Abstract
E-learning activities are becoming more and more common. Whilst it is well known that the physical presence of others motivates individuals to engage in perceptual and learning tasks, systematic investigations comparing the effects of physical and virtual co-presence of others on knowledge acquisition are still scarce. Here we investigate the effects of physical and virtual co-presence of others on explicit and implicit learning. In Experiment 1 (discovery sample), retrieval accuracy in a spatial memory task and EEG indexes (mismatch negativity-MMN) of implicit perceptual learning were recorded when participants were alone or in presence of another individual. In Experiment 2 (replicating sample), we added a “virtual” condition, where the same tasks were performed during a video-conference call. In both experiments, MMN was demonstrated to encode for perceptual learning as revealed by the significant correlation with Bayesian Surprise (a consolidated information-theoretic index of Bayesian learning). Furthermore, In Experiments 1 and 2 physical co-presence systematically ameliorated memorization performances and increased MMN indexes related to implicit learning. These positive effects were absent in the virtual condition, thus suggesting that only physical, but not virtual co-presence is effective in potentiating learning dynamics.
Similar content being viewed by others
Introduction
Although the use of on-line education offers significant practical, economic and safety (as during the Covid-19 pandemic) advantages to both students and teachers, the effects of physical vs. virtual presence of others (i.e., peers and teachers) on learning outcomes is still poorly investigated. A better understanding of the differences and similarities between ‘in presence’ and online learning is urgently needed to orient present and future decisions of teachers, institutions and policymakers regarding the use of online teaching activities.
Sharing information with others is vital for collective coordination1 and we might have evolved to prefer information that is shared2. Indeed, social psychology revealed that sharing mental states with others3 favors the encoding of novel information, emotions and sensations4, because sensory outcomes of shared vs. solo experiences are amplified5,6,7, so that people devote greater cognitive resources to co-attended stimuli8,9 which undergo deeper processing10,11. In general, co-presence positively affects cognitive functions, such as attention to a target stimulus12,13, social and behavioural learning9, language acquisition14, memory15,16,17,18, intrinsic motivation19, dissonance reduction20 and behavioural control21. Overall, these findings are generally referred to as shared attention effects17. To date, however, no experimental evidence of the electrophysiological correlates of such effects has been provided yet22.
Attentional resources are particularly likely to be directed to information that is co-attended with significant others, such as in-group members13,15,18, partners23 and caregivers1,24. Indeed, “psychological closeness”23 between co-experiencers moderates the amplification of shared experiences. Boothby et al.6, modulated psychological closeness by manipulating social (strangers vs. friends) and physical (being in the same room vs. being connected via live video feed) distance and found that only socially and physically closer co-experiencer showed amplified experiences. These results suggest that stimuli that are remotely co-experienced may not attract human attention as when shared with physically present co-attendants.
Other studies, however, suggest that virtual co-presence can mimic shared attention effects. Shteynberg et al.9 falsely told their participants that another participant was participating online, and they demonstrated that experience that is shared with a sham similar virtual other (who appeared to have chosen the same colour avatar as the participant) can foster social imitation, emotional intensification8 and the recall of a list of co-attended words18. Similarly, it has been recently shown that adult online learning of factual information is better for live interactive presentations than recorded video presentations25. These studies, however, were focused on the in-group/out-group variable or other social factors and did not directly compare physical and online co-presence conditions and the magnitude of the corresponding effects.
In the present study, we investigated whether (1) the physical presence of others, relative to their absence, potentiates memory, as well as implicit learning of sensory regularities; (2) the electrophysiological investigation of perceptual learning dynamics may uncover the underlying neural mechanisms of shared attention effects; and (3) the virtual presence of others (e.g. during a video-call) has a comparable effect to physical presence on memorization and perceptual learning.
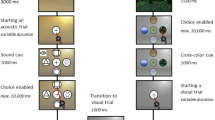
In Experiment 1 (discovery sample), participants (N = 18) performed a spatial memorization task (Experiment 1a) and an EEG task (1b) in two different ‘presence’ conditions: Solo (alone) and Other (together with a confederate—see Fig. 1). In Experiments 2a–b (replicating sample), we replicated the same tasks, with the inclusion of an additional Virtual condition (the confederate participated via video-call). Behavioural Exp. 1a–2a consisted of a traditional spatial memory task (similar to the popular “memory” game), where participants were asked to memorize and then recall the spatial locations of common objects. In EEG Exp. 1b–2b, participants were asked to listen to a stream of higher or lower frequency (Hz) sounds, while we registered their mismatch negativity (MMN) response, a differential wave obtained by subtracting the neural response to Standard events from that of Deviant events26. The MMN response to frequency—in Hz—deviant sounds, peaking between 150 and 250 ms post-stimulus onset, is thought to result from the activations of the posterior auditory cortex (superior temporal gyrus) and the inferior frontal gyrus27,28, and is often interpreted as a neurophysiological signature of implicit perceptual learning, i.e. indexing the update of the predictive models of the sensory environment29,30,31. Therefore, to verify such an association between MMN and perceptual learning in the present paradigm, in both EEG experiments we performed a point-by-point correlation between single-trial amplitude fluctuations and Bayesian Surprise, a well-known theoretic index of perceptual learning30,32 (see “Methods” and SI).
The top panel represents the experimental set-up across conditions in the two experiments. In Experiment 2, the screen used for the video-call was shown to the participant just before each experiment and subsequently placed next to the participant were the confederate sat in the Other condition. The screen is here represented in front of the participant for display reasons only. The screen was shown to the participant for a few seconds and then positioned next to the participants in the same position as in the Other condition (see “Stimuli and experimental design”). The left bottom panel represents the pictures employed in the memorization phase of the memory task. The right bottom panel represents Standard and Deviant auditory stimuli employed in the MMN roving paradigm. Human figures were created with the Adobe software Mixamo (https://www.mixamo.com).
