular workload in the absence of changes in BP or metabolic profiles at the age of 21 years.
Data availability
Access to the deidentified data of this study can be provided upon reasonable request to the corresponding author from qualified researchers who are adequately experienced in human subject confidentiality protocols.
References
Rosengren, A. et al. Socioeconomic status and risk of cardiovascular disease in 20 low-income, middle-income, and high-income countries: The prospective urban rural epidemiologic (PURE) study. Lancet Glob. Health 7, e748–e760 (2019).
World Health Organization. Nutrition in the WHO African Region. WHO|Regional Office for Africa. www.afro.who.int/publications/nutrition-who-african-region (2017).
United Nations Children’s Fund (UNICEF). Malawi Multiple Indicator Cluster Survey Report 2019–2020. UNICEF|Malawi MICS Snapshots of Key Findings www.unicef.org/malawi/reports/malawi-multiple-indicator-cluster-survey-report (2021).
Barker, D. J. Fetal origins of coronary heart disease. BMJ 311, 171–174 (1995).
Blanton, L. V. et al. Gut bacteria that prevent growth impairments transmitted by microbiota from malnourished children. Science 351, 3311 (2016).
Wells, J. C. K. The capacity-load model of non-communicable disease risk: Understanding the effects of child malnutrition, ethnicity and the social determinants of health. Eur. J. Clin. Nutr. 72, 688–697 (2018).
Sudfeld, C. R. et al. Linear growth and child development in low- and middle-income countries: A meta-analysis. Pediatrics 135, e1266–e1275 (2015).
Hales, C. N. & Barker, D. J. P. The thrifty phenotype hypothesis. Br. Med. Bull. 60, 5–20 (2001).
Silverman, J. A. et al. The effects of malnutrition on cardiac function in African children. Arch. Dis. Child. 101, 166–171 (2016).
Papaioannou, T. G. et al. Non-invasive 24 hour ambulatory monitoring of aortic wave reflection and arterial stiffness by a novel oscillometric device: The first feasibility and reproducibility study. Int. J. Cardiol. 169, 57–61 (2013).
Hametner, B. et al. Assessment of model based (input) impedance, pulse wave velocity, and wave reflection in the asklepios cohort. PLoS ONE 10, e0141656 (2015).
Hametner, B. et al. Wave reflection quantification based on pressure waveforms alone—Methods, comparison, and clinical covariates. Comput. Methods Prog. Biomed. 109, 250–259 (2013).
Wassertheurer, S. et al. A new oscillometric method for pulse wave analysis: Comparison with a common tonometric method. J. Hum. Hypertens. 24, 498–504 (2010).
Bikia, V. et al. Noninvasive cardiac output and central systolic pressure from cuff-pressure and pulse wave velocity. IEEE J. Biomed. Health Inform. 24, 1968–1981 (2020).
Feistritzer, H.-J. et al. Comparison of an oscillometric method with cardiac magnetic resonance for the analysis of aortic pulse wave velocity. PLoS ONE 10, e0116862 (2015).
Weiss, W. et al. Oscillometric estimation of central blood pressure: Validation of the Mobil-O-Graph in comparison with the SphygmoCor device. Blood Press. Monit. 17, 128–131 (2012).
Franssen, P. M. L. & Imholz, B. P. M. Evaluation of the Mobil-O-Graph new generation ABPM device using the ESH criteria. Blood Press. Monit. 15, 229–231 (2010).
Papaioannou, T. G. et al. Accuracy and precision of cardiac output estimation by an automated, brachial cuff-based oscillometric device in patients with shock. Proc. Inst. Mech. Eng. H J. Eng. Med. 234, 1330–1336 (2020).
Godoy, A., Contreras, A. & Tabares, A. Agreement analysis of stroke volume and cardiac output measurement between a oscillometric device and transthoracic echocardiogram in normotensive individuals: A preliminary report. Braz. J. Anesthesiol. https://doi.org/10.1016/j.bjane.2021.09.006 (2021).
Luzardo, L. et al. 24-h ambulatory recording of aortic pulse wave velocity and central systolic augmentation: A feasibility study. Hypertens. Res. 35, 980–987 (2012).
Kulmala, T. et al. Socio-economic support for good health in rural Malawi. East Afr. Med. J. 77, 168–171 (2000).
