Abstract
Old age and spinal surgery significantly increase the risk of postoperative hyponatremia. However, detailed analyses of postoperative hyponatremia after spinal surgery in elderly patients are lacking. Therefore, we retrospectively reviewed the records of 582 consecutive patients aged > 60 years who underwent spinal surgery to evaluate the frequency, risk factors, and symptoms of postoperative hyponatremia after spinal surgery in the elderly population. Postoperative hyponatremia was defined as a postoperative blood sodium level < 135 meq/L at postoperative day (POD)1, POD3, and/or after POD6. A total of 92 (15.8%) patients showed postoperative hyponatremia. On a multivariate analysis, a diagnosis of a spinal tumor/infection, decompression and fusion surgery, and lower preoperative sodium levels were significant independent factors of postoperative hyponatremia (p = 0.014, 0.009, and < 0.001, respectively). In total, 47/92 (51%) cases could have been symptomatic; vomiting was noted in 34 cases (37%), nausea in 19 cases (21%), headache in 14 cases (15%), and disturbances in consciousness, including delirium, in ten cases (21%); all incidences of these symptoms were significantly higher in elderly patients with postoperative hyponatremia than in the matched control group without postoperative hyponatremia (p < 0.05, respectively). Additionally, the length of stay was 2 days longer in patients than in the matched controls (p = 0.002).
Similar content being viewed by others
Introduction
The global average life expectancy increased by 5.5 years from 2000 to 20161. Reflecting this trend, patients treated with surgical therapy for spinal diseases are becoming older in many countries. In fact, a report from Japan, which has the greatest aging population worldwide, demonstrated that the proportion of patients aged > 70 years among all spinal surgical cases increased from 25% in 2004 to 40% in 2015 and that the mean age at surgery increased from 54.6 years in 2004 to 63.7 years in 20152.
Although progress in surgical techniques and equipment, as well as perioperative clinical care, have rendered surgery safer, it is still necessary to pay attention to postoperative complications, especially in elderly patients. One of the most common, however silently, complication is postoperative hyponatremia. Mild-to-severe hyponatremia may present with symptoms such as nausea, headache, gait instability, attention impairment, and falls, which may be considered as normal postoperative reactions3,4,5. Moreover, even mild hyponatremia in hospitalized patients is significantly correlated with 1-year and 5-year mortality6. A previous study reported that for every 1-mEq/L decrease in the sodium level below 135 mEq/L, mortality increases by 23%7. Furthermore, hyponatremia is correlated with perioperative complications, including pneumonia, wound infections, and major coronary events, as well as prolonged hospitalization8.
The prevalence of hyponatremia after general orthopedic surgery is as high as 30%9. Although several factors, including diuretic use, hypotonic fluids, and nonsteroidal anti-inflammatory drugs, have been reported as risk factors of postoperative hyponatremia10,11,12,13,14, the major factors that significantly increase the risk of postoperative hyponatremia are older age and spinal surgery9. However, no study has examined the frequency, risk factors, and potential symptoms of postoperative hyponatremia in elderly patients undergoing spinal surgery. Thus, this study aimed to (1) determine the frequency and risk factors for postoperative hyponatremia in this population and (2) evaluate the clinical symptoms and outcomes of postoperative hyponatremia by comparing patients with and without it.
Methods
Ethical statements
This study comprised a retrospective analysis of prospectively collected data on patients who underwent spine surgery in our institution. All study participants provided informed consent, and the study protocol was approved by the Institutional Review Board of Osaka City University (approval number: 3170). All methods were performed in accordance with the Declaration of Helsinki and the Ethical Guidelines for Medical and Health Research Involving Human Subjects in Japan. No funds were received in support of this work. No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Patient population
We retrospectively reviewed the medical records of 600 consecutive patients who underwent spinal surgery under general anesthesia at our institution between January 2018 and July 2020, were aged > 60 years by the time of operation, and had complete blood test data preoperatively, at postoperative day (POD)1, POD3, and POD6. Among these patients, those who underwent hemodialysis for chronic renal failure or underwent spinal surgery twice within 1 month, regardless of whether it was expected or unexpected, were excluded from analysis. Finally, a total of 582 patients were included in the current study (279 women and 303 men; mean age at surgery, 73.5 ± 7.4 years).
