Abstract
IL-27 is an IL-12 family cytokine with immune regulatory properties, capable of modulating inflammatory responses, including autoimmunity. While extensive studies investigated the major target cells of IL-27 mediating its functions, the source of IL-27 especially during tissue specific autoimmune inflammation has not formally been examined. IL-27p28 subunit, also known as IL-30, was initially discovered as an IL-27-specific subunit, and it has thus been deemed as a surrogate marker to denote IL-27 expression. However, IL-30 can be secreted independently of Ebi3, a subunit that forms bioactive IL-27 with IL-30. Moreover, IL-30 itself may act as a negative regulator antagonizing IL-27. In this study, we exploited various cell type specific IL-30-deficient mouse models and examined the source of IL-30 in a T cell mediated autoimmune neuroinflammation. We found that IL-30 expressed by infiltrating and CNS resident APC subsets, infiltrating myeloid cells and microglia, is central in limiting the inflammation. However, dendritic cell-derived IL-30 was dispensable for the disease development. Unexpectedly, in cell type specific IL-30 deficient mice that develop severe EAE, IL-30 expression in the remaining wild-type APC subsets is disproportionately increased, suggesting that increased endogenous IL-30 production may be involved in the severe pathogenesis. In support, systemic recombinant IL-30 administration exacerbates EAE severity. Our results demonstrate that dysregulated endogenous IL-30 expression may interfere with immune regulatory functions of IL-27, promoting encephalitogenic inflammation in vivo.
Similar content being viewed by others
Introduction
IL-27 is an IL-12 family heterodimeric cytokine composed of p28 (also known as IL-30) and Ebi3 subunits, and binds the IL-27 specific receptor, a heterodimeric surface receptor complex made of IL-27Rα and gp1301, 2. IL-27 mediates highly diverse, even opposing, pro- and anti-inflammatory roles by supporting Tbet/IFNγ expression in developing Th1 cells and by inhibiting Rorc expression and Th17 differentiation, respectively3,4,5. Another well appreciated anti-inflammatory function of IL-27 operates through IL-10 induction from activated CD4 T cells6, 7. IL-10-producing Foxp3− helper T (known as Tr1) cells are thought to play an important role in suppressing inflammation and in maintaining tolerance in many conditions8, 9. Indeed, mice deficient in IL-27Rα subunit are highly susceptible to Th17-mediated autoimmune inflammation, experimental autoimmune encephalomyelitis (EAE), and the susceptibility is thought to be attributed to the deficit in the development of IL-10-producing CD4 T cells10. However, IL-27 directly acting on Foxp3+ Treg cells supports Treg cells’ ability to suppress autoimmune inflammation via Lag3-dependent and Tr1-independent mechanisms11.
Being the IL-27-specific subunit, IL-30 was measured as a surrogate to assess IL-27 production. The primary source of IL-30 is cells of myeloid origin, including monocytes, macrophages, and dendritic cells2, and signals triggering IL-30 secretion are mostly of innate immunity. TLR3 and TLR4 have previously been shown to trigger IL-30 expression in dendritic cells via an IRF3-dependent mechanism12, and IFNγ signal can augment the expression13, 14. IFNβ, a widely used immune suppressive cytokine for the treatment of autoimmunity, is another signal inducing IL-30 production and inhibiting Th17 differentiation15. However, little is known regarding the precise source and immune regulatory functions of IL-30 especially during tissue specific autoimmune inflammation, such as EAE.
There is emerging evidence that IL-30 may exhibit an immune regulatory function distinct from that of IL-2716, 17. Caspi and colleagues utilized IL-30-overexpressing mouse model to show that IL-30 inhibits T cell differentiation to Th1 and Th17 lineage cells and the development of autoimmunity18. IL-30 also negatively regulates humoral and cellular responses during parasite infection, independent of its role as an IL-27 subunit19. In the context of tumorigenesis, IL-30 has been suggested to play a pro-tumorigenic role, in part by supporting cancer stem-like cell survival, vascularization, and proliferation20, 21. However, the nature of such pro-inflammatory and pro-tumorigenic functions of IL-30 remains largely unclear22.
The current study aimed at identifying the source of IL-30 and its potential role during autoimmune inflammation in the central nervous system (CNS). We utilized cell type specific Il27p28−/− mouse models and found that IL-30 expressed by myeloid cells and microglia but not by dendritic cells plays an important role in limiting autoimmune inflammation. Unexpectedly, we also noted that exacerbated inflammation seen in those cell type specific IL-30-deficient mice was associated with drastic overexpression of Il27p28 mRNA in otherwise unaffected wild type CNS-infiltrating and resident APC subsets, possibly resulting in disproportionate IL-30 production. Systemic administration of recombinant IL-30 alone into mice with ongoing EAE similarly aggravated the disease progression, suggesting that IL-30 itself can enhance encephalitogenic immune activity. Increased IL-30 expression was associated with decrease in Treg cell expression of Lag3, an indicative of IL-27 signals, suggesting that IL-30 may antagonize IL-27’s action on Treg cells in vivo. Yet, IL-30 showed no measurable biological activity on activated T cells as determined by its ability to phosphorylate Stat1 and Stat3 or to antagonize IL-27 activity in vitro. Therefore, IL-30 may exert a distinct function to antagonize IL-27 and to support autoimmune inflammatory T cell responses in vivo.
Results
Inflammatory cytokine mRNA expression in EAE
We first measured cytokine gene expression in the brain and spinal cords during EAE. Three time points were chosen: days 8, 14, and 21 post induction, representing the disease onset, peak, and partial recovery, respectively. Il27p28 mRNA expression mirrored the disease activity, and it peaked around day 14, showing > 10-fold increase in both tissue sites compared to those of naïve mice (Fig. 1a). The Ebi3 subunit mRNA expression displayed a similar pattern as the Il27p28 (Fig. 1a). Il12p40 mRNA expression in the spinal cord was markedly increased with the similar kinetics, reaching ~ 100-fold increase over naïve tissue, although its expression in the brain was not observed (Fig. 1a). On the other hand, Il12p35 or Il23p19 mRNA expression only slightly increased (Fig. 1a and data not shown). Expression of inflammatory cytokines, such as, Tnfa, Ifng, Il17a, Il1b, and Il6, as well as of key transcription factors, Tbx21 and Rorc, followed the similar pattern (Fig. 1b,c). Foxp3 mRNA expression substantially increased at the peak of the disease and was maintained thereafter, demonstrating Treg accumulation in the tissue, likely involved in inflammatory resolution23. We also measured inflammatory chemokines, such as Ccl2, Ccl3, Ccl7, Cxcl1, Cxcl9, and Cxcl10. Again, the expression pattern showed similar kinetics, with the greater magnitude in the spinal cord (Supp Fig. 1). Myeloid cells capable of presenting antigens, including macrophages and dendritic cells, are the primary source of IL-12 family cytokines including IL-27. To examine relative sources of each cytokine during autoimmune inflammation in the CNS, we FACS sorted different APC subsets from the inflamed CNS tissues; CD45high CD11bhigh infiltrating myeloid cells, CD45int CD11bhigh microglia, and CD45low cells that include astrocytes and oligodendrocytes, at the peak of the disease, and cytokine gene expression was determined. While both infiltrating myeloid cells and microglia similarly expressed all the tested IL-12 family cytokines, the level of Il27p28 and Ebi3 mRNA expression was particularly greater than any other subunits examined (Fig. 1d). These results prompted us to investigate the central source of IL-27, especially IL-27p28 (referred to as IL-30 hereafter) subunit during the development of autoimmune neuroinflammation.
