Abstract
For large herbivores living in highly dynamic environments, maintaining range fidelity has the potential to facilitate the exploitation of predictable resources while minimising energy expenditure. We evaluate this expectation by examining how the seasonal range fidelity of African elephants (Loxodonta africana) in the Kruger National Park, South Africa is affected by spatiotemporal variation in environmental conditions (vegetation quality, temperature, rainfall, and fire). Eight-years of GPS collar data were used to analyse the similarity in seasonal utilisation distributions for thirteen family groups. Elephants exhibited remarkable consistency in their seasonal range fidelity across the study with rainfall emerging as a key driver of space-use. Within years, high range fidelity from summer to autumn and from autumn to winter was driven by increased rainfall and the retention of high-quality vegetation. Across years, sequential autumn seasons demonstrated the lowest levels of range fidelity due to inter-annual variability in the wet to dry season transition, resulting in unpredictable resource availability. Understanding seasonal space use is important for determining the effects of future variability in environmental conditions on elephant populations, particularly when it comes to management interventions. Indeed, over the coming decades climate change is predicted to drive greater variability in rainfall and elevated temperatures in African savanna ecosystems. The impacts of climate change also present particular challenges for elephants living in fragmented or human-transformed habitats where the opportunity for seasonal range shifts are greatly constrained.
Similar content being viewed by others
Introduction
Movement studies across a range of mammal species have shown that animals often return to familiar sites to access predictable resources1,2,3. Maintaining range fidelity enables individuals to maximise nutritional and energetic intake by exploiting the location of known foraging opportunities while minimising the risk of potential threats2,4,5. For example, female mule deer (Odocoileus hemionus) exhibit high range philopatry during the summer months when they give birth, raise offspring and accrue fat reserves6, while site fidelity in southern Australian bottlenose dolphins (Tursiops cf. australis) has been linked with highly productive habitats and long-lasting social bonds7. Determining the extent of range fidelity exhibited by individual animals can provide important insights into how these movement decisions can scale up to influence population dynamics and community level processes8,9.
Large herbivores inhabiting seasonally dynamic environments may demonstrate high levels of range fidelity due to the predictability of resources in specific habitats during a given season10. This is a common driver of migratory behaviour among species that consistently track resources across the landscape during the spring green-up—referred to as ‘surfing the green wave’11. However, exploratory movement is less of a risk to animals occupying relatively homogeneous habitats due to dependability in conditions, and range fidelity is therefore a less common strategy for these species12. Similarly, animals inhabiting unpredictable environments are expected to exhibit ‘nomadic’ behaviour with comparatively low consistency in movement behaviour over space and time11,13. Ultimately, these movement strategies are shaped by a combination of extrinsic variables that drive resource quality and predictability in the environment and the intrinsic factors related to an individual’s physiology, behavioural plasticity and life history14,15.
Fidelity is not just limited to range location, but is also dependent upon variation in range size1,2. An individual may change the size of its range while maintaining the core area of habitat use, consequently presenting relatively high range fidelity, but with a change in the degree of range overlap1. The nutritional, energetic and reproductive demands of an animal are fundamental in driving range size and overlap16,17. However, these are further influenced by a myriad of extrinsic and intrinsic factors that play out over time and space. For large herbivores, these include habitat quality, population density, the abundance of predators, human disturbance, sociality, body size, digestive physiology and life history1,18,19.
The movement behaviour of African savanna elephants (Loxodonta africana) is predominantly driven by their considerable energetic and nutritional requirements20,21, as well as their dependence on surface water availability22,23,24. Elephants are true mixed feeders, demonstrating a seasonal dietary shift from grazing in the summer wet season to browsing in the winter dry season25,26. This is because vegetation productivity is higher in the wet season, which is associated with increased nutritional value and digestibility of plant material25,27,28. Moreover, an inverse relationship between rainfall and movement autocorrelation has been exhibited, showing that once released from resource limitations in the dry season, elephants become less constrained in their space use due to increased resource availability29,30,31.
By adapting their movements to correspond with seasonal resource availability, elephants are able to minimise the energetic costs associated with moving through the heterogeneous savanna landscape32,33,34, while balancing critical physiological requirements, such as thermoregulation35. Fire meanwhile, is a key ecological driver that can shape the structure and species composition of the savanna through the removal of plant biomass, which temporarily reduces primary productivity until a green flush of vegetation is stimulated36. Elephant distribution patterns have been shown to be influenced by fire return period, with family groups selecting for areas experiencing lower fire frequency37.
Quantifying an individual’s space use over time is crucial for understanding both their consistency in movement behaviour and predictability of the habitats that they occupy2,38,39. Through the generation of utilisation distributions (UDs), we are now able to quantify the probability of occurrence, and thus, concentration of space use for an individual across temporal scales40,41. However, there are a limited number of studies that have considered the intraspecific similarity in UDs across multiple, recurrent seasons, with fewer still explicitly attempting to identify the key drivers of fidelity in range use (but see2,10,42).
Identifying the environmental context in which individuals are driven to make their movement decisions, allows ecologists to make informed predictions about the effects of fluctuations in resource availability or anthropogenic disturbance on large herbivore space use over time, which is crucial in the face of unprecedented and rapid environmental change6,10,31. These predictions are vital not only to species conservation, but also in determining the effects that species such as the elephant may have on their ecosystems, including altering habitat structure43,44,45. The aim of this study was to identify the consequences of spatial and temporal resource variability on seasonal range fidelity of African elephant family groups in Kruger National Park, South Africa. The objectives were to determine (1) the overall consistency in seasonal space use within and across multiple years, and (2) identify the key environmental variables driving seasonal range fidelity.
