Abstract
The concept of developmental origin of health and disease has ignited a search for mechanisms and health factors influencing normal intrauterine development. Sleep is a basic health factor with substantial individual variation, but its implication for early prenatal development remains unclear. During the embryonic period, the yolk sac is involved in embryonic nutrition, growth, hematopoiesis, and likely in fetal programming. Maternal body measures seem to influence its size in human female embryos. In this prospective, longitudinal observational study of 190 healthy women recruited before natural conception, we assessed the effect of prepregnant sleep duration (actigraphy) on the fetal crown-rump-length (CRL) and yolk sac size (ultrasound). All women gave birth to a live child. The prepregnancy daily sleep duration had an effect on the male yolk sac and CRL at the earliest measurement only (7 weeks). I.e., the yolk sac diameter decreased with increasing sleep duration (0.22 mm·h−1d−1, 95%CI [0.35-0.09], P < 0.01), and CRL decreased (0.92 mm·h−1d−1, 95%CI [1.77-0.08], P = 0.03). Since there was no association at the second measurement (10 weeks), and in the group of female fetuses at any measure point, we suggest a sex- and time-dependent embryonic adaptation to sleep generated differences in the intrauterine environment in normal pregnancies.
Similar content being viewed by others
Introduction
It is widely accepted that sleep is essential for our health and physiologic homeostasis. This is certainly the case for pregnancy, as sleep disturbances are associated with pregnancy complications1, such as preeclampsia, gestational diabetes, preterm birth, and prolonged birth duration2,3,4,5,6. Ethical reasons restrict in vivo experiments, so studies of sleep effects on human embryonic physiology and development are scarce1,7. This is a dilemma, since physiology and its normal variation lay the foundation for the understanding of pathology and disease. Why is, for example, more than one nightshift per week associated with a 30% higher risk of early pregnancy loss?8 What are the pathophysiologic mechanisms? And, how much of regular sleep is needed in pregnancy?
In vitro experiments in assisted reproduction and animal studies demonstrate a possible circadian rhythm link to embryonic development and epigenetic programming via melatonin, cortisol, and sex steroids9,10,11,12,13,14,15,16,17. Further, deep sleep influences the growth hormone secretion in humans18, which in turn has notable effects on fertility and the intra-uterine environment19. Likewise, sleep disturbances (such as insomnia) are linked to HPA-axis activation20 and rise in cortisol levels21—a stress response with capability to impair fertility during critical periods, ovulation, implantation, and placental growth and development22. Nevertheless, animal studies, in vitro studies, and studies on infertile cohorts seeking health services cannot necessarily reflect the normal physiology of healthy women; this is where we can employ ultrasound as a non-invasive method.
In pregnancy, the human yolk sac is one of the first structures that can be assessed by ultrasound23. It has a complex surface structure that ensures embryonal nutrition and growth before the placenta is sufficiently developed24,25, and its size is associated with fetal anomalies and unfavorable pregnancy outcomes26,27,28. Previous animal studies have demonstrated yolk sac size variation related to environmental factors, e.g., noise, temperature, and nutrition27,29,30. Further, women’s body composition, i.e., lower weight and height have been linked to larger yolk sac31, while human sleep with its large-scale physiologic variation has not been a focus of research32,33.
Thus, we hypothesized that maternal sleep has an effect on embryonic development in humans, and aimed at assessing the effect of maternal sleep recordings on measurements of yolk sac size and crown-rump-length (CRL).
Method
The present study is a prospective, longitudinal observational study of the relation of actigraphy measured sleep in healthy women with ultrasound assessed early fetal development. It is embedded in the ongoing CONIMPREG research program which collects data of healthy women from before conception, through pregnancy, and until the child reaches the age of 5 years.
Study cohort
The participants were recruited during the period 2014–2020 by means of social media (targeted Facebook® advertisements) and posters. Healthy non-smoking women aged 20–35 years with a BMI of 18–30 kg/m2 were eligible provided they had an uncomplicated obstetric history and no chronic diseases or fertility problems. The participants did not use contraceptives at study entry, including the preceding month. The participants who did not conceive during any of the 6 monitored cycles allotted to them, discontinued the participation (Fig. 1). The study was approved by the Regional Committee for Medical Research Ethics South East Norway (REK South East, ref. 2013/856a). Written informed consent was obtained from all participants and all research was performed in accordance with relevant guidelines and regulations.
(a) Flowchart of the study population with the participant exclusions—*other: 1 twin pregnancy, 2 abortions > 18 weeks (fetal anomaly), and 1 irregular menstrual cycle; **missing data: Pregnancies with incomplete entry data (N = 54) and pregnant < 13 weeks at time of data extraction (N = 8). (b) Timing of measurements and recordings.