Cytokine gene expression in the CNS during EAE. (a–c) EAE was induced in C57BL/6 mice as described in the “Methods”. RNA was isolated from the brain and spinal cords at disease onset (day 8 post immunization), acute phase (day 14 post immunization) and remission phase (day 21 post immunization). n = 4–6 per group. mRNA expression of IL-12 family genes, cytokines and transcription factors were measured by qRT-PCR. Data were normalized by Gapdh gene expression and compared to that of naive mice. (d) CD45high CD11bhigh (infiltrating myeloid cells), CD45int CD11bhigh (microglia) and CD45low (including astrocyte and oligodendrocyte) cells were sorted from the CNS at the peak of disease (day 17 post immunization) and expression of the indicated genes was measured by qPCR. Gene expression was normalized by Gapdh and compared to that of naïve mice. The results shown represent two independent experiments. *p < 0.05; **p < 0.01; ***p < 0.001; as determined by Mann–Whitney nonparametric test.
Myeloid cell-derived IL-30 regulates encephalitogenic immune responses
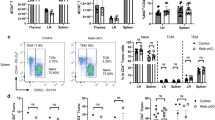
Cells of myeloid origin, especially monocytes and macrophages, are an important source of IL-301, 24. To interrogate the contribution of myeloid cell-derived IL-27 to EAE pathogenesis, we utilized myeloid cell-specific IL-30−/− (LysMCre Il27p28fl/fl) mice. The lack of Il27p28 mRNA expression in macrophages in LysMCre Il27p28fl/fl mice was validated by qPCR (Supp Fig. 2). Myeloid cell-specific IL-30−/− mice developed severe EAE, although they appear to recover from the initial paralysis similarly to that of wild type mice (Fig. 2a). Severe acute disease was further reflected by increased CD4 T cells infiltrating the CNS and histopathological assessment at the peak of the disease (Fig. 2b,d). CNS accumulation of Foxp3+ Treg cells was significantly greater in these mice, although Treg cell proportion or Foxp3 expression was found comparable (Fig. 2c and data not shown). Treg cell expression of ICOS, GITR, and CD25 remained unchanged regardless of myeloid cell-derived IL-30 (data not shown). CD4 T cells expressing inflammatory cytokines were substantially increased in the CNS tissues of LysMCre Il27p28fl/fl mice (Fig. 2e), consistent with severe EAE phenotypes in these mice. In support, serum cytokine levels, especially IL-6, IFNγ, IL-17A, and TNFα, were significantly increased in myeloid cell specific IL-30−/− mice (Fig. 2f). mRNAs for encephalitogenic cytokines and key transcription factors were similarly elevated in these mice (Fig. 2g), and inflammatory chemokine expression in the CNS tissues was also elevated in mice with myeloid cell specific IL-30 deletion (Supp Fig. 3). CNS expression of Il27p28 and Il12p40 mRNAs was greater in the absence of myeloid cell-derived IL-30, which may be attributed to elevated expression of IL-30-inducing cytokines such as IFNγ (Fig. 2g), although Il23p19 and Il12p35 mRNA expression was similar (Fig. 2g). Increased Il27p28 mRNA expression in LysMCre Il27p28fl/fl mice was further examined by comparing the expression in FACS sorted CNS APC subsets. Unexpectedly, we observed that Il27p28 mRNA expression was drastically increased in microglia and CD45low cells in this condition (Fig. 2h). While the LysM promoter can target microglia, the targeting efficiency is only partial25, 26. Goldman et al. elegantly reported that LysMCre-induced gene targeting efficiency in microglia is at ~ 40%27. Therefore, it is possible that Il27p28 mRNA expression observed in microglia from LysMCre Il27p28fl/fl mice appears to be derived from LysM-independent microglia subsets. Il27p28 mRNA expression in infiltrating myeloid cells was not found, corroborating IL-30 deficiency of myeloid cells (Fig. 2h). Of note, Ebi3 mRNA expression was also increased in microglia and CD45low cells (Fig. 2h), while myeloid cell expression of the Ebi3 mRNA remained unchanged. Therefore, myeloid lineage cells could be an important source of IL-27, capable of modulating EAE pathogenesis.
EAE in myeloid cell-specific Il27p28−/− mice. LysMWT (n = 7) and LysMCre Il27p28fl/fl (LysMΔIl27p28) (n = 11) mice were induced for EAE. (a) EAE clinical score. (b,c) The numbers of CNS infiltrating CD4+ and CD4+Foxp3+ Treg cells, and the mean fluorescence intensity (MFI) of Foxp3 was determined by flow cytometry at day 17 post immunization. (d) At day 18 postimmunization, tissue sections of the spinal cords were stained with H&E. Arrows indicate infiltration of inflammatory cells. × 200 magnification. (e) Flow cytometry analysis of GM-CSF, IFNγ, IL-17, and TNFα CD4+ T cells from the CNS of EAE mice (day 17 post immunization). (f) The levels of IL-6, IFNγ, TNFα and IL-17A in the serum of EAE mice (day 17 post immunization) were measured using Cytometric Bead Array. Each serum sample was analyzed in duplicates. (g) qPCR analysis of the indicated mRNAs in the brain and spinal cords from naïve, LysMWT, and LysMΔIl27p28 mice 17 days post immunization. Gene expression was normalized by Gapdh and compared to that of naïve mice. n = 3 per group. (h) qPCR analysis of the indicated mRNAs in freshly sorted CD45high CD11bhigh (infiltrating myeloid cells), CD45int CD11bhigh (microglia) and CD45low (astrocyte and oligodendrocyte) cells from LysMWT or LysMΔIl27p28 mice 17 days post immunization. n = 3 per group. *p < 0.05; **p < 0.01; ***p < 0.001; as determined by Mann–Whitney nonparametric test.