Ndekha, M. et al. Seasonal variation in the dietary sources of energy for pregnant women in Lungwena, rural Malawi. Ecol. Food Nutr. 38, 605–622 (2000).
Maleta, K., Virtanen, S. M., Espo, M., Kulmala, T. & Ashorn, P. Childhood malnutrition and its predictors in rural Malawi. Paediatr. Perinat. Epidemiol. 17, 384–390 (2003).
Maleta, K., Virtanen, S., Espo, M., Kulmala, T. & Ashorn, P. Timing of growth faltering in rural Malawi. Arch. Dis. Child. 88, 574–578 (2003).
Teivaanmäki, T., Cheung, Y. B., Kortekangas, E., Maleta, K. & Ashorn, P. Transition between stunted and nonstunted status: Both occur from birth to 15 years of age in Malawi children. Acta Paediatr. 104, 1278–1285 (2015).
Teivaanmäki, T. et al. Height gain after two-years-of-age is associated with better cognitive capacity, measured with Raven’s coloured matrices at 15-years-of-age in Malawi: Height gain is associated with cognitive capacity. Matern. Child Nutr. 13, e12326 (2017).
Choudhary, M. K. et al. Posture-related differences in cardiovascular function between young men and women: Study of noninvasive hemodynamics in rural Malawi. JAHA 11, e022979 (2022).
World Health Organization. WHO Child Growth Standards: Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-Age: Methods and Development. www.who.int/publications-detail-redirect/924154693X (2006).
Inker, L. A. et al. Estimating glomerular filtration rate from serum creatinine and cystatin C. N. Engl. J. Med. 367, 20–29 (2012).
Gorlin, R., McMillan, I. K. R., Medd, W. E., Matthews, M. B. & Daley, R. Dynamics of the circulation in aortic valvular disease. Am. J. Med. 18, 855–870 (1955).
Kangas, P. et al. Changes in hemodynamics associated with metabolic syndrome are more pronounced in women than in men. Sci. Rep. 9, 1–11 (2019).
Kangas, P. et al. Increased cardiac workload in the upright posture in men: Noninvasive hemodynamics in men versus women. J. Am. Heart Assoc. 5, e002883 (2016).
Weber, T. et al. Validation of a brachial cuff-based method for estimating central systolic blood pressure. Hypertension 58, 825–832 (2011).
Wei, W., Tölle, M., Zidek, W. & van der Giet, M. Validation of the mobil-O-Graph: 24 h-blood pressure measurement device. Blood Press. Monit. 15, 225–228 (2010).
Hametner, B. et al. Oscillometric estimation of aortic pulse wave velocity: Comparison with intra-aortic catheter measurements. Blood Press. Monit. 18, 173–176 (2013).
Suojanen, L. J. et al. Individual changes of central blood pressure in response to upright posture: Different hemodynamic phenotypes. J. Hypertens. 39, 2403–2412 (2021).
Beevers, G., Lip, G. Y. H. & O’Brien, E. Blood pressure measurement: Part I—Sphygmomanometry: Factors common to all techniques. BMJ 322, 981–985 (2001).
Netea, R. T., Smits, P., Lenders, J. W. & Thien, T. Does it matter whether blood pressure measurements are taken with subjects sitting or supine?. J. Hypertens. 16, 263–268 (1998).
CDC. Section ‘Functional-Assessments’ on the Web-Page Clinical Resources | STEADI | CDC Injury Center 2021. www.cdc.gov/steadi/materials.html.
Laurent, S. et al. Expert consensus document on arterial stiffness: Methodological issues and clinical applications. Eur. Heart J. 27, 2588–2605 (2006).
Townsend, R. R. et al. Recommendations for improving and standardizing vascular research on arterial stiffness: A scientific statement from the American Heart Association. Hypertension 66, 698–722 (2015).
Gennser, G., Rymark, P. & Isberg, P. E. Low birth weight and risk of high blood pressure in adulthood. BMJ 296, 1498–1500 (1988).
Lurbe, E., Torro, I., Rodríguez, C., Alvarez, V. & Redón, J. Birth weight influences blood pressure values and variability in children and adolescents. Hypertension 38, 389–393 (2001).
Lurbe, E., Torro, M. I., Carvajal, E., Alvarez, V. & Redón, J. Birth weight impacts on wave reflections in children and adolescents. Hypertension 41, 646–650 (2003).