Coherently with previous behavioural studies, we expected to observe improved explicit and implicit learning in the Other as compared to the Solo condition, expressed by better memorization performances (Exp. 1a) and larger MMN responses (Exp. 1b), respectively. Moreover, if virtual presence similarly influences memorization as physical presence, in Experiments 2 we expect to observe comparable results in Other and Virtual conditions.
Methods
Experiment 1 (discovery sample)
Participants
Eighteen healthy right-handed subjects participated in Experiment 1 (13 females; mean age: 26.95 years, ± 2.98 SD; mean years of education: 18.12, ± 1.75 SD). Participants were homogeneous in terms of education, most were graduate university students. All participants gave their written informed consent to participate in the study, which conformed to the standards required by the Declaration of Helsinki and was approved by the Ethics Committee of the University of Turin (Prot. n. 121724—01/03/18). Participants were not compensated for taking part in the experiment.
Sample size (N = 18) was a priori determined through a power analysis based on the effect size obtained in a pilot experiment identical to the main behavioural experiment, involving 6 additional participants and exploring spatial memorization accuracies (ACC) between Other and Solo conditions (Cohen’s d = 0.823; α = 0.05; required power = 0.95). Additionally, since there is not an established method for estimating the required sample of properly powered point-by-point cluster-based statistics (see “Data analysis”) in EEG studies, we followed Brysbaert's guidelines33 which suggest that when power is unknown one should to assume a medium effect sizes (d = 0.5), and run the required amount of participants according to the specific design, which in our case is a repeated measure test of a main effect (see Table 7 in Brysbaert33; required sample = 18; required power = 0.8). The resulting required sample was similar to the ones in previous studies where we employed the same EEG analyses34,35,36. Moreover, to maximize statistical power and reduce the probability of false positives37 we planned the collection of two independent discovery and replicating samples (see “Experiment 2 (replicating sample)”).
Stimuli and experimental design
The experiment was based on a within-subject design. Each participant performed the experiment in two experimental conditions. The experiment (Fig. 1) consisted of two sessions, each divided into two different conditions (i.e., Solo and Other). In the Solo condition participants were alone in the experimental room during the tasks. In the Other condition, subjects sat next to another individual. For each condition participants had to perform two different tasks: a behavioural ecological task (i.e. ‘memory task’) and an electrophysiological task (i.e. ‘EEG Mismatch Negativity task’). The two experimental tasks were performed in two different days. The order of presentation of both conditions and tasks was randomized across subjects.
To test explicit learning and memorization, in the memory task we employed the memory V.R. test, an object-location memory test developed in collaboration with SynArea Consultants Srl (Turin, Italy). Object location tests have been proved to be an effective assessment for explicit memory competences38. The task was developed using Unity software (https://unity3d.com). The visual stimuli employed were 16 everyday-life object images (i.e., ball, glass, book) presented in a 4 rows × 4 columns pattern. The memory V.R. test consisted in two phases, one devoted to memorization and the other to recall (see “Memory task”). Importantly, in the Other condition, the confederate sat next to 4 participants only in the memorization phase.
The EEG MMN task was designed to investigate implicit perceptual learning. Converging evidence from electrophysiological studies suggests that the MMN elicited by the presentation of sounds that deviate from a pattern established by the preceding inputs39,40, is typically considered a neurobiological marker of implicit perceptual learning of sensory regularities41,42. If co-presence of another subject modulates implicit perceptual learning, we expect to observe significantly different MMNs in the two scenarios.
EEG was registered while subjects listened to sound sequences. Deviant and Standard sounds were presented according to the classic roving paradigm with two sounds differing in their frequency (Hz; high-pitch and low-pitch). For further details refer to the SI.
During the experiments, participants sat at a table with eyes open, in front of a 53 cm (21 inches) computer screen. The screen centre was aligned with the subjects’ trunk midline. Each arm of the participant arms was resting on the corresponding leg during the MMN roving paradigm. In the Other condition, the confederate sat on a chair one meter away from the participant, so that they could easily see the computer screen and hear the sounds played via the loudspeakers. Both participants and confederates were asked to remain silent and fixate the computer screen during the experiments.
Memory task
Subjects performed the memory task twice, once for each condition. Each of the two condition had a total duration of 27 min. Each experimental block started with the memorization phase (duration: 2 min), where participants were asked to pay attention to the location of the objects (see Fig. 1) and try to memorize it. Subsequently, subjects performed a distraction task for 5 min, to avoid any rehearsal effects. In the final recall phase, participants were asked to place the 16 objects in the exact position displayed in the memorization phase. Each object placement had to be completed within 10 s, and was tracked by a green bar displayed on the screen under the object. The experimental block (memorization phase, distraction task and recall phase) was repeated three times for each condition (i.e., Solo and Other), each block presenting a different random-generated objects layout. In the Other condition, participants were told they were going to memorize locations together with their confederate, but for logistic reasons both of them were going to recall them in separate rooms. We specified that it was not a competition and that their data were not going to be compared. The presentation software automatically registered reaction times and response accuracy.
EEG MMN task
Here we investigated whether and how implicit perceptual learning is modulated by contextual factors, such as the presence of another person. To test this hypothesis, in each of the two sessions, one for each condition, participants performed 2 runs of a standard roving paradigm while we registered their EEG activity. Sound sequences consisted of trains of 1152 stimuli per run. During the Other condition, participants and confederates were simply asked to remain silent, look straight ahead at a central fixation cross on the screen and listen to the sound sequences; during the Solo condition, participants performed the same EEG MMN task alone. The order of presentation of the two runs (total duration: approximately 40 min) was randomized across participants, as to exclude any specific sequence effect. Differently from traditional oddball paradigms43, where the repeated presentation of standard sounds is occasionally interrupted by the occurrence of physically different deviant sounds, in roving paradigms44,45 different stimuli (high-pitch and low-pitch intervals in our case) can represent both Deviant and Standard stimuli (Fig. 1).