DC-derived IL-30 is dispensable
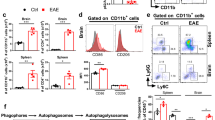
IL-27 produced by DCs promotes the generation of IL-10+ CD4 T cells capable of attenuating autoimmune inflammation28. Utilizing IL-30-floxed mouse model, it was concluded that DC-derived IL-27 plays a role in antitumor immunity by regulating NK and NKT cell recruitment and activation29, 30. To test the role of DC-derived IL-27 in EAE, DC-specific IL-30−/− (CD11cCre Il27p28fl/fl) mice were used. qPCR analysis validated DC-specific IL-30 deficiency in these mice (Supp Fig. 2). Despite the involvement of DC-derived IL-27 in T cell immunity, we found that DC-specific IL-30 deletion did not affect EAE pathogenesis, as both disease onset and the clinical severity remained similar to those of wild type control mice (Fig. 3a). CD4 T cells infiltrating the CNS tissues were comparable in both proportions and absolute numbers (Fig. 3b). Likewise, Foxp3+ Treg cell accumulation in the CNS was also comparable (Fig. 3c). Treg cell expression of surface markers associated with the suppressive function, ICOS, GITR, and CD25, was similar regardless of DC-derived IL-30 (Fig. 3d). We then measured CD4 T cell expression of encephalitogenic cytokines by flow cytometry. As shown in Fig. 3e, intracellular expression of GM-CSF, IFNγ, IL-17, and TNFα in CNS infiltrating CD4 T cells was similar in both proportions and absolute numbers. Consistent with these findings, expression of Ifng, Il17a, and Il1b mRNA in the CNS tissues was also similar between WT and DC-specific IL-30−/− groups (Fig. 3f). Serum cytokine levels measured by cytokine bead array (CBA) were comparable between the groups (Fig. 3g). Inflammatory chemokine and IL-12 family cytokine expression in the tissues was similar between the groups (Supp Fig. 4a,b). Lastly, CNS APC subsets were FACS sorted as above, and IL-12 family cytokine gene expression was determined. Consistent with the EAE severity and overall immune responses, we found no differences in cytokine gene expression between the groups (Fig. 3h and not shown). Therefore, DC-derived IL-30 plays little role in regulating encephalitogenic immune responses.
EAE in DC-specific Il27p28−/− mice. CD11cWT (n = 9) and CD11cCre Il27p28fl/fl (CD11cΔIl27p28) (n = 10) were induced for EAE. (a) Time course of the development of EAE. (b–d) The numbers of CNS-infiltrating CD4+ and CD4+Foxp3+ Treg cells, and the mean fluorescence intensity (MFI) of Foxp3, CD44, ICOS, GITR and CD25 were determined by flow cytometry at day 17 post immunization. (e) Flow cytometry analysis of GM-CSF, IFN-γ, IL-17, and TNFα CD4+ T cells from the CNS of EAE mice (day 17 post immunization). (f) RNAs isolated from the brain and spinal cords at day 17 post immunization were analyzed for the expression of Ifng, Il17a, and Il1b. n = 3 per group. Gene expression was normalized by Gapdh and compared to that of naïve mice. (g) The levels of IL-2, IL-4, IL-6, IFNg, TNFa and IL-17A in the serum of EAE mice (day 17 post immunization) were measured using the Cytometric Bead Array. Each serum sample was analyzed in duplicates. (h) qPCR analysis of the indicated mRNAs in freshly sorted CD45high CD11bhigh (infiltrating myeloid cells), CD45int CD11bhigh (microglia) and CD45low (astrocyte and oligodendrocyte) cells from CD11cWT and CD11cΔIl27p28 mice with EAE. *p < 0.05; **p < 0.01; ***p < 0.001; as determined by Mann–Whitney nonparametric test.
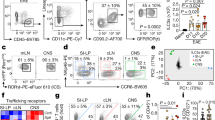
EAE in microglia-specific Il27p28−/− mice. Cx3cr1WT (n = 9) and Cx3cr1Cre Il27p28fl/f (Cx3cr1ΔIl27p28) (n = 10) were induced for EAE. (a) EAE clinical scores. (b,c) Total numbers of CNS-infiltrating CD4+ and CD4+Foxp3+ Treg cells, and the mean fluorescence intensity (MFI) of Foxp3 was determined by flow cytometry at day 17 post immunization. (d) At day 18 postimmunization, tissue sections of the spinal cords were stained with H&E. Arrows indicate infiltration of inflammatory cells. × 200 magnification. (e) Flow cytometry analysis of GM-CSF, IFNγ, IL-17, and TNFα CD4+ T cells from the CNS of EAE mice (day 17 post immunization). (f) qPCR analysis of the indicated mRNAs in the brain and spinal cords from naïve, Cx3cr1WT, and Cx3cr1ΔIl27p28 mice 17 days post immunization. Gene expression was normalized by Gapdh and compared to that of naïve mice. n = 3–5 per group. (g) qPCR analysis of the indicated mRNAs in freshly sorted CD45high CD11bhigh (infiltrating myeloid cells), CD45int CD11bhigh (microglia) and CD45low (including astrocyte and oligodendrocyte) cells from Cx3cr1WT or Cx3cr1ΔIl27p28 mice 17 days post immunization. n = 3 per group. *p < 0.05; **p < 0.01; ***p < 0.001; as determined by Mann–Whitney nonparametric test.
Microglia-derived IL-30 plays a similar regulatory role in EAE
Microglia are resident CNS glial cells capable of producing IL-2731, 32. To target microglia expression of IL-27, Cx3cr1Cre Il27p28fl/f mice were used33. Microglia specific IL-30−/− mice exhibited severe EAE analogous to myeloid cell specific IL-30−/− mice (Fig. 4a). Assessment of total CD4 and Foxp3+ Treg cell CNS infiltration as well as of histopathology further support severe EAE phenotypes (Fig. 4b–d). Treg cell associated surface marker and Foxp3 expression remained unchanged in microglia-specific IL-30−/− mice (Fig. 4c and data not shown). CNS infiltrating CD4 T cell expression of inflammatory cytokines was markedly increased, further supporting greater susceptibility of these mice (Fig. 4e). We measured inflammatory cytokine and chemokine gene expression by qPCR and confirmed that microglia-specific IL-30 deficiency results in greater increase of the expression (Fig. 4f and data not shown). Il27p28 mRNA expression in the CNS of microglia-specific IL-30−/− mice was significantly elevated compared to that of wild type mice (data not shown). Also observed was that Il27p28 mRNA expression in FACS sorted different CNS APC subsets, i.e., infiltrating myeloid cells and CD45low cells, was drastically increased in microglia-specific IL-30−/− mice (Fig. 4g). The complete lack of Il27p28 mRNA expression in sorted microglia further validated microglia-specific IL-30 deficiency (Fig. 4g). Significant increase of Ebi3 mRNA expression was also seen in infiltrating monocytes and CD45low cells but not in microglia (Fig. 4g). Therefore, microglia-derived IL-30 also appears to play a regulatory role in EAE pathogenesis.