Data collection
The current study includes data from four consecutive study visits (Fig. 1). At the first visit (i.e., at inclusion and before the participants attempted to conceive), prepregnant height was measured manually using a wall-mounted stadiometer34 and weight was measured digitally using hand-to-foot bioelectrical impedance analysis (model BC-418, Tanita, Tokyo, Japan). The body fat percentage was estimated using the instrument’s computer software, and the lean body mass was calculated by subtracting the body fat mass from the total body weight. Measurements were performed as recommended by the manufacturer35 and immediately followed by actigraphy recording. Following inclusion, the woman monitored her menstrual cycles by recording events, such as bleeding, sexual intercourse, and pregnancy tests. Once she had a positive pregnancy test, she reported to the study office and was scheduled for the second visit at gestational age (GA) 7 ± 1 weeks calculated from her last menstrual period (LMP). At that second visit, the gestational age was confirmed using transvaginal ultrasound scanning. The embryonic CRL and secondary yolk sac were assessed36. Later in pregnancy, at the third visit, 10 ± 1 weeks, the ultrasound measurements were repeated and at the fourth visit, 13 ± 1 weeks (end of the 1st trimester), maternal body composition and sleep duration were re-assessed along with the sonographic measurement of the CRL (Fig. 1).
Actigraphy
Sleep duration was recorded with the SenseWear Mini Armband, model MF-SW, BodyMedia, Pittsburgh, PA, USA. The monitor was worn on the upper posterior aspect of the nondominant arm for 4 days37 and the recording started at midnight. The SenseWear actigraph is a wireless, noninvasive monitor with a sampling rate of 32/sec and incorporates triaxial accelerometry, heat flux, galvanic skin response, skin temperature, and near-body temperature, and takes into account information on sex, age, height, and weight38. Sleep and wakefulness are discriminated based on motion and skin temperature in 1-min epochs. Sleep raw data were processed and summarized using the manufacturer’s analysis software (SenseWear Professional, version 8.0.0.2903, Body Media) and exported into Excel (Microsoft Office, Excel version 2016, Redmond, WA, USA). This actigraph has been compared with other models and standard measurement methods in clinical and experimental settings39,40,41,42,43,44. Recently, our group used this monitor to measure sleep in pregnancy with results in good agreement with other actigraphy studies33. The sleep efficiency (SE) was calculated by the software as sleep proportion of the time lying down.
Embryonic and fetal assessment
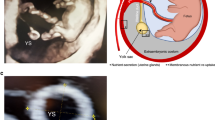
The fetal crown-rump-length and yolk sac size were examined by seven obstetricians and one certified midwife using a transvaginal ultrasound transducer, 6–12 MHz, Voluson Expert E8; (GE Medical Systems, Kretz Ultrasound, Zipf, Austria). The yolk sac diameter was determined as the average of two perpendicular diameters repeated thrice31. Yolk sac growth rate was calculated as the difference in yolk sac diameter divided by the number of days between the two measurements.
Statistics
Statistical analysis was performed using SPSS (version 24, Armonk, NY, USA), R (Foundation for Statistical Computing, version 4.1, Vienna, Austria), and R-studio (Integrated development for R, Boston, MA, USA) software. Sampling days were excluded from the statistical analyses when the data loss exceeded 6% of a single day. Mean, standard deviation (SD) with minimum and maximum value were calculated for each continuous variable, and frequencies and proportions were calculated for categorical variables. When the distribution was asymmetric the median and interquartile range were reported. In addition, the 95% confidence intervals (CIs) of the mean were calculated for sleep parameters, the number of recorded days, the frequency of weekend days, and the CRL and yolk sac size with their GA at the measurements.
Linearity assumptions and normal distribution of the residuals were ascertained, and ordinary least square linear and quantile regression models were used to analyze the yolk sac size association with the sleep duration before pregnancy and the sleep duration at the end of the first trimester (week 13). Results were compared with results from robust regression methods (iterated reweighted least squares by Huber weights and bi-square weighting) and heteroskedastic methods (sandwich variance estimators). Calculations were carried out with and without model outliers (Cooks distance > 4 times). Mixed models were used for the repeated measurements analysis. The regression models were calculated with and without fetal sex stratification and stratification from lower to higher quartiles of the sleep duration. In sub analysis, we controlled for GA, maternal age, parity, physical activity level before conception, and maternal body composition parameters (i.e., height, weight, body mass index, lean body mass, and body fat percent). Those factors were added one by one to the primary model (yolk sac or CRL size by total sleep duration) and were included if they were altering the effect-size of the association notably. As measures of fit, the adjusted R–squared and Akaike information criterion were calculated. Differences between the regression models were tested using analysis of variance methods. The different strata were also compared with paired and unpaired Student’s t-test or nonparametric tests.
