Abstract
The oceans play a major role in the earth’s climate by regulating atmospheric CO2. While oceanic primary productivity and organic carbon burial sequesters CO2 from the atmosphere, precipitation of CaCO3 in the sea returns CO2 to the atmosphere. Abiotic CaCO3 precipitation in the form of aragonite is potentially an important feedback mechanism for the global carbon cycle, but this process has not been fully quantified. In a sediment-trap study conducted in the southeastern Mediterranean Sea, one of the fastest warming and most oligotrophic regions in the ocean, we quantify for the first time the flux of inorganic aragonite in the water column. We show that this process is kinetically induced by the warming of surface water and prolonged stratification resulting in a high aragonite saturation state (ΩAr ≥ 4). Based on these relations, we estimate that abiotic aragonite calcification may account for 15 ± 3% of the previously reported CO2 efflux from the sea surface to the atmosphere in the southeastern Mediterranean. Modelled predictions of sea surface temperature and ΩAr suggest that this process may weaken in the future ocean, resulting in increased alkalinity and buffering capacity of atmospheric CO2.
Similar content being viewed by others
Introduction
The production of CaCO3 minerals (namly, aragonite and calcite) plays an important role in regulating the ocean’s carbon budget1,2. Whereas most of the CaCO3 production in the oceans is biogenic, a fraction forms in abiotic reactions. To date, abiotic CaCO3 production in the form of aragonite has been observed in certain localities, such as the Bahamas or the Persian Gulf, where specific conditions are present. However, as ocean acidification intensifies, the production of abiotic CaCO3 will diminish and may have already diminished significantly, due to the reduction in the saturation state of the carbonate ion. Here we show, for the first time, the occurrence of abiotic aragonite production in the eastern Mediterranean, under different conditions than previously observed and a possible analogue to future ocean conditions under the effects of global warming. According to the proposed mechanism, the abiotic production of CaCO3 in surface waters may be enhanced by warming and stratification of the oceans, rather than additional seeding or water mass mixing. In contrast, ocean acidification may inhibit the abiotic production of surface waters, countering the positive effect of warming. Based on our observations, we argue that abiotic aragonite production is more ubiquitous than previously considered and delineate the production mechanism. Based on state-of-the-art climate models, we illustrate that the potential that this mechanism is diminishing and discuss its impact.
Background
The exchange of CO2 between the ocean and the atmosphere has been considered to act as a major feedback mechanism that helps regulate planetary climate1,2. Currently, it is estimated that ca. 25% of anthropogenic CO2 emitted to the atmosphere annually is absorbed by the ocean3,4. The ensuing chemical reaction of the absorbed CO2 with seawater produces carbonic acid, resulting in a process known as ocean acidification5. The production of carbonic acid not only reduces seawater's pH, but also shifts the speciation of dissolved inorganic carbon (DIC = CO2 + HCO3− + CO3−2) in seawater away from carbonate (CO3−2) to bi-carbonate (HCO3−), according to the following stoichiometric equation – CO2 + H2O + CO3−2 → 2HCO3−.
The speciation of DIC in seawater is also influenced by temperature through its effect on the thermodynamic dissociation constants, where warming shifts the carbonate system towards CO3−2. Most of the transformation of DIC into solid phase, be it organic or inorganic, is biologically-driven6. Where, biologically mediated assimilation of DIC into inorganic carbon solid phase (CaCO3) removes Ca+2 from seawater and results also in a reduction of seawater total alkalinity (TA). This process is also non conservative with salinity changes7. While the organic sink (“biological pump”) removes carbon from the atmosphere, the inorganic sink (precipitation of carbonate minerals), consumes TA and shifts the carbonate system towards CO2 and its degassing from the ocean surface to the atmosphere8, acting as a positive feedback to global warming2,9. Precipitation of both crystal forms of CaCO3 (aragonite and calcite) in the ocean is mostly biogenic10 and is a highly complex process, affected by environmental conditions, such as temperature, dissolved nutrient levels and, most noteworthy, the ratio of the Ca+2 and CO3−2 ion activity product to the solubility constant of CaCO3, also known as the CaCO3 saturation state (Ω)9.