In vivo administration of recombinant IL-30 exacerbates encephalitogenic inflammation
IL-30 can be secreted independently of Ebi334. Therefore, aberrant susceptibility to EAE seen in the Il27p28 gene deficient animals in this study may stem from defective IL-27 and/or IL-30 secretion from the targeted myeloid cells or microglia. We were particularly intrigued by the unexpected elevation of Il27p28 mRNA expression in ‘wild-type’ CNS APC subsets within those cell type specific IL-30−/− mice. As such increase did not occur in DC-specific IL-30−/− mice where the disease severity remained unchanged, we posit that elevated Il27p28 mRNA expression may be associated with severe disease progression. IL-30 may function as a natural antagonist of gp130-mediated signaling34. In support, it was previously reported that secreted IL-30 could inhibit the biological functions of IL-2722. Therefore, IL-30 may antagonize anti-inflammatory roles of IL-27, driving encephalitogenic inflammation. To test this possibility, we induced EAE in B6 mice and then administered recombinant IL-27 or IL-30 via a mini-osmotic pump once the mice developed noticeable clinical signs. As shown in Fig. 5a, IL-27 administered substantially dampened the EAE severity, as previously reported35. By contrast, IL-30 administration exacerbated the clinical severity of the recipient mice (Fig. 5a). In support of the disease severity, CNS infiltrating CD4 T cells were diminished by IL-27 administration, while IL-30 administration substantially increased the infiltration (Fig. 5b). Inflammatory cytokine expression by infiltrating CD4 T cells was similarly affected by IL-27 and IL-30 administered in vivo. TNFα-, IL-17-, GM-CSF-, and IFNγ-expressing CD4 T cell accumulation in the CNS was markedly diminished by IL-27, while the accumulation was dramatically increased following IL-30 administration (Fig. 5c). Inflammatory chemokine mRNA expression in the CNS followed similar pattern and was significantly diminished by IL-27 but rather increased by IL-30 (Fig. 5d). We previously showed that Foxp3+ Treg cells are the primary target cells of IL-27 in vivo and that IL-27 induces Lag3 expression in Treg cells36. Consistent with the previous findings, IL-27 administered increased Lag3 expression in Treg cells compared to that of sham treated controls, while IL-30 administration significantly diminished Treg cell expression of Lag3 (Fig. 5e). Likewise, we observed that Treg cell Lag3 expression was significantly diminished in microglia-specific IL-30−/− mice (Fig. 5f), in which Il27p28 mRNA expression was dysregulated and the mice developed severe EAE (Fig. 4a,f). We measured IL-30 protein secretion from the CNS homogenates and found that the level was significantly increased in microglia-specific IL-30−/− compared to that in wild type mice with EAE (Fig. 5g). Unexpectedly, however, we also noticed elevated IL-27 heterodimer secretion from the CNS homogenates (Fig. 5h). Since Treg cell Lag3 expression was lower in this condition (Fig. 5f), these results suggest that IL-30 produced may compete with IL-27 for its binding to the receptor and to dampen inflammatory responses in part via Treg cells in vivo. The competition may result in elevated IL-27 levels in the CNS homogenates.
In vivo IL-30 administration develops significantly exacerbated EAE. C57BL/6 mice were induced for EAE. Osmotic pumps containing IL-27 (400 ng, n = 5), IL-30 (400 ng, n = 5) were subcutaneously implanted or sham surgery (n = 4) was performed at 12 days post induction. (a) EAE score. (b) Total CD4+ T cell numbers in the CNS at 22 days post induction. (c) The numbers of GM-CSF, IFNγ, IL-17, and TNFα+ CD4 T cells were determined by intracellular cytokine staining at day 22 post immunization. (d) qPCR analysis of the indicated chemokine expression in the brain from the sham, IL-27-pump and IL-30-pump group. Gene expression was normalized by Gapdh and compared to that of sham surgery group. (e) Lag3 expression of CNS infiltrating Treg cells was determined by flow cytometry. (f) Cx3cr1WT and Cx3cr1ΔIl27p28 mice induced for EAE as described in Fig. 4 were used to measure CNS infiltrating Treg cell expression of Lag3. (g,h) IL-30 and IL-27 levels in the CNS homogenates were measured by ELISA and were normalized to those of naïve wild type mice. n = 7 per group. *p < 0.05; **p < 0.01; ***p < 0.001; as determined by Mann–Whitney nonparametric test.
IL-30 does not display regulatory properties in vitro
Although IL-30 alone fails to support T cell proliferation or IFNγ production37, IL-30 reportedly inhibits the production of IL-17 and IL-10 triggered by IL-27 or IL-6 stimulation in activated T cells34, 38. In addition, IL-30 also induces LPS-induced TNFα and IP-10 production in monocytes39. Since we observed that IL-30 may be able to antagonize IL-27’s regulatory function in vivo, we sought to test if IL-30 expresses regulatory properties. Naïve CD4 and CD8 T cells were stimulated in the presence of recombinant IL-27 or IL-30. As expected, IL-27 rapidly phosphorylated both Stat1 and Stat3 in CD4 and CD8 T cells (Fig. 6a and data not shown). By contrast, IL-30 stimulation had little effects on Stat phosphorylation (Fig. 6a). The lack of Stat phosphorylation by IL-30 stimulation led us to further examine its ability to induce or to antagonize IFNγ or IL-10 expression in activated T cells. CD4 T cells stimulated under Th1 or Th17 polarization condition substantially upregulated Ifng mRNA expression in response to IL-27 but not to IL-30 (Fig. 6b and data not shown). Likewise, IL-27 induced robust Il10 mRNA expression in both developing Th1 and Th17 type CD4 T cells, while IL-30 failed to do so (Fig. 6c and data not shown). Pre-stimulation with IL-27 effectively inhibited IL-27-induced Stat phosphorylation, whereas IL-30 pre-stimulation had no impact on interfering with IL-27-induced Stat phosphorylation (Fig. 6d). Therefore, IL-30 alone does not appear to alter cytokine expression in activated T cells in vitro. IL-30 may influence T cell immunity through APCs, as Ebi3 mRNA expression can be elevated especially when Il27p28 mRNA expression was increased. In bone marrow-derived macrophages, neither IL-27 nor IL-30 had the ability to induce IL-12 or IL-27 expression (Supp Fig. 5).
IL-30 stimulation in CD4 T cell activation in vitro. (a) FACS sorted CD4+ naïve cells were stimulated with recombinant IL-27, IL-30. Phosphorylated STAT1 and STAT3 expression was determined by flow cytometry at 10- and 30-min following stimulation. (b,c) Naive CD4 T cells were stimulated under Th1 polarization conditions in the presence of IL-27 (filled bar) or IL-30 (open bar) (0–50 ng/mL) for 3 days. Ifng and Il10 mRNA expression was determined by qPCR. (d) Naïve CD4 T cells were incubated with media (Nil), 50 ng IL-27, or 50 ng IL-30 for one hour. The cells were then washed and restimulated with IL-27. Stat1 and Stat3 phosphorylation was determined by flow cytometry at 15 and 30 min following stimulation. The data shown are representative of two independent experiments.
Discussion
In this study, we took advantage of cell type specific IL-30-deficient mouse models to investigate the sources of IL-30 and to gain closer insights into the immune regulatory functions of IL-27 (and of IL-30 itself) in autoimmune inflammation. Consistent with the previous reports, we found that infiltrating myeloid cell-derived IL-30 is critical to limit autoimmune inflammation in the CNS. Interestingly, other CNS resident APC subsets, such as microglia, were equally important in modulating inflammatory responses within the CNS, as targeting IL-30 expression in microglia also confers greater susceptibility to autoimmunity. Unexpectedly, however, we found that mice deficient in IL-30 specifically in DCs were not susceptible to the disease, suggesting a dispensable role of DC-derived IL-30 in EAE pathogenesis.