Surgical indication
The surgical indication and approach are decided on a case-by-case basis by the treating physicians. Generally, patients with relatively slow progressive diseases such as lumbar spinal stenosis or cervical spondylotic myelopathy are initially treated with conservative therapy. Surgery is performed if patients do not achieve satisfactory improvement. Patients with radiological instability are treated with fusion surgery15,16, and those with emergent symptoms such as severe palsy, severe myelopathy, infection, and progressive tumors are treated with adequate surgery immediately. In all cases in this study, seven surgeons were involved in the decision making regarding surgical indication and approach in the experimental period; however, the final surgical indication and approach were decided through a consensus of all surgeons in our institution.
Perioperative clinical care
Postoperative intravenous infusions
As part of our clinical pathway, we administered 1000 mL of acetated Ringer's solution and 1000 mL of glucose-acetated maintenance solution postoperatively from the day of surgery to the next day with some adjustments based on each patient’s weight, renal function, and blood loss. When a patient's food intake exceeded 50%, intravenous treatment was considered as complete on the next morning.
Postoperative painkillers
The standard care protocol included the routine use of celecoxib 200 mg/day as a painkiller for 7 days postoperatively. The use of additional painkillers, such as acetaminophen, opioids, or non-steroidal anti-inflammatory drugs (oral or intravenous administration), was allowed as needed. In patients with chronic renal dysfunction, the pharmacological dose was adjusted based on the creatinine clearance value.
Collected data
Preoperative data
Data regarding the age at surgery; sex; height; weight; body mass index (BMI); comorbidities (diabetes mellitus, rheumatoid arthritis, and osteoporosis); use of medications that could affect blood sodium levels, including steroids, anticoagulants, and angiotensin II receptor blockers (ARBs)17; and type of spinal disease, that is, degenerative lumbar disease, degenerative thoracic disease, degenerative cervical disease, degenerative scoliosis, spinal tumor/infection; and so on (including implant removal) were collected.
Operative data
Operative data regarding the total operative time, blood loss (mL), surgical method (decompression or decompression and fusion), and surgical complications (dural tear) were collected.
Postoperative data
Postoperative data regarding the potential symptoms of postoperative hyponatremia, including vomiting, nausea, leg edema, headache, and a disturbance of consciousness, were evaluated retrospectively from medical records. Data on the length of hospital stay, discharge to home rate, and incidence of systemic complications, including circulatory disease and respiratory disease, were collected.
Blood test data
Blood test data regarding the estimated glomerular filtration rate (eGFR) and levels of hemoglobin (Hb) (g/dL), hematocrit (Hct) (%), platelet (Plt) (× 10,000/μ), albumin (Alb) (g/dL), urea nitrogen (UN) (mg/dL), creatinine (Cre) (mg/dL), sodium (Na) (mEq/L), potassium (K) (mEq/L), and hemoglobin A1c (HbA1c) (%) were collected. Blood tests were performed preoperatively, and on POD1, POD3, and POD6 in all patients. Blood tests performed after POD6 were evaluated as “after POD6”.
Definition of hyponatremia
Postoperative hyponatremia, according to clinical guidelines, was defined as a postoperative blood sodium level < 135 meq/L at POD1, POD3, or after POD618,19. When patients showed hyponatremia < 135 meq/L preoperatively but not postoperatively, the patient was considered to show non-postoperative hyponatremia. Additionally, when patients showed hyponatremia < 135 meq/L both preoperatively and postoperatively, regardless of the POD, the patient was considered to show postoperative hyponatremia.
Study design & statistical analysis
The incidence of postoperative hyponatremia was determined. Subsequently, three analyses were performed.
-
1.
Risk factor analysis
To calculate the risk factors of postoperative hyponatremia, all patients were divided into two groups; the postoperatively hyponatremia and control groups. Preoperative, operative, and postoperative factors, as well as preoperative blood test data, were compared between groups using the Mann–Whitney U test for continuous variables and the chi-squared test for categorical variables. Subsequently, variables with a significance of p < 0.05 on the univariate analysis were included in a multivariate analysis as explanatory variables; postoperative hyponatremia was set as the objective variable. The adjusted odds ratio (aOR) and 95% confidence intervals (CIs) of dependent variables were calculated.
-
2.
Potential symptoms and outcomes analysis
To adjust the differences of backgrounds between the postoperative hyponatremia and control groups, the matched control group was created using propensity score matching. To estimate the propensity score, a logistic regression model was fitted using the patient’s age, sex, type of spinal disease, and surgical method data. The nearest-neighbor matching procedure was used, with the restriction that the matched propensities had to be within 0.01 units of each other. To identify the characteristics associated with the postoperative hyponatremia group, the clinical symptoms and clinical outcomes of patients in this group were compared with participants in the matched control group using the chi-squared test.