Oceanic surface waters are mostly supersaturated with respect to CaCO3 (Ω > 1) and therefore, one would rightfully expect the abiotic precipitation of CaCO3 to be a common occurrence and an important process in the oceanic carbon cycle and climate regulation. However, to date, large spatial scale abiotic CaCO3 precipitation (usually aragonite) in the marine environment have been observed only during events referred to as “whitening events”, when CaCO3 is abiotically precipitated in surface waters, turning them milky white in relatively shallow waters11,12,13. Such events occur almost exclusively in the Persian Gulf and the Bahamas. While the specific mechanism driving whitening events is still a highly contested topic14,15,16, extremely high levels of Ω seem to be an important factor triggering them.
The abiotic nucleation of CaCO3 in seawater is strongly inhibited kinetically17,18. Suspended particles, which provide a mineral surface area for nucleation (“seeding”), were also suggested to play an important control for carbonate precipitation from seawater16,19. However, “seeded” experiments have concluded that temperature controls the mineralogy of the precipitating phase and that the precipitation rate is proportional to the saturation state20,21. Hence, vast low latitude oceanic regions, where the saturation state is high22 and most CO2 degassing occurs23, are suspected to accommodate abiotic precipitation of aragonite. Yet, this process remains largely undetected, as it is often concealed by the larger biological signal8,9.
Abiotic aragonite precipitation
As the ocean warms and stratification intensifies, primary productivity decreases, most notably in oligotrophic regions25,26. This would mean that the carbon cycle in these regions may sway from biological mediation towards greater abiotic (chemical and physical) control. This process is already ongoing in the southeastern Mediterranean27,28 making it a good model system for the warmer stratified future of the oceans. Like most oceanic basins, the region is experiencing acidification, yet, the rate of acidification is relatively low due to increasing alkalinity (supplemental 1, Fig. S1.1). In contrast, the summertime sea surface temperature has risen at an exceptionally high rate of up to 1 °C/decade over the last three decades29,30, substantially higher than warming rates in other low latitude tropical seas31. Additionally, the oligotrophic state of the region results in one of the lowest primary productivities of any marine water body in the world32.
In a sediment-trap study that we conducted in the southeastern Mediterranean Sea (Fig. 1), we quantify for the first time the flux of inorganic precipitation of aragonite in the water column during the summer (supplemental 2). Unlike calcite, there are only a few sources of biogenic aragonite in the study area, e.g. pteropod blooms and resuspension of eroded aragonite shells of benthic organisms – none of which were present in the traps based on microscopic examination of the material in the traps and adjacent surface sediments. The Sr/Ca ratios in the traps ranged between 6.8 and 11.4 in the summer months when aragonite was present (Fig. 2; supplemental 1, Table S1.1). The Sr/Ca ratio in the sediment was found to be ~ 5.0 and biogenic sources such as pteropods were found to have ratios ranging between 2.3 and 3.6 (supplement 1, Table S1.2). As a result, neither of these sources can explain the higher Sr/Ca ratio in the traps. However, these values agree well with the Sr/Ca ratio (~ 9.0) of abiotic aragonite precipitated in lab experiments from seawater in the range of summertime temperatures in the study area33. This is still lower than the maximum value of Sr/Ca = 11.4, where the excess Sr is likely sourced from Saharan dust, which has a Sr/Ca ratio of ~ 3034,35. However, the dust is aragonite-free and its supply rate is low during summer36. Thus, neither dust nor resuspension, given the low wave activity during summer, explain the relatively high Sr/Ca ratio in the traps. Thus, it is reasonable to conclude that it is caused by abiotic precipitation of aragonite (supplemental 1, Figs. S1.2–S1.3).
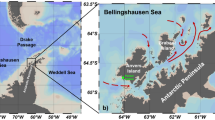
(a) Location map of the Mediterranean Sea showing sea surface temperature (SST) in °C for August 2016, Data from MODIS|Aqua (https://oceancolor.gsfc.nasa.gov/). (b) bathymetric map of the Israeli shelf24 showing the location of the Hadera and THEMO1 stations. (c) SST temperature prolife along the transect shown in (a).
Record from the Hadera station showing (from the bottom), temperature (at 26 m) and inferred pCO2, mean monthly salinity for the given month and inferred alkalinity, calculated saturation index for aragonite, aragonite fraction of total carbonate, flux of aragonite to the trap, inferred total CO2 degassing from aragonite precipitation, Sr/Ca ratio of the sediment in the trap, and acoustic backscatter. Shaded areas indicate periods when seawater ΩAr > 4 (see Fig. 3).
