Abstract
Thoracic sympathetic nerve block (TSNB) has been widely used in the treatment of neuropathic pain. To reduce block failure rates, TSNB is assisted with several modalities including fluoroscopy, computed tomography, and ultrasonography. The present study describes our experience assessing the usefulness of thoracoscopy in TSNB for predicting compensatory hyperhidrosis before sympathectomy in primary hyperhidrosis. From September 2013 to October 2021, TSNB was performed under local anesthesia using a 2-mm thoracoscope in 302 patients with severe primary hyperhidrosis. Among the 302 patients, 294 were included for analysis. The target level of TSNB was T3 in almost all patients. The mean procedure time was 21 min. Following TSNB, the mean temperature of the left and right palms significantly changed from 31.5 to 35.3 °C and from 31.5 to 34.8 °C, respectively. With TSNB, primary hyperhidrosis was relieved in all patients. Pneumothorax occurred in six patients, in which no chest tube insertion was required. One patient developed hemothorax and was discharged the next day after small-bore catheter drainage. Transient ptosis developed in 10 patients and improved within a day in all patients. Our experiences showed that thoracoscopic TSNB is accurate, safe, and feasible to block the thoracic sympathetic nerve in patients with severe primary hyperhidrosis.
Similar content being viewed by others
Introduction
Thoracoscopic sympathectomy is the most effective and definitive treatment for severe primary hyperhidrosis when conservative treatments have failed1,2,3. However, patients are hesitant to undergo thoracoscopic sympathectomy due to the possible occurrence of postoperative compensatory hyperhidrosis, which happens in 50–90% of patients, and is particularly likely in patients with craniofacial hyperhidrosis4,5. Since there is no effective treatment for postoperative compensatory hyperhidrosis after definite sympathectomy, many authors have proposed several strategies including limited level sympathectomy6, lower level sympathectomy7,8, sympathetic nerve clipping9 and temporary thoracoscopic sympathetic block10,11.
Thoracic sympathetic nerve block (TSNB) can be done either in an ultrasound (US)-guided manner12, in a computed tomography (CT)-guided manner13, or thoracoscopically10. The US-guided and CT-guided approaches are less invasive than the thoracoscopic approach. However, these approaches appear to be less accurate than the thoracoscopic approach, which allows the surgeons to visualize a targeted sympathetic nerve in their own eyes. In 2009, we started conducting thoracoscopic TSNB under local anesthesia with a 2-mm single incision not only to reduce invasiveness but also to enhance our accurateness during the surgical procedure11.
The purpose of this study is to share our surgical experience with TSNB, and to examine the accuracy, safety and feasibility of this technique.
Results
Among 302 patients with severe primary hyperhidrosis who underwent TSNB using thoracoscopy, 294 patients met the study criteria. Eight patients including seven patients with compensatory hyperhidrosis and one patient with multi-level sympathetic nerve blocks were excluded from this study. The mean age of the remaining 294 patients, including 182 males and 112 females, was 29 years (Table 1). The diagnoses were primary palmar hyperhidrosis in 258 patients (87.8%) and craniofacial hyperhidrosis in 36 patients (12.2%).
TSNBs were done at the T3 level in almost all patients. Only left or right ipsilateral TSNB was performed in 12 patients due to intraoperative pain (eight patients) or pleural adhesions (four patients). The mean procedure times of TSNB were 21 ± 8 min. The mean temperatures of the left and right palms following TSNB significantly changed from 31.5 to 35.3 °C (P < 0.0005) and from 31.5 to 34.8 °C (P < 0.0005), respectively (Table 2, Fig. 1). All patients maintained oxygen saturation levels above 90%, and there was no need for intubation during TSNB. Either palmar or craniofacial symptoms was relieved in all patients. During TSNB, there were no severe complications, such as injury of the intrathoracic organs including lung and vessels. After TSNB, pneumothorax was found in six patients (2.0%), and all the patients improved without intervention and were discharged postoperatively after one day. Hemothorax was found after TSNB with routine chest X-ray in one patient (0.3%), who was discharged the next day after the insertion of a small-bore catheter (14 G). Temporary ptosis occurred in 10 patients (3.4%) and they all improved with one day of observation.