Ethical declaration and informed consent
The study was approved by: Regional Comittee for Medical Research Ethics South East Norway (REK South East, ref. 2013/856a). E-mail: rek-sorost@medisin.uio.no. Written informed consent was obtained from all participants and all research was performed in accordance with relevant guidelines and regulations.
Results
Of 436 eligible participants, 190 (43.6%) conceived with a successful pregnancy and provided sufficient data to be included in the present study (Fig. 1). Their demographic information and obstetric outcomes are provided in Tables 1 and 2.
During actigraphy, 92.1% of all days before conception and 93.7% at the end of the first trimester were successfully recorded. The yolk sac was measured totally 358 times, and the CRL 559 times (Fig. 2). The summary statistics of the actigraphy recording, yolk sac diameter, and CRL are shown in Tables 3 and 4.
The median time from the first sleep recording to estimated day of conception (i.e., 14 days after LMP) was 36 days (IQR 10–75). The majority conceived within the two first menstrual cycles. The total sleep duration before conception was 38 min shorter than at the end of the first trimester (95%CI [28.6–47.2], P < 0.01), the total length of actigraphy and the frequency of recorded weekend days did not differ before and after conception.
Prepregnant sleep duration, but not the other sleep parameters before pregnancy had an effect on the yolk sac size (Supplementary Table S1). Generally, the effect on the yolk sac and CRL was weaker when the relation to measurement time was not taken into account (Supplementary Tables S1 and S2). The following analyses were stratified, all grouped according to time of the measurement at week 7, week 10, and week 13 for the CRL (Table 5 and 6).
Relation between daily sleep duration and yolk sac size
At the first ultrasound measurement (week 7), but not at the second (week 10), we found a sex-dependent negative effect of the prepregnant sleep duration on the yolk sac of male embryos (Fig. 3 and Table 5), i.e., longer sleep before pregnancy was associated with smaller yolk sac sizes.
Effect of daily total sleeping time before pregnancy (a), and effect of the daily total sleeping time at 13 weeks (b) on the first (upper row) and second yolk sac measurement (lower row) in naturally conceived healthy pregnancies presented with regression line and its 95% confidence interval. The first column represents the total dataset and the second and third the analysis according to embryonic sex.
It was noted that the sleep duration recorded later, at the end of the first trimester (week 13), was similarly linked to the yolk sac size at 7 weeks, but here, the effect was weaker (Fig. 3 and Table 5).
There was a considerable variation in the yolk sac growth rate between the two measurement time points (Supplementary Fig. S1), but no association between the average growth rate and sleep duration (Supplementary Table S3).
Grouping of the sleep duration in quartiles, however, suggested that the yolk sac size was larger for the lowest sleep quartile than for the highest (Fig. 4); for these quartiles—lowest and highest—the sleep-yolk sac association also tended to be stronger.
Raw-data-descriptive-inferential-statistics plot of the yolk sac size at week 7 (N = 166) grouped according to prepregnant sleep duration quartiles: Low sleep duration range (4 h 30 min–6 h 28 min); middle sleep duration range (6 h 29 min–7 h 40 min); high sleep duration range (7 h 42 min–9 h 19 min)—* Kruskal–Wallis test (p = 0.03; § Post-hoc-test (Dunn’s test), p = 0.038.
Further, the relation between sleep duration before pregnancy and the embryonic yolk sac at week 7 was consistent across the yolk sac deciles (Supplementary Figs. S2 and S3), and still present after adjustment for maternal characteristics and GA (Supplementary Tables S4 and S5). In sub-analyses, these adjustments were made for all models (i.e., yolk sac at 7 and 10 weeks), with preconception sleep duration and sleep duration at the end of the 1st trimester (13 weeks) as predictor, all stratified and unstratified by fetal sex. In none of the models these adjustments changed our conclusions.
Maternal sleep duration and embryo size through the first trimester
Similar to the association between yolk sac diameter and sleep duration, the embryonic CRL was shorter when the prepregnant sleep duration increased. Stratification by the time of measurement and fetal sex revealed a comparable pattern, i.e., the effect was confined to male embryos at week 7, not later (week 10 or week 13) (Table 6). The sleep duration at the end of the 1st trimester (week 13) was not associated with any of the CRL measurements (males or females) at week 7, week 10, or week 13. Adjustments for maternal age, parity and body composition did not change these results significantly.