Methods
Study site
Kruger National Park (KNP) is located in South Africa and extends over 19,485 km2 of heterogeneous savanna landscape (Fig. 1). The southern region of KNP receives the most rain, ranging from 500 to 700 mm year−1, while the northern section receives approximately 300–500 mm year−146,47. The east of KNP is formed of nutrient-rich basaltic soils, with the west comprised of less-fertile granite soils47. Northern KNP is dominated by Mopane woodlands47,48, and in the south, Combretum and Acacia. There is broad leaf savanna in the west and fine leaf in the east47. KNP experiences hotter summer wet seasons with temperatures reaching up to 33 °C, and cooler winter dry seasons with temperatures dropping to around 6 °C47. In 2015, the elephant population was estimated to number a minimum of 17,086 individuals, with a growth of 4.2% per annum since 200349.
Movement data
Location data were collected from 13 female elephants between 2006 and 2013 using Global Positioning Satellite (GPS) Collars (see Fig. 1 and Table S1) (Africa Wildlife Tracking cc., South Africa). The collaring procedure is explained in more detail in Birkett et al.29. Each of the collared individuals were selected from a different family group (adult females and their dependent young). Collars recorded locations of the animal every thirty minutes with an average estimated positional dilution of precision (PDOP) value < 2.029 and a maximum circular error probability (CEP) of 4 m. These values are indicative of overall low position estimation error. The methods were approved by the University of KwaZulu-Natal Ethics Committee and conducted in accordance with institutional guidelines.
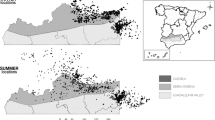
Home ranges (90% Kernel) of all thirteen elephant family groups in KNP and associated private reserves using the data collected over the duration of the study period (see Table S1 for annual and seasonal range size comparisons for each family). The red box on the inset map highlights the location of KNP in South Africa. This map was created in RStudio52 using the ‘maps’ package50 database, while the coordinates of the study area were projected from the WGS84 datum to UTM zone 36S.
Utilisation distribution (UD: the probability of animal occurrence in space and time) estimations were calculated using the dynamic Brownian bridge movement model (dBBMM), implemented using the package ‘move’51 in Rstudio52. The dBBMM builds on the original Brownian bridge movement model (BBMM;53) by incorporating both temporal and behavioural characteristics of movement paths into home range estimation40. Such features of a movement model are key for estimating the space use of a species like the African elephant, which is known for making highly directional movements over long distances towards known areas of resource abundance54,55. After visual data inspection and recommendations by Kranstauber et al.40, we specified our moving window size as 31 (equivalent to 15.5 h), with a margin size of 11 (equivalent to 5.5 h). These parameters were based on the number of GPS locations recorded per 24 h for an individual elephant, while also ensuring that sudden changes in movement variance associated with environmental events, such as the increases in temperature, onset of seasonal rainfall and fire could be detected.
Environmental data
Seasons were classified as summer (December–February), autumn (March–May), winter (June–August) and spring (September–November). These were defined using phenological research into green-up dates56 and the analyses of solar radiation in KNP57. By using four seasons, we were also able to capture the transitional periods between the traditional wet (spring & summer) and dry (autumn & winter) seasons. Environmental factors considered in this study included green vegetation density, rainfall and mean daily temperature. Datasets for vegetation quality were accessed through the Environmental-Data Automated Track Annotation System (Env-DATA) provided by Movebank 58. Summaries of the Moderate Resolution Imaging Spectroradiometer (MODIS) enhanced vegetation index (EVI) (250 × 250 m resolution)—a measure of vegetation greenness—were obtained every 16 days59 and the mean value for each seasonal UD was calculated.
SANParks collected monthly rainfall measurements from twenty-three weather stations across KNP enabling rainfall to be interpolated for each season. Average measurements of rainfall were calculated for each seasonal UD. We also included the average rainfall for the preceding season for each UD, due to the influence of rainfall on savanna primary productivity over subsequent weeks and months. These values were assigned to the variable “lag rainfall”. Temperature records were obtained from the South African Weather Service, with the mean daily values calculated for each season. Prescribed burning was conducted in KNP between the years of 2005–2013, across all seasons. Three measurements for fire were calculated for each seasonal UD: the percentage of area burned, percentage of area burned in the last fire before the season started and the time since last fire. Surface water was not included in our analysis, as it is readily available across KNP in the form of rivers, waterholes and pumped dams, and while no doubt a key driver of elephant movement, the relative availability is not expected to vary significantly between seasons60.
We predicted that rainfall would have the greatest influence on seasonal range fidelity, with increased levels of rainfall resulting in lower fidelity during the transition from dry to wet season as forage abundance and quality improved30,33. Whereas higher rainfall in the transition from wet to dry season would result in greater range fidelity as foraging opportunities were maintained into the winter61. Similarly, we anticipated that an increase in primary productivity from the dry to wet season would result in decreased fidelity due to reduced constraints on movement29,62. But conditions that led to primary productivity being maintained from summer into winter would lead to higher fidelity as summer ranges continued to provide key forage resources27. We also expected lower range fidelity when the second season experiences an increase in temperature, as this is a key driver of vegetation phenology and growth56. Fire is more complex to predict, as burn events can cause significant disturbance and displace animals from preferred habitats63. However, the post-fire flush of vegetation may also provide important foraging opportunities depending upon the season37,64. Finally, it is important to note that elephant ranging behaviour may well be driven by a combination of these environmental variables. We therefore employed an information-theoretic approach to evaluate a set of candidate models and quantify the contribution of each variable in explaining the range fidelity observed across seasonal comparisons.