Recently, it was suggested that abiotic aragonite precipitation may be caused by seeding, where aragonite crystallizes on suspended nucleation particles37,38. In this model, a high load of nucleation seeds/surface area promotes increased precipitation9. The saturation state, while elevated (≥ 4), does not appear to be sufficient for spontaneous nucleation of aragonite from seawater39. Therefore, our results support utilization of preexisting nuclei in the water mass by a sustained high aragonite saturation state during summer. Acoustic backscatter data from the ADCP adjacent to the sediment trap (Fig. 2), a proxy for turbidity in the water, indicated highest turbidity between January and May, and much lower turbidity during the summer months. This trend was mirrored in the total mass and CaCO3 fluxes in the sediment traps (supplement 1; Table S1.1) decreased from 82 and 15.8 gr m−2 d−1 in January 2016 to 2.9 and 0.7 gr m−2 d−1 in August 2016, respectively. As such, we would argue that even in stratified, low-turbidity environments, precipitation nuclei are not a limiting factor for inorganic precipitation of aragonite.
The other major factor for the precipitation of aragonite is the seawater CaCO3 saturation state. Abiotic nucleation of aragonite has been demonstrated to be possible with ΩAr ≈ 4, given induction time (the period of time that is necessary to initiate a reaction) of around two months37. To maintain stable ΩAr conditions, a prolonged period of water column stability is required which is consistent with the stratification of the surface water. Our observations show that aragonite formation started in late-spring and continued throughout the summer months, between May and October 2016 (Fig. 2), corresponding to the time of year when warming and weaker mechanical mixing causes intense stratification of the surface layer (Fig. 3). At its apex, the gradient across the seasonal thermocline was on the order of 10 °C (supplement 1; Fig. S1.4). As this layer concentrates heat at the surface, pCO2 levels reach super saturation in excess of 450 μatm (Fig. 2). During winter, the upper water column is mixed (Fig. 3), temperature and ΩAr are lower (Fig. 2), while biogenic production and sediment load are higher, suppressing abiotic formation of aragonite (Fig. 4a). The absence of abiotic aragonite in the sediment traps during the autumn of 2015 when ΩAr was at times > 4, is likely the result of two notable (4–6 °C) cooling events that occurred in August and September 2015. These events lasted several days (Fig. 2) and broke the stratification, resetting the required induction time of ΩAr. Notably, 2015 was an anomalous year with the least number of days with ΩAr > 4 in the last decade (supplemental 1, Fig. S1.5). Additionally, while nearly constant ΩAr > 4 conditions in local surface waters prevail throughout the summertime, when stratification is most intense (supplement 1, Fig. S1.6), when that stratification is broken, as in winter (supplement 1, Fig. S1.7), values of ΩAr drop below 4. The aragonite found in the traps in late May to early June 2016 likely had formed higher in the water column as the stratification and super-saturation conditions began already in early April at shallower depths and extended into the summer28 (Fig. 3), allowing for aragonite formation (Fig. 4b).
Schematic model for aragonite formation in the Eastern Mediterranean. During cooling events and vertical mixing of the water column (a), the cooler water with increased productivity acts as a CO2 sink. During prolonged periods of warming (b), the water column becomes thermally stratified, CO2 degasses due to super saturation, CO3−2 and ΩAr increase, and abiotic aragonite precipitation occurs and intensify CO2 supersaturation and degassing.
The estimated flux of aragonite based on the trap data from 2016 would be ~ 0.20 ± 0.04 mol m−2 year−1 for the deployment period. Assuming that this is a representative flux of inorganic carbonate production, this would correspond to a CO2 efflux8 of 0.12 ± 0.01 mol CO2 m−2 year−1 (supplemental 4). The total annual net CO2 flux to the atmosphere in the southeastern Mediterranean Sea was previously estimated at 0.85 ± 0.27 mol m−228. This suggests that the abiotic aragonite precipitation may account for ~ 15 ± 3% of the annual net CO2 flux from the sea surface to the atmosphere in the southeastern Mediterranean. Relatively stable and consistent sea surface temperature and salinity conditions in the region40 allow for the assumption that this represents at least the region of the Levant Basin and possibly extends beyond it in the southeastern Mediterranean. This net annual calcification flux per unit area is only ~ 0.5% of CaCO3 flux estimated for coral reefs41, but the potential area of production is orders of magnitude larger, making it a significant source of atmospheric CO2.
