Abstract
Alcohol use is an important health issue and has been suggested to contribute to the burden produced by obesity. Both alcohol use and obesity are subject to sex differences. The available studies on the relationship between alcohol use and body mass index (BMI) report inconsistent results with positive, negative, and null findings which requests a meta-analytic approach. Therefore, we conducted a meta-analysis of case–control, cohort, and cross-sectional studies. The systematic literature search and data extraction was performed by 3 independent raters. We conducted sex-separated meta-analyses and -regressions to investigate how alcohol consumption associates with BMI. Our systematic literature search resulted in 36 studies with 48 data sets (Nmen = 172,254; kmen = 30; Nwomen = 24,164; kwomen = 18; Nunknown sex = 672,344; kunknown sex = 24). Alcohol use was associated with higher BMI in men (g = 0.08 [0.07; 0.09]) and lower BMI in women (g = − 0.26 [− 0.29; − 0.22]). Moreover, we found the amount of daily alcohol intake in men (β = 0.001 [0.0008; 0.0014]) and ethnicity in women (g[Caucasians] = − 0.45 versus g[Asians] = − 0.05; z = 11.5, p < 0.0001) to moderate these effects. We here identified sex-diverging relationships between alcohol use and BMI, found daily alcohol intake and ethnicity to sex-specifically moderate these effects, and argue that sex-specific choice of beverage type and higher amount of daily alcohol use in men than in women account for these observations. Future research is needed to provide empirical evidence for the underlying mechanisms.
Similar content being viewed by others
Introduction
For 2016, the World Health Organization (WHO)1 estimates a total alcohol per capita consumption of 6.4 L. Harmful use of alcohol accounted for approximately 3 million deaths (= 5.3% of all deaths) and 132.6 million disability-adjusted life years (DALYs) (= 5.1% of all DALYs). Moreover, high prevalence rates for harmful alcohol use and dependence of 8.8% and 8.2% are estimated in the WHO European Region and the WHO Region of the Americas. Women vary from men in their alcohol use patterns and risk of alcohol use disorder (AUD); also, sex differences in mechanisms underlying alcohol use and AUD have been reported2,3,4,5,6,7,8,9,10,11,12,13,14,15,16.
Obesity is a health concern, particularly in the Western world. As alcohol contains much energy (29 kJ per gram alcohol) and most people in Europe (59.9%) and the Americas (54.1%) drink alcohol1, it is speculated that alcohol contributes to the obesity problem. However, the association between alcohol intake and body mass index (BMI) is not that simple. Ethanol intake inhibits different insulin actions in the body17 with the possible long-term consequence of a generalized insulin resistance17,18 contributing to overweight. Furthermore, obesity and AUD share common etiopathogenetic mechanisms such as mesolimbic dysfunction19,20,21,22 with lower dopamine D2 receptor availability23, increased cue-reactivity24,25, and genetics26,27. Similar to alcohol use, BMI and obesity vary significantly between women and men with higher rates of obesity in the female group28.
A systematic review published in 2011 addressed the association between alcohol intake and gain of weight29. The authors’ evaluation of large cross-sectional and well-powered prospective studies with long follow-up periods revealed contradictory results. However, they assumed a positive association to be more likely in men (vs. women) and in heavy drinkers, whereas there were negative or no associations in moderate consumers. Indeed, also studies that were published more recently demonstrate positive and negative relationships as well as null findings30,31,32. Negative associations might be explained by the greater thermogenic effect of alcohol than carbohydrates or fat with a lower net efficiency33. Initial evidence also indicates that sex differences (e.g., in choice of beverage type and drinking patterns, which influence the energy intake) might account for the ambiguous relationship between alcohol intake and BMI29. A more recent systematic review and meta-analysis published in 2022 showed that alcohol consumption increased the risk of obesity in adults 2.05 times compared to non-drinkers34, but did not distinguish between moderate vs. heavy drinkers or men vs. women. Another recent meta-analysis that focused on overweight, obese, or abdominally obese subjects again reports higher odds of obesity and overweight in heavy drinkers, but did not include normal weight samples35.
Thus, we here present a systematic review and meta-analysis to investigate how alcohol consumption is related to BMI while also exploring the moderating influence of sex, the amount of alcohol consumed, the percentage of smokers, and differences between ethnicities. Since the existing literature is inconclusive, we decided to follow an exploratory approach.
Materials and methods
Search strategy and study selection
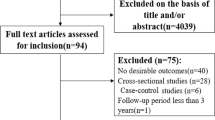
We conducted a two-step literature search from study inception until February 4th, 2021, using PubMed and Google Scholar including case–control, cohort, and cross-sectional studies. In the course of the review process, we updated this literature search from study inception until September 9th, 2022. Due to the enormous number of results when applying a full-text search strategy (> 1,150,000 hits), we limited our search strategy exclusively to titles. The search terms alcohol dependence, alcohol use disorder, binge drinking, alcohol, social consumption, social drinking, withdrawal, alcohol intoxication, seizure, delirium, AUDIT, CAGE, beer, wine, liquor were used to represent alcohol intake and were combined with the search terms BMI and body mass index. In a second step, the reference lists of retrieved articles were searched manually for further eligible titles. All abstracts were screened applying the selection criteria that are detailed in our coding protocol (Supplementary Table S1). The remaining articles were checked for eligibility according to the Preferred Reporting Items for Systemic Reviews and Meta-analyses (PRISMA) statement36 on the basis of a full-text review and the entire literature search was summarized according to these PRISMA guidelines37.
Data extraction
The data extraction process strictly followed our approach described in Siegmann et al.11 and Siegmann et al.38 and was performed by two out of three investigators (C. M., E. S., M. M.) for different portions of the publications each. All recorded variables can be found in the previously defined coding protocol (Supplementary Table S1). Disagreement was resolved by discussion and compromise on the eventually extracted values. We assessed the risk of bias with either the Newcastle–Ottawa Scale for case–control studies39 or an adaptation of the Newcastle–Ottawa Scale for cohort studies39, which was specifically designed for cross-sectional studies by Herzog et al.40 in their systematic review. The final risk of bias values were obtained by averaging the extractors’ values.