High-pitch and low-pitch intervals were presented in consecutive trains of alternating pitch with a constant inter-stimulus interval of 1 s (in accordance with previous studies employing similar inter-trials30). Any time a change in the stimulation stream occurs (i.e., the passage from a high-pitch to a low-pitch stimulus train or vice versa), the first stimulus of the new train constitutes a Deviant event, since it differs from the preceding train of stimuli, which are therefore considered Standard46. The length of the trains of high-pitch and low-pitch intervals was chosen according to a pseudo-random order, so that both the number of presentations and the average value of the Bayesian surprise (see “Data analysis”) were equal across pitch types (i.e. high or low; Fig. 1). Moreover, the ratio between Standard (80%), and Deviant (20%) trials was kept constant across runs. Differently from traditional oddball paradigms, in roving protocols each stimulus type has exactly the same probability of occurrence, thus allowing to dissociate genuine effects of Bayesian perceptual learning from rarity-driven modulations.
Data analysis
Behavioral analyses
Single subjects’ (N = 18) mean accuracies corresponding to the Solo and Other condition from the memory task were compared with a paired sample two-tailed t-test. Behavioural results were corrected for multiple comparisons (total number of tests = 4; see Exp. 2a) by a Benjamini–Hochberg procedure with a false discovery rate of 5%.
Bayesian perceptual surprise computation
For a detailed description of the mathematical computations please refer to the Supplementary Information. Similarly to previous studies30,32,35,47, to relate single-trial EEG signals to Bayesian perceptual learning, we computed Bayesian surprise values for each single trial using a sequential Bayesian learning algorithm of stimulus probabilities, thus obtaining 1152 estimated surprise values (i.e., the total number of sounds composing the sequence presented in each session of the EEG MMN task).
Statistical analyses
ERPs belonging to the same condition (i.e., Solo or Other) and to the same type (i.e., standard vs. deviant) were then averaged, to obtain four average waveforms for each subject (i.e., Solo Standard, Solo Deviant, Other Standard, Other Deviant). For preprocessing details please refer to SI.
In the present study we employed point-by-point statistical tests. It is worth noting that point-by-point analyses represent a statistical approach common in EEG studies48,49,50,51,52, directed to highlight possible differences between conditions across the whole epoch time-course, without any a-priori assumption. One statistical comparison for each time point composing a waveform was performed. In order to correct for multiple comparisons, a cluster-based permutation testing approach (1000 random permutations; alpha level = 0.05; percentile of mean cluster sum = 95; minimum number of adjacent channels = 2) was employed to each point-by-point analysis53. Significant clusters were based on both temporal contiguity and spatial adjacency of a minimum of two electrodes. The so obtained clusters of significance represent the result of the point-by-point analyses, corrected by permutation testing. Hence, pre-processed epochs and Bayesian surprise values corresponding to single trials served as input for a point-by-point trial-by-trial correlation analysis49,54. For each participant and for each condition separately, the analysis computed the correlation between Bayesian surprise and trial-by-trial (N = 1152) fluctuations of the EEG signal registered at single channels. The outcome of the correlation analysis was two 1 s long time series of r-values for each channel and for each subject (from 0.15 s pre-onset to 0.85 s post-onset). This constituted the input for two group-level two-tailed point-by-point t-tests with permutation-based correction for multiple comparisons (1000 permutations; alpha level = 0.05; percentile of mean cluster sum = 95; minimum number of adjacent channels = 2). For the Solo condition and the Other condition separately, the test compared single subjects’ correlation coefficients against the constant 0 at each time point. This allowed us to identify time-clusters containing signal amplitudes which significantly correlated with Bayesian surprise indexing model update. Supposedly, these clusters contain waveforms that are significantly modulated by Bayesian perceptual learning. We expected to find greater correlation values around the time interval of the MMN, N2, P300, N4 indexes, in correspondence of the centro-parietal electrodes.
Subsequently, to explore the presence of a significant modulatory effect of co-presence on the MMN index, we computed MMN differential waves for each subject and for the two conditions separately. MMN responses were obtained by subtracting the ERP elicited by standard intervals (in this analysis we included only the last standard trial for each stimuli train occurring before deviant trials30; N = 208) from that elicited by deviant intervals26. Single participants’ MMN registered on single channels were then entered in group-level analyses. Since we were interested in testing for possible differences in MMN responses corresponding to the two conditions, we performed a point-by-point t-test49, with clustersize-based permutation correction for multiple comparisons based on temporal consecutivity (1000 permutations; alpha level = 0.05; percentile of mean cluster sum = 95; minimum number of adjacent channels = 2), on differential MMN (Deviant-Standard) values. The test compared single subjects’ MMN amplitudes corresponding to the Solo and Other conditions at each time point, for each channel separately. This allowed us to identify time-clusters containing MMN indexes of implicit perceptual learning which significantly differed between Solo and Other conditions.
Experiment 2 (replicating sample)
Participants
Eighteen healthy right-handed subjects participated in the experiment (9 women, age mean = 26.22; ± SD = ± 1.55; years of education, mean: 17.73; ± SD: ± 1.27). All participants gave their written informed consent to participate in the study, which conformed to the standards required by the Declaration of Helsinki and was approved by the Ethics Committee of the University of Turin (Prot. n. 121724—01/03/18). Participants were not compensated for taking part in the experiment.
Stimuli and experimental design
All procedures were identical to those employed in Experiment 1, except for the addition of a third condition, i.e., the Virtual condition, both in the behavioural and in the electrophysiological task. In this Virtual condition, the same confederate of the Other condition performed the memorization phase of the memory task and listened to the sound sequences together with the subject via a video-call (i.e. using Skype). To avoid confounding effects by keeping the different conditions (Other and Virtual) perceptually identical, after an initial phase where subjects were shown their partners on the computer screen (video-calls were started on a lap-top while the experiment was run on a different computer), the screen was placed next to the participant, in the same position where the confederate was in the Other condition. Participants were told that their confederate could see and hear the presented stimuli and, similarly to the Other condition, that confederates were going to perform the recall phase of the memory task separately for logistic reasons. Because time spent together and shared experiences can influence the psychological distance between participants and confederate, which in turn can amplify shared attention effects6, procedure were kept as similar as possible between the Other and Virtual conditions: in both conditions participants and confederates were asked to look at each other for a few seconds and say hello to each other. After this minimal familiarization, (which was conceived mainly to exclude the possibility that participants believed that the videocall was registered and no one was taking part to the experiment together with them) participants were asked to look straight ahead at the fixation cross. Similarly to Experiment 1, both the participant and the confederate were asked to remain silent throughout the experiments.