This is the first time that the relation between warming, stratification, abiotic aragonite precipitation and CO2 release is demonstrated in any marine system. The observed link between surface warming and CO2 efflux due to abiotic aragonite precipitation provide a new mechanism by which warming reduces the southeastern Mediterranean Sea buffering capacity for sequestering atmospheric CO2 (and possibly even becoming a CO2 source28). This is a novel feedback mechanism of the carbonate system under global warming.
Implications
More broadly, it was recently shown that much of the lower latitude surface ocean is characterized by ΩAr ≥ 4, and that ΩAr is positively correlated with temperature22,42,43. As the lower latitude ocean warms, much of that heat is stored in the surface waters, resulting in enhanced stratification and oligotrophy25,26,31. These three properties (warming, stratification and oligotrophy) will likely modify the balance of the carbon system, thereby affecting the ocean’s capacity to store CO244. Warming, in particular, is of significant importance as it affects the apparent rate of CaCO3 precipitation through the kinetic rate coefficient KT and order of reaction nT (which are temperature dependent; supplement 3; Fig. S3.1). Thus, if the temperature increases KT and nT faster than ocean acidification decreases ΩAr, the net apparent CaCO3 precipitation rate may increase. However, atmospheric buildup of CO2 is faster than warming in terms of their combined and individual effects on KT and ΩAr (supplement 3). Under the influence of warming alone, global potential aragonite production rate would increase, but due to the effect of CO2 induced acidification, they have been and are expected to continue to drop (Fig. 5). As a result, the abiotic aragonite sink may be weakening, resulting in reduced TA uptake and non-conservative build-up of TA in the oceans as suggested by the Hawaiian Ocean Time Series salinity normalized TA time series (supplement 1, Fig. S1.8). In turn, this TA build-up could potentially increase the ocean’s buffering capacity for increasing atmospheric CO2, increasing oceanic uptake. However, it should be noted that decadal changes in terrestrial runoff, deep mixing and evaporation regimes in and around the Pacific could also explain the non-conservative long-term trend in salinity45. Regardless, the exact contribution of abiotic aragonite precipitation in the upper water column cannot be fully quantified at this time—but it is a feedback mechanism that should be further investigated and incorporated into planetary models to move towards a more complete description of the ocean/atmosphere system.
The change in \(R_{ai}\) (abiotic aragonite precipitation rate) over time from model simulations. \(R_{ai}\) (black line), \(R_{ai}^{SST - only}\)(red line) and \(R_{ai}^{\Omega - only}\)(blue line) from CMIP6 Historical and ScenarioMIP SSP2-4.5 for the eastern Mediterranean. The \(R_{ai}\) scenario reflects the ‘true’ CMIP prediction, whereas the \(R_{ai}^{SST - only}\) and \(R_{ai}^{\Omega - only}\) scenarios fix \(\Omega_{Ar}\) and SST, respectively to 1850–1899 climatological values. The top, central and bottom panels are UKESM1-0-LL, GFDL-CM4, and CanESM5, respectively. Shaded area indicate the range of error while the lines represents the annual mean (of monthly mean \(R_{ai}\) calculated across the eastern Mediterranean) and the shaded region spans the annual range.
In the geological record, shifts between periods of aridity and strong oceanic stratification with high aragonite/calcite ratios and periods of high humidity, cooler mixed water column conditions with low aragonite/calcite ratios, are evident in low latitude sediment cores42. Taking into account that surface seawater pCO2 is mostly temperature-dependent, and that ocean temperatures will continue to increase in the near future46, a reduction in the ocean’s capacity to absorb CO2 is to be expected under prolonged stratification. In both Earth’s past and near future, warming due to the earth orbital changes could enhance the abiotic carbonate formation, potentially resulting in a greater release of CO2 from the surface waters. The switch from cooler waters (glacial/icehouse periods), where carbon is removed by the biological pump to warm (interglacial/greenhouse period), stratified and oligotrophic surface water, where abiotic aragonite is precipitated may have altered the oceanic buffering capacity. This means that the surface water will become a much less effective sink and under extreme scenarios, larger areas of the surface ocean may become a CO2 source. However, our proposed feedback mechanism suggests a future reduction in abiotic aragonite precipitation under the increasing effect of ocean acidification. The unprecedented rate of anthropogenic climate change is not paired with a comparable increase in continental weathering. In the geological past, weathering may have provided TA to sustain this mechanism, at least in epicontinental seas which could decouple with respect to Ca2+ and Mg2+ from the ocean47.