Statistical analysis
All analyses were conducted and all figures were made using the metafor package41 within the open-source software environment R, version 4.1.142 and GraphPad Prism 8.4.3 (Graph Pad Soft- ware Inc., San Diego, CA, USA).
We estimated the standardized mean difference (Hedges’ g) in BMI among drinking subjects and control subjects and, in a second step, explored the influence of sex. Following our coding protocol (Supplementary Table S1) we distinguished between data from males, data from females, and data from studies not reporting sex-separated measures (“unknown group”).
In order to combine studies reporting different measures of effect, correlative and odds ratio data were transformed into Hedges’ g using common transformation formulas43. The drinking group was characterized by various measures: (1) alcohol consumption levels in grams per day, (2) blood alcohol levels, (3) frequency of alcohol drinking, (4) score in the AUDIT questionnaire44, (5) being diagnosed with AUD by a psychiatrist, (6) score in the Obsessive–Compulsive Drinking Scale, German version (OCDS-G)45, (7) frequency of hospital readmissions following withdrawal treatment, and (8) being classified as binge drinkers according to a definition by Patrick et al.46. The control group either consisted of never-drinkers and non-binge-drinkers or was defined as consuming < 1 drink per week, drinking < 1 day per week, or scoring below the cut-off value of the AUDIT questionnaire44.
Owing to our study design which compared more than one variation of drinking status (e.g., light, moderate, and heavy drinking) against a single control group, the meta-analytic effect size estimates are correlated and non-independent, respectively. To account for this repeated usage of one common control group, we performed a multivariate random-effects meta-analysis recommended for the analysis of multiple-treatment studies47,48. The Q-statistic is reported as a measure for heterogeneity. We decided against conventional ad-hoc approaches, e.g., averaging multiple reported effects, since these would have resulted in loss of useful information for moderator analyses47. We ran prespecified multivariate meta-regressions for the moderators drinking amount in grams per day, percentage of smokers in the drinking group, and study quality (i.e., the risk of bias in these studies). We also tested whether drinking amount is suited as a quadratic predictor. All meta-regressions were Bonferroni-corrected for multiple testing. Since ethnicity is a variable that influences both alcohol-related behavior49,50 as well as BMI or body fat51, we investigated the difference in the outcome measure among the ethnicities Caucasian, Asian, African, and Hispanic in a prespecified subgroup analysis. Based on the aforementioned results by Sayon-Orea et al.29 concerning the influence of different beverage types, we planned to run another prespecified subgroup analysis distinguishing between beer, wine, liquor, and mixed alcohol consumption.
Small study effects were assessed by visual detection of asymmetries in a contour-enhanced funnel plot52,53. The sensitivity of our analysis was evaluated by comparing models with and without effect sizes which we assume to be influential outliers54. They were detected following an exploratory data analysis55. p < 0.05 (2-sided) was considered statistically significant.
Results
Eligible studies
The literature search is summarized in the PRISMA flow chart (Fig. 1). We identified 36 articles13,31,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89 comprising 48 independent samples. The characteristics of all included studies are detailed in Table 1.
Meta-analytic results and moderator analyses
Since sensitivity analyses revealed one substantially influential outlier78, we conducted our analysis without this study. When analyzing all of the studies together we found a very small positive association between alcohol use and BMI (g = 0.01, 95% CI [0.01; 0.02]) with a high heterogeneity index (Q = 1139.74, p < 0.0001). Including the factor sex as a moderator revealed a significant moderating effect for male (z = 0.08, p < 0.0001) and female sex (z = − 0.26, p < 0.0001) while the group of studies with unknown sex distribution remained non-significant (z = 0.004, p = 0.27). This indicates that sex substantially influences the effect size and we therefore conducted sex-separated analyses: while in men the association was slightly positive (g = 0.08, 95% CI [0.07; 0.09]), we found a negative effect in women (g = − 0.26, 95% CI [− 0.29; − 0.22]). In both samples, heterogeneity was rather high (Q = 237.90 for women, Q = 106.21 for men, p < 0.0001 for both) indicating that these models are not yet of adequate fit for the data and require further moderator analyses. The sex-separated results in comparison to the overall results are shown in Figs. 2 and 3.
Forest plot of the standardized mean difference in BMI among male drinking and non-drinking subjects. This plot shows the results of the individual studies examining males together with their 95% confidence interval (CI). The weight of each study contributing to the overall effect is illustrated by the size of the square. The summary polygons at the bottom of the plot show the results from the multivariate meta-analytic model for (1) the subgroup of males and for (2) all included studies.
Forest plot of the standardized mean difference in BMI among female drinking and non-drinking subjects. This plot shows the results of the individual studies examining females together with their 95% confidence interval (CI). The weight of each study contributing to the overall effect is illustrated by the size of the square. The summary polygons at the bottom of the plot show the results from the multivariate meta-analytic model for (1) the subgroup of females and for (2) all included studies.
Bearing this sex-diverging result in mind, we first analyzed the interaction of sex and ethnicity before conducting a subgroup analysis for this moderator. It revealed a significant interaction of Asian ethnicity with female sex (z = 0.39, p < 0.0001) again suggesting that analyses concerning ethnicity should be performed sex-separately. No African and only one Hispanic study provided sex-separated data; therefore, sex-separated subgroup analyses were limited to Asian vs. Caucasian participants. In men, these ethnic groups did not differ significantly (z = 1.35, p = 0.51), whereas in women, the negative association between alcohol consumption and BMI was more pronounced in Caucasian than in Asian participants (g = − 0.45 compared to g = − 0.05; z = 11.5, p < 0.0001).
Due to insufficient data, it was not possible to compute the moderator analysis distinguishing between different beverage types: only one study64 reported separate BMI data for beer, wine, and liquor drinkers.
Meta-regression analyses
Since all our meta-analytic results revealed a strong influence of sex, we also performed sex-separated meta-regression analyses. The threshold for significant results was Bonferroni-corrected at p = 0.006.