Finally, the interval from actigraphy sleep measurement before pregnancy to LMP/estimated conception was neither influencing the CRL-sleep-association nor the yolk sac-sleep-association (i.e., the number of menstrual cycles to achieve pregnancy was irrelevant for these relations).
Discussion
This study demonstrates a sex- and time-dependent relation between prepregnant sleep duration and human embryonic development. The association between a shorter maternal sleep duration before pregnancy and a larger yolk sac and CRL was confined to male embryos at 7 weeks of gestation.
However, this effect on 7-weeks’ embryos could be traced later in pregnancy. I.e., although sleep duration assessed at 13 weeks was 38 min longer, it still had a distributional pattern that significantly linked it to the yolk sac size at 7 weeks.
A strength of the study is the prospective longitudinal design to address our study questions. Secondly, our participants were healthy women who conceived naturally without any influence of hormonal treatment commonly used in assisted reproduction. However, our study did not discriminate daytime and nocturnal sleep duration. The total daily sleep duration included daytime naps and does not necessarily reflect the sleep quality and quantity at night. In addition, we cannot be certain that the sleep patterns recorded before pregnancy, although close to conception (Table 2), were continued into early pregnancy. An additional use of sleep diaries would have strengthened the study and provided a deeper insight. On the other hand, the fact that the sleep duration pattern at 13 weeks was similarly related to yolk sac and embryo at 7 weeks as was prepregnant sleep duration, suggests that the sleep pattern was similarly distributed through the entire period.
Stress, physical and psychological, are known confounders with quite similar hormonal responses, but the present study was not designed to disentangle such factors from sleep effects. The study population, however, consisted of healthy women with no history of chronic diseases or risk factors, and the chance for being under chronic physical and psychological stress should therefore be low.
There is some evidence from animal studies supporting an effect of environmental factors on the yolk sac (e.g. temperature, nutrition, and noise)29,30,45,46, but extrapolation to human conditions may not be warranted as yolk sac development and implantation mechanisms differ between species25,47,48. However, a human study found that the yolk sac diameter was influenced by maternal body size31. They reported a smaller yolk sac at 8–12 weeks when maternal prepregnant height or weight was high. The relation was sex-dependent, i.e., shown only in female fetuses. Conversely, adjustment for maternal body composition parameters, including height and weight, did not change our estimates of the relation between daily sleep duration and yolk sac size (Supplementary Tables S4 and S5). We suggest to consider this rather as a concept than contradiction. The first trimester is a developmental rush hour. From conception and implantation to the end of week 13, myriads of developmental steps are taking place with different time windows opening and closing at high frequency. In the current study the sleep effect on the yolk sac was present at week 7, but not 3 weeks later supporting this notion.
Our findings can be explained by at least two different biological models. First, the increase of the yolk sac size could compensate for a less favorable intrauterine environment due to a shorter sleep duration. This concept has been suggested before (in connection with maternal body size) and implies that the human yolk sac can compensate for reduced access to maternal nutrients by means of a larger surface31. It is in line with the time-dependent and early effect on the yolk sac at 7 weeks: early nutrition depends foremost on the yolk sac; later, there is a gradual transition to a hemotrophic placental supply24,25. The similar association of the embryonic CRL at week 7 (Table 6) may appear inconsistent with such a model of yolk sac compensation, but could be explained by a delay of the compensatory effect.
A second model would be that there is a systematic shift of the embryonic or fetal age related to the measured sleep duration. I.e., both time of ovulation with the fertile window (day 8–15)49 and time of implantation vary (i.e., day 6–12 after ovulation)50. The influence of sleep on the HPA-axis, estradiol, melatonin, and other hormones20,51 can cause menstrual cycle changes11,18,20,21,51,52,53,54 and earlier ovulation. In addition, the interval from ovulation to implantation is inversely correlated with the first trimester measurement of the fetal CRL55. Thus, an association between the prepregnant sleep duration with early ovulation or shorter ovulation–implantation interval would result in a systematically larger yolk sac diameter and fetal CRL at any estimated GA by the LMP-method. This theory provides an explanation why both the yolk sac size and the CRL are sleep-related in early gestation. Later when size variation increases, due to individual embryonic and yolk sac growth or because the yolk sac dissolves25,56, these sleep-related findings may not be traceable.