We therefore suggest that the feedback between warming, acidification, and induced CO2 release due to abiotic aragonite precipitation can be regarded as a potential feedback to global warming, adding to a growing list of feedbacks such as reduced primary productivity in the ocean, desertification, and melting of permafrost46, and therefore should be accounted for in future estimation of ocean evolution in response to climate change.
Methods
Samples were collected from sediment traps deployed 2 km offshore of Hadera, Israel at 25 m bottom depth. Analysis of sediment samples was carried out using X-ray fluorescence (XRF) and X-ray diffraction (XRD). Seawater carbonate chemistry parameters were estimated from in situ measurements of temperature and salinity from the ISRAMR data base, which were converted to pCO2 and total alkalinity respectively. See supplement 2 for more information on methods. Global state of the carbon system was estimated from CMIP6 products, see supplement 3 for more information.
Data availability
Scripts, results and additional material for model simulations are available via a the Figshare repository at https://doi.org/10.6084/m9.figshare.15121131; all other material in the supplement are available at https://doi.org/10.6084/m9.figshare.1972181. Oceanographic data used in this manuscript is available through the ISRAMAR (https://isramar.ocean.org.il/isramar2009/) databases. see https://themo.haifa.ac.il/ for more information on the THEMO project. Additional notes on data are listed in supplements 2 and 3.
References
Marinov, I. & Sarmiento, J. L. The role of the oceans in the global carbon cycle: An overview. In The Ocean Carbon Cycle and Climate, vol 40 (eds Follows, M. & Oguz, T.) 251–295 (Springer NATO Science Series (Series IV: Earth and Environmental Sciences), 2004). https://doi.org/10.1007/978-1-4020-2087-2_8
Williams, R. G., Katavouta, A. & Goodwin, P. Carbon-cycle feedbacks operating in the climate system. Curr. Clim. Chang. Reports 5, 282–295 (2019).
Sabine, C. L. & Tanhua, T. Estimation of anthropogenic CO2 inventories in the ocean. Ann. Rev. Mar. Sci. 2, 175–198 (2010).
Friedlingstein, P. et al. Global carbon budget 2021. Earth Syst. Sci. Data 14, 1917–2005 (2022).
Caldeira, K. & Wickett, M. E. Anthropogenic carbon and ocean pH. Nature 425, 365–365 (2003).
Galvez, M. E., Fischer, W. W., Jaccard, S. L. & Eglinton, T. I. Materials and pathways of the organic carbon cycle through time. Nat. Geosci. 13, 535–546 (2020).
Steiner, Z., Turchyn, A. V., Harpaz, E. & Silverman, J. Water chemistry reveals a significant decline in coral calcification rates in the southern Red Sea. Nat. Commun. 9, 3615 (2018).
Frankignoulle, M., Canon, C. & Gattuso, J.-P. Marine calcification as a source of carbon dioxide: Positive feedback of increasing atmospheric CO2. Limnol. Oceanogr. 39, 458–462 (1994).
Renforth, P. & Henderson, G. Assessing ocean alkalinity for carbon sequestration. Rev. Geophys. 55, 636–674 (2017).
Milliman, J. D. Production and accumulation of calcium carbonate in the ocean: Budget of a nonsteady state. Glob. Biogeochem. Cycles 7, 927–957 (1993).
Wells, A. J. & Illing, L. V. Present-day precipitation of calcium carbonate in the Persian Gulf. In Deltaic and Shallow Marine Deposits (ed. van Straaten, L. M. J. U.) 429–435 (Elsevier, 1964).
Long, J., Hu, C. & Robbins, L. Whiting events in SW Florida coastal waters: A case study using MODIS medium-resolution data. Remote Sens. Lett. 5, 539–547 (2014).
Broecker, W. S. & Takahashi, T. Calcium carbonate precipitation on the Bahama Banks. J. Geophys. Res. 71, 1575–1602 (1966).