The meta-regression analysis concerning the amount of alcohol in gram per day revealed a significant moderating influence on the effect sizes of the overall sample (β = 0.0009, p < 0.0001) and of men (β = 0.001, p < 0.0001). This result suggests that in men with every additional gram of alcohol per day the association of alcohol use and BMI increases by 0.001. In women, the meta-regression was not significant (β = 0.002, p = 0.07). The addition of a quadratic term did not explain for more variance than the linear term (no U-shaped association detectable) (data not shown). In our sample, the mean alcohol consumption was 23.14 g/day for men and 13.82 g/day for women.
Ethanol intake and smoking was correlated in our sample, insofar that the amount of alcohol (g/day) and the percentage of smokers in the drinking group was moderately associated for males (r = 0.39, p = 0.097) and highly associated for females (r = 0.92, p = 0.003). The meta-regression regarding the percentage of smokers revealed a non-significant influence on the effect size (males: β = 0.06, p = 0.05; females: β = − 0.52, p = 0.09).
The mean study quality assessed via the Newcastle–Ottawa-Scale39,40 was 5.37 ± 0.79. The meta-regression analyses regarding study quality remained non-significant for the male sample (β = − 0.019, p = 0.11), but suggest an influence of the studies’ risk of bias on the female (β = 0.350, p < 0.0001) and the overall effect size (β = − 0.038, p < 0.0001).
Small study effects and sensitivity analyses
When visually examining the funnel plot (Supplementary Fig. S1), no evidence of small study effects or publication bias was detectable. Sensitivity analyses revealed one influential outlier78 whose inclusion biased the results substantially, especially in terms of heterogeneity (result before exclusion: g = − 0.04, 95% CI [− 0.05, − 0.04], Q = 115,516.38; after exclusion: g = 0.01, 95% CI [0.01; 0.02], Q = 1139.74). Therefore, this study was excluded from all analyses.
Discussion
To our knowledge, this is the first systematic literature search and meta-analysis to investigate how alcohol use associates with BMI in normal to overweight individuals while focusing on possible sex differences. We found a sex-diverging relationship of small size: whereas alcohol use was slightly related to higher BMI in men, it was more strongly linked to lower BMI in women. Thus, our meta-analytic findings provide empirical evidence to confirm the previous assumption of a systematic review29 that alcohol use is to a small extent, but positively associated with BMI in men; to this state of knowledge, we also add that alcohol use in women is related to a lower BMI. This also matches data of a recent study indicating that higher BMI is related to an increased risk for hospital readmissions in male in-patients with AUD, while it tends to be protective in female in-patients90. When analyzing both sexes together, our results match previous meta-analyses suggesting that higher alcohol intake associates with higher BMI34,35. In terms of practical relevance, the small effect sizes of up to Hedges’ g = − 0.26 mean that the BMI of approximately 60% of female drinkers is below the average BMI of non-drinking women91. One has to bear in mind that with a non-overlap of approximately 15% between drinkers and non-drinkers the practical impact of these findings is small91. Nevertheless, these effects raise the question which mechanisms underlie the sex-diverging relationships between alcohol use and BMI. In moderator analyses, we found significant and sex-separated effects of the amount of daily ethanol intake and ethnicity.
The amount of alcohol consumed daily influenced the BMI in men. This supports the assumption that caloric intake due to alcohol consumption leads to higher BMI in men. Sayon-Orea et al.29 suggest in their review that the caloric impact of alcohol use (represented by a positive association of BMI and alcohol use) is only evident in subjects who drink more often and in larger quantity. On average, males consume more alcohol than females1 which also holds true for the here analyzed sample (mean[males] = 23.14 g/day; mean[females] = 13.82 g/day). It is possible that a part of the sex-diverging effect found here is attributable to the differences in the mean ethanol intake between men and women. Accordingly, we did not observe such a significant moderating effect in the female samples, which suggests that either their drinking quantity was too low or that mechanisms other than calorie supply are relevant in women. A further explanation of the positive association found in men might be the toxic effect of alcohol on different body functions. Regular alcohol consumption can lead to a state of generalized insulin resistance by inhibiting, for example, glucose disposal or insulin release in men17. This resistance is commonly paralleled by higher body weight18,92.
We aimed at testing whether sex differences in choice of beverage type account for the sex diverging relationships of alcohol use and BMI. However, we were not able to provide meta-analytic support of this association here, as our systematic literature search identified only one early paper that provided beverage type-specific data64. This study indicates higher BMI in liquor than in wine drinkers of both sexes. Similarly, Sayon-Orea et al.29 found that beer and liquor consumption (≥ 7 drinks/week) are associated with weight gain, whereas no such effect was found for wine consumption. We also analyzed correlations between beverage-type specific alcohol drinking and BMI in an additional data set7 and found a positive correlation between liquor intake and BMI in male patients with AUD (see Supplementary Fig. S2). As men prefer liquor7,93, the higher caloric intake associated with liquor consumption (which is more often found in men vs. women) might account for our observation of higher BMI in men with alcohol use vs. men who deny alcohol consumption. Furthermore, the sex-specific choice of beverage type might also help to explain why our meta-analysis demonstrates lower BMI in women with alcohol use. Consumption of wine, but not use of beer or liquor, is related to more frequent exercising in both sexes94 and to a more healthy dietary behavior95,96. Choice of beverage type was also suggested to associate with intake of fat, carbohydrate, and vitamins64. Women more often choose wine7,93 and thus the alcohol intake in women is expected to be related to more frequent exercising and possibly to a more healthy life-style which might account for the here observed lower BMI in alcohol-using women. However, future research is needed to study the mechanisms underlying the identified sex-diverging association between alcohol use and BMI.
A higher percentage of smokers in the drinking group amplified the sex-diverging association of alcohol use and BMI but remained non-significant. Usually, smoking is associated with weight loss and lower BMI80,97 as in our female sample. However, more recent studies with large sample sizes suggest that this relationship is less clear, especially in obese persons98,99, corresponding to the results in our male sample. Weinland et al.13 also reported an amplifying effect of active smoking status which might be due to a positive association of smoking and alcohol consumption in general61,80. In our study, these two variables were highly correlated, as well. It is possible that the results of this meta-regression reflect the results for subjects drinking in higher quantity since subgroups comprising more smokers consumed more alcohol here.