IL-30 was initially considered an IL-27-specific subunit; thus, mice deficient in or overexpressing the Il27p28 gene have been used to interrogate the immune regulatory functions of IL-27. Mice overexpressing the Il27p28 gene are resistant to autoimmune diseases, and it was proposed that such resistance originates from IL-27’s ability to antagonize inflammatory T cell responses, particularly Th1 and Th17 immunity18. Likewise, mice deficient in IL-30 are highly susceptible to autoimmune inflammation and express heightened Th17 immunity5. Therefore, it was reasonably concluded that IL-30’s ability to control inflammatory responses is through its role as an IL-27 subunit. It was then discovered that IL-30 can be secreted in the absence of the Ebi3 and is able to modulate immune responses34. Tagawa and colleagues reported that IL-30 alone can mediate immune regulatory functions, suppressing allogenic T cell responses22. More recently, Park et al. reported that IL-30 can act as a negative regulator of both B and T cell responses during T. gondii infection, independently of IL-2719. IL-30 may antagonize other cytokines, such as IL-27 or IL-6 that utilizes the gp130 surface receptor for the signaling34. Although IL-30 stimulation itself does not trigger any detectable Stat phosphorylation in vitro, its presence may be sufficient to hinder Stat1 and Stat3 phosphorylation induced by IL-27 or IL-634. Indeed, we found that IL-30 stimulation alone does not induce Stat phosphorylation in T cells. However, we also noted that IL-30 fails to interfere with IL-27-induced Stat1/3 phosphorylation or IL-27-stimulated gene expression in vitro. Petes et al. previously reported that IL-30-induced Stat1 and Stat3 phosphorylation can be bimodal and a delayed second phosphorylation event occurs at later time points in THP-1 cells39. When primary monocytes were used, delayed and weak Stat1 but not Stat3 phosphorylation was similarly observed39. However, we found no signs of delayed Stat phosphorylation in T cells (data not shown). Moreover, neither IL-27 nor IL-30 altered cytokine expression in bone marrow-derived macrophages. While the reason underlying the discrepancy is not clear, concentrations used may account for the difference, as the previous study used higher IL-30 concentration. It is worth noting that IL-30 can form complexes with other subunits besides Ebi3, such as p40, CLF, and IL-30 itself40. IL-30 also forms a complex with sIL-6Rα to trigger ‘IL-30 trans-signaling’41, 42, although the studies were mostly performed using cell lines and/or the IL-30-sIL6Rα fusion protein. Thus, the precise working mechanism underlying IL-30-induced immune regulation especially in vivo requires further investigation. Of note, sIL-6Rα is also involved in IL-6 trans-signals, mediating pro-inflammatory functions of IL-6 in many disease conditions43, 44. While the levels of serum sIL-6Rα are found increased and correlated with disease activity in MS patients45, the mechanism by which sIL-6Rα potentially contributes to the inflammation (either via IL-6 or IL-30) remains to be determined. IL-30 may signal through gp130 homodimers without soluble IL-6Rα only at high concentrations41. While IL-30 does not impact T cells and macrophages in vitro, our data suggest that IL-30 may act on other target cells, partly Treg cells, to modulate inflammatory responses in vivo.
Finding dysregulated Il27p28 mRNA expression in some cell type specific IL-30−/− mice with severe disease is of particular interest. What does trigger such an aberrant expression? IFNγ has previously been reported to induce IL-30 expression in myeloid cells46, and we likewise found elevated IFNγ expression in infiltrating CD4 T cells. IL-30 expression may thus be directly correlated with the disease severity and inflammation. Disproportionate expression of Il27p28 mRNA may result in excessive IL-30, which may interfere with IL-27’s function to inhibit the inflammation. As a result, IFNγ production by autoreactive T cells continuously increases, further amplifying IL-30 expression. Although we found no evidence that IL-30 alone is able to induce Stat phosphorylation and gene expression in T cells in vitro, IL-30 may be able to do so in vivo, in part, based on its ability to antagonize IL-27’s function to modulate Treg cell Lag3 expression. Expression of Treg cell markers, ICOS, GITR, CD25, and Foxp3 remained unchanged in cell type-specific IL-30−/− mice or in IL-27 or IL-30 treatment. We previously showed that Tim-3, CD39, and CTLA4 expression in Treg cells is not affected by the lack of IL-27 signaling35. However, we noticed that Lag3 expression on Treg cells may be balanced by IL-27 and IL-30. Therefore, IL-30-induced effects may be operated in part by reducing Lag3 expression in Treg cells, although we cannot exclude the possibility that IL-30 itself poses unknown immune regulatory functions in vivo, because the receptor gp130 is expressed on multiple cell types including myeloid cells, B cells, and endothelial cells, etc.47. Alternatively, IL-30 may form a complex with other subunits, such as cytokine-like factor 1 (CLF1) or soluble IL-6Rα48, and it is possible that different complexes may be preferentially formed depending on the type of cells. Whether these complexes mediate different functions remains to be determined. Of note, increased Ebi3 mRNA expression was observed in cells expressing aberrant Il27p28 mRNA expression. Since such dysregulated Ebi3 expression appears to be associated with Il27p28, increase in Ebi3 expression may be the result of cellular activation49, 50. However, it is important to emphasize that the fold increase was far greater in Il27p28 compared to that in the Ebi3 expression. Neutralizing or blocking antibodies against IL-30 cannot be used to investigate this possibility, because it will affect both IL-30 and IL-27. One may require a system in which IL-30 itself cannot be secreted unless it forms the heterodimeric IL-27 complex. Feige and colleagues recently reported a discrepant IL-30 secretion pattern between human and mice51. A single amino acid difference is sufficient to alter the secretion pattern of IL-30. Unlike mouse IL-30, which has two cysteine residues that enable a stable secretion of IL-30 independent of Ebi3, human IL-30 has only one cysteine residue. Thus, in the absence of Ebi3, IL-30 fails to be secreted; instead, it retains in the ER and ultimately gets degraded51, suggesting that Ebi3-independent IL-30 secretion will not occur in humans. We thus speculate that the in vivo functions of the IL-27:IL-30 axis are different in humans and mice. Further investigation will be necessary to examine the regulatory functions of IL-30.
DCs, especially cDC1 type subsets that express the chemokine receptor XCR1, express IL-30 when immunized with a combination adjuvant, poly I:C and agonistic anti-CD40 Ab52. Our finding that DC-derived IL-30 plays little role in limiting EAE pathogenesis and encephalitogenic immune responses suggests that DCs may not be the primary source of IL-30 during autoimmune inflammation in the CNS. DC-derived IL-30 may be important in the secondary lymphoid tissues during priming event as seen in the spleen following intravenous immunization with adjuvants52, and IL-30-derived from inflammatory monocytes/macrophages or tissue resident APC subsets may be more crucial in limiting immune responses in the target tissues.
