Abstract
The prognostic significance of absolute lymphocyte count (ALC) and absolute neutrophil count (ANC) remains unclear in patients with postoperative pneumonia (POP). The study objectives were to investigate the prognostic effects of ALC and ANC in POP patients, and to evaluate the time courses of ALC and ANC during hospitalization. This post-hoc analysis of a single-center prospective observational study evaluated consecutive POP patients, and comparatively analyzed community-acquired pneumonia (CAP) patients to highlight features of POP. In total, 228 POP patients and 1027 CAP patients were assessed. Severe lymphopenia (ALC < 500 cells/μL) at diagnosis was associated with worse 90-day survival in both types of pneumonia. In POP patients, neutrophilia (ANC > 7500 cells/μL) was associated with better survival, whereas CAP patients with neutrophilia tended to have a lower survival rate. Prolonged lymphopenia and delayed increase in neutrophils were characteristic time-course changes of non-survivors in POP. The time courses of ALC and ANC between survivors and non-survivors in POP trended differently from those in CAP. Our study showed