We here also provide first meta-analytic evidence that ethnicity modulates the sex-separated relationship between alcohol use and BMI. The effect size of the lower BMI values in alcohol-drinking vs. control women was stronger in the Caucasian than in the Asian subsample. It is well-established that genetics influences the response to alcohol and the vulnerability to develop AUD100,101, and this might explain the here observed inter-ethnic variation of the relationship between alcohol use and BMI.
Limitations and strengths
There are some limitations to this study. First, the literature search was restricted to titles resulting in a smaller number of eligible studies. We tried to extend the literature search to abstracts or a full-text search, but the number of results was too high to be economically screened for eligibility. Additionally, the literature search only identified two studies addressing patients with AUD13,59; thus, it remains to be shown whether the here reported associations could be generalized to AUD. Third, we could not properly test for an inverted U-shaped relationship between the amount of alcohol consumption and BMI since we lacked studies with highly consuming participants. Even if there was an inverted U-shaped association between these two variables, our dataset with light to moderate drinkers would only be able to detect the ascending slope up to the point of inflection. Fourth, all effect sizes computed in this study are small following Cohen102. Consequently, they do not reach the extent of the minimal clinically relevant difference defined by Sayon-Orea et al.29. Fifth, the usage of a multivariate meta-analytic model might also involve some limitations103. It assumes, for example, that missing values are missing at random which is not always true, especially when they are missing due to non-significance. Furthermore, additional modeling assumptions are required and harder to verify than in univariate meta-analyses. Applying the multivariate model also entails numerous strengths. It uses more information improving statistical properties such as smaller mean-square error and greater precision104. It also allows for accounting for covariances between pooled estimates and for reducing reporting bias when some outcomes are selectively missing. A detailed summary of advantages and disadvantages of multivariate meta-analysis can be found in Jackson et al.103. Further strengths of this study are the strict adherence to the standardized PRISMA guidelines36,37 (see Supplementary Table S2), the probable absence of small study effects, the reasonably robust results revealed by our sensitivity analyses and by the stability of our results after updating the literature search, and the rather representative control group. In case–control studies, a control group is usually defined by the strict absence of the variable of interest105. Concerning the variable alcohol use, this is not typical for the average population where, for example, only 23.5% are lifetime abstainers in the European region1. Our control subjects are defined by different measures (see section “Statistical analyses”) and therefore, are not classed among ”super healthy” controls. Thus, our results can more easily be generalized to the general population105. This also holds true for our drinking group with especially women being comparable to the general population in terms of their mean ethanol intake (14 g/day here compared to 15 g/day among females worldwide1). Additionally, this is, up to our knowledge, the first meta-analysis specifically examining the association of alcohol consumption and BMI for different ethnicities.
Conclusion
As far as we know, this is the first meta-analysis to show that alcohol use associates with higher BMI in men and lower BMI in women. The effect size is lower in female Asians than Caucasians and it is influenced by the amount of alcohol consumption per day. However, our effect sizes did not reach the extent of the minimal clinically relevant difference defined by a previous review29. The differences in average daily alcohol consumption between men and women as well as sex-specific choice of beverage linked to life style factors such as exercise and intake of fat and carbohydrates might account for these sex-diverging effects. This interpretation requests validation by future studies. Our findings lay the foundation of further studies investigating mechanisms underlying alcohol use.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
World Health Organization. Global Status Report on Alcohol and Health 2018 (World Health Organization, 2019).
Hansson, A. C. et al. Oxytocin reduces alcohol cue-reactivity in alcohol-dependent rats and humans. Neuropsychopharmacology 43, 1235–1246. https://doi.org/10.1038/npp.2017.257 (2018).
Hansson, A. C. & Spanagel, R. No changes in the oxytocin system in alcohol-dependent female rodents and humans: Towards a sex-specific psychopharmacology in alcoholism. Addict. Biol. 26, e12945. https://doi.org/10.1111/adb.12945 (2021).
Lenz, B. et al. Prenatal and adult androgen activities in alcohol dependence. Acta Psychiatr. Scand. 136, 96–107. https://doi.org/10.1111/acps.12725 (2017).
Lenz, B., Köllner, M. G., Mühle, C., Weinland, C. & Kornhuber, J. Basic human body dimensions relate to alcohol dependence and predict hospital readmission. J. Clin. Med. 8, 2076. https://doi.org/10.3390/jcm8122076 (2019).
Lenz, B. et al. Sex hormone activity in alcohol addiction: Integrating organizational and activational effects. Progress Neurobiol. 96, 136–163. https://doi.org/10.1016/j.pneurobio.2011.11.001 (2012).
Lenz, B. et al. Oxytocin blood concentrations in alcohol use disorder: A cross-sectional, longitudinal, and sex-separated study. Eur. Neuropsychopharmacol. 51, 55–67. https://doi.org/10.1016/j.euroneuro.2021.04.015 (2021).
Mühle, C., Barry, B., Weinland, C., Kornhuber, J. & Lenz, B. Estrogen receptor 1 gene variants and estradiol activities in alcohol dependence. Progress Neuro-psychopharmacol. Biol. Psychiatry. 92, 301–307. https://doi.org/10.1016/j.pnpbp.2019.01.008 (2019).
Mühle, C., Weinland, C., Gulbins, E., Lenz, B. & Kornhuber, J. Peripheral acid sphingomyelinase activity is associated with biomarkers and phenotypes of alcohol use and dependence in patients and healthy controls. Int. J. Mol. Sci. 19, 4028. https://doi.org/10.3390/ijms19124028 (2018).
Müller, C. P., Mühle, C., Kornhuber, J. & Lenz, B. Sex-dependent alcohol instrumentalization goals in non-addicted alcohol consumers versus patients with alcohol use disorder: Longitudinal change and outcome prediction. Alcoholism 45, 577–586. https://doi.org/10.1111/acer.14550 (2021).
Siegmann, E.-M., Bouna-Pyrrou, P., Lenz, B. & Kornhuber, J. Digit ratio (2D:4D) in relation to substance and computer use: A meta-analysis. J. Neural Transmission. 126, 623–636. https://doi.org/10.1007/s00702-019-02002-2 (2019).