Box plot of the temperature of the palm before and after TSNB (thoracic sympathetic nerve block). The horizontal line indicates the median, the box is the interquartile range (IQR), and the whiskers extend to the upper adjacent value (large value = 75th percentile + 1.5 × IQR) and the lower adjacent value (smallest value = 25th percentile − 1.5 × IQR).
Discussion
TSNB has been widely used over the past decades as an anesthetic technique, not only to prevent persistent postoperative pain after thoracic surgery, but also to treat neuropathic pain disorders, such as complex regional pain syndrome, phantom limb pain, post-herpetic neuralgia, and ischemic vascular disease14,15,16,17. In recent studies, the role of TSNB has expanded to include different purposes other than pain practice. As an example, it can be applied to improve coronary microcirculation in rats with chronic heart failure. Sun and his colleagues reported that high thoracic sympathetic block improved myocardial capillary spasm and growth18. TSNB can also be applied as a predictive procedure for compensatory hyperhidrosis before sympathectomy in primary hyperhidrosis, based on the effect of sympathetic block on increasing skin perfusion and temperature through vasodilation at the anesthetized sites10,11,19,20. Miller et al.10 performed a temporary TSNB in 25 patients to predict whether compensatory hyperhidrosis would occur after sympathectomy in primary hyperhidrosis. All patients had temporary relief of primary hyperhidrosis after TSNB. Three patients had temporary compensatory hyperhidrosis after TSNB, and one of them experienced severe compensatory hyperhidrosis and did not proceed with sympathectomy. The remaining two patients experienced mild compensatory hyperhidrosis after TSNB and also had it after sympathectomy. Their results showed that temporary TSNB is a reversible and accurate procedure for determining compensatory hyperhidrosis after sympathectomy.
Over the past decades, there have been several approaches to blocking thoracic sympathetic nerve: the landmark-based approach, US-guided approach, CT-guided approach, fluoroscopically, and thoracoscopically10,11,12,13,21,22,23. A classic technique that does not use image guidance is a landmark-based approach that elicits a loss of resistance21. Other techniques include simple advancement over or under transverse process for 1 to 1.5 cm, using a nerve stimulator, pressure monitoring and X-ray direct vision24,25,26,27. Research examining a landmark-based approach TSNB found that the failure rate varies from 6.8 to 10%28,29,30. Like the landmark-based approach with the aid of X ray direct vision, TSNB can be conducted under imaging guidance, such as US-guidance, CT-guidance, and under fluoroscopy12,22,23. TSNB under fluoroscopy and the CT-guided approach results in inevitable radiation exposure and uncomfortable posture of the operator and patient during the procedures12. In addition, TSNB with the CT-guided approach is not practical in clinical practice. Under fluoroscopy, TSNB can be done by detecting bony surfaces as landmarks for introducing the needle for block; however, other structures such as vascular and soft tissue structures cannot be seen, which increase the risk of injuries of adjacent structures. Kim and his colleagues have reported that most patients (80%) achieved a temperature increase (≥ 1.5 °C) on the palm after TSNB under fluoroscopy at the T2 spinal level, which was superior to TSNB under the US-guided approach (20.0%)31. On the other hand, the US-guided approach is preferable over the previously mentioned approaches for checking the surrounding structures, including vascular and soft tissue structures, in detail to reduce complications32,33. A recent study by Kim and his colleagues reported that TSNB under the US-guided approach achieved a temperature increase (≥ 1.5 °C) between the ipsilateral and contralateral hands in 7 of 12 patients (58.3%), and minimized complications including pneumothorax12. It is true that the US-guided and CT-guided approaches are less invasive and that US-guided approach can be done at one’s bedside, while TSNB is done under thoracoscopy in the operating room.
From the perspective of accuracy and safety, thoracoscopic TSNB has the advantages of directly targeting the sympathetic nerve in our own eyes, leading to a success rate of approximately 100%10,11. Our results have shown that the mean temperature of the left and right palms following TSNB under thoracoscopy increased more than 1.5 °C in all patients. To further reduce the inconvenience to patients, we share here our experience of TSNB under local anesthesia with a 2-mm single incision not only to reduce invasiveness but also to enhance our accurateness during the surgical procedure. Even though we cannot directly compare our results with those of TSNB using other modalities, direct vision seems to be superior to imaging guidance. Our result has shown that pneumothorax occurred in 6 patients (n = 6/294). However, chest tube catheter drainage was not necessary, because pneumothorax was a result of not completely draining air from the chest cavity after TSNB. Only one patient developed hemothorax after TSNB, which was presumed to have occurred after removal of the trocar from the insertion site of the 2 mm surgical trocar. However, the patient was able to be discharged the next day after small-bore catheter drainage. Temporary ptosis occurred in 10 patients, which is probably caused by the block cranially extending into the sympathetic chain with the mixture injected at the T3 level. Our result has shown that TSNB under thoracoscopy is not inferior to the US-guided approach in terms of complications.