Statistical analysis of movement
All statistical analyses were conducted in the R environment for statistical computing. Family group movement tracks were divided into segments (“bursted”) categorised by season (summer, autumn, winter, spring) and year, via the ‘move’ package51. Seasonal UDs were calculated for each family group according to their “bursted” seasonal movements, before measuring similarity in UD rasters with the Earth Mover’s Distance (EMD) function51. The EMD quantifies the similarity between UDs by calculating a measure of minimal amount of effort taken to shape one UD into another. This provides a spatially explicit comparison between UDs, delivering a more informed comparison than the volume of intersection (VI) or Bhattacharyya’s affinity, because it considers spatial proximity rather than just exclusive overlap41.
EMD values were compared across all paired combinations for each family group, including consecutive seasons within the same year (e.g. winter 2008 and spring 2008) and the same seasons across consecutive years (e.g. summer 2008 and summer 2009). EMD values were divided by the threshold value (the largest distance that the united locations of each UD contained), and converted to lie between zero and one, with zero representing two identical UDs.
We fitted generalised linear mixed effects models using the ‘glmmTMB’ package in R65 with EMD included as the response variable. Fixed effects included the difference in environmental variables for each season-year pair of UDs. These were calculated by subtracting values of the first season from the second (e.g. spring 2008 rainfall – winter 2008 rainfall). For example, negative values in rainfall difference indicates lower rainfall in the second season compared to the first, and vice versa. The environmental variables included: EVI (vegetation greenness: scale 0–1), mean daily temperature (°C), rainfall (converted from mm to m), lag rainfall (converted from mm to m), percentage of UD burned within season (BIS) (%), percentage of UD burned in last fire before season (BLS) (%), and time since last burn in UD before season (TSLB) (days).
Testing for correlation between fixed effects demonstrated multicollinearity between rainfall, temperature and EVI for consecutive seasonal comparisons within the same year. These fixed effects (with a correlation coefficient greater than 0.4) were excluded from the same models. However, no evidence of collinearity was found between fixed effects when comparing seasonal space use across years. Thirty-three candidate models were generated a priori for EMD for consecutive seasonal comparisons within the same year (Table S2a), and thirty-nine for the same seasonal comparisons between consecutive years (Table S2b). All models included elephant family identity as a random effect to account for repeated observations of the same group. We also included three interactions (season * rainfall, season * temperature and season * EVI) to determine how variation in environmental conditions across seasonal comparisons influenced range fidelity.
Akaike’s Information Criterion adjusted for small sample size (AICc) was used for model selection66. Model averaging was conducted on the models that accounted for ≥ 0.95 of the AICc weight using the ‘AICcmodavg’ package67. The significance for each environmental variable that featured in the top models was assessed by extracting the β estimates and 95% confidence intervals (CI) and establishing whether these CIs overlapped zero.
Ethics approval
Ethical approval for the collaring of elephants was obtained from the University of Kwazulu-Natal Animal Ethics sub-committee (Ref. 009/10/Animal). This research was also part of a registered and approved SANParks project, in association with Kruger National Park and Scientific Services (Ref: BIRPJ743).
Results
The average EMD value among all family groups was 0.13 (SE ± 0.006) for consecutive seasonal comparisons within the same year and 0.12 (SE ± 0.007) for the same seasonal comparisons across consecutive years (scale is 0–1, with zero representing two identical UDs; see Fig. 2). For within and between year seasonal space use comparisons the EMD values ranged from 0.01 to 0.45 (n = 166) and 0.01–0.39 (n = 135), respectively. Over 80% of seasonal comparisons had an EMD value of 0.2 or less, indicating that the elephants exhibited a high level of seasonal range fidelity within and between years.
Consecutive seasons within the same year
Mean EMD remained consistently low during these seasonal transitions with no significant difference in range fidelity (Fig. 3a and Table S3). Consistency in space use for consecutive seasons in the same year was best explained by a model that included an interaction between rainfall and season, accounting for 28% of the AICc weight (Table 1). Model averaging indicated that difference in rainfall was the only variable found to be significant in predicting EMD (Table 2). Range fidelity increased when there was greater rainfall in the second season for autumn–winter and summer-autumn comparisons (Fig. 4).
Seasonal space use comparisons of elephants in KNP for (a) consecutive seasons within the same year and (b) the same seasons between consecutive years. Means are indicated by the ◊ symbol. Significant differences in Earth Mover’s Distance (EMD) values for seasonal space use comparisons across years (significance codes: ‘*’ 0.05 ‘***’ 0.001).
The relationship between difference in rainfall for consecutive seasons within the same year and Earth Mover’s Distance (EMD) for (a) summer and autumn (b) autumn and winter (c) winter and spring and (d) spring and summer comparisons. Negative values in rainfall difference indicates lower rainfall in the second season compared to the first, and vice versa. Lower values in EMD indicate higher fidelity.
The same seasons between consecutive years
Mean EMD values were highest for autumn comparisons than any other season (spring: EMD = 0.09 ± 0.01; winter: EMD = 0.10 ± 0.01; summer: EMD = 0.12 ± 0.01; autumn: EMD = 0.16 ± 0.02), indicating that elephant family groups showed the least range fidelity from one autumn to the next. Additionally, EMD values were similar for both spring and winter comparisons (Fig. 3b). EMD values in autumn were significantly higher than in both winter (p < 0.001) and spring (p < 0.001), and significantly higher in summer than in spring (p < 0.05) (Fig. 3b and Table S4).
An analysis of EMD for the same season across consecutive years produced eight top models (Table 3). Our top model for explaining consistency in seasonal space use across consecutive years included an additive model for season and time since last burn within range area, accounting for just under a third of the 95% AICc weight (Table 3). Model averaging revealed that EMD values in spring, summer and winter were all significantly different when compared with autumn (Table 4).