Weinland, C., Braun, B., Mühle, C., Kornhuber, J. & Lenz, B. Cloninger type 2 score and Lesch typology predict hospital readmission of female and male alcohol-dependent inpatients during a 24-month follow-up. Alcoholism 41, 1760–1767. https://doi.org/10.1111/acer.13468 (2017).
Weinland, C., Mühle, C., Kornhuber, J. & Lenz, B. Body mass index and craving predict 24-month hospital readmissions of alcohol-dependent in-patients following withdrawal. Progress Neuro-psychopharmacol. Biol. Psychiatry. 90, 300–307. https://doi.org/10.1016/j.pnpbp.2018.11.019 (2019).
Weinland, C., Mühle, C., Kornhuber, J. & Lenz, B. Crossed eye/hand laterality and left-eyedness predict a positive 24-month outcome in alcohol-dependent patients. Alcoholism 43, 1308–1317. https://doi.org/10.1111/acer.14051 (2019).
Weinland, C., Mühle, C., Kornhuber, J. & Lenz, B. Progesterone serum levels correlate negatively with craving in female postmenopausal in-patients with alcohol use disorder: A sex- and menopausal status-separated study. Progress Neuro-psychopharmacol. Biol. Psychiatry. 110, 110278. https://doi.org/10.1016/j.pnpbp.2021.110278 (2021).
Weinland, C., Mühle, C., von Zimmermann, C., Kornhuber, J. & Lenz, B. Sulphated dehydroepiandrosterone serum levels are reduced in women with alcohol use disorder and correlate negatively with craving: A sex-separated cross-sectional and longitudinal study. Addict. Biol. 27, e13135. https://doi.org/10.1111/adb.13135 (2022).
Boden, G., Chen, X., DeSantis, R. A. & Kendrick, Z. Ethanol inhibits insulin action on lipolysis and on insulin release in elderly men. Am. J. Physiol. 265, 197–202. https://doi.org/10.1152/ajpendo.1993.265.2.E197 (1993).
de La Monte, S., Derdak, Z. & Wands, J. R. Alcohol, insulin resistance and the liver–brain axis. J. Gastroenterol. Hepatol. 27(Suppl 2), 33–41. https://doi.org/10.1111/j.1440-1746.2011.07023.x (2012).
Volkow, N. D. & Wise, R. A. How can drug addiction help us understand obesity?. Nat. Neurosci. 8, 555–560. https://doi.org/10.1038/nn1452 (2005).
Bowirrat, A. & Oscar-Berman, M. Relationship between dopaminergic neurotransmission, alcoholism, and Reward Deficiency syndrome. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 132B, 29–37. https://doi.org/10.1002/ajmg.b.30080 (2005).
Volkow, N. D., Fowler, J. S., Wang, G.-J. & Goldstein, R. Z. Role of dopamine, the frontal cortex and memory circuits in drug addiction: Insight from imaging studies. Neurobiol. Learn. Memory. 78, 610–624. https://doi.org/10.1006/nlme.2002.4099 (2002).
Verdejo-Román, J., Vilar-López, R., Navas, J. F., Soriano-Mas, C. & Verdejo-García, A. Brain reward system’s alterations in response to food and monetary stimuli in overweight and obese individuals. Hum. Brain Mapping. 38, 666–677. https://doi.org/10.1002/hbm.23407 (2017).
Martinez, D. et al. Alcohol dependence is associated with blunted dopamine transmission in the ventral striatum. Biol. Psychiatry. 58, 779–786. https://doi.org/10.1016/j.biopsych.2005.04.044 (2005).
Filbey, F. M., Myers, U. S. & DeWitt, S. Reward circuit function in high BMI individuals with compulsive overeating: Similarities with addiction. Neuroimage 63, 1800–1806. https://doi.org/10.1016/j.neuroimage.2012.08.073 (2012).
Garcia-Garcia, I. et al. Reward processing in obesity, substance addiction and non-substance addiction. Obes. Rev. 15, 853–869. https://doi.org/10.1111/obr.12221 (2014).
Lichenstein, S. D. et al. Familial risk for alcohol dependence and developmental changes in BMI: The moderating influence of addiction and obesity genes. Pharmacogenomics 15, 1311–1321. https://doi.org/10.2217/pgs.14.86 (2014).
Ehlers, C. L. & Wilhelmsen, K. C. Genomic screen for substance dependence and body mass index in southwest California Indians. Genes Brain Behav. 6, 184–191. https://doi.org/10.1111/j.1601-183X.2006.00246.x (2007).
Flegal, K. M., Kruszon-Moran, D., Carroll, M. D., Fryar, C. D. & Ogden, C. L. Trends in obesity among adults in the United States, 2005 to 2014. JAMA 315, 2284–2291. https://doi.org/10.1001/jama.2016.6458 (2016).
Sayon-Orea, C., Martinez-Gonzalez, M. A. & Bes-Rastrollo, M. Alcohol consumption and body weight: A systematic review. Nutr. Rev. 69, 419–431. https://doi.org/10.1111/j.1753-4887.2011.00403.x (2011).
Nies, M. A., Sun, L., Kazemi, D., Carriker, A. & Dmochowski, J. Relationship of body mass index to alcohol consumption in college freshmen. TheScientificWorldJOURNAL 2012, 849018. https://doi.org/10.1100/2012/849018 (2012).
Bouna-Pyrrou, P., Mühle, C., Kornhuber, J., Weinland, C. & Lenz, B. Body mass index and serum levels of soluble leptin receptor are sex-specifically related to alcohol binge drinking behavior. Psychoneuroendocrinology 127, 105179. https://doi.org/10.1016/j.psyneuen.2021.105179 (2021).
Wills, A. G., Evans, L. M. & Hopfer, C. Phenotypic and genetic relationship between BMI and drinking in a sample of UK adults. Behav. Genet. 47, 290–297. https://doi.org/10.1007/s10519-017-9838-2 (2017).
Schutz, Y. Role of substrate utilization and thermogenesis on body-weight control with particular reference to alcohol. Proc. Nutr. Soc. 59, 511–517. https://doi.org/10.1017/s0029665100000744 (2000).