Data analysis
Statistical analyses were identical to those employed in Experiment 1, with the only exception that the point-by-point correlation analysis was performed three times, once for each condition (i.e. Solo, Other; Virtual). Coherently, three point-by-point t-tests confronted MMNs values corresponding to three conditions (Other vs. Solo, Other vs. Virtual, Virtual vs. Solo).
The datasets analysed (Single subjects’ results from Experiment 1 and 2) during the current study and the experiment code generated by the e-prime presentation software are available in the Mendeley repository [Sarasso, Pietro (2022), “Whith or without you—MMN in virtual and physical co-presence”, Mendeley Data, V2, https://doi.org/10.17632/9sj977x4j4.2]. Neither of these experiments was formally preregistered.
Results
Experiment 1
Spatial memory task
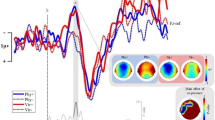
The effect of physical co-presence was evaluated by comparing memorization accuracies in the Solo condition (M = 34.62 (72.125%); SD = 8.2) with accuracies from the Other condition (M = 39.17 (81.25%); SD = 3.73). A paired sample t-test has been used for this purpose. Significant improvements were observed in memorization performances corresponding to the Other condition compared to the Solo condition, [t(17) = − 2.32, p = 0.033, dz = 0.58; Fig. 2a]. The present p value survived Benjamini–Hochberg correction for multiple comparisons (total number of performed tests = 4; false discovery rate 5%).
Whisker Plots in (A) depict behavioural results from Experiment 1a corresponding to the Solo (light blue) and Other (blue) conditions. Dots represent single subjects’ mean accuracies. Panel (B) shows average waveforms and peak amplitudes distribution on the scalp for relevant components (N1, N2, P3, N4) from Experiment 1b. Dotted and solid lines represent average ERPs corresponding to Standard and Deviant trials respectively. MMN differential waveforms are displayed in (C) (bottom-right) and show a negative peak at approximately 210 ms post-onset. Scalpmaps represent p values distribution across the scalp from the point-by-point t-test. Scalpmaps show results from the t-test at 200 and 300 ms corresponding to the effect peak latency of the two significant time clusters (193–215 ms and 290–317 ms) surviving permutation testing. Panel (D) (left-bottom) represents average r values from the point-by-point correlation analysis. Correlation between Bayesian Surprise and trial-by-trial amplitudes peaked at 210 ms post onset, corresponding to the MMN peak latency. Human figures in panel A were created with the Adobe software Mixamo (https://www.mixamo.com).
Trial-by-trial correlation with Bayesian surprise
The correlation analysis between single trial amplitudes and Bayesian Surprise indicated that r values peaked over frontal electrodes (Fz) at similar latencies for Other and Solo conditions (Fig. 2d). At Fz, r values corresponding to the Other and Solo both significantly differed from the costant 0 in three significant clusters which survived cluster correction, corresponding to the latencies of the MMN, the N2-P3a complex and the N4 components of the auditory evoked potential. In the following lines we report the latency of significant clusters at Fz for the Other condition (parentheses indicate p values and t values at peak latencies): 130–244 ms (p < 0.001; t = − 6.39); 262–375 ms (p < 0.001; t = 5.67); 499–497 ms (p < 0.001; t = − 6.43). In the following lines we report the latency of significant clusters at Fz for the Solo condition: 143–240 ms (p < 0.001; t = − 6.1); 265–383 ms (p < 0.001; t = 6.04); 418–551 ms (p < 0.001; t = − 4.4). These results confirmed our prediction, indicating that MMN (peaking at 210 ms post-onset) indexes Bayesian perceptual learning in our study. Furthermore, and in accordance with data from the literature, we also found a correlation between model updating following surprising stimuli (i.e. sensory surprise) and the P3 and N400 components55,56.
MMN results
ERPs elicited by the Other and Solo conditions on Fz (where MMN peak amplitudes are registered) are reported in Fig. 2b. Grand-average waveforms were comparable with previous studies on auditory frequency processing40,57. For both Other and Solo conditions, the MMN differential wave obtained by subtracting Standard from Deviant average response, showed a negative peak over frontocentral at approximately 210 ms postonset, coherently with previous findings40 (Fig. 2c). Crucially, the point-bypoint t-test performed on MMN differential waves (Other vs. Solo) revealed two significant time-clusters surviving cluster correction. The first centro-parietal cluster spanned across C3, Cp5, Cp1 and P7 electrodes at 193–215 ms post-onset (Fig. 2c), which corresponds to the MMN peak latency. This result confirmed that, as expected, MMN waveforms were significantly larger in the Other condition compared to the Solo condition. The scalp location of the effect of condition on MMN differential waves is coherent with previous studies that localized the source of MMN responses to frequency deviance in the superior temporal gyrus27,28. Müller et al. PET study58 showed that when contrasting frequency deviant and frequency standard sounds the largest magnitude of activation to frequency deviant sounds emerged in the lower part of the left posterior superior temporal gyrus. The second cluster surviving cluster correction is centred over frontal electrodes (Fp1, Fpz, Fp2, Fz, F4, Fc5) at 290–317 ms post-onset.
As it can be visualised in Fig. 2b this is possibly due to a more negative rebound of the P2 component following the presentation of Standard trials in the Solo condition (following a larger P2 component in the Other condition).
Experiment 2
Behavioral memory task
Mean accuracies (Fig. 3a) peaked in the Other condition [M = 38.11 (79.4%); SD = 6.4] and were reduced in the Solo condition [M = 33.72 (70.25%); SD = 9.47] and in the and Virtual condition [M = 30.61 (63.77%); SD = 10.39].