In summary, we report that IL-30 expresses a regulatory property to promote autoimmune inflammation in vivo, which could partially be mediated by interfering with regulatory function of IL-27. Further investigation is needed to understand its precise immune modulatory functions, through which a novel approach targeting IL-30 and/or IL-27 may be developed.
Materials and methods
Animals
All the animal experiments were conducted in accordance to ARRIVE guidelines and approved by the institutional animal care and use committees (IACUC) of Northwestern University (protocol #IS00015862). In addition, all the methods were performed in accordance with the relevant guidelines and regulations. C57BL/6, CD11c (Itgax)Cre (strain #8068), GfapCre (strain #24098), LysM (Lyz2)Cre (strain #4781), and Cx3cr1Cre (strain #25524) mice were purchased from the Jackson Laboratory (Bar Harbor, ME). Il27p28fl/fl mice were previously reported29. All mice were bred in a specific pathogen-free facility at Northwestern University Feinberg School of Medicine.
EAE induction
Mice were subcutaneously injected with 200 μL of an emulsion containing 300 μg of MOG35–55 peptide (BioSynthesis, Lewisville, TX) and equal volume of Complete Freund's adjuvant supplemented with 5 mg/mL of Mycobacterium tuberculosis strain H37Ra (Difco, Detroit, MI). Additionally, mice were intraperitoneally injected with 200 ng of pertussis toxin (Sigma, St. Louis, MO) at the day of immunization and 48 h later. Disease development was analyzed daily and scored on a 0–5 scale: 0, no clinical signs; 1, limp tail, 2, hind limb weakness, 3, hind limb paralysis, 4, hind limb paralysis and front limb weakness, 5, moribund or death.
Osmotic pump implantation
Mice anesthetized with Ketamine and Xylazine were subcutaneously implanted with a mini-osmotic pump (#1007D, Alzet Durect, Cupertino, CA) as previously described11. This pump system has a reservoir volume of 100 μL and allows for the continuous delivery of the content for 7 days without the need for external connections or frequent handling of animals. Pumps containing 400 ng of rIL-27 or rIL-30 (R&D Systems, Minneapolis, MN) were implanted at day 12 post immunization. Mice with sham surgery were used as controls.
Flow cytometry
Mononuclear cells from the CNS of EAE mice were isolated by Percoll gradient centrifugation as previously described53. The cells were then stained with anti-CD4 (RM4–5), anti-CD44 (IM7), anti-CD25 (PC61.5), anti-GITR (DTA-1), anti-Foxp3 (FJK-16s), and anti-ICOS (C398.4A) antibodies. For intracellular staining, harvested cells were stimulated ex vivo with PMA (10 ng/mL, Millipore-Sigma) and ionomycin (1 μM, Millipore-Sigma) for 4 h in the presence of 2 μM monensin (Calbiochem) during the last 2 h of stimulation. Cells were immediately fixed with 4% paraformaldehyde, permeabilized, and stained with anti-IL-17 (TC11-18H10), anti-IFNγ (XMG1.2), anti-TNFα (TN3-19), anti-GM-CSF (MP1-22E9) antibodies. All the antibodies were purchased from eBioscience (San Diego, CA), BD PharMingen (San Diego, CA), and Biolegend (San Diego, CA). In some experiments cells were stimulated with recombinant IL-27 or IL-30 and phosphorylated Stat1 (4a) and Stat3 (LUVNKLA) expression was determined by flow cytometry. Samples were acquired using a FACSCelesta (BD Bioscience) and analyzed using a FlowJo (Treestar, Ashland, OR). Cytokine-expressing effector CD4 T cells were enumerated by flow cytometry. CNS APC subsets were sorted based on CD45 and CD11b expression using a FACSMelody cell sorter (BD Bioscience). Sorted cells were subjected to gene expression by qPCR as described below.
Cytometric beads array
Serum cytokines were determined using Cytometric Beads Array (BD Biosciences) according to the manufacturer's instructions. The data were analyzed using the CBA software, and the standard curve for each cytokine was generated using the mixed cytokine standard.
Real-time quantitative PCR
Mice with EAE were euthanized and perfused with PBS. The brain and spinal cords were isolated and total RNA was extracted using a TRIzol reagent according to the manufacturer’s instructions (Invitrogen). In some experiments using bone marrow-derived macrophages, bone marrow cells harvested from the tibia and femur were cultured with L929-media (15% volume) for 6 days. Bone marrow generated macrophages were then harvested (> 95% express CD11b). Harvested cells were then stimulated with IL-27 or IL-30, and cytokine gene expression was determined by qPCR as below. cDNA was then obtained using a MMLV reverse transcriptase (Promega, location). qPCR analysis was performed using a QuantStudio 3 Real-Time PCR System (Applied Biosystems, Waltham, MA) using a Radiant qPCR mastermix (Alkali Scientific, Fort Lauderdale, FL) or SYBR green mastermix (Applied Biosystems). The data were normalized by housekeeping Gapdh gene and then compared to the control group. Primers used for the study are listed in Supplementary Table 1.
Sequential protein extraction
The protein extraction was performed as previously reported54. In brief, mice were euthanized and perfused with cold PBS (10 mM, pH 7.4). The brains and spinal cords were then removed. Soluble protein fractions were obtained from the whole cortex and spinal cord using sequential protein extraction. Fractions were obtained by homogenization of the cortex and spinal cord with a dounce homogenizer in buffer including protease inhibitor cocktail (2 mL/200 μg of tissues; Thermo Scientific, 1862209). After centrifugation for 1.5 h at 43,000 rpm in tube (#349622, 3.5 mL, 13 × 51 mm tubes) for high-speed centrifugation from Beckman-Coulter, the supernatants were obtained and aliquoted and stored at − 80 °C. To measure cytokines in the brain and spinal cord soluble fractions, IL-30 (R&D Systems, Minneapolis, MN) and IL-27 (LEGEND MAXTM, BioLegend) ELISA kits were used according to the manufacturer’s instructions.
Histological analysis
Mice were euthanized on the 18th day after EAE induction by intracardiac fixative perfusion using 4% paraformaldehyde (PFA). Spinal column was fixed in 4% PFA for 3 days and spines were washed with tap water and placed in 14% EDTA (pH 7.3, Sigma) for decalcification with shaking for 2 weeks at 4 °C. Tissues were then treated in ethanol/xylene and paraffin-embedded. 5-µm sections were mounted on glass slides and stained with hematoxylin and eosin (H&E).
Statistical analysis
Statistical significance was determined by the Mann–Whitney test using Prism software (GraphPad, San Diego, CA). p < 0.05 was considered statistically significant.
Data availability
Any further data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Kourko, O., Seaver, K., Odoardi, N., Basta, S. & Gee, K. IL-27, IL-30, and IL-35: A cytokine triumvirate in cancer. Front. Oncol. 9, 969. https://doi.org/10.3389/fonc.2019.00969 (2019).
Yoshida, H. & Hunter, C. A. The immunobiology of interleukin-27. Annu. Rev. Immunol. 33, 417–443. https://doi.org/10.1146/annurev-immunol-032414-112134 (2015).