The advantages of thoracoscopic TSNB are the facts that the injection level is accurate, the injection is done under direct vision, and the procedure can be completed before the temperature change of the palm. On the other hand, thoracoscopic TSNB has several disadvantages. First, it is more invasive than the image-assisted procedure, as it requires a tiny skin incision. Second, if there is pleural adhesion, the procedure is difficult. Third, it is difficult for the patient to breathe during the procedure because it is performed under local anesthesia with the lung collapsed. Fourth, complications such as pneumothorax may occur after the procedure due to lung injury caused by thoracoscopic insertion. To predict the effect of sympathectomy for the treatment of hyperhidrosis and the occurrence of compensatory hyperhidrosis, it is recommended to show the same results after the predictive procedure. Therefore, considering the advantages and disadvantages of thoracoscopic TSNB, this modality was used considering accuracy more important than invasiveness to predict compensatory hyperhidrosis before sympathectomy in primary hyperhidrosis. However, if US-guided TSNB is not significantly different from thoracoscopic TSNB in terms of accuracy, it would be better to use US-guided manner.
The limitations of this study are the facts that it was a retrospective study, was conducted at a single institution, was not performed in patients with various diseases, and did not include a comparison with TSNB performed by other modalities. In conclusion, our experiences showed that TSNB using thoracoscopy was accurate, safe and feasible for blocking the thoracic sympathetic nerve in patients with severe primary hyperhidrosis. However, further studies are needed to compare these results with those of TSNB perfomed for other diseases and with the use of other modalities, US-guided or CT-guided manner, in the future.
Methods
From September 2013 to October 2021, 302 consecutive patients with severe primary hyperhidrosis underwent TSNB using thoracoscopy performed by the same surgeon. The patients were included in this study who had severe primary palmar and craniofacial hyperhidrosis. We excluded patients with compensatory hyperhidrosis or recurrent hyperhidrosis who had undergone previous treatments as well as patients who required multi-level sympathetic nerve blocks due to the presence of multiple symptom sites other than palmar or craniofacial.
Anesthesia
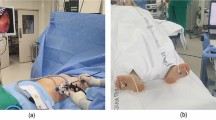
All patients underwent TSNB under local anesthesia in an operating room. Spontaneous respiration was maintained and supported by oxygen (3–5 L/min via mask), case by case, without endotracheal tube. Standard monitoring included electrocardiogram, non-invasive blood pressure, pulse oximetry, and respiratory rate. Temperature probes (Skin Temperature Probe D-S18A, Exacon Scientific, Roskild, Denmark) were applied on both palms.
Surgical technique: TSNB
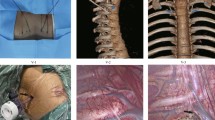
The patients were placed in a prone position, and a doughnut-shaped cushion was applied to their forehead. Both arms were abducted at 90 degrees and both elbows were flexed at less than 90 degrees. A single surgical incision that was approximately 2-mm long was made at the fifth intercostal space in the mid-axillary line after local anesthesia (0.5% lidocaine) was injected from the skin to the pleura. A surgical trocar (MiniPort 2 mm, Tyco Healthcare UK Ltd., Gosport, UK) was inserted. After the insufflation of CO2 into the pleural space, the 2-mm thoracoscope was positioned. With visualization by thoracoscope, a spinal needle (23 Gx89mm, Hakko Co. Ltd., Chikuma, Japan) was safely introduced onto the targeted sympathetic nerve immediately above the parietal pleura through the intercostal space from the patient’s back (Fig. 2A). We used about 10 mL of a mixture (ropivacaine 37.5 mg, dexamethasone 5 mg and 0.05 mL of 0.1% epinephrine solution) that we injected through the spinal needle around both target sympathetic chains and ganglia (Fig. 2B). This local anesthetic mixture was injected slowly, followed by repeated aspiration to avoid the intravascular administration through the spinal needle around both target sympathetic chains and ganglia. After confirming that the temperature of both hands started to rise, we drained out the air through the trocar while the patient was engaging in the Valsalva maneuver, and the trocar was removed. Patients were discharged home on the day of the operation, and they did not have any complications requiring chest tube insertion after the procedure.