The present study also demonstrated a fetal sex-dependent sleep-yolk sac association that was present only for male embryos. Weinberg et al. reported already in 1995 that women with a short follicular phase before conception tended to give birth to males57. It is tempting to propose that a similar mechanism could explain the present results. Nevertheless, sexual dimorphism alone could explain sex-specific environmental effects including a sex-specific plasticity that depends on embryonic age58. That means, time windows for developmental influence may differ between the sexes as seen in the present and a previous study on yolk sac size31. Male embryos at 7 weeks in the present study were sensitive to maternal sleep, while female embryos after 8 weeks in the previous study were sensitive to maternal body height and weight, which could be analogue to the different timelines for development of girls and boys during postnatal life.
The present findings are not derived from extreme or pathologic cases but represent the healthy majority of pregnancies and point out that human development exhibit a measurable sensitivity to environmental factors already during the embryonic period. Note that the current yolk sac changes were associated with the sleep duration before pregnancy. Epigenetic programming has been linked both to sleep9,10,11,12,13,14,15,16,17 and immune progenitor cells produced in the yolk sac59. This opens the possibility of influencing and conditioning individual health development of the offspring right from the periconceptional period.
The present results made us also realize how rapid the pace of development is during the 1st trimester with sex and GA specific time windows that were susceptible to maternal and environmental effects (i.e., sleep and body composition). Future studies need to consider a correspondingly rapid frequency of observational windows to discern effects.
In addition, the two suggested explanatory models require further investigation; (a) the yolk sac compensation of a less favorable intrauterine environment due to a shorter prepregnant sleep duration and/or (b) an earlier ovulation or implantation due to a shorter prepregnant sleep duration. A precise determination of time of ovulation and implantation would be a good starting point50.
Conclusion
Normal variation in prepregnant sleep does have an effect on embryonic development in healthy women; a shorter sleep duration corresponds to a larger yolk sac and CRL. Two features stand out; there is a sex-difference with an effect that appears to be exclusively among males, and the effect seems to operate during a short time window—around 7 weeks of pregnancy—underscoring the rapid pace of development that dominates the embryonic period.
Data availability
All data generated or analysed during this study are included in this published article (and its supplementary information files).
Change history
03 April 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41598-024-58405-z
References
Romero, R. & Badr, M. S. A role for sleep disorders in pregnancy complications: Challenges and opportunities. Am. J. Obstet. Gynecol. 210, 3–11. https://doi.org/10.1016/j.ajog.2013.11.020 (2014).
Williams, M. A. et al. Associations of early pregnancy sleep duration with trimester-specific blood pressures and hypertensive disorders in pregnancy. Sleep 33, 1363–1371. https://doi.org/10.1093/sleep/33.10.1363 (2010).
Facco, F. L. et al. Objectively measured short sleep duration and later sleep midpoint in pregnancy are associated with a higher risk of gestational diabetes. Am. J. Obstet. Gynecol. 217, 447.e1-447.e13. https://doi.org/10.1016/j.ajog.2017.05.066 (2017).
Facco, F. L. et al. Association of adverse pregnancy outcomes with self-reported measures of sleep duration and timing in women who are nulliparous. J Clin Sleep Med. 14, 2047–2056. https://doi.org/10.5664/jcsm.7534 (2018).
Okun, M. L., Schetter, C. D. & Glynn, L. M. Poor sleep quality is associated with preterm birth. Sleep 34, 1493–1498. https://doi.org/10.5665/sleep.1384 (2011).
Lee, K. A. & Gay, C. L. Sleep in late pregnancy predicts length of labor and type of delivery. Am. J. Obstet. Gynecol. 191, 2041–2046. https://doi.org/10.1016/j.ajog.2004.05.086 (2004).
Warland, J., Dorrian, J., Morrison, J. L. & O’brien, L. M. Maternal sleep during pregnancy and poor fetal outcomes: A scoping review of the literature with meta-analysis. Sleep Med. Rev. 41, 197–219. https://doi.org/10.1016/j.smrv.2018.03.004 (2018).
Begtrup, L. M. et al. Night work and miscarriage: A danish nationwide register-based cohort study. Occup. Environ. Med. 76, 302–308. https://doi.org/10.1136/oemed-2018-105592 (2019).
Torres-Farfan, C. et al. Maternal melatonin effects on clock gene expression in a nonhuman primate fetus. Endocrinology 147, 4618–4626. https://doi.org/10.1210/en.2006-0628 (2006).
Lanoix, D., Beghdadi, H., Lafond, J. & Vaillancourt, C. Human placental trophoblasts synthesize melatonin and express its receptors. J. Pineal Res. 45, 50–60. https://doi.org/10.1111/j.1600-079x.2008.00555.x (2008).