Discussion
The results from our study indicate high seasonal range fidelity across all thirteen elephant family groups. Indeed, although there were significant differences in seasonal range fidelity across years (particularly during the autumn months), the average EMD values within and between years remained low and remarkably consistent. Given the comparatively long-term duration of our study, these results indicate that the elephants within KNP tend to concentrate their space use in familiar areas, demonstrating high fidelity in both seasonal ranging patterns and spatial use. Through the maintenance of range fidelity, species living in heterogeneous environments are able to maximise their individual fitness by exploiting predictable resources3,10,68. Developing an understanding of the conditions that influence similarity in space use is vital for population management, particularly for an ecosystem engineer that can range over hundreds and even thousands of km2 but is also sensitive to changes in temporal resource availability69,70.
For African elephants, strategically maintaining high range fidelity could minimise energy expenditure through detailed knowledge on predictable resource availability. Certainly, movements have been shown to be strongly shaped by energetic and nutritional considerations at the landscape scale. For example, elephants in northern Kenya avoid comparatively steep slopes despite the availability of abundant forage on higher ground71, while elephants in the semi-arid Etosha National Park have been shown to access the closest waterhole 90% of the time, demonstrating a detailed spatial knowledge over large scales that facilitates optimal movement55. Moreover, a recent study in KNP revealed facultative shuttling (moving frequently between water and safer sites) towards water sources, with a greater proportion of time spent near water at peak temperatures during the day, especially in the dry season35. These results demonstrate how elephants are able to modify their movements in response to thermal stress, whilst utilising their considerable spatial memory to efficiently access key resources55,62,72.
It is important to highlight that the consistent availability of surface water in KNP may facilitate the high seasonal range fidelity observed across the elephant family groups in this study. However, a relative abundance of water could also release elephants to range more widely and exploit a variety of habitats compared with arid regions where access to water can constrain movements, especially in the dry season27. In support of this, analysis of fine-scale movements of elephants to and from water in KNP revealed comparatively low-level fidelity to specific water points35. By essentially accounting for surface water in our study, it was possible to explore consistency in space-use as a function of environmental conditions and forage resource availability that has relevance beyond the study population.
At the group level, this high seasonal fidelity within and across years, will inevitably result in heterogeneity in habitat use within ranges due to variation in foraging intensity and primary productivity70,73. It is reasonable to suggest that this may well translate to pronounced variation in elephant activity across the national park, with family groups remaining consistent in their space use and thus concentrating their foraging on vegetation in the core of the range, while other habitats experience lower browsing pressure. The high range fidelity demonstrated in our study could play a pivotal role in enhancing the biodiversity of KNP. Indeed, research has continued to support the ‘habitat heterogeneity hypothesis’, suggesting that heterogeneity increases biodiversity through greater niche provision74,75,76,77. However, it is worth noting that at the population-level elephant distribution patterns in KNP may have become more homogenous over the past three decades as a function of population growth37. From 1985 to 2004 the annual census revealed significant signs of clustering and sexual segregation. During the subsequent decade this was no longer observed and the elephants were distributed throughout KNP habitat types, albeit concentrated closer to major rivers37. These annual counts can only provide a relatively coarse snapshot of elephant distribution and further investigation should consider if elephant ranges correspond with threatened habitats, or endemic species that may be unable to adapt to intense browsing pressure24,78.
For consecutive seasons within the same year, rainfall was found to be the only significant driver of seasonal range fidelity. During summer to autumn and autumn to winter transitions, family groups maintained site fidelity when rainfall remained comparatively high in the second season. The winter months are typically the driest, while autumn represents the transition from the wet to the dry season and an elephant dietary shift from grass to browse26,79. Patterns of higher fidelity following these seasonal transitions is likely due to exploitation of aseasonal rainfall (i.e. unusually wet conditions at a time that is typically dry) and the subsequent retention of higher quality vegetation, such as grasses in the autumn and palatable deciduous trees in the winter, which has been shown to influence ungulate population dynamics80,81.
Interestingly, Thaker et al.35 revealed that the daily distance moved by elephants in KNP was consistent across both wet and dry seasons, which was attributed to high density and accessibility of waterholes and surface water in the south of KNP. Given that the elephants are largely range faithful, it is possible that, with a continuation of rainfall into the dry season, elephants are less inclined to adjust their seasonal ranges due to familiarity with the locations and availability of key resources54,55. Indeed, elephants appear to be very effective at integrating long term information on environmental conditions into their movement strategies34.
The lowest levels of seasonal range fidelity between years occurred in the autumn months, while EMD values for winter, spring and summer indicated higher fidelity and greater associated familiarity with spatiotemporal resource availability. As demonstrated by Birkett et al.29, and supported by our intra-annual analyses, the breakpoints at which seasonal transitions occur are subject to considerable variation across years. This variability in EMD during the autumn months is indicative of the unpredictability in location and longevity of resources moving into the dry season56. Seasonal dietary shift is a primary adaptation of elephants to fluctuations in resource availability, with grass predominantly being consumed in the wet season and an increasing dependence on woody browse during the winter months25,26,82. Reliance on these resources continues into the spring until elephants are released from the dry season and the first green-up of vegetation occurs26,83. Climate change is likely to amplify variation in the timing of key phenological processes84, which may have significant effects on elephant movement. This is highlighted by the significant difference in fidelity across consecutive years between spring and summer, possibly attributable to the previous early flush of some deciduous trees in spring and an early release in constrained movements, even without rain.
The elephants in KNP did not significantly alter their space use to either avoid or utilise burn patches within their seasonal ranges. However, previous studies demonstrating that elephants initially avoid burned areas in the short term and return once the vegetation recovers37,63. It is therefore possible that our temporal scale for analysis was too coarse to highlight the short-term effects of fire on movement. Similarly, change in temperature was not highlighted as a key predictor of range fidelity. Indeed, research has found that elephants in KNP respond to “landscape of thermal stress” at finer temporal scales by periodically shuttling to water during the hottest times of the day35.