Whisker Plots in (A) depict behavioural results from Experiment 2a corresponding to the Solo (light blue), Virtual (purple) and Other (blue) conditions. Dots represent single subjects’ mean accuracies. Panel (B) shows average waveforms and peak amplitudes distribution on the scalp for relevant components (N1, N2, P3, N4) from Experiment 2b. Dotted and solid lines represent average ERPs corresponding to Standard and Deviant trials respectively. MMN differential waveforms are displayed in (C) (bottom-right) and show a negative peak at approximately 210 ms post-onset. Scalpmaps represent p values from point-by-point t-tests (Solo vs. Other; Virtual vs. Other), corresponding to the effect peak latency of significant time clusters (Solo vs. Other: 110–180 ms and 191–231 ms; Virtual vs. Other: 110–135 ms and 235–295 ms) surviving permutation testing. Panel (D) (left-bottom) represents average r values from the point-by-point correlation analysis. Correlation between Bayesian Surprise and trial-by-trial amplitudes peaked at 210 ms post onset, corresponding to the MMN peak latency. Human figures in panel A were created with the Adobe software Mixamo (https://www.mixamo.com).
The effect of physical and virtual co-presence was further evaluated by comparing memorization accuracies corresponding to the three different conditions by means of three two-tailed paired sample t-tests (Other vs. Solo; Other vs. Virtual; Virtual vs. Solo). Significant enhancements in memorization performances were observed in the Other condition compared to the Solo condition [t(17) = 2.59; p = 0.019, dz = 0.73] and in the Other condition compared to Virtual condition [t(17) = 4.07; p < 0.001, dz = 1.06]. Memorization performances in the Virtual and Solo conditions were not significantly different [t(17) = 1.31; p = 0.207; dz = 0.31]. All significant p values survived Benjamini–Hochberg correction for multiple comparisons (total number of performed tests = 4; false discovery rate 5%).
Trial-by-trial correlation with Bayesian surprise
As in Experiment 1, the trial-by-trial correlation analysis between single trial amplitudes and Bayesian Surprise indicated that r values peaked over fronto-central electrodes at three different latencies, similarly for the Other, Solo and Virtual conditions (Fig. 3d). Significant clusters corresponded to the latencies of the MMN, the N2-P3a complex and the N4 components of the auditory evoked potential. At Fz, results from the trial-by-trial correlation analysis performed on EEG responses following the presentation of the three conditions, Other, Solo and Virtual intervals evidenced three significant clusters (i.e. r values significantly differed from the constant 0). For the Other condition: 59–249 ms (p < 0.001; t = − 9.27); 264–347 ms (p < 0.001; t = 4.88); 405–522 ms (p < 0.001; t = − 5.84). For the Solo condition: 129–247 ms (p < 0.001; t = − 6.71); 265–362 ms (p < 0.001; t = 5.66); 407–497 ms (p < 0.001; t = − 5.78). For the Virtual condition: 129–236 ms (p < 0.001; t = − 5.72); 257–336 ms (p < 0.001; t = 8.52); 419–555 ms (p < 0.001; t = − 4.18). These results confirmed our prediction, indicating that MMN indexes Bayesian perceptual learning in our study. Furthermore, and in accordance with data from the literature, we also found a correlation between model updating following surprising stimuli and the P3 and N400 components55,56.
MMN results
ERPs elicited during the Other, Solo and Virtual conditions intervals over Fz are reported in Fig. 3b. Grand-average waveforms were similar to Experiment 1 and comparable with previous studies on auditory frequency processing40. For Other, Solo and Virtual intervals, MMN waveforms showed a negative peak over fronto-central electrodes at approximately 220 ms post-onset (Fig. 3c). The point-by-point t-test comparing MMN waveforms registered over Fz during the Other and Solo conditions (Other vs Solo) revealed two early significant clusters corresponding to the N1 and MMN latencies, and a later significant frontal cluster at approximately 700 ms post onset. The first smaller cluster, corresponding to the N1 negative peak spanned across frontal electrodes (Fp1, Fpz, F7, F3, Fc5) at 110–177 ms post-onset. The second cluster including the time interval between 191 and 231 ms extending over a broad set of frontal, central and parietal electrodes (Fp1, Fpz, Fp2, F7, F3, Fz, FC5, FC1, FC2, FCz, T7, C3, Cz, CP1,CP2, CP6, P3, Pz, P4, Oz, O2 and POz) corresponded to the MMN peak latency. The point by point t-test comparing MMN registered during the Other and Virtual conditions revealed a single significant cluster including the time interval between 235 and 295 ms post-onset, corresponding to the descending portion of the MMN negative peak. The significant cluster spanned across frontal channels. More specifically, the results were significant over: FP1, Fpz, F3, Fz, F4, F8.
The t-test comparing Solo and Virtual conditions (Solo vs. Virtual) was not statistically significant. Overall, as expected, MMN waveforms were significantly larger in the Other condition (Fig. 3c).
Discussion
In Experiment 1 (discovery sample), we demonstrated that in the Other condition, as compared to the Solo condition: (1) memorization performance was significantly higher (Exp. 1a) and (2) the amplitude of MMN responses was significantly greater (Exp. 1b). These results indicate the presence of an enhancement of both high- and low-level perceptual learning processes in case of the physical co-presence of a confederate (Fig. 2). In Experiment 2 (replicating sample), we replicated the presence of a significant difference between Other and Solo conditions, both at a behavioural and EEG level. Surprisingly, in the Virtual condition, memorization performance was significantly lower (Exp. 2a) and MMN responses were significantly reduced (Exp. 2b), as compared to the Other condition. No difference was found between the Virtual and the Solo condition (Exp. 2a–b) (Fig. 3). That is, coherently with previous studies5, the presence of a virtual confederate does not improve implicit and explicit learning as compared to physical presence.
Furthermore, in Exp. 1b and 2b, the correlation between the EEG signal and the Bayesian Surprise index, showed a significant (Figs. 2d and 3d) negative peak within the time window including the MMN (≈ 210 ms post-onset), thus confirming that MMN actually reflects implicit learning mechanisms.