Wulandari, A. N. & Hanida, A. N. Meta-analysis: Relationship between alcohol consumption and age on obesity in adults. J. Epidemiol. Public Health. 7, 153–166. https://doi.org/10.26911/jepublichealth.2022.07.02.01 (2022).
Golzarand, M., Salari-Moghaddam, A. & Mirmiran, P. Association between alcohol intake and overweight and obesity: A systematic review and dose–response meta-analysis of 127 observational studies. Crit. Rev. Food Sci. Nutr. 62, 8078–8098. https://doi.org/10.1080/10408398.2021.1925221 (2022).
Liberati, A. et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. BMJ 339, b2700. https://doi.org/10.1016/j.jclin (2009).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 6, e100097. https://doi.org/10.1016/j.ijsu.2010.02.007 (2010).
Siegmann, E.-M. et al. Digit ratio (2D:4D) and transgender identity: New original data and a meta-analysis. Sci. Rep. 10, 19326. https://doi.org/10.1038/s41598-020-72486-6 (2020).
Wells, G. A. et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 3 November 2018.
Herzog, R. et al. Are healthcare workers’ intentions to vaccinate related to their knowledge, beliefs and attitudes? A systematic review. BMC Public Health 13, 1–17. https://doi.org/10.1186/1471-2458-13-154 (2013).
Viechtbauer, W. Conducting meta-analyses in R with the metafor package. J. Stat. Softw. 36, 1–48. https://doi.org/10.18637/jss.v036.i03 (2010).
R Core Team. R: A Language and Environment for Statistical Computing}. https://www.R-project.org/ (2021). Accessed 4 January 2019.
Borenstein, M., Hedges, L. V., Higgins, J. P. & Rothstein, H. R. Introduction to Meta-analysis 45–49 (Wiley, 2009).
Saunders, J. B., Aasland, O. G., Babor, T. F., La Fuente, J. R. de & Grant, M. Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption—II. Addiction (Abingdon, England). 88, 791–804. https://doi.org/10.1111/j.1360-0443.1993.tb02093.x (1993).
Mann, K. & Ackermann, K. Die OCDS-G: Psychometrische Kennwerte der deutschen Version der Obsessive Compulsive Drinking Scale. SUCHT 46, 90–100. https://doi.org/10.1024/suc.2000.46.2.90 (2000).
Patrick, M. E. et al. Extreme binge drinking among 12th-grade students in the United States: Prevalence and predictors. JAMA Pediatr. 167, 1019–1025. https://doi.org/10.1001/jamapediatrics.2013.2392 (2013).
Cheung, M.W.-L. A guide to conducting a meta-analysis with non-independent effect sizes. Neuropsychol. Rev. 29, 387–396. https://doi.org/10.1007/s11065-019-09415-6 (2019).
Gleser, L. J. & Olkin, I. Stochastically dependent effect sizes. in The Handbook of Research Synthesis and Meta-analysis (ed. Cooper), 2nd ed. (Russell Sage Foundation, 2009), pp. 357–376.
Agarwal, D. P. & Goedde, H. W. Medicobiological and genetic studies on alcoholism. Role of metabolic variation and ethnicity on drinking habits, alcohol abuse and alcohol-related mortality. Clin. Investigator 70, 465–477. https://doi.org/10.1007/BF00210227 (1992).
Wall, T. L., Luczak, S. E. & Hiller-Sturmhöfel, S. Biology, genetics, and environment: Underlying factors influencing alcohol metabolism. Alcohol Res. 38, 59–68 (2016).
WHO expert consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 363, 157–163. https://doi.org/10.1016/S0140-6736(03)15268-3 (2004).
Peters, J. L., Sutton, A. J., Jones, D. R., Abrams, K. R. & Rushton, L. Contour-enhanced meta-analysis funnel plots help distinguish publication bias from other causes of asymmetry. J. Clin. Epidemiol. 61, 991–996. https://doi.org/10.1016/j.jclinepi.2007.11.010 (2008).
Sterne, J. A. & Egger, M. Funnel plots for detecting bias in meta-analysis. J. Clin. Epidemiol. 54, 1046–1055. https://doi.org/10.1016/S0895-4356(01)00377-8 (2001).
Viechtbauer, W. & Cheung, M. W. Outlier and influence diagnostics for meta-analysis. Res. Synthesis Methods 1, 112–125. https://doi.org/10.1002/jrsm.11 (2010).
Greenhouse, J. B. & Iyengar, S. Sensitivity analysis and diagnostics. in The Handbook of Research Synthesis and Meta-analysis (ed. Cooper), 2nd ed. (Russell Sage Foundation, 2009), pp. 417–433.
Cronce, J. M., Bedard-Gilligan, M. A., Zimmerman, L., Hodge, K. A. & Kaysen, D. Alcohol and binge eating as mediators between posttraumatic stress disorder symptom severity and body mass index. Obesity (Silver Spring, Md.) 25, 801–806. https://doi.org/10.1002/oby.21809 (2017).
Ahaneku, G. I., Ahaneku, J. E., Osuji, C. U., Oguejiofor, C. O. & Opara, P. C. (2014) Lipid patterns, alcohol intake and BMI of adult Nigerians in a sub-urban slum in Enugu, Nigeria. Pan Afr. Med. J. 18, 37. https://doi.org/10.11604/pamj.2014.18.37.1926.
Wakabayashi, I. Increased body mass index modifies associations between alcohol intake and blood cholesterol profile. Eur. J. Clin. Investig. 42, 179–185. https://doi.org/10.1111/j.1365-2362.2011.02568.x (2012).
Gazdzinski, S., Durazzo, T. C., Mon, A. & Meyerhoff, D. J. Body mass index is associated with brain metabolite levels in alcohol dependence—A multimodal magnetic resonance study. Alcoholism Clin. Exp. Res. 34, 2089–2096. https://doi.org/10.1111/j.1530-0277.2010.01305.x (2010).
Gearhardt, A. N. & Corbin, W. R. Body mass index and alcohol consumption: Family history of alcoholism as a moderator. Psychol. Addictive Behav. 23, 216–225. https://doi.org/10.1037/a0015011 (2009).