-
3.
Characteristics of the symptomatic hyponatremia analysis
TO identify the factors related to symptomatic hyponatremia, the postoperative hyponatremia group was further classified into two subgroups; the symptomatic group, which included hyponatremia patients with potential symptoms, and the asymptomatic group, which included hyponatremia patients without any potential symptoms. Preoperative data, operative data, and blood test data were compared between the symptomatic and asymptomatic groups using the Mann–Whitney U test for continuous variables and the chi-squared test for categorical variables.
Continuous variables were represented by the average ± 1.0 standard deviation when showing normal distribution; otherwise, they were represented as medians with the 1st and 3rd quartiles. All analyses were performed using SPSS version 23 (SPSS, Chicago, IL, USA). A p-value of < 0.05 was considered statistically significant.
Results
Incidence of postoperative hyponatremia
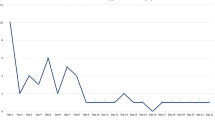
Of the 582 patients in this analysis, 92 patients (15.8%) showed postoperative hyponatremia, regardless of onset timing. Among these, 21 patients showed hyponatremia both preoperatively and postoperatively, 43 patients showed hyponatremia from POD1, 10 patients showed hyponatremia from POD3, and 18 patients showed hyponatremia after POD6 (Fig. 1).
Risk factors of postoperative hyponatremia
There were no significant differences between both groups in preoperative factors, with the exception of anticoagulant use (Table1); patients with preoperative hyponatremia took anticoagulants more frequently than control participants (p < 0.001). Regarding surgical characteristics, the method and spinal disease were significantly different between both groups (p = 0.002 and 0.010, respectively; Table 2). Regarding preoperative laboratory data, there were significant differences between groups in some preoperative blood test parameters, including Hct, Alb, and Na (p = 0.002, 0.013, and < 0.001, respectively; Table 3). The multivariate analysis, which included significant variables taken from the univariate analysis, revealed a diagnosis of spinal tumor/infection, decompression and fusion surgery, and preoperative lower Na level as significant independent factors (p = 0.014, 0.009, and < 0.001, respectively; Table 4). Namely, patients who underwent spinal surgery for tumor/infection had a significantly higher incidence of postoperative hyponatremia than those who underwent spinal surgery for lumbar disease (aOR, 3.15; p = 0.014). Patients who underwent decompression and fusion surgery had a significantly higher incidence of postoperative hyponatremia than those who underwent decompression surgery (aOR, 2.15; p = 0.009). Patients with a lower preoperative sodium level had a significantly higher incidence of postoperative hyponatremia than patients with higher preoperative sodium levels (aOR, 0.64; p < 0.001).
Potential symptoms and outcomes of postoperative hyponatremia
After matching age, sex, type of spinal disease, and surgical method, 92 patients were extracted to the matched control group from the control group using propensity score analysis. There were no significant differences between the postoperative hyponatremia group and matched control group in age (p = 0.340), sex (p = 0.659), type of spinal disease (p = 0.819), and surgical method (p = 0.769). In the postoperative hyponatremia group, 34 patients demonstrated postoperative vomiting (37%), 19 patients demonstrated nausea (21%), 14 patients demonstrated headache (15%), 6 patients demonstrated leg edema (7%), and 10 patients had disturbance of consciousness including delirium (11%). All such incidences of potential symptoms, except for leg edema, were significantly higher in the postoperative hyponatremia group than the matched control group (Table 5). Regarding clinical outcomes, no significant differences in discharge to home rates and perioperative systemic complications were observed between both groups. However, the postoperative hyponatremia group demonstrated a 2-day longer hospital stay than the matched control group (p = 0.022).
Factors relating with symptomatic hyponatremia
Forty-seven patients were included in the symptomatic group, and 45 patients were included in the asymptomatic group. In the comparison between the symptomatic and asymptomatic groups, no significant differences were noted (Table 6).