Overall, our results demonstrate that, even in the absence of a joint action18,59,60, physical co-presence is sufficient to enhance both higher-level memory encoding and lower-level perceptual learning, expressed by a distinctive electrophysiological pattern, i.e., the significant enhancement of MMN waveforms. Such effects are generally interpreted as a consequence of the fact that shared attention on a specific sensory event makes it more valuable as compared to when the same event is not co-attended2. The present results demonstrate that this sensory up-weighting impacts not only on higher level memory encoding15,61 but also on low-level perceptual learning. More specifically, as shown by the significant correlation with the Bayesian Surprise index (which quantifies the informational value conveyed by each stimulus), the amplitude of MMN precisely indexes the sensory information weighting according to its estimated relevance62,63. Therefore, the significantly greater amplitudes of MMN responses in the Other vs. Solo condition may be interpreted as the result of a sensory up-weighting driven by physical co-presence. At a neurophysiological level, it has been suggested that this top-down control of sensory weighting may be implemented through the disinhibition of the post-synaptic gain of pyramidal cells, which are tuned to the attended sensory modality63,64.
In Exp. 2b the virtual co-presence of others did not enhance explicit memory encoding and perceptual learning as compared to physical presence, suggesting that attentional mechanisms acted as if subjects were alone. Previous research suggests that the effect of shared attention on memory can be reduced by psychological distance5,6,23, which operationalizes how “close” a partner is in time, space and relational dimensions2. Accordingly, as it has been suggested for previous similar results5, partners connected via videocall might have been perceived more distant psychologically, or relationally, and therefore failed to activate the shared-attention mechanisms responsible for memory enhancement. More explicitly, sitting together in the same room might be that sort of shared subjective experience that is able to modulate intergroup relations and psychological closeness65.
It might be argued that differences in learning outcomes across conditions, rather than being driven by the specific enhancement of perceptual learning dynamics, are related to changes in the general arousal level. In other words, the state of physiological reactivity of the subject might be simply heightened by the physical presence of the confederate. Importantly, general arousal-driven effects should equally affect responses to Standard and Deviant stimuli, rather than be solely focused on change detection responses. However, our control analysis performed on arousal-related components of the evoked potentials (such as N1) elicited by the responses to Standard sounds revealed no significant difference across conditions (see SI). Instead, significant modulations were focused on the responses to Deviant sounds. Crucially, this finding suggests the presence of a neural mechanism, specifically directed to optimize the processing of novel information.
The present results contribute to cumulative theoretical knowledge on social cognition, suggesting that during physical but not virtual presence of conspecifics, more neural resources are selectively devoted to surprising sensory states, thus amplifying mismatch responses. This neural up-weighting of sensory information may represent the underlying mechanism of shared attention effects, allowing individuals to best attune to relevant, shared information and groups to consolidate common knowledge18. The present results, if confirmed, might be of relevance in teaching and rehabilitation research. Future studies should be specifically directed to investigate the role of psychological/relational, contextual and technological variables that modulate perceived psychological distancing in physical and virtual settings, in order to maximize the learning advantages brought by shared attention mechanisms.
References
Tomasello, M., Carpenter, M., Call, J., Behne, T. & Moll, H. Understanding and sharing intentions: The origins of cultural cognition. Behav. Brain Sci. 28, 675–735 (2005).
Wagner, U., Giesen, A., Knausenberger, J. & Echterhoff, G. The joint action effect on memory as a social phenomenon: The role of cued attention and psychological distance. Front. Psychol. https://doi.org/10.3389/fpsyg.2017.01697 (2017).
Koike, T. et al. Neural substrates of shared attention as social memory: A hyperscanning functional magnetic resonance imaging study. Neuroimage 125, 401–412 (2016).
Decety, J. & Fotopoulou, A. Why empathy has a beneficial impact on others in medicine: Unifying theories. Front. Behav. Neurosci. https://doi.org/10.3389/fnbeh.2014.00457 (2015).
Boothby, E. J., Smith, L. K., Clark, M. S. & Bargh, J. A. Psychological distance moderates the amplification of shared experience. Personal. Soc. Psychol. Bull. https://doi.org/10.1177/0146167216662869 (2016).
Boothby, E. J., Clark, M. S. & Bargh, J. A. Shared experiences are amplified. Psychol. Sci. https://doi.org/10.1177/0956797614551162 (2014).
Shteynberg, G., Bramlett, J. M., Fles, E. H. & Cameron, J. The broadcast of shared attention and its impact on political persuasion. J. Pers. Soc. Psychol. https://doi.org/10.1037/pspa0000065 (2016).
Shteynberg, G. et al. Feeling more together: Group attention intensifies emotion. Emotion https://doi.org/10.1037/a0037697 (2014).
Shteynberg, G. & Apfelbaum, E. P. The power of shared experience: Simultaneous observation with similar others facilitates social learning. Soc. Psychol. Personal. Sci. https://doi.org/10.1177/1948550613479807 (2013).
Craik, F. I. M. & Lockhart, R. S. Levels of processing: A framework for memory research. J. Verbal Learn. Verbal Behav. https://doi.org/10.1016/S0022-5371(72)80001-X (1972).
Craik, F. I. & Tulving, E. Depth of processing and the retention of words in episodic memory. J. Exp. Psychol. Gen. https://doi.org/10.1037/0096-3445.104.3.268 (1975).
Böckler, A., Knoblich, G. & Sebanz, N. Effects of a coactor’s focus of attention on task performance. J. Exp. Psychol. Hum. Percept. Perform. https://doi.org/10.1037/a0027523 (2012).
He, X., Lever, A. G. & Humphreys, G. W. Interpersonal memory-based guidance of attention is reduced for ingroup members. Exp. Brain Res. https://doi.org/10.1007/s00221-011-2698-8 (2011).
Lytle, S. R. & Kuhl, P. K. Social interaction and language acquisition: Toward a neurobiological view. In The Handbook of Psycholinguistics. 615–634 (Wiley Blackwell, 2018). https://doi.org/10.1002/9781118829516.ch27.