Palatini, P., Mos, L., Saladini, F. & Rattazzi, M. Blood pressure hyperreactivity to standing: A predictor of adverse outcome in young hypertensive patients. Hypertension 79, 984–992 (2022).
Roman, M. J. & Devereux, R. B. Association of central and peripheral blood pressures with intermediate cardiovascular phenotypes. Hypertension 63, 1148–1153 (2014).
Vlachopoulos, C. et al. Prediction of cardiovascular events and all-cause mortality with central haemodynamics: A systematic review and meta-analysis. Eur. Heart J. 31, 1865–1871 (2010).
Frohlich, E. D. Hyperdynamic circulation and hypertension. Postgrad. Med. 52, 68–73 (1972).
Jiang, X., Srinivasan, S. R., Urbina, E. & Berenson, G. S. Hyperdynamic circulation and cardiovascular risk in children and adolescents. Circulation 91, 1101–1106 (1995).
Reddy, Y. N. V., Melenovsky, V., Redfield, M. M., Nishimura, R. A. & Borlaug, B. A. High-output heart failure: A 15-year experience. J. Am. Coll. Cardiol. 68, 473–482 (2016).
O’Rourke, M. F. & Hashimoto, J. Mechanical factors in arterial aging: A clinical perspective. J. Am. Coll. Cardiol. 50, 1–13 (2007).
Wilenius, M. et al. Central wave reflection is associated with peripheral arterial resistance in addition to arterial stiffness in subjects without antihypertensive medication. BMC Cardiovasc. Disord. 16, 131 (2016).
Arterial Stiffness’ Collaboration. Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: Establishing normal and reference values. Eur. Heart J. 31, 2338–2350 (2010).
Weber, T. et al. Wave reflections, assessed with a novel method for pulse wave separation, are associated with end-organ damage and clinical outcomes. Hypertension 60, 534–541 (2012).
Laslett, L. J. et al. The worldwide environment of cardiovascular disease: Prevalence, diagnosis, therapy, and policy issues. J. Am. Coll. Cardiol. 60, S1–S49 (2012).
Havranek, E. P. et al. Social determinants of risk and outcomes for cardiovascular disease: A scientific statement from the American Heart Association. Circulation 132, 873–898 (2015).
Kulmala, T. et al. The importance of preterm births for peri- and neonatal mortality in rural Malawi. Paediatr. Perinat. Epidemiol. 14, 219–226 (2000).
USAID-DHF-Program. Malawi Malaria Indicator Survey 2017. www.dhsprogram.com/publications/publication-MIS28-MIS-Final-Reports.cfm.
Schwartz, J. E., Feig, P. U. & Izzo, J. L. Pulse wave velocities derived from cuff ambulatory pulse wave analysis: Effects of age and systolic blood pressure. Hypertension 74, 111–116 (2019).
Acknowledgements
The authors are deeply grateful to the study participants, the Lungwena team for data collection, and Paula Liikanen, research nurse, for invaluable technical assistance in setting up the recording system on-location in Lungwena, Malawi.
Funding
This work was financially supported by grants from the Foundation for Pediatric Research, Academy of Finland, Tampere University, and Nutriset S.A.S., Malaunay, France to PA and UA, Competitive State Research Financing of the Expert Responsibility Area of Tampere University Hospital to IP (ref. 9AA062, 9AB057), Finnish Foundation for Cardiovascular Research to IP, and Sigrid Jusélius Foundation to IP.
Author information
Authors and Affiliations
Contributions
R.-M.P.: Formal Analysis, Visualization, Writing—Original draft. M.K.C.: Formal analysis, Methodology. T.T.: Formal analysis, Methodology. C.M.: Investigation. K.M.: Investigation. O.N.: Methodology, Validation. P.A.: Funding Acquisition, Conceptualization, Resources, Project Administration. U.A.: Funding Acquisition, Conceptualization, Resources, Project Administration. I.P.: Methodology, Formal analysis, Writing—original draft, Funding Acquisition. All authors: Writing—Review & Editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Penninkangas, RM., Choudhary, M.K., Mangani, C. et al. Low length-for-age Z-score within 1 month after birth predicts hyperdynamic circulation at the age of 21 years in rural Malawi. Sci Rep 13, 10283 (2023). https://doi.org/10.1038/s41598-023-37269-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37269-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.