An illustrative case
The patient was a 77-year-old woman who underwent L4-5 lateral interbody fusion for lumbar spinal canal stenosis with instability. The patient showed mild hyponatremia (129 mEq/L) on POD3. Hence, she was treated with oral salt loading (3 g/day) until POD7 when her blood sodium level improved to 131 mEq/L. However, disturbance of consciousness suddenly appeared after rehabilitation at POD12 (E2V1M4 of Glasgow Coma Scale). Severe hyponatremia (116 mEq/L) and hyperkalemia (5.3 mEq/L) in the blood test, but normal natrium (70 mmol/L) and kalium (37 mmol/l) concentration in a urine test, were found. There were no abnormal findings on radiography examinations such as brain magnetic resonance imaging. Endocrinologists diagnosed this patient with mineralocorticoid responsive hyponatremia of the elderly (MRHE), which is a condition in which elderly patients taking ARBs and non-steroidal anti-inflammatory drugs (NSAIDs) develop severe hyponatremia following dehydration.20 This patient routinely took NSAIDs and ARBs and seemed to be dehydrated after rehabilitation. Her medications were discontinued, and we administered 1.7% saline at a dose of 45 mL/h. The next day, her disturbance of consciousness improved (E4V5M6), and blood sodium concentration was elevated to be 124 mEq/L. Treatment using 1.2% saline administration, oral salt loading (3 g/day), and prescribed fludrocortisone was continued. Finally, the blood sodium level reached 133 mEq/L on POD22, and the patient was discharged.
Discussion
In the current study, we found that the incidence of hyponatremia after spinal surgery in elderly patients was 15.8%. The risk factors for postoperative hyponatremia included a specific disease type (tumor/infection) and surgical method (decompression and fusion) and low preoperative sodium levels. Furthermore, more than 50% of elderly patients with postoperative hyponatremia could be symptomatic, and symptoms include disturbances in consciousness. The length of hospital stay was 2 days longer in patients with hyponatremia than in matched control patients.
The frequency of postoperative hyponatremia in the field of orthopedics in previous reports was approximately 30%.9 However, in the current study, which selected patients with two major high-risk factors of preoperative hyponatremia, the incidence was 15.8%. This discrepancy could be due to advances in anesthesia and perioperative management, including infusion management, clinical pathways, and medication selection. Regarding the onset of postoperative hyponatremia, a previous report demonstrated that 93% of cases were found within 48 h9. In contrast, in the current study, approximately 20% of preoperative hyponatremia cases were observed after POD6. The postulated reason for this difference is that the previous report was from the United States, whereas the postoperative hospital stay was shorter than that in the current study. Therefore, the observation period may have been shortened, consequently increasing the ratio of cases with early-onset hyponatremia.
Old age, spinal surgery, and hip surgery have been reported as risk factors for postoperative hyponatremia20. In the current study, which focused on elderly patients who underwent spinal surgery, the risk factors for postoperative hyponatremia were a diagnosis of spinal tumor/infection, decompression and fusion surgery, and low preoperative sodium level. The incidence of postoperative complications is known to be higher in decompression and fusion surgery than in decompression surgery, partially because of the higher invasiveness of fusion surgery21,22,23,24. Furthermore, the incidence of hyponatremia is higher with more invasive surgeries9,25,26,27. These data suggest that hyponatremia is more likely to occur in decompression and fusion surgery than in decompression alone due to higher surgical invasiveness. Regarding disease type, a previous report revealed that 21% of patients with a severe infection and 14% of patients with a malignant tumor showed hyponatremia27. This suggests that a spinal tumor or spinal infection sets the potential for hyponatremia in the patient background. Additionally, adding surgical invasion to background details may make hyponatremia apparent via MRHE or syndrome of inappropriate secretion of antidiuretic hormone3.
We retrospectively evaluated vomiting, nausea, headache, leg edema and disturbances in consciousness, including delirium, which were reported as potential symptoms of postoperative hyponatremia21. We found that the incidence of such potential symptoms except for leg edema were significantly higher in patients with hyponatremia than in the matched controls. In a prospective multicenter study, the incidence of observed symptoms of hyponatremia were reported as follows: vomiting, 30%; nausea, 44%; headache, 27%; general fatigue, 59%; and disturbances in consciousness, 5%28. The incidences of symptoms of postoperative hyponatremia in the current study (vomiting, 37%; nausea, 21%; headache, 15%; and disturbances in consciousness, 11%) were similar to those in a previous report. Additionally, we found that the length of hospital stay was 2 days longer in patients with hyponatremia than in the matched controls. Although the retrospective nature of this study rendered it difficult to exclude information bias, the finding that 50% of patients with hyponatremia were likely to be symptomatic may comprise an important message for spine physicians.