Statistical analysis
We retrospectively analyzed the data of patients who underwent TSNB using thoracoscopy. Categorical variables are presented as counts and percentages. Continuous variables are presented as means with standard deviations, and they were analyzed using the dependent-sample t-test. Statistical significance was defined by P values < 0.05. All statistical analyses were performed using IBM SPSS ver. 27.0 (IBM Corp., Armonk, NY, USA),
Ethics approval
The study was approved by the Institutional Review Board of Incheon St. Mary’s Hospital, College of Medicine, the Catholic University of Korea (IRB approval number: OC22RISI0137). The study was performed in accordance with the Declaration of Helsinki. The requirement for informed consent was waived by the Institutional Review Board of Incheon St. Mary’s Hospital, College of Medicine, the Catholic University of Korea (IRB approval number: OC22RISI0137) due to the retrospective nature of the current study.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Tetteh, H. A. et al. Primary palmoplantar hyperhidrosis and thoracoscopic sympathectomy: A new objective assessment method. Ann Thorac Surg. 87(1), 267–74 (2009) (discussion 74-5).
Baumgartner, F. J., Bertin, S. & Konecny, J. Superiority of thoracoscopic sympathectomy over medical management for the palmoplantar subset of severe hyperhidrosis. Ann. Vasc. Surg. 23(1), 1–7 (2009).
Bryant, A. S. & Cerfolio, R. J. Satisfaction and compensatory hyperhidrosis rates 5 years and longer after video-assisted thoracoscopic sympathotomy for hyperhidrosis. J. Thorac. Cardiovasc. Surg. 147(4), 1160–3.e1 (2014).
Dumont, P., Denoyer, A. & Robin, P. Long-term results of thoracoscopic sympathectomy for hyperhidrosis. Ann. Thorac. Surg. 78(5), 1801–1807 (2004).
Moon, D. H., Kang, D. Y., Kim, D. W., Kang, M. K. & Lee, S. Early results of new endoscopic thoracic sympathectomy for craniofacial hyperhidrosis. J. Thorac. Dis. 10(6), 3627–3631 (2018).
Weksler, B., Blaine, G., Souza, Z. B. & Gavina, R. Transection of more than one sympathetic chain ganglion for hyperhidrosis increases the severity of compensatory hyperhidrosis and decreases patient satisfaction. J. Surg. Res. 156(1), 110–115 (2009).
Joo, S., Lee, G. D., Haam, S. & Lee, S. Comparisons of the clinical outcomes of thoracoscopic sympathetic surgery for palmar hyperhidrosis: R4 sympathicotomy versus R4 sympathetic clipping versus R3 sympathetic clipping. J. Thorac. Dis. 8(5), 934–941 (2016).
Soares, T. J., Dias, P. G. & Sampaio, S. M. Impact of video-assisted thoracoscopic sympathectomy and related complications on quality of life according to the level of sympathectomy. Ann. Vasc. Surg. 63, 63–7.e1 (2020).
Raveglia, F., Angeli, M. C. & Scarci, M. Anatomical clipping of sympathetic nerve to reduce compensatory sweating in primary hyperhidrosis: A novel technique. Shanghai Chest 3, 28 (2019).
Miller, D. L. & Force, S. D. Temporary thoracoscopic sympathetic block for hyperhidrosis. Ann. Thorac. Surg. 85(4), 1211–4 (2008) (discussion 5-6).
Lee, J. et al. Thoracoscopic sympathetic block to predict compensatory hyperhidrosis in primary hyperhidrosis. J. Thorac. Dis. 13(6), 3509–3517 (2021).
Kim, J. et al. Ultrasound-guided thoracic paravertebral block as a sympathetic blockade for upper extremity neuropathic pain: A prospective pilot study. J. Pain Res. 13, 3395–3403 (2020).
Yang, H. et al. CT-guided chemical thoracic sympathectomy versus botulinum toxin type A Injection for palmar hyperhidrosis. Thorac. Cardiovasc. Surg. 67(5), 402–406 (2019).