Eskenazi, T., Doerrfeld, A., Logan, G. D., Knoblich, G. & Sebanz, N. Your words are my words: Effects of acting together on encoding. Q. J. Exp. Psychol. https://doi.org/10.1080/17470218.2012.725058 (2013).
Richardson, D. C. et al. Joint perception: Gaze and social context. Front. Hum. Neurosci. https://doi.org/10.3389/fnhum.2012.00194 (2012).
Shteynberg, G. Shared attention. Perspect. Psychol. Sci. 10, 579–590 (2015).
Shteynberg, G. A silent emergence of culture: The social tuning effect. J. Pers. Soc. Psychol. https://doi.org/10.1037/a0019573 (2010).
Carr, P. B. & Walton, G. M. Cues of working together fuel intrinsic motivation. J. Exp. Soc. Psychol. https://doi.org/10.1016/j.jesp.2014.03.015 (2014).
Sarasso, P., Ronga, I., Neppi-Modona, M. & Sacco, K. The role of musical aesthetic emotions in social adaptation to the Covid-19 pandemic. Front. Psychol. 12, 445 (2021).
Peterburs, J., Liepelt, R., Voegler, R., Ocklenburg, S. & Straube, T. It’s not me, it’s you—Differential neural processing of social and non-social nogo cues in joint action. Soc. Neurosci. https://doi.org/10.1080/17470919.2017.1403374 (2019).
Mairon, N., Nahum, M., Stolk, A., Knight, R. T. & Perry, A. Behavioral and EEG measures show no amplifying effects of shared attention on attention or memory. Sci. Rep. https://doi.org/10.1038/s41598-020-65311-7 (2020).
Boothby, E. J., Smith, L. K., Clark, M. S. & Bargh, J. A. The world looks better together: How close others enhance our visual experiences. Pers. Relatsh. 24, 694–714 (2017).
Reid, V. M., Striano, T., Kaufman, J. & Johnson, M. H. Eye gaze cueing facilitates neural processing of objects in 4-month-old infants. NeuroReport 15, 2553–2555 (2004).
De Felice, S., Vigliocco, G. & de Hamilton, A. F. C. Social interaction is a catalyst for adult human learning in online contexts. Curr. Biol. 31, 4853-4859.e3 (2021).
Näätänen, R., Paavilainen, P., Rinne, T. & Alho, K. The mismatch negativity (MMN) in basic research of central auditory processing: A review. Clin. Neurophysiol. 118, 2544–2590 (2007).
Jääskeläinen, I. P. et al. Human posterior auditory cortex gates novel sounds to consciousness. Proc. Natl. Acad. Sci. U. S. A. https://doi.org/10.1073/pnas.0303760101 (2004).
Molholm, S., Martinez, A., Ritter, W., Javitt, D. C. & Foxe, J. J. The neural circuitry of pre-attentive auditory change-detection: An fMRI study of pitch and duration mismatch negativity generators. Cereb. Cortex https://doi.org/10.1093/cercor/bhh155 (2005).
Lieder, F., Daunizeau, J., Garrido, M. I., Friston, K. J. & Stephan, K. E. Modelling trial-by-trial changes in the mismatch negativity. PLoS Comput. Biol. 9, (2013).
Ostwald, D. et al. Evidence for neural encoding of Bayesian surprise in human somatosensation. Neuroimage 62, 177–188 (2012).
Stefanics, G., Heinzle, J., Attila Horváth, A. & Enno Stephan, K. Visual mismatch and predictive coding: A computational single-trial ERP study. J. Neurosci. https://doi.org/10.1523/JNEUROSCI.3365-17.2018 (2018).
Baldi, P. & Itti, L. Of bits and wows: A Bayesian theory of surprise with applications to attention. Neural Netw. 23, 649–666 (2010).
Brysbaert, M. How many participants do we have to include in properly powered experiments? A tutorial of power analysis with reference tables. J. Cogn. 2, 16 (2019).
Sarasso, P. et al. Memorisation and implicit perceptual learning are enhanced for preferred musical intervals and chords. Psychon. Bull. Rev. https://doi.org/10.3758/s13423-021-01922-z (2021).
Sarasso, P. et al. Nice and easy: Mismatch negativity responses reveal a significant correlation between aesthetic appreciation and perceptual learning. J. Exp. Psychol. Gen. https://doi.org/10.1037/xge0001149 (2021).
Sarasso, P. et al. Preferred music listening is associated with perceptual learning enhancement at the expense of self-focused attention. Psychon. Bull. Rev. https://doi.org/10.3758/s13423-022-02127-8 (2022).
Strong, R. W. & Alvarez, G. Using simulation and resampling to improve the statistical power and reproducibility of psychological research. PsyArXiv Prepr. https://doi.org/10.31234/osf.io/2bt6q (2019).
Stevenson, R. F. et al. Hippocampal CA1 gamma power predicts the precision of spatial memory judgments. Proc. Natl. Acad. Sci. U. S. A. https://doi.org/10.1073/pnas.1805724115 (2018).
Garrido, M. I., Kilner, J. M., Stephan, K. E. & Friston, K. J. The mismatch negativity: A review of underlying mechanisms. Clin. Neurophysiol. https://doi.org/10.1016/j.clinph.2008.11.029 (2009).
Sams, M., Paavilainen, P., Alho, K. & Näätänen, R. Auditory frequency discrimination and event-related potentials. Electroencephalogr. Clin. Neurophysiol. Evoked Potentials https://doi.org/10.1016/0168-5597(85)90054-1 (1985).
Chang, M., Iizuka, H., Naruse, Y., Ando, H. & Maeda, T. Unconscious learning of auditory discrimination using mismatch negativity (MMN) neurofeedback. Sci. Rep. 4, 6729 (2014).
Perez, V. B. et al. Mismatch negativity is a sensitive and predictive biomarker of perceptual learning during auditory cognitive training in schizophrenia. Neuropsychopharmacology 42, 2206 (2017).