Research comparing elephant space use has largely focussed on calculating areas of explicit spatial overlap31,85. However, this approach does not account for the proximity in space use between seasonal distributions, resulting in space use similarity being oversimplified. The Earth mover’s distance used in our study has enabled the integration of spatially explicit comparisons of seasonal range use alongside accurate depictions of environmental variability. We have highlighted that rainfall has a significant influence on range fidelity during the dry season. Notably, rainfall in the previous season was not found to be a significant driver of fidelity, which suggests that the elephants in KNP are responding to current forage availability, rather than longer-term changes in plant phenology. However, as rapid advances in technology are enabling ever more detailed movement data to be collected and analysed86,87, there is the potential for future studies to explore range shifts that track the measurable effects of climate change across multiple years and populations. Likewise, in this study we only considered the resource availability and environmental conditions within the area of the UD. An important next step is to incorporate elephant perception of key ecological and environmental drivers that lie beyond the boundaries of the range, but which likely shape movement decisions, nonetheless.
Although no detailed demographic information relating to the composition of family groups within this study is available, a recent study demonstrated that range shifts and expansions of elephants in northern Kenya were predominantly attributed to generational turnover, with loss of mature adults significant in predicting increase in range size and centroid shift62. Family groups also demonstrated a clear shift in movement patterns away from increased poaching levels, and towards areas of greater primary productivity62. These results highlight the role of both environmental and anthropogenic drivers on intraspecific philopatry. However, within KNP, the capacity for anthropogenic disturbance is limited (beyond tourist activity in rest camps and along park roads). Therefore, a strong focus on the effects of environmental variation on elephant movement patterns remains key to future management plans and biodiversity conservation within the park.
Understanding the drivers of elephant space use is useful in predicting movement patterns that are likely to result from the longer-term effects of climate change88,89, particularly given they are keystone species capable of shaping habitat structure and ecosystem function44,90,91. Specifically, it is predicted that climate change will lead to increased variability in rainfall patterns and higher temperatures in African savannas92,93. The elephants in KNP have demonstrated their ability to detect and respond to aseasonal rainfall patterns by adjusting their ranging behaviour. Dry seasons that experience below average rainfall are likely to drive reduced range fidelity as elephants seek out remaining foraging opportunities, a situation which could become exacerbated with increased drought frequency. Ultimately, the altered movement and foraging behaviour of elephants at the population-level could have cascading effects on vegetation45,94 other herbivore species95, and predators96,97.
Identifying areas of concentrated elephant space use allows scientists and managers to highlight potential areas of human-wildlife conflict and habitats that are vulnerable to sustained grazing and browsing pressure. Indeed, such highly concentrated utilisation may shape biodiversity, especially if these areas overlap with those of range-restricted species. Such predictions are also very useful in assisting with the sustainable management of elephant populations, including the preservation of landscape connectivity and vital movement corridors98,99,100. We have demonstrated that rainfall is the overriding factor influencing elephant range fidelity in KNP where surface water availability is abundant and human disturbance comparatively low. This has considerable implications for elephant conservation as the effects of climate change accelerate and environmental conditions become less predictable across an already fragmented species range.
Data availability
Data are archived on the Movebank (movebank.org) website: “African Elephant Slotow Kruger”.
References
Richard, E., Said, S., Hamann, J. L. & Gaillard, J. M. Daily, seasonal and annual variations in individual home range overlap of two sympatric spacies of deer. Can. J. Zool. 92, 853–859 (2014).
Sorensen, A. A., Stenhouse, G. B., Bourbonnais, M. L. & Nelson, T. A. Effects of habitat quality and anthropogenic disturbance on grizzly bear (Ursus arctos horribilis) home-range fidelity. Can. J. Zool. 93, 857–865 (2015).
van Beest, F. M., Rivrud, I. M., Loe, L. E., Milner, J. M. & Mysterud, A. What determines variation in home range size across spatiotemporal scales in a large browsing herbivore?. J. Anim. Ecol. 80, 771–785 (2011).
Naidoo, R., Du, P., Weaver, G. S. L. C., Jago, M. & Wegmann, M. Factors affecting intraspecific variation in home range size of a large African herbivore. Landsc. Ecol. 27, 1523–1534 (2012).
Bose, S. et al. Implications of fidelity and philopatry for the population structure of female black-tailed deer. Behav. Ecol. 28, 983–990 (2017).
Northrup, J. M., Anderson, C. R. Jr. & Wittemyer, G. Environmental dynamics and anthropogenic development alter philopatry and space-use in a North American cervid. Divers. Distrib. 22, 547–557 (2016).
Passadore, C., Möller, L., Diaz-aguirre, F. & Parra, G. J. High site fidelity and restricted ranging patterns in southern Australian bottlenose dolphins. Ecol. Evol. 8, 242–256 (2018).
Morales, J. M. et al. Building the bridge between animal movement and population dynamics. Philos. Trans. R. Soc. B Biol. Sci. 365, 2289–2301 (2010).
Shaw, A. K. Causes and consequences of individual variation in animal movement. Mov. Ecol. 8, 1–12 (2020).
Morrison, T. A. et al. Drivers of site fidelity in ungulates. J. Anim. Ecol. 00, 1–12 (2021).
Abrahms, B. et al. Emerging perspectives on resource tracking and animal movement ecology. Trends Ecol. Evol. 36, 308–320 (2021).
Barraquand, F. & Benhamou, S. Animal movements in heterogeneous landscapes: Identifying profitable places and homogeneous movement bouts. Ecology 89, 3336–3348 (2008).
Mueller, T. & Fagan, W. F. Search and navigation in dynamic environments: From individual behaviors to population distributions. Oikos 117, 654–664 (2008).