Andreae, M. H. & Andreae, D. A. Regional anaesthesia to prevent chronic pain after surgery: A Cochrane systematic review and meta-analysis. Br. J. Anaesth. 111, 711–720 (2013).
Day, M. Sympathetic blocks: The evidence. Pain Pract. 8(2), 98–109 (2008).
Kumar, V., Krone, K. & Mathieu, A. Neuraxial and sympathetic blocks in herpes zoster and postherpetic neuralgia: An appraisal of current evidence. Reg. Anesth. Pain Med. 29, 454–461 (2004).
Choi, E., Nahm, F. S. & Lee, P. B. Sympathetic block as a new treatment for lymphedema. Pain Physician 18(4), 365–372 (2015).
Sun, G., Liu, F. & Xiu, C. High thoracic sympathetic block improves coronary microcirculation disturbance in rats with chronic heart failure. Microvasc. Res. 122, 94–100 (2019).
Kakuyama, M., Toda, H., Osawa, M. & Fukuda, K. The bilateral effect of stellate ganglion block on the facial skin blood flow. Reg. Anesth. Pain Med. 25, 389–392 (2000).
Stevens, R. A. et al. The relative increase in skin temperature after stellate ganglion block is predictive of a complete sympathectomy of the hand. Reg. Anesth. Pain Med. 23, 266–270 (1998).
Karmakar, M. K. Thoracic paravertebral block. Anesthesiology 95(3), 771–780 (2001).
Hogan, Q. H., Erickson, S. J. & Abram, S. E. Computerized tomography (CT) guided stellate ganglion blockade. Anesthesiology 77, 596–599 (1992).
Baig, S., Moon, J. Y. & Shankar, H. Review of sympathetic blocks: Anatomy, sonoanatomy, evidence, and techniques. Reg. Anesth. Pain Med. 42(3), 377–391 (2017).
Greengrass, R. et al. Paravertebral block for breast cancer surgery. Can. J. Anaesth. 43, 858–866 (1996).
Naja, M. Z. et al. Varying anatomical injection points within the thoracic paravertebral space: Effect on spread of solution and nerve blockade. Anaesthesia 59, 459–463 (2004).
Richardson, J., Cheema, S. P., Hawkins, J. & Sabanathan, S. Thoracic paravertebral space location. A new method using pressure measurement. Anaesthesia. 51(2), 137–9 (1996).
Richardson, J., Lönnqvist, P. A. & Naja, Z. Bilateral thoracic paravertebral block: Potential and practice. Br. J. Anaesth. 106, 164–171 (2011).
Coveney, E. et al. Use of paravertebral block anesthesia in the surgical management of breast cancer: Experience in 156 cases. Ann. Surg. 227, 496–501 (1998).
Lönnqvist, P. A., MacKenzie, J., Soni, A. K. & Conacher, I. D. Paravertebral blockade: Failure rate and complications. Anaesthesia 50, 813–815 (1995).
Pusch, F. et al. Single-injection paravertebral block compared to general anaesthesia in breast surgery. Acta Anaesthesiol. Scand. 43, 770–774 (1999).
Kim, Y. H., Kim, S. Y., Lee, Y. J. & Kim, E. D. A prospective, randomized cross-over trial of T2 paravertebral block as a sympathetic block in complex regional pain syndrome. Pain Physician 22, E417-e424 (2019).
Narouze, S. Ultrasound-guided stellate ganglion block: Safety and efficacy. Curr. Pain Headache Rep. 18, 424 (2014).
Ardon, A. E., Lee, J., Franco, C. D., Riutort, K. T. & Greengrass, R. A. Paravertebral block: Anatomy and relevant safety issues. Korean J. Anesthesiol. 73(5), 394–400 (2020).
Author information
Authors and Affiliations
Contributions
J.W.H. and J.Y.J. contributed to the study conception and design. Material preparation and data collection were performed by E.E. and J.Y.J. Data collection and analysis were performed by J.Y.J., C.B.P., E.E., and S.S.P. The first draft of the manuscript was written by J.W.H., S.A., and J.Y.J., and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Han, J.W., Ahn, S., Jeong, J.Y. et al. Use of thoracoscopy for thoracic sympathetic nerve block in primary hyperhidrosis. Sci Rep 13, 1402 (2023). https://doi.org/10.1038/s41598-023-28727-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-28727-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.