Skrzypczak, M., Szwed, A., Pawlińska-Chmara, R. & Skrzypulec, V. Body mass index, waist to hip ratio and waist/height in adult Polish women in relation to their education, place of residence, smoking and alcohol consumption. Homo 59, 329–342. https://doi.org/10.1016/j.jchb.2008.06.003 (2008).
Kleiner, K. D. et al. Body mass index and alcohol use. J. Addictive Diseases 23, 105–118. https://doi.org/10.1300/J069v23n03_08 (2004).
Männistö, S. et al. Reported alcohol intake, diet and body mass index in male smokers. Eur. J. Clin. Nutr. 50, 239–245 (1996).
Männistö, S., Uusitalo, K., Roos, E., Fogelholm, M. & Pietinen, P. Alcohol beverage drinking, diet and body mass index in a cross-sectional survey. Eur. J. Clin. Nutr. 51, 326–332. https://doi.org/10.1038/sj.ejcn.1600406 (1997).
Ishizaki, M. et al. Erythrocyte Na+/K(+)-ATPase and membrane and serum lipid profiles: As related to alcohol, body mass index and blood pressure. Clin. Exp. Hypertension (New York, NY” 1993) 16, 741–759. https://doi.org/10.3109/10641969409078023 (1994).
Kauffmann, F., Annesi, I., Neukirch, F., Oryszczyn, M. P. & Alperovitch, A. The relation between snoring and smoking, body mass index, age, alcohol consumption and respiratory symptoms. Eur. Respir. J. 2, 599–603 (1989).
Hooper, L. M., Doehler, K., Jankowski, P. J. & Tomek, S. E. Patterns of self-reported alcohol use, depressive symptoms, and body mass index in a family sample: The buffering effects of parentification. Family J. 20, 164–178. https://doi.org/10.1177/1066480711435320 (2012).
Cho, M.-K. & Kim, M. Y. Differences in SBP, BMI, and stress with AUDIT score in adolescents. TONURSJ 12, 228–237. https://doi.org/10.2174/1874434601812010228 (2018).
Amorim, T. C., Amorim, M. E., Guedes, L. A. & Lima, S. Evaluation of alcohol addiction among Brazilian Northeast medical students through the alcohol use disorders identification test and the relation with body mass index and smoking. Rev. Bras. Clin. Med. 10, 398–401 (2012).
Remus, D. Body mass index and alcohol consumption influences (physical education). Int. J. Sci. Res. 3, 484–485 (2014).
Sebelien, M. B. BMI status and intake of red meat, dietary fiber and alcohol in colorectal cancer patients prior to diagnosis. An interim analysis. Master thesis. University of Oslo, 2013.
Ventus, D. & Jern, P. Lifestyle factors and premature ejaculation: Are physical exercise, alcohol consumption, and body mass index associated with premature ejaculation and comorbid erectile problems?. J. Sexual Med. 13, 1482–1487. https://doi.org/10.1016/j.jsxm.2016.08.007 (2016).
Santangelo, O. E. et al. Is there a link between BMI and depressive symptomatology, risky consumption of alcohol and anxious symptomatology? Study in a sample of university students. Minerva Psichiatr. https://doi.org/10.23736/S0391-1772.19.02011-9 (2019).
Roggi, C., Silva, S., Maccarini, L., Mazzei, B. & Meloni, C. Alcohol, smoking and body mass index in a sample of adults from the general population. Ann. Ig. 4, 359–367 (1992).
Janssens, J. P. et al. Overweight, obesity and beer consumption—Alcohol drinking habits in Belgium and body mass index. Arch Public Health 59, 223–238 (2001).
Nishigaki, D. et al. Body mass index modifies the association between frequency of alcohol consumption and incidence of hypertension in men but not in women: A retrospective cohort study. Hypertension Res. 43, 322–330. https://doi.org/10.1038/s41440-019-0382-8 (2020).
Booranasuksakul, U., Singhato, A., Rueangsri, N. & Prasertsri, P. (2019) Association between Alcohol Consumption and Body Mass Index in University Students. Asian/Pacific Island Nursing J. 4, 57–65. https://doi.org/10.31372/20190401.1035.
Rask-Andersen, M., Karlsson, T., Ek, W. E. & Johansson, Å. Gene-environment interaction study for BMI reveals interactions between genetic factors and physical activity, alcohol consumption and socioeconomic status. PLoS Genet. 13, e1006977. https://doi.org/10.1371/journal.pgen.1006977 (2017).
Yi, S.-W., Hong, J.-S., Yi, J.-J. & Ohrr, H. Impact of alcohol consumption and body mass index on mortality from nonneoplastic liver diseases, upper aerodigestive tract cancers, and alcohol use disorders in Korean older middle-aged men: Prospective cohort study. Medicine 95, e4876. https://doi.org/10.1097/md.0000000000004876 (2016).
Shaikh, R. A., Siahpush, M., Singh, G. K. & Tibbits, M. Socioeconomic status, smoking, alcohol use, physical activity, and dietary behavior as determinants of obesity and body mass index in the United States: Findings from the National Health Interview Survey. Int. J. MCH AIDS. 4, 22–34. https://doi.org/10.21106/ijma.53 (2015).
Yue, F. et al. Association of alcohol consumption with the impaired β-cell function independent of body mass index among Chinese men. Endocrine J. 59, 425–433. https://doi.org/10.1507/endocrj.ej12-0003 (2012).
Loomba, R., Bettencourt, R. & Barrett-Connor, E. Synergistic association between alcohol intake and body mass index with serum alanine and aspartate aminotransferase levels in older adults: The Rancho Bernardo Study. Alimentary Pharmacol. Therapeutics 30, 1137–1149. https://doi.org/10.1111/j.1365-2036.2009.04141.x (2009).
Kawamoto, R. et al. Alcohol consumption is associated with decreased insulin resistance independent of body mass index in Japanese community-dwelling men. Tohoku J. Exp. Med. 218, 331–337. https://doi.org/10.1620/tjem.218.331 (2009).
Agarwal, K., Demiral, S. B., Manza, P., Volkow, N. D. & Joseph, P. V. Relationship between BMI and alcohol consumption levels in decision making. Int. J. Obesity. 45, 2455–2463. https://doi.org/10.1038/s41366-021-00919-x (2021).