Sawyer, H., Merkle, J. A., Middleton, A. D., Dwinnell, S. P. H. & Monteith, K. L. Migratory plasticity is not ubiquitous among large herbivores. J. Anim. Ecol. 88, 450–460 (2019).
Shakeri, Y. N., White, K. S. & Waite, J. N. Staying close to home: Ecological constraints on space use and range fidelity in a mountain ungulate. Ecol. Evol. 11, 11051–11064 (2021).
Damuth, J. Home range, home range overlap, and species energy use among herbivorous mammals. Biol. J. Linn. Soc. 15, 185–193 (1981).
Lindstedt, S. L., Miller, B. J. & Buskirk, S. W. Home range, time, and body size in mammals. Ecol. Soc. Am. 67, 413–418 (1986).
Ofstad, E. G., Herfindal, I., Solberg, E. J. & Sæther, B. E. Home ranges, habitat and body mass: Simple correlates of home range size in ungulates. Proc. R. Soc. B Biol. Sci. 283, 20161234 (2016).
Gehr, B. et al. Stay home, stay safe—Site familiarity reduces predation risk in a large herbivore in two contrasting study sites. J. Anim. Ecol. 89, 1329–1339 (2020).
Sach, F., Dierenfeld, E. S., Langley-Evans, S. C., Watts, M. J. & Yon, L. African savanna elephants (Loxodonta africana) as an example of a herbivore making movement choices based on nutritional needs. PeerJ 7, 1–27 (2019).
Pretorius, Y. et al. Diet selection of African elephant over time shows changing optimization currency. Oikos 121, 2110–2120 (2012).
Chamaillé-Jammes, S., Valeix, M. & Fritz, H. Managing heterogeneity in elephant distribution: Interactions between elephant population density and surface-water availability. J. Appl. Ecol. 44, 625–633 (2007).
Purdon, A. & van Aarde, R. J. Water provisioning in Kruger National Park alters elephant spatial utilisation patterns. J. Arid Environ. 141, 45–51 (2017).
Shannon, G., Matthews, W. S., Page, B. R., Parker, G. E. & Smith, R. J. The affects of artificial water availability on large herbivore ranging patterns in savanna habitats: A new approach based on modelling elephant path distributions. Divers. Distrib. 15, 776–783 (2009).
Kos, M. et al. Seasonal diet changes in elephant and impala in mopane woodland. Eur. J. Wildl. Res. 58, 279–287 (2012).
Shannon, G., Mackey, R. L. & Slotow, R. Diet selection and seasonal dietary switch of a large sexually dimorphic herbivore. Acta Oecologica 46, 48–55 (2013).
Loarie, S. R., van Aarde, R. J. & Pimm, S. L. Elephant seasonal vegetation preferences across dry and wet savannas. Biol. Conserv. 142, 3099–3107 (2009).
Scogings, P. F. et al. Seasonal variations in nutrients and secondary metabolites in semi-arid savannas depend on year and species. J. Arid Environ. 114, 54–61 (2015).
Birkett, P. J., Vanak, A. T., Muggeo, V. M. R., Ferreira, S. M. & Slotow, R. Animal perception of seasonal thresholds: Changes in elephant movement in relation to rainfall patterns. PLoS ONE 7, 1–8 (2012).
Cushman, S. A., Chase, M. & Griffin, C. Elephants in space and time. Oikos 109, 331–341 (2005).
Bohrer, G., Beck, P. S., Ngene, S. M., Skidmore, A. K. & Douglas-Hamilton, I. Elephant movement closely tracks precipitation-driven vegetation dynamics in a Kenyan forest-savanna landscape. Mov. Ecol. 2, 1–12 (2014).
Purdon, A., Mole, M. A., Chase, M. J. & van Aarde, R. J. Partial migration in savanna elephant populations distributed across southern Africa. Sci. Rep. 8, 1–11 (2018).
Shannon, G., Page, B. R., Duffy, K. J. & Slotow, R. The ranging behaviour of a large sexually dimorphic herbivore in response to seasonal and annual environmental variation. Austral Ecol. 35, 731–742 (2010).
Tsalyuk, M., Kilian, W., Reineking, B. & Getz, W. M. Temporal variation in resource selection of African elephants follows long-term variability in resource availability. Ecol. Monogr. 89, 1–19 (2019).
Thaker, M., Prins, H. H. T., Slotow, R., Vanak, A. T. & Gupte, P. R. Fine-scale tracking of ambient temperature and movement reveals shuttling behavior of elephants to water. Front. Ecol. Evol. 7, 1–12 (2019).
Govender, N., Trollope, W. S. W. & Van Wilgen, B. W. The effect of fire season, fire frequency, rainfall and management on fire intensity in savanna vegetation in South Africa. J. Appl. Ecol. 43, 748–758 (2006).
MacFadyen, S., Hui, C., Verburg, P. H. & Van Teeffelen, A. J. A. Spatiotemporal distribution dynamics of elephants in response to density, rainfall, rivers and fire in Kruger National Park, South Africa. Divers. Distrib. 25, 880–894 (2019).
Edwards, M. A., Nagy, J. A. & Derocher, A. E. Low site fidelity and home range drift in a wide-ranging, large Arctic omnivore. Anim. Behav. 77, 23–28 (2009).
Switzer, P. Site fidelity in predictable and unpredictable habitats. Evol. Ecol. 7, 533–555 (1993).
Kranstauber, B., Kays, R., Lapoint, S. D., Wikelski, M. & Safi, K. A dynamic Brownian bridge movement model to estimate utilization distributions for heterogeneous animal movement. J. Anim. Ecol. 81, 738–746 (2012).
Kranstauber, B., Smolla, M. & Safi, K. Similarity in spatial utilization distributions measured by the earth mover’s distance. Methods Ecol. Evol. 8, 155–160 (2017).