The clinical implications relevant to spine surgeons of this study are as follows: hyponatremia in elderly patients who undergo spinal surgery is not uncommon and can have adverse outcomes, and approximately 50% of patients with postoperative hyponatremia are likely to be symptomatic. Moreover, we have revealed some risk factors of postoperative hyponatremia in elderly patients. Due to differences in healthcare systems among countries, spinal surgeons might not be able to follow-up with patients for a long time postoperatively. However, by sharing the current findings with home doctors and attending surgeons, patients who show postoperative hyponatremia may more likely be treated with adequate early and appropriate treatment.
Several limitations of this study should be addressed. First, as described above, this was a retrospective study. Hence, the postoperative interventions were not identical among patients, data on perioperative infusions were not analyzed, and there might be some unreliability in symptom data. Secondly, we could not evaluate the causes of postoperative hyponatremia or adequateness of interventions after the onset of hyponatremia. Third, we analyzed data from only one institution. Therefore, a prospective multicenter study to evaluate the incidence, cause, and adequate therapy for postoperative hyponatremia in the elderly population is necessary.
Conclusion
The incidence of hyponatremia after spinal surgery in the elderly population is nearly 16%, and risk factors include a diagnosis of a spinal tumor/infection, fusion surgery, and low preoperative sodium levels. Additionally, more than 50% of patients with postoperative hyponatremia could be symptomatic, and such patients need a 2-day longer length of hospital stay than matched controls. By sharing the current findings with home doctors and attending surgeons, patients who show postoperative hyponatremia can more likely receive appropriate treatment with adequate timing.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Word Health Organization. https://apps.who.int/iris/bitstream/handle/10665/311696/WHO-DAD-2019.1-eng.pdf (2019).
Kobayashi, K., Ando, K., Nishida, Y., Ishiguro, N. & Imagama, S. Epidemiological trends in spine surgery over 10 years in a multicenter database. Eur. Spine J. 27, 1698–1703. https://doi.org/10.1007/s00586-018-5513-4 (2018).
Amini, A. & Schmidt, M. H. Syndrome of inappropriate secretion of antidiuretic hormone and hyponatremia after spinal surgery. Neurosurg. Focus 16, E10. https://doi.org/10.3171/foc.2004.16.4.11 (2004).
Tambe, A. A., Hill, R. & Livesley, P. J. Post-operative hyponatraemia in orthopaedic injury. Injury 34, 253–255. https://doi.org/10.1016/s0020-1383(02)00256-5 (2003).
Renneboog, B., Musch, W., Vandemergel, X., Manto, M. U. & Decaux, G. Mild chronic hyponatremia is associated with falls, unsteadiness, and attention deficits. Am. J. Med. 119(71), e71-78. https://doi.org/10.1016/j.amjmed.2005.09.026 (2006).
Waikar, S. S., Mount, D. B. & Curhan, G. C. Mortality after hospitalization with mild, moderate, and severe hyponatremia. Am. J. Med. 122, 857–865. https://doi.org/10.1016/j.amjmed.2009.01.027 (2009).
Wald, R., Jaber, B. L., Price, L. L., Upadhyay, A. & Madias, N. E. Impact of hospital-associated hyponatremia on selected outcomes. Arch. Intern. Med. 170, 294–302. https://doi.org/10.1001/archinternmed.2009.513 (2010).
Leung, A. A. et al. Preoperative hyponatremia and perioperative complications. Arch. Intern. Med. 172, 1474–1481. https://doi.org/10.1001/archinternmed.2012.3992 (2012).
Hennrikus, E. et al. Prevalence, timing, causes, and outcomes of hyponatremia in hospitalized orthopaedic surgery patients. J. Bone Jt. Surg. Am. 97, 1824–1832. https://doi.org/10.2106/JBJS.O.00103 (2015).
McPherson, E. & Dunsmuir, R. A. Hyponatraemia in hip fracture patients. Scott. Med. J. 47, 115–116. https://doi.org/10.1177/003693300204700506 (2002).
Hornick, P. & Allen, P. Acute hyponatraemia following total hip replacement. Br. J. Clin. Pract. 44, 776–777 (1990).
Egom, E. E., Chirico, D. & Clark, A. L. A review of thiazide-induced hyponatraemia. Clin. Med. (Lond.) 11, 448–451. https://doi.org/10.7861/clinmedicine.11-5-448 (2011).