Swart, P. K., Oehlert, A. M., Mackenzie, G. J., Eberli, G. P. & Reijmer, J. J. G. The fertilization of the Bahamas by Saharan dust: A trigger for carbonate precipitation?. Geology 42, 671–674 (2014).
Purkis, S. et al. Hydrodynamic control of whitings on Great Bahama Bank. Geology 45, 939–942 (2017).
Morse, J. W., Gledhill, D. K. & Millero, F. J. CaCO3 precipitation kinetics in waters from the great Bahama bank.. Geochim. Cosmochim. Acta 67, 2819–2826 (2003).
Iglesias-Rodriguez, M. D. et al. Progress made in study of ocean’s calcium carbonate budget. Eos Trans. Am. Geophys. Union 83, 365–375 (2002).
Burton, E. A. & Walter, L. M. Relative precipitation rates of aragonite and Mg calcite from seawater: Temperature or carbonate ion control?. Geology 15, 111 (1987).
Bustos-Serrano, H., Morse, J. W. & Millero, F. J. The formation of whitings on the Little Bahama Bank. Mar. Chem. 113, 1–8 (2009).
Morse, J. W., Wang, Q. & Tsio, M. Y. Influences of temperature and Mg: Ca ratio on CaCO3 precipitates from seawater. Geology 25, 85 (1997).
Inskeep, W. P. & Bloom, P. R. An evaluation of rate equations for calcite precipitation kinetics at less than 0.01 atm and pH greater than 8. Geochim. Cosmochim. Acta 49, 2165–2180 (1985).
Jiang, L.-Q. et al. Climatological distribution of aragonite saturation state in the global oceans. Glob. Biogeochem. Cycles 29, 1656–1673 (2015).
Takahashi, T. et al. Climatological mean and decadal change in surface ocean pCO2, and net sea–air CO2 flux over the global oceans. Deep Sea Res. Part II Top. Stud. Oceanogr. 56, 554–577 (2009).
Kanari, M. et al. Sediment transport mechanisms revealed by quantitative analyses of seafloor morphology: New evidence from multibeam bathymetry of the Israel exclusive economic zone. Mar. Pet. Geol. 114, 104224 (2020).
Roxy, M. K. et al. A reduction in marine primary productivity driven by rapid warming over the tropical Indian Ocean. Geophys. Res. Lett. 43, 826–833 (2016).
Behrenfeld, M. J. et al. Climate-driven trends in contemporary ocean productivity. Nature 444, 752–755 (2006).
Sisma-Ventura, G., Yam, R. & Shemesh, A. Recent unprecedented warming and oligotrophy of the eastern Mediterranean Sea within the last millennium. Geophys. Res. Lett. 41, 5158–5166 (2014).
Sisma-Ventura, G., Bialik, O. M., Yam, R., Herut, B. & Silverman, J. pCO2 variability in the surface waters of the ultra-oligotrophic Levantine Sea: Exploring the air–sea CO2 fluxes in a fast warming region. Mar. Chem. https://doi.org/10.1016/j.marchem.2017.06.006 (2017).
Ozer, T., Gertman, I., Kress, N., Silverman, J. & Herut, B. Interannual thermohaline (1979–2014) and nutrient (2002–2014) dynamics in the Levantine surface and intermediate water masses, SE Mediterranean Sea. Glob. Planet. Change 151, 60–67 (2017).
Skliris, N. et al. Decadal scale variability of sea surface temperature in the Mediterranean Sea in relation to atmospheric variability. Ocean Dyn. 62, 13–30 (2012).
Polovina, J. J., Howell, E. A. & Abecassis, M. Ocean’s least productive waters are expanding. Geophys. Res. Lett. 35, L03618 (2008).
Reich, T. et al. Seasonal dynamics of phytoplankton and bacterioplankton at the ultra-oligotrophic southeastern Mediterranean Sea. bioRxiv https://doi.org/10.1101/2021.03.24.436734 (2021).
Gaetani, G. A. & Cohen, A. L. Element partitioning during precipitation of aragonite from seawater: A framework for understanding paleoproxies. Geochim. Cosmochim. Acta 70, 4617–4634 (2006).
Rahav, E. et al. The impact of atmospheric dry deposition associated microbes on the southeastern Mediterranean Sea surface water following an intense dust storm. Front. Mar. Sci. 3, 127 (2016).