Zhang, L. et al. Effects of melatonin administration on embryo implantation and offspring growth in mice under different schedules of photoperiodic exposure. Reprod. Biol. Endocrinol. 15, 78. https://doi.org/10.1186/s12958-017-0297-7 (2017).
Voiculescu, S. E., Zygouropoulos, N., Zahiu, C. D. & Zagrean, A. M. Role of melatonin in embryo fetal development. J. Med. Life 7, 488–492 (2014).
Olcese, J. M. Melatonin and female reproduction: An expanding universe. Front. Endocrinol. (Lausanne) 11, 85. https://doi.org/10.3389/fendo.2020.00085 (2020).
Ivanov, D. et al. Melatonin, its beneficial effects on embryogenesis from mitigating oxidative stress to regulating gene expression. Int. J. Mol. Sci. 22, 5885. https://doi.org/10.3390/ijms22115885 (2021).
Unfer, V., Raffone, E., Rizzo, P. & Buffo, S. Effect of a supplementation with myo-inositol plus melatonin on oocyte quality in women who failed to conceive in previous in vitro fertilization cycles for poor oocyte quality: A prospective, longitudinal, cohort study. Gynecol. Endocrinol. 27, 857–861. https://doi.org/10.3109/09513590.2011.564687 (2011).
Hardeland, R. Melatonin and chromatin. Melatonin Res. 2, 67–93 (2019).
Hsu, C. N. & Tain, Y. L. Light and circadian signaling pathway in pregnancy: Programming of adult health and disease. Int. J. Mol. Sci. 21, E2232. https://doi.org/10.3390/ijms21062232 (2020).
Van Cauter, E., Plat, L. & Copinschi, G. Interrelations between sleep and the somatotropic axis. Sleep 21, 553–566 (1998).
Li, J., Chen, Q., Wang, J., Huang, G. & Ye, H. Does growth hormone supplementation improve oocyte competence and ivf outcomes in patients with poor embryonic development? A randomized controlled trial. BMC Pregnancy Childbirth 20, 310. https://doi.org/10.1186/s12884-020-03004-9 (2020).
Kloss, J. D., Perlis, M. L., Zamzow, J. A., Culnan, E. J. & Gracia, C. R. Sleep, sleep disturbance, and fertility in women. Sleep Med. Rev. 22, 78–87. https://doi.org/10.1016/j.smrv.2014.10.005 (2015).
Vgontzas, A. N. et al. Chronic insomnia is associated with nyctohemeral activation of the hypothalamic-pituitary-adrenal axis: Clinical implications. J. Clin. Endocrinol. Metab. 86, 3787–3794. https://doi.org/10.1210/jcem.86.8.7778 (2001).
Nakamura, K., Sheps, S. & Arck, P. C. Stress and reproductive failure: Past notions, present insights and future directions. J. Assist. Reprod. Genet. 25, 47–62. https://doi.org/10.1007/s10815-008-9206-5 (2008).
Mantoni, M. & Pedersen, J. F. Ultrasound visualization of the human yolk sac. J. Clin. Ultrasound 7, 459–460 (1979).
Burton, G. J., Hempstock, J. & Jauniaux, E. Nutrition of the human fetus during the first trimester—a review. Placenta 22(Suppl A), S70-7. https://doi.org/10.1053/plac.2001.0639 (2001).
Ross, C. & Boroviak, T. E. Origin and function of the yolk sac in primate embryogenesis. Nat. Commun. 11, 3760. https://doi.org/10.1038/s41467-020-17575-w (2020).
Lindsay, D. J. et al. Yolk sac diameter and shape at endovaginal us: Predictors of pregnancy outcome in the first trimester. Radiology 183, 115–118. https://doi.org/10.1148/radiology.183.1.1549656 (1992).
Cepni, I. et al. Significance of yolk sac measurements with vaginal sonography in the first trimester in the prediction of pregnancy outcome. Acta Obstet. Gynecol. Scand. 76, 969–972. https://doi.org/10.3109/00016349709034911 (1997).
Oh, J. S., Wright, G. & Coulam, C. B. Gestational sac diameter in very early pregnancy as a predictor of fetal outcome. Ultrasound Obstet. Gynecol. 20, 267–269. https://doi.org/10.1046/j.1469-0705.2002.00774.x (2002).
Lara, R. A. & Vasconcelos, R. O. Impact of noise on development, physiological stress and behavioural patterns in larval zebrafish. Sci. Rep. 11, 6615. https://doi.org/10.1038/s41598-021-85296-1 (2021).