Malin, J. W., Kolstad, K., Hozack, W. J. & Rothman, R. H. Thiazide-induced hyponatremia in the postoperative total joint replacement patient. Orthopedics 20, 681–683 (1997).
Lane, N. & Allen, K. Hyponatraemia after orthopaedic surgery. BMJ 318, 1363–1364. https://doi.org/10.1136/bmj.318.7195.1363 (1999).
Kobayashi, Y. et al. Clinical outcomes of minimally invasive posterior decompression for lumbar spinal stenosis with degenerative spondylolisthesis. Spine (Phila Pa 1976) 46, 1218–1225. https://doi.org/10.1097/BRS.0000000000003997 (2021).
Tamai, K. et al. Anterior cervical discectomy and fusion provides better surgical outcomes than posterior laminoplasty in elderly patients with C3–4 level myelopathy. Spine (Phila Pa 1976) 42, 548–555. https://doi.org/10.1097/BRS.0000000000001874 (2017).
Correia, L. et al. Severe hyponatremia in older patients at admission in an internal medicine department. Arch. Gerontol. Geriatr. 59, 642–647. https://doi.org/10.1016/j.archger.2014.08.002 (2014).
Verbalis, J. G. et al. Diagnosis, evaluation, and treatment of hyponatremia: Expert panel recommendations. Am. J. Med. 126, S1-42. https://doi.org/10.1016/j.amjmed.2013.07.006 (2013).
Spasovski, G. et al. Clinical practice guideline on diagnosis and treatment of hyponatraemia. Eur. J. Endocrinol. 170, G1-47. https://doi.org/10.1530/EJE-13-1020 (2014).
Ishikawa, S. et al. Close association of urinary excretion of aquaporin-2 with appropriate and inappropriate arginine vasopressin-dependent antidiuresis in hyponatremia in elderly subjects. J. Clin. Endocrinol. Metab. 86, 1665–1671. https://doi.org/10.1210/jcem.86.4.7426 (2001).
Gill, G. et al. Characteristics and mortality of severe hyponatraemia—A hospital-based study. Clin. Endocrinol. (Oxf.) 65, 246–249. https://doi.org/10.1111/j.1365-2265.2006.02583.x (2006).
Deyo, R. A. et al. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA 303, 1259–1265. https://doi.org/10.1001/jama.2010.338 (2010).
Ciol, M. A., Deyo, R. A., Howell, E. & Kreif, S. An assessment of surgery for spinal stenosis: Time trends, geographic variations, complications, and reoperations. J. Am. Geriatr. Soc. 44, 285–290. https://doi.org/10.1111/j.1532-5415.1996.tb00915.x (1996).
Deyo, R. A., Ciol, M. A., Cherkin, D. C., Loeser, J. D. & Bigos, S. J. Lumbar spinal fusion: A cohort study of complications, reoperations, and resource use in the Medicare population. Spine (Phila Pa 1976) 18, 1463–1470 (1993).
Chung, H. M., Kluge, R., Schrier, R. W. & Anderson, R. J. Postoperative hyponatremia. A prospective study. Arch. Intern. Med. 146, 333–336 (1986).
Crestanello, J. A. et al. Postoperative hyponatremia predicts an increase in mortality and in-hospital complications after cardiac surgery. J. Am. Coll. Surg. 216, 1135-1143e1131. https://doi.org/10.1016/j.jamcollsurg.2013.02.010 (2013).
Upadhyay, A., Jaber, B. L. & Madias, N. E. Incidence and prevalence of hyponatremia. Am. J. Med. 119, S30-35. https://doi.org/10.1016/j.amjmed.2006.05.005 (2006).
Nigro, N. et al. Symptoms and characteristics of individuals with profound hyponatremia: A prospective multicenter observational study. J. Am. Geriatr. Soc. 63, 470–475. https://doi.org/10.1111/jgs.13325 (2015).
Author information
Authors and Affiliations
Contributions
Conception: Y.K. and K.T. Organization: K.T. Execution: Y.K., O.M., H.H. and K.T. Statistical analysis: S.T. Design: K.T. and H.T. Review and critique: H.H., H.T., M.H., H.T., A.S. and H.N. Manuscript: Writing of the first draft: Y.K. Review and critique: All authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kinoshita, Y., Tamai, K., Oka, M. et al. Prevalence, risk factors, and potential symptoms of hyponatremia after spinal surgery in elderly patients. Sci Rep 12, 18622 (2022). https://doi.org/10.1038/s41598-022-23583-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-23583-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.