Krom, M., Cliff, R., Eijsink, L., Herut, B. & Chester, R. The characterisation of Saharan dusts and Nile particulate matter in surface sediments from the Levantine basin using Sr isotopes. Mar. Geol. 155, 319–330 (1999).
Offer, Z. Y. & Goossens, D. T. years of aeolian dust dynamics in a desert region (Negev desert, Israel): Analysis of airborne dust concentration, dust accumulation and the high-magnitude dust events. J. Arid Environ. 47, 211–249 (2001).
Wurgaft, E., Steiner, Z., Luz, B. & Lazar, B. Evidence for inorganic precipitation of CaCO3 on suspended solids in the open water of the Red Sea. Mar. Chem. 186, 145–155 (2016).
Pan, Y. et al. The role of Mg2+ in inhibiting CaCO3 precipitation from seawater. Mar. Chem. 237, 104036 (2021).
Sun, W., Jayaraman, S., Chen, W., Persson, K. A. & Ceder, G. Nucleation of metastable aragonite CaCO3 in seawater. Proc. Natl. Acad. Sci. 112, 3199–3204 (2015).
Grodsky, S. A., Reul, N., Bentamy, A., Vandemark, D. & Guimbard, S. Eastern Mediterranean salinification observed in satellite salinity from SMAP mission. J. Mar. Syst. 198, 103190 (2019).
Albright, R., Langdon, C. & Anthony, K. R. N. Dynamics of seawater carbonate chemistry, production, and calcification of a coral reef flat, central Great Barrier Reef. Biogeosciences 10, 6747–6758 (2013).
Caldeira, K. & Akai, M. Ocean storage. In IPCC Special Report on Carbon Dioxide Capture and Storage (eds Metz, B. et al.) 277–317 (Cambridge University Press, 2005).
Gregor, L. & Gruber, N. OceanSODA-ETHZ: A global gridded data set of the surface ocean carbonate system for seasonal to decadal studies of ocean acidification. Earth Syst. Sci. Data 13, 777–808 (2021).
Hallenberger, M. et al. Increased fluvial runoff terminated inorganic aragonite precipitation on the Northwest Shelf of Australia during the early Holocene. Sci. Rep. 9, 18356 (2019).
Du, Y. et al. Decadal trends of the upper ocean salinity in the tropical Indo-Pacific since mid-1990s. Sci. Rep. 5, 16050 (2015).
IPCC. Special Report on the Ocean and Cryosphere in a Changing Climate. (The Intergovernmental Panel on Climate Change, United Nations, 2019).
Bialik, O. M. et al. Mg isotope response to dolomitization in hinterland-attached carbonate platforms: Outlook of δ 26 Mg as a tracer of basin restriction and seawater Mg/Ca ratio. Geochim. Cosmochim. Acta 235, 189–207 (2018).
Acknowledgements
This research was funded by the Israel Ministry of Energy Contract No. 21417028 to TK and Onn Crouvi (GSI). We thank Barak Herut from IOLR who headed the “Israel continental shelf research” and obtained the funds for executing the sediment trap research. OMB was supported by Marie Skłodowska Curie fellowship (101003394—RhodoMalta) and is currently supported by the German (GEOMAR) - Israeli (University of Haifa) Helmholtz International Laboratory: The Eastern Mediterranean Sea Centre - An Early-Warning Model-System for our Future Oceans (EMS FORE). Farther acknowledgments are noted in supplement 2.
Author information
Authors and Affiliations
Contributions
O.M.B.—Conceptualization, contributed data and analysis tools, carried out analysis and took part in writing of the manuscript. G.S.V.—Conceptualization, contributed data and analysis tools, carried out analysis and took part in writing of the manuscript. N.V.V.—Carried out analysis and contributed to the writing of this manuscript. J.S.—Contributed analysis tools and contributed to the writing of this manuscript. T.K.—Conceptualization, conceived the design of the data collection, obtained funds, contributed data and analysis tools, carried out analysis and took part in writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bialik, O.M., Sisma-Ventura, G., Vogt-Vincent, N. et al. Role of oceanic abiotic carbonate precipitation in future atmospheric CO2 regulation. Sci Rep 12, 15970 (2022). https://doi.org/10.1038/s41598-022-20446-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-20446-7
This article is cited by
-
Availability and type of energy regulate the global distribution of neritic carbonates
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.