Mikec, M. et al. Influence of environmental and nutritional stressors on yolk sac utilization, development of chicken gastrointestinal system and its immune status. World’s Poult. Sci. J. 62, 31–40. https://doi.org/10.1079/wps200582 (2006).
Odland Karlsen, H. et al. The human yolk sac size reflects involvement in embryonic and fetal growth regulation. Acta Obstet. Gynecol. Scand. 98, 176–182. https://doi.org/10.1111/aogs.13466 (2018).
Lee, K. A., Zaffke, M. E. & Mcenany, G. Parity and sleep patterns during and after pregnancy. Obstet Gynecol. 95, 14–18. https://doi.org/10.1016/s0029-7844(99)00486-x (2000).
Vietheer, A., Kiserud, T., Lie, R. T., Haaland, Ø. A. & Kessler, J. Sleep and physical activity from before conception to the end of pregnancy in healthy women: A longitudinal actigraphy study. Sleep Med. https://doi.org/10.1016/j.sleep.2021.04.028 (2021).
Lohman, T. Anthropometric Standardization Reference Manual (Human kinetics book, 1988).
Corporation, T. Body Composition Analyser BC-418-MA. Instruction Manual, (Tanita Corporation of America. https://www.tanita.com/es/.downloads/download/?file=855638086&fl=en_US,
Robinson, H. P. Sonar measurement of fetal crown-rump length as means of assessing maturity in first trimester of pregnancy. Br. Med. J. 4, 28–31. https://doi.org/10.1136/bmj.4.5883.28 (1973).
Matthews, C. E., Ainsworth, B. E., Thompson, R. W. & Bassett, D. R. Sources of variance in daily physical activity levels as measured by an accelerometer. Med. Sci. Sports Exerc. 34, 1376–1381. https://doi.org/10.1097/00005768-200208000-00021 (2002).
Liden, C. B. et al. Benefits of the sensewear armband over other physical activity and energy expenditure measurement techniques. White Papers Body Media 1, 1–14 (2001).
Sunseri, M. et al. The sensewear armband as a sleep detection device. White Papers Body Media 1–9
Peterson, B. T. et al. Comparison of actigraphy and polysomnography to assess effects of zolpidem in a clinical research unit. Sleep Med. 13, 419–424. https://doi.org/10.1016/j.sleep.2011.12.003 (2012).
Soric, M. et al. Validation of a multi-sensor activity monitor for assessing sleep in children and adolescents. Sleep Med. 14, 201–205. https://doi.org/10.1016/j.sleep.2012.11.003 (2013).
O’driscol, D. M., Turton, A. R., Copland, J. M., Strauss, B. J. & Hamilton, G. S. Energy expenditure in obstructive sleep apnea: Validation of a multiple physiological sensor for determination of sleep and wake. Sleep Breath. 17, 139–146. https://doi.org/10.1007/s11325-012-0662-x (2013).
Sharif, M. M. & Bahammam, A. S. Sleep estimation using bodymedia’s sensewear armband in patients with obstructive sleep apnea. Ann. Thorac. Med. 8, 53–57. https://doi.org/10.4103/1817-1737.105720 (2013).
Shin, M., Swan, P. & Chow, C. M. The validity of actiwatch2 and sensewear armband compared against polysomnography at different ambient temperature conditions. Sleep Sci. 8, 9–15. https://doi.org/10.1016/j.slsci.2015.02.003 (2015).
Fiksen, Ø. & Folkvord, A. Maternal effects and the benefit of yolk supply in cod larvae in different environments—a simulation model. ICES Council Meeting, 1–6 (1999)
Watkins, A. J. et al. Adaptive responses by mouse early embryos to maternal diet protect fetal growth but predispose to adult onset disease. Biol. Reprod. 78, 299–306. https://doi.org/10.1095/biolreprod.107.064220 (2008).
Burton, G. J., Cindrova-Davies, T. & Turco, M. Y. Review: Histotrophic nutrition and the placental-endometrial dialogue during human early pregnancy. Placenta 102, 21–26. https://doi.org/10.1016/j.placenta.2020.02.008 (2020).
Carter, A. M. Unique aspects of human placentation. Int. J. Mol. Sci. 22, 8099. https://doi.org/10.3390/ijms22158099 (2021).
Wilcox, A. J., Dunson, D. & Baird, D. D. The timing of the “fertile window” in the menstrual cycle: Day specific estimates from a prospective study. BMJ 321, 1259–1262. https://doi.org/10.1136/bmj.321.7271.1259 (2000).