Näätänen, R., Gaillard, A. W. K. & Mäntysalo, S. Early selective-attention effect on evoked potential reinterpreted. Acta Psychol. https://doi.org/10.1016/0001-6918(78)90006-9 (1978).
Baldeweg, T., Klugman, A., Gruzelier, J. & Hirsch, S. R. Mismatch negativity potentials and cognitive impairment in schizophrenia. Schizophr. Res. 69, 203–217 (2004).
Rosch, R. E., Auksztulewicz, R., Leung, P. D., Friston, K. J. & Baldeweg, T. Selective prefrontal disinhibition in a roving auditory oddball paradigm under N-methyl-D-aspartate receptor blockade. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 4, 140–150 (2019).
McCleery, A. et al. Parsing components of auditory predictive coding in schizophrenia using a roving standard mismatch negativity paradigm. Psychol. Med. 49, 1195–1206 (2019).
Itti, L. & Baldi, P. Bayesian surprise attracts human attention. Vis. Res. 49, 1295–1306 (2009).
Harris, A. M., Dux, P. E. & Mattingley, J. B. Detecting unattended stimuli depends on the phase of prestimulus neural oscillations. J. Neurosci. https://doi.org/10.1523/JNEUROSCI.3006-17.2018 (2018).
Novembre, G. et al. Saliency detection as a reactive process: Unexpected sensory events evoke cortico-muscular coupling. J. Neurosci. https://doi.org/10.1523/JNEUROSCI.2474-17.2017 (2018).
Ronga, I., Valentini, E., Mouraux, A. & Iannetti, G. D. Novelty is not enough: Laser-evoked potentials are determined by stimulus saliency, not absolute novelty. J. Neurophysiol. https://doi.org/10.1152/jn.00464.2012 (2013).
Sarasso, P. et al. Beauty in mind: Aesthetic appreciation correlates with perceptual facilitation and attentional amplification. Neuropsychologia 136, 107282 (2020).
Sarasso, P. et al. Aesthetic appreciation of musical intervals enhances behavioural and neurophysiological indexes of attentional engagement and motor inhibition. Sci. Rep. 9, 1–14 (2019).
Maris, E. & Oostenveld, R. Nonparametric statistical testing of EEG- and MEG-data. J. Neurosci. Methods https://doi.org/10.1016/j.jneumeth.2007.03.024 (2007).
Pyasik, M. et al. I’m a believer: Illusory self-generated touch elicits sensory attenuation and somatosensory evoked potentials similar to the real self-touch. Neuroimage 229, 117727 (2021).
Bennett, D., Murawski, C. & Bode, S. Single-trial event-related potential correlates of belief updating. eNeuro https://doi.org/10.1523/ENEURO.0076-15.2015 (2015).
Kolossa, A., Kopp, B. & Fingscheidt, T. A computational analysis of the neural bases of Bayesian inference. Neuroimage https://doi.org/10.1016/j.neuroimage.2014.11.007 (2015).
Garrido, M. I., Teng, C. L. J., Taylor, J. A., Rowe, E. G. & Mattingley, J. B. Surprise responses in the human brain demonstrate statistical learning under high concurrent cognitive demand. NPJ Sci. Learn. 1, 16006 (2016).
Müller, B. W., Jüptner, M., Jentzen, W. & Müller, S. P. Cortical activation to auditory mismatch elicited by frequency deviant and complex novel sounds: A PET study. Neuroimage https://doi.org/10.1006/nimg.2002.1176 (2002).
Echterhoff, G. & Kopietz, R. The socially shared nature of memory: From joint encoding to communication. In Collaborative Remembering: Theories, Research, and Applications (2018).
Elekes, F., Bródy, G., Halász, E. & Király, I. Enhanced encoding of the co-actor’s target stimuli during a shared non-motor task. Q. J. Exp. Psychol. https://doi.org/10.1080/17470218.2015.1120332 (2016).
Wagner, U. & Echterhoff, G. When does oxytocin affect human memory encoding? The role of social context and individual attachment style. Front. Hum. Neurosci. 12 (2018).
Garrido, M. I., Rowe, E. G., Halász, V. & Mattingley, J. B. Bayesian mapping reveals that attention boosts neural responses to predicted and unpredicted stimuli. Cereb. Cortex https://doi.org/10.1093/cercor/bhx087 (2018).
Heilbron, M. & Chait, M. Great expectations: Is there evidence for predictive coding in auditory cortex?. Neuroscience https://doi.org/10.1016/j.neuroscience.2017.07.061 (2017).
Auksztulewicz, R. & Friston, K. Attentional enhancement of auditory mismatch responses: A DCM/MEG study. Cereb. Cortex 25, 4273–4283 (2015).
Pinel, E. C., Long, A. E., Johnson, L. C. & Yawger, G. C. More about when I’s meet: The intergroup ramifications of I-sharing, Part II. Pers. Soc. Psychol. Bull. 44, 1601–1614 (2018).
Funding
This study was funded by Finpiemonte (Grant "Rehome" n. 320-36).
Author information
Authors and Affiliations
Contributions
P.S. and I.R. developed the study concept. All authors contributed to the study design. Testing and data collection were performed by E.D., P.B. and I.L.. P.S., I.R., I.L. and P.B. performed the data analysis and interpretation under the supervision of M.N.-M. and K.S. N.R. provided mathematical advice. P.S. and I.R. drafted the manuscript, and K.S., A.C. and M.N.-M. provided critical revisions. All authors approved the final version of the manuscript for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sarasso, P., Ronga, I., Del Fante, E. et al. Physical but not virtual presence of others potentiates implicit and explicit learning. Sci Rep 12, 21205 (2022). https://doi.org/10.1038/s41598-022-25273-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-25273-4
This article is cited by
-
Shared attention in virtual immersive reality enhances electrophysiological correlates of implicit sensory learning
Scientific Reports (2024)
-
Crossing the distance: University student newcomer socialization in online semesters—a case study
Unterrichtswissenschaft (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.