Yoshimura, T. et al. Two-sided roles of IL-27: Induction of Th1 differentiation on naive CD4+ T cells versus suppression of proinflammatory cytokine production including IL-23-induced IL-17 on activated CD4+ T cells partially through STAT3-dependent mechanism. J. Immunol. 177, 5377–5385. https://doi.org/10.4049/jimmunol.177.8.5377 (2006).
Cao, Y., Doodes, P. D., Glant, T. T. & Finnegan, A. IL-27 induces a Th1 immune response and susceptibility to experimental arthritis. J. Immunol. 180, 922–930. https://doi.org/10.4049/jimmunol.180.2.922 (2008).
Diveu, C. et al. IL-27 blocks RORc expression to inhibit lineage commitment of Th17 cells. J. Immunol. 182, 5748–5756. https://doi.org/10.4049/jimmunol.0801162 (2009).
Pot, C. et al. Cutting edge: IL-27 induces the transcription factor c-Maf, cytokine IL-21, and the costimulatory receptor ICOS that coordinately act together to promote differentiation of IL-10-producing Tr1 cells. J. Immunol. 183, 797–801. https://doi.org/10.4049/jimmunol.0901233 (2009).
Zhang, H. et al. An IL-27-driven transcriptional network identifies regulators of IL-10 expression across T helper cell subsets. Cell Rep. 33, 108433. https://doi.org/10.1016/j.celrep.2020.108433 (2020).
Pot, C., Apetoh, L. & Kuchroo, V. K. Type 1 regulatory T cells (Tr1) in autoimmunity. Semin. Immunol. 23, 202–208. https://doi.org/10.1016/j.smim.2011.07.005 (2011).
Zeng, H., Zhang, R., Jin, B. & Chen, L. Type 1 regulatory T cells: A new mechanism of peripheral immune tolerance. Cell. Mol. Immunol. 12, 566–571. https://doi.org/10.1038/cmi.2015.44 (2015).
Batten, M. et al. Interleukin 27 limits autoimmune encephalomyelitis by suppressing the development of interleukin 17-producing T cells. Nat. Immunol. 7, 929–936. https://doi.org/10.1038/ni1375 (2006).
Kim, D. et al. Cutting edge: IL-27 attenuates autoimmune neuroinflammation via regulatory T cell/Lag3-dependent but IL-10-independent mechanisms in vivo. J. Immunol. 202, 1680–1685. https://doi.org/10.4049/jimmunol.1800898 (2019).
Molle, C. et al. IL-27 synthesis induced by TLR ligation critically depends on IFN regulatory factor 3. J. Immunol. 178, 7607–7615. https://doi.org/10.4049/jimmunol.178.12.7607 (2007).
Blahoianu, M. A., Rahimi, A. A., Kozlowski, M., Angel, J. B. & Kumar, A. IFN-gamma-induced IL-27 and IL-27p28 expression are differentially regulated through JNK MAPK and PI3K pathways independent of Jak/STAT in human monocytic cells. Immunobiology 219, 1–8. https://doi.org/10.1016/j.imbio.2013.06.001 (2014).
Rajaiah, R., Puttabyatappa, M., Polumuri, S. K. & Moudgil, K. D. Interleukin-27 and interferon-gamma are involved in regulation of autoimmune arthritis. J. Biol. Chem. 286, 2817–2825. https://doi.org/10.1074/jbc.M110.187013 (2011).
Ramgolam, V. S., Sha, Y., Jin, J., Zhang, X. & Markovic-Plese, S. IFN-beta inhibits human Th17 cell differentiation. J. Immunol. 183, 5418–5427. https://doi.org/10.4049/jimmunol.0803227 (2009).
Di Carlo, E. Decoding the role of interleukin-30 in the crosstalk between cancer and myeloid cells. Cells 9, 615. https://doi.org/10.3390/cells9030615 (2020).
Min, B., Kim, D. & Feige, M. J. IL-30(dagger) (IL-27A): A familiar stranger in immunity, inflammation, and cancer. Exp. Mol. Med. 53, 823–834. https://doi.org/10.1038/s12276-021-00630-x (2021).
Chong, W. P. et al. IL-27p28 inhibits central nervous system autoimmunity by concurrently antagonizing Th1 and Th17 responses. J. Autoimmun. 50, 12–22. https://doi.org/10.1016/j.jaut.2013.08.003 (2014).
Park, J. et al. Impact of interleukin-27p28 on T and B cell responses during toxoplasmosis. Infect. Immun. 87, e00455-19. https://doi.org/10.1128/IAI.00455-19 (2019).
Di Meo, S. et al. Interleukin-30 expression in prostate cancer and its draining lymph nodes correlates with advanced grade and stage. Clin. Cancer Res. 20, 585–594. https://doi.org/10.1158/1078-0432.CCR-13-2240 (2014).
Sorrentino, C. et al. Interleukin-30/IL27p28 shapes prostate cancer stem-like cell behavior and is critical for tumor onset and metastasization. Cancer Res. 78, 2654–2668. https://doi.org/10.1158/0008-5472.CAN-17-3117 (2018).
Shimozato, O. et al. The secreted form of p28 subunit of interleukin (IL)-27 inhibits biological functions of IL-27 and suppresses anti-allogeneic immune responses. Immunology 128, e816–e825. https://doi.org/10.1111/j.1365-2567.2009.03088.x (2009).
Korn, T. et al. Myelin-specific regulatory T cells accumulate in the CNS but fail to control autoimmune inflammation. Nat. Med. 13, 423–431. https://doi.org/10.1038/nm1564 (2007).
Dibra, D., Cutrera, J. J. & Li, S. Coordination between TLR9 signaling in macrophages and CD3 signaling in T cells induces robust expression of IL-30. J. Immunol. 188, 3709–3715. https://doi.org/10.4049/jimmunol.1100883 (2012).
Casali, B. T. & Reed-Geaghan, E. G. Microglial function and regulation during development, homeostasis and Alzheimer’s disease. Cells 10, 957. https://doi.org/10.3390/cells10040957 (2021).
Wieghofer, P., Knobeloch, K. P. & Prinz, M. Genetic targeting of microglia. Glia 63, 1–22. https://doi.org/10.1002/glia.22727 (2015).
Goldmann, T. et al. A new type of microglia gene targeting shows TAK1 to be pivotal in CNS autoimmune inflammation. Nat. Neurosci. 16, 1618–1626. https://doi.org/10.1038/nn.3531 (2013).
Awasthi, A. et al. A dominant function for interleukin 27 in generating interleukin 10-producing anti-inflammatory T cells. Nat. Immunol. 8, 1380–1389. https://doi.org/10.1038/ni1541 (2007).
Zhang, S. et al. High susceptibility to liver injury in IL-27 p28 conditional knockout mice involves intrinsic interferon-gamma dysregulation of CD4+ T cells. Hepatology 57, 1620–1631. https://doi.org/10.1002/hep.26166 (2013).