Wartmann, F., Juarez, C. & Fernandez-duque, E. Size, site fidelity, and overlap of home ranges and core areas in the socially monogamous owl monkey (Aotus azarae) of Northern Argentina. Int. J. Primatol. 35, 919–939 (2014).
Pringle, R. M. Elephants as agents of habitat creation for small vertebrates at the patch scale. Ecology 89, 26–33 (2008).
Valeix, M. et al. Elephant-induced structural changes in the vegetation and habitat selection by large herbivores in an African savanna. Biol. Conserv. 144, 902–912 (2011).
Coverdale, T. C. et al. Elephants in the understory: opposing direct and indirect effects of consumption and ecosystem engineering by megaherbivores. Ecology 97, 3219–3230 (2016).
Gertenbach, W. Rainfall patterns in the Kruger National Park. Koedoe 23, 35–43 (1980).
Venter, F. J., Scholes, R. J. & Eckhardt, H. C. The abiotic template and its associated vegetation pattern. In The Kruger Experience (eds du Toit, J. T. et al.) 83–129 (Island Press, 2003).
Young, K. D., Ferreira, S. M. & van Aarde, R. J. The influence of increasing population size and vegetation productivity on elephant distribution in the Kruger National Park. Austral Ecol. 34, 329–342 (2009).
Ferreira, S. M., Greaver, C. & Simms, C. Elephant population growth in Kruger National Park, South Africa, under a landscape management approach. Koedoe 59, 1–6 (2017).
Brownrigg, R. Package ‘Maps’: Draw Geographical Maps (2022).
Kranstauber, B. & Smolla, M. Move: Visualizing and analyzing animal track data. R package version 2.1.0 (2013).
R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. URL https://www.R-project.org/ (2017).
Horne, J. S., Garton, E. O., Krone, S. M. & Lewis, J. S. Analyzing animal movement using Brownian bridges. Ecology 88, 2354–2363 (2007).
Wato, Y. A. et al. Movement patterns of African elephants (Loxodonta africana) in a semi-arid savanna suggest that they have information on the location of dispersed water sources. Front. Ecol. Evol. 6, 1–8 (2018).
Polansky, L., Kilian, W. & Wittemyer, G. Elucidating the significance of spatial memory on movement decisions by African savannah elephants using state-space models. Proc. R. Soc. B Biol. Sci. 282, 1–7 (2015).
Archibald, S. & Scholes, R. J. Leaf green-up in a semi-arid African savanna–separating tree and grass responses to environmental cues. J. Veg. Sci. 18, 583–594 (2007).
Majozi, N. P. et al. Analysing surface energy balance closure and partitioning over a semi-arid savanna FLUXNET site in Skukuza, Kruger National Park, South Africa. Hydrol. Earth Syst. Sci. 21, 3401–3415 (2017).
Dodge, S. et al. The environmental-data automated track annotation (Env-DATA) system: Linking animal tracks with environmental data. Mov. Ecol. 1, 1–14 (2013).
Didan, K. MOD13Q1 MODIS/terra vegetation indices 16-day L3 global 250m SIN Grid V006. NASA EOSDIS Land Process. DAAC https://doi.org/10.5067/MODIS/MOD13Q1.006 (2015).
Redfern, J. V., Grant, C. C., Gaylard, A. & Getz, W. M. Surface water availability and the management of herbivore distributions in an African savanna ecosystem. J. Arid Environ. 63, 406–424 (2005).
Young, K. D., Ferreira, S. M. & van Aarde, R. J. Elephant spatial use in wet and dry savannas of southern Africa. J. Zool. 278, 189–205 (2009).
Goldenberg, S. Z., Douglas-Hamilton, I. & Wittemyer, G. Inter-generational change in African elephant range use is associated with poaching risk, primary productivity and adult mortality. Proc. R. Soc. B Biol. Sci. 285, 1–8 (2018).
Woolley, L.-A. et al. Population and individual elephant response to a catastrophic fire in Pilanesberg National Park. PLoS ONE 3, 1–10 (2008).
Eby, S. L., Anderson, T. M., Mayemba, E. P. & Ritchie, M. E. The effect of fire on habitat selection of mammalian herbivores: The role of body size and vegetation characteristics. J. Anim. Ecol. 83, 1196–1205 (2014).
Brooks, M. E. et al. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J. 9, 378–400 (2017).
Burnham, K. P. & Anderson, D. R. Model Selection and Multimodal Inference: A Practical Information-Theoretic Approach (Springer, 2002).
Mazerolle, M. J. AICcmodavg: Model Selection and Multimodel Inference Based on (Q)AIC(c) (2020).
van Moorter, B. et al. Memory keeps you at home: A mechanistic model for home range emergence. Oikos 118, 641–652 (2009).
Guldemond, R. A. R., Purdon, A. & van Aarde, R. J. A systematic review of elephant impact across Africa. PLoS ONE 12, 1–12 (2017).
Abraham, J. O., Goldberg, E. R., Botha, J. & Staver, A. C. Heterogeneity in African savanna elephant distributions and their impacts on trees in Kruger National Park, South Africa. Ecol. Evol. 11, 5624–5634 (2021).
Wall, J., Douglas-Hamilton, I. & Vollrath, F. Elephants avoid costly mountaineering. Curr. Biol. 16, 527–529 (2006).
Presotto, A., Fayrer-Hosken, R., Curry, C. & Madden, M. Spatial mapping shows that some African elephants use cognitive maps to navigate the core but not the periphery of their home ranges. Anim. Cogn. 22, 251–263 (2019).
Landman, M., Schoeman, D. S., Hall-Martin, A. J. & Kerley, G. I. H. Understanding long-term variations in an elephant piosphere effect to manage impacts. PLoS ONE 7, 1–11 (2012).
Fahrig, L. et al. Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol. Lett. 14, 101–112 (2011).