Crovetto, M. et al. Association of weekend alcohol consumption with diet variables, body mass index, cardiovascular risk and sleep. Human Nutr. Metab. 27, 200140. https://doi.org/10.1016/j.hnm.2022.200140 (2022).
Gao, C. et al. Four-way decomposition of effect of alcohol consumption and body mass index on lipid profile. Int. J. Environ. Res. Public Health. https://doi.org/10.3390/ijerph182413211 (2021).
Hashimoto, Y. et al. Effect of body mass index on the association between alcohol consumption and the development of chronic kidney disease. Sci. Rep. 11, 20440. https://doi.org/10.1038/s41598-021-99222-y (2021).
Innes, H. et al. Characterizing the risk interplay between alcohol intake and body mass index on cirrhosis morbidity. Hepatology (Baltimore, MD) 75, 369–378. https://doi.org/10.1002/hep.32123 (2022).
Kokubun, K., Pineda, J. C. D. & Yamakawa, Y. Unhealthy lifestyles and brain condition: Examining the relations of BMI, living alone, alcohol intake, short sleep, smoking, and lack of exercise with gray matter volume. PLoS ONE 16, e0255285. https://doi.org/10.1371/journal.pone.0255285 (2021).
Hoffmann, S. et al. Body mass index interacts with sex to predict readmission in in-patients with alcohol use disorder. Addiction Biol. 28, e13239. https://doi.org/10.1111/adb.13239 (2023).
Kim, H.-Y. Statistical notes for clinical researchers: Effect size. Restorative Dentistry Endodontics 40, 328–331. https://doi.org/10.5395/rde.2015.40.4.328 (2015).
Schindler, T. H. et al. Relationship between increasing body weight, insulin resistance, inflammation, adipocytokine leptin, and coronary circulatory function. J. Am. College Cardiol. 47, 1188–1195. https://doi.org/10.1016/j.jacc.2005.10.062 (2006).
Klatsky, A. L., Armstrong, M. A. & Kipp, H. Correlates of alcoholic beverage preference: Traits of persons who choose wine, liquor or beer. Br. J. Addiction 85, 1279–1289. https://doi.org/10.1111/j.1360-0443.1990.tb01604.x (1990).
Watten, R. G. Sports, physical exercise and use of alcohol. Scand. J. Med. Sci. Sports 5, 364–368. https://doi.org/10.1111/j.1600-0838.1995.tb00060.x (1995).
Hansel, B. et al. Relationships between consumption of alcoholic beverages and healthy foods: The French supermarket cohort of 196,000 subjects. Eur. J. Prevent. Cardiol. 22, 215–222. https://doi.org/10.1177/2047487313506829 (2015).
Johansen, D., Friis, K., Skovenborg, E. & Grønbaek, M. Food buying habits of people who buy wine or beer: Cross sectional study. BMJ 332, 519–522. https://doi.org/10.1136/bmj.38694.568981.80 (2006).
Seeley, R. J. & Sandoval, D. A. Weight loss through smoking. Nature 475, 176–177. https://doi.org/10.1038/475176a (2011).
Wang, Q. Smoking and body weight: Evidence from China health and nutrition survey. BMC Public Health 15, 1238. https://doi.org/10.1186/s12889-015-2549-9 (2015).
Courtemanche, C., Tchernis, R. & Ukert, B. The effect of smoking on obesity: Evidence from a randomized trial. J. Health Econ. 57, 31–44. https://doi.org/10.1016/j.jhealeco.2017.10.006 (2018).
Enoch, M.-A. Genetic influences on the development of alcoholism. Curr. Psychiatry Rep. 15, 412. https://doi.org/10.1007/s11920-013-0412-1 (2013).
Enoch, M.-A. Genetic influences on response to alcohol and response to pharmacotherapies for alcoholism. Pharmacol. Biochem. Behav. 123, 17–24. https://doi.org/10.1016/j.pbb.2013.11.001 (2014).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences 2nd edn. (Routledge, 1988).
Jackson, D., White, I. R. & Riley, R. D. Multivariate Meta-Analysis. In Handbook of Meta-Analysis, edited by C. H. Schmid, T. Stijnen & I. R. White (Chapman and Hall/CRC, First edition. | Boca Raton : Taylor and Francis, [2020] | Series: Chapman & Hall/CRC handbooks of modern statistical methods, 2020), pp. 163–186.
Jackson, D., Riley, R. & White, I. R. Multivariate meta-analysis: Potential and promise. Stat. Med. 30, 2481–2498. https://doi.org/10.1002/sim.4172 (2011).
Jablensky, A. Research methods in psychiatric epidemiology: An overview. Aust. New Zealand J. Psychiatry 36, 297–310. https://doi.org/10.1046/j.1440-1614.2002.01035.x (2002).
Heinz, A. et al. Addiction Research Consortium: Losing and regaining control over drug intake (ReCoDe)-From trajectories to mechanisms and interventions. Addiction Biol. 25, e12866. https://doi.org/10.1111/adb.12866 (2020).
Funding
Open Access funding enabled and organized by Projekt DEAL. This work was funded in part by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation)-Project-ID 402170461-TRR265106. CM is a member of the research training group 2162 'Neurodevelopment and Vulnerability of the Central Nervous System' funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation; 270949263/GRK2162). This work was supported by intramural grants from the University Hospital of the Friedrich-Alexander University Erlangen-Nurnberg (FAU). We acknowledge financial support by Deutsche Forschungsgemeinschaft and Friedrich-Alexander-Universität Erlangen-Nürnberg within the funding programme “Open Access Publication Funding”. The funders had no role in the study design, data collection, analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Conception: B.L., C.M., C.W., E.S. Acquisition: C.M., E.S., M.M. Analysis: B.L., C.M., E.S. Interpretation: B.L., C.M., E.S., F.K., J.K. Drafting of the manuscript: B.L., C.M., E.S. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Siegmann, EM., Mazza, M., Weinland, C. et al. Meta-analytic evidence for a sex-diverging association between alcohol use and body mass index. Sci Rep 12, 21869 (2022). https://doi.org/10.1038/s41598-022-25653-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-25653-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.