Wei, J. et al. Critical role of dendritic cell-derived IL-27 in antitumor immunity through regulating the recruitment and activation of NK and NKT cells. J. Immunol. 191, 500–508. https://doi.org/10.4049/jimmunol.1300328 (2013).
de Aquino, M. T. et al. IL-27 limits central nervous system viral clearance by promoting IL-10 and enhances demyelination. J. Immunol. 193, 285–294. https://doi.org/10.4049/jimmunol.1400058 (2014).
Sonobe, Y. et al. Production of IL-27 and other IL-12 family cytokines by microglia and their subpopulations. Brain Res. 1040, 202–207. https://doi.org/10.1016/j.brainres.2005.01.100 (2005).
Yona, S. et al. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38, 79–91. https://doi.org/10.1016/j.immuni.2012.12.001 (2013).
Stumhofer, J. S. et al. A role for IL-27p28 as an antagonist of gp130-mediated signaling. Nat. Immunol. 11, 1119–1126. https://doi.org/10.1038/ni.1957 (2010).
Do, J. et al. Treg-specific IL-27Ralpha deletion uncovers a key role for IL-27 in Treg function to control autoimmunity. Proc. Natl. Acad. Sci. U.S.A. 114, 10190–10195. https://doi.org/10.1073/pnas.1703100114 (2017).
Do, J. S. et al. An IL-27/Lag3 axis enhances Foxp3+ regulatory T cell-suppressive function and therapeutic efficacy. Mucosal Immunol. 9, 137–145. https://doi.org/10.1038/mi.2015.45 (2016).
Pflanz, S. et al. IL-27, a heterodimeric cytokine composed of EBI3 and p28 protein, induces proliferation of naive CD4+ T cells. Immunity 16, 779–790. https://doi.org/10.1016/s1074-7613(02)00324-2 (2002).
Stumhofer, J. S. et al. Interleukin 27 negatively regulates the development of interleukin 17-producing T helper cells during chronic inflammation of the central nervous system. Nat. Immunol. 7, 937–945. https://doi.org/10.1038/ni1376 (2006).
Petes, C., Mariani, M. K., Yang, Y., Grandvaux, N. & Gee, K. Interleukin (IL)-6 inhibits IL-27- and IL-30-mediated inflammatory responses in human monocytes. Front. Immunol. 9, 256. https://doi.org/10.3389/fimmu.2018.00256 (2018).
Hasegawa, H. et al. Expanding diversity in molecular structures and functions of the IL-6/IL-12 heterodimeric cytokine family. Front. Immunol. 7, 479. https://doi.org/10.3389/fimmu.2016.00479 (2016).
Garbers, C. et al. An interleukin-6 receptor-dependent molecular switch mediates signal transduction of the IL-27 cytokine subunit p28 (IL-30) via a gp130 protein receptor homodimer. J. Biol. Chem. 288, 4346–4354. https://doi.org/10.1074/jbc.M112.432955 (2013).
Yang, X. et al. Soluble IL-6 receptor and IL-27 subunit p28 protein complex mediate the antiviral response through the type III IFN pathway. J. Immunol. 197, 2369–2381. https://doi.org/10.4049/jimmunol.1600627 (2016).
Rose-John, S. IL-6 trans-signaling via the soluble IL-6 receptor: Importance for the pro-inflammatory activities of IL-6. Int. J. Biol. Sci. 8, 1237–1247. https://doi.org/10.7150/ijbs.4989 (2012).
Kallen, K. J. The role of transsignalling via the agonistic soluble IL-6 receptor in human diseases. Biochim. Biophys. Acta 1592, 323–343. https://doi.org/10.1016/s0167-4889(02)00325-7 (2002).
Padberg, F. et al. CSF and serum levels of soluble interleukin-6 receptors (sIL-6R and sgp130), but not of interleukin-6 are altered in multiple sclerosis. J. Neuroimmunol. 99, 218–223. https://doi.org/10.1016/s0165-5728(99)00120-4 (1999).
Murugaiyan, G., Mittal, A. & Weiner, H. L. Identification of an IL-27/osteopontin axis in dendritic cells and its modulation by IFN-gamma limits IL-17-mediated autoimmune inflammation. Proc. Natl. Acad. Sci. U.S.A. 107, 11495–11500. https://doi.org/10.1073/pnas.1002099107 (2010).
Tanaka, T. et al. Interleukin-27 induces the endothelial differentiation in Sca-1+ cardiac resident stem cells. Cytokine 75, 365–372. https://doi.org/10.1016/j.cyto.2015.06.009 (2015).
Crabe, S. et al. The IL-27 p28 subunit binds cytokine-like factor 1 to form a cytokine regulating NK and T cell activities requiring IL-6R for signaling. J. Immunol. 183, 7692–7702. https://doi.org/10.4049/jimmunol.0901464 (2009).
Hooper, K. M. et al. Prostaglandin E2 inhibition of IL-27 production in murine dendritic cells: A novel mechanism that involves IRF1. J. Immunol. 198, 1521–1530. https://doi.org/10.4049/jimmunol.1601073 (2017).
Pirhonen, J., Siren, J., Julkunen, I. & Matikainen, S. IFN-alpha regulates Toll-like receptor-mediated IL-27 gene expression in human macrophages. J. Leukoc. Biol. 82, 1185–1192. https://doi.org/10.1189/jlb.0307157 (2007).
Muller, S. I. et al. A folding switch regulates interleukin 27 biogenesis and secretion of its alpha-subunit as a cytokine. Proc. Natl. Acad. Sci. U.S.A. 116, 1585–1590. https://doi.org/10.1073/pnas.1816698116 (2019).
Kilgore, A. M. et al. IL-27p28 production by XCR1(+) dendritic cells and monocytes effectively predicts adjuvant-elicited CD8(+) T cell responses. Immunohorizons 2, 1–11. https://doi.org/10.4049/immunohorizons.1700054 (2018).
Hwang, M. et al. Distinct CD4 T-cell effects on primary versus recall CD8 T-cell responses during viral encephalomyelitis. Immunology 144, 374–386. https://doi.org/10.1111/imm.12378 (2015).
Boza-Serrano, A., Yang, Y., Paulus, A. & Deierborg, T. Innate immune alterations are elicited in microglial cells before plaque deposition in the Alzheimer’s disease mouse model 5xFAD. Sci. Rep. 8, 1550. https://doi.org/10.1038/s41598-018-19699-y (2018).
Funding
This study was supported by grants from NIH AI125247 and NMSS RG 1411-02051 (to B.M.).
Author information
Authors and Affiliations
Contributions
D.K. designed and performed most of the experiments, analyzed the data, and wrote the manuscript. S.K. and M.K. performed experiments shown in part of the Figs. 5 and 6. Z.Y. provided key reagents. B.M. designed the experiments, analyzed the data, and wrote the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, D., Kim, S., Kang, Ms. et al. Cell type specific IL-27p28 (IL-30) deletion in mice uncovers an unexpected regulatory function of IL-30 in autoimmune inflammation. Sci Rep 13, 1812 (2023). https://doi.org/10.1038/s41598-023-27413-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-27413-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.