Hamm, M. & Drossel, B. Habitat heterogeneity hypothesis and edge effects in model metacommunities. J. Theor. Biol. 426, 40–48 (2017).
Katayama, N. et al. Landscape heterogeneity-biodiversity relationship: Effect of range size. PLoS ONE 9, 1–8 (2014).
Tews, J. et al. Animal species diversity driven by habitat heterogeneity/diversity: The importance of keystone structures. J. Biogeogr. 31, 79–92 (2004).
O’Connor, T. G., Goodman, P. S. & Clegg, B. A functional hypothesis of the threat of local extirpation of woody plant species by elephant in Africa. Biol. Conserv. 136, 329–345 (2007).
Codron, J. et al. Elephant (Loxodonta africana) diets in Kruger National Park, South Africa: Spatial and landscape differences. J. Mammal. 87, 27–34 (2006).
Mduma, S. A. R., Sinclair, A. R. E. & Hilborn, R. Food regulates the Serengeti wildebeest: A 40-year record. J. Anim. Ecol. 68, 1101–1122 (1999).
Ogutu, J. O. & Owen-Smith, N. ENSO, rainfall and temperature influences on extreme population declines among African savanna ungulates. Ecol. Lett. 6, 412–419 (2003).
Codron, J. et al. Landscape-scale feeding patterns of African elephant inferred from carbon isotope analysis of feces. Oecologia 165, 89–99 (2011).
Woolley, L.-A., Millspaugh, J. J., Woods, R. J., Page, B. R. & Slotow, R. Intraspecific strategic responses of African elephants to temporal variation in forage quality. J. Wildl. Manag. 73, 827–835 (2009).
Dube, K. & Nhamo, G. Evidence and impact of climate change on South African national parks. Potential implications for tourism in the Kruger National Park. Environ. Dev. 33, 1–11 (2020).
Tshipa, A. et al. Partial migration links local surface-water management to large-scale elephant conservation in the world’s largest transfrontier conservation area. Biol. Conserv. 215, 46–50 (2017).
Nathan, R. et al. Big-data approaches lead to an increased understanding of the ecology of animal movement. Science (80-.) 375, 1–12 (2022).
Kays, R., Crofoot, M. C., Jetz, W. & Wikelski, M. Terrestrial animal tracking as an eye on life and planet. Science (80-.) 348, 1222–1232 (2015).
Mpakairi, K. S., Ndaimani, H., Tagwireyi, P., Zvidzai, M. & Madiri, T. H. Futuristic climate change scenario predicts a shrinking habitat for the African elephant (Loxodonta africana): Evidence from Hwange National Park, Zimbabwe. Eur. J. Wildl. Res. 66, 1–10 (2020).
Staver, A. C., Wigley-Coetsee, C. & Botha, J. Grazer movements exacerbate grass declines during drought in an African savanna. J. Ecol. 107, 1482–1491 (2019).
Asner, G. P., Vaughn, N., Smit, I. P. J. & Levick, S. Ecosystem-scale effects of megafauna in African savannas. Ecography (Cop.) 39, 240–252 (2016).
Shannon, G. et al. Relative impacts of elephant and fire on large trees in a savanna ecosystem. Ecosystems 14, 1372–1381 (2011).
Mole, M. A., DÁraujo, S. R., van Aarde, R. J., Mitchell, D. & Fuller, A. Coping with heat: Behavioural and physiological responses of savanna elephants in their natural habitat. Conserv. Physiol. 4, 1–11 (2016).
Ncongwane, K. P., Botai, J. O., Sivakumar, V., Botai, C. M. & Adeola, A. M. Characteristics and long-term trends of heat stress for South Africa. Sustainability 13, 1–20 (2021).
Lagendijk, G., Mackey, R. L., Page, B. R. & Slotow, R. The effects of herbivory by a mega- and mesoherbivore on tree recruitment in sand forest, South Africa. PLoS ONE 6, 1–9 (2011).
Wells, H. B. M. et al. Experimental evidence that effects of megaherbivores on mesoherbivore space use are influenced by species’ traits. J. Anim. Ecol. 90, 2510–2522 (2021).
Thaker, M. et al. Minimizing predation risk in a landscape of multiple predators: Effects on the spatial distribution of African ungulates. Ecology 92, 398–407 (2011).
Fležar, U. et al. Simulated elephant-induced habitat changes can create dynamic landscapes of fear. Biol. Conserv. 237, 267–279 (2019).
Brennan, A. et al. Characterizing multispecies connectivity across a transfrontier conservation landscape. J. Appl. Ecol. 57, 1700–1710 (2020).
Roever, C. L., van Aarde, R. J. & Leggett, K. Functional connectivity within conservation networks: Delineating corridors for African elephants. Biol. Conserv. 157, 128–135 (2013).
Green, S. E., Davidson, Z., Kaaria, T. & Doncaster, C. P. Do wildlife corridors link or extend habitat? Insights from elephant use of a Kenyan wildlife corridor. Afr. J. Ecol. 56, 860–871 (2018).
Funding
The study was supported by funding from the Amarula Trust and University of Kwazulu-Natal.
Author information
Authors and Affiliations
Contributions
R.B.R., L.S.C. and G.S. conceived and designed the research. R.S. provided the elephant movement data, N.G. provided the fire data, R.B.R. performed the statistical analysis, R.B.R., L.S.C., R.S., M.T., A.V.T. and G.S. interpreted the results. R.B.R. wrote the manuscript; all authors contributed ideas and provided editorial revisions.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Burton-Roberts, R., Cordes, L.S., Slotow, R. et al. Seasonal range fidelity of a megaherbivore in response to environmental change. Sci Rep 12, 22008 (2022). https://doi.org/10.1038/s41598-022-25334-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-25334-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.