Wilcox, A. J., Baird, D. D. & Weinberg, C. R. Time of implantation of the conceptus and loss of pregnancy. N. Engl. J. Med. 340, 1796–1799. https://doi.org/10.1056/NEJM199906103402304 (1999).
Carlomagno, G., Minini, M., Tilotta, M. & Unfer, V. From implantation to birth: Insight into molecular melatonin functions. Int. J. Mol. Sci. 19, 2802. https://doi.org/10.3390/ijms19092802 (2018).
Baker, F. C. & Driver, H. S. Circadian rhythms, sleep, and the menstrual cycle. Sleep Med. 8, 613–622. https://doi.org/10.1016/j.sleep.2006.09.011 (2007).
Lawson, C. C. et al. Rotating shift work and menstrual cycle characteristics. Epidemiology 22, 305–312. https://doi.org/10.1097/EDE.0b013e3182130016 (2011).
He, C. et al. Melatonin-related genes expressed in the mouse uterus during early gestation promote embryo implantation. J. Pineal Res. 58, 300–309. https://doi.org/10.1111/jpi.12216 (2015).
Mahendru, A. A. et al. Impact of ovulation and implantation timing on first-trimester crown-rump length and gestational age. Ultrasound Obstet. Gynecol. 40, 630–635. https://doi.org/10.1002/uog.12277 (2012).
Blaas, H. G., Eik-Nes, S. H. & Bremnes, J. B. The growth of the human embryo. A longitudinal biometric assessment from 7 to 12 weeks of gestation. Ultrasound Obstet. Gynecol. 12, 346–354. https://doi.org/10.1046/j.1469-0705.1998.12050346.x (1998).
Weinberg, C. R., Baird, D. D. & Wilcox, A. J. The sex of the baby may be related to the length of the follicular phase in the conception cycle. Hum. Reprod. 10, 304–307. https://doi.org/10.1093/oxfordjournals.humrep.a135932 (1995).
Deegan, D. F. & Engel, N. Sexual dimorphism in the age of genomics: How, when, where. Front Cell Dev. Biol. 7, 186. https://doi.org/10.3389/fcell.2019.00186 (2019).
Chen, S., Yang, J., Wei, Y. & Wei, X. Epigenetic regulation of macrophages: From homeostasis maintenance to host defense. Cell Mol. Immunol. 17, 36–49. https://doi.org/10.1038/s41423-019-0315-0 (2020).
Acknowledgements
The authors express their gratitude to the women who participated in this study and their families. The study was funded by Western Norway Health Trust, University of Bergen, Norway, and Wayne University and National Institute of Health, USA. Dr Hemamalini Rajkumar contributed in establishing the study, collecting data and data-handling. Rita Sollien and Norunn Solvang (Reg. midwives) and Carol Cook (MBE) managed inclusions, logistics, measurements, and data collection. Cathrine Ebbing (PhD), Henriette Odland-Karlsen (PhD), and Synnøve Lian Johnsen (PhD) contributed with data collection and valuable advices.
Funding
Open access funding provided by University of Bergen. The study received financial support from the Western Norway Health Trust, Norway; University of Bergen, Norway; Wayne University, Detroit, Michigan, USA, and National Institute of Health, USA.
Author information
Authors and Affiliations
Contributions
A.V.: Conceptualization, methodology, investigation, software, data curation, visualization, validation, formal analysis, interpretation, writing—original draft preparation, review and editing; T.K.: Project administration, conceptualization, methodology, investigation, data curation, formal analysis, interpretation, visualization, writing—review & editing. R.T.L.: Methodology, formal analysis, validation, visualization, writing—review & editing. Ø.A.H.: Methodology, software, formal analysis, validation, visualization, writing, review and editing. J.K.: Project administration, methodology, investigation, formal analysis, validation, visualization, writing—review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained errors in the Abstract. “I.e., the yolk sac diameter decreased with increasing sleep duration (0.22 mm·h−1d−1, 95%CI [0.35–0.09], P < 0.01), and CRL increased (0.92 mm·h−1d−1, 95%CI [1.77–0.08], P = 0.03).” It now reads: “I.e., the yolk sac diameter decreased with increasing sleep duration (0.22 mm·h−1d−1, 95%CI [0.35-0.09], P < 0.01), and CRL decreased (0.92 mm·h−1d−1, 95%CI [1.77-0.08], P = 0.03).”
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vietheer, A., Kiserud, T., Haaland, Ø.A. et al. Effect of maternal sleep on embryonic development. Sci Rep 12, 17099 (2022). https://doi.org/10.1038/s41598-022-21516-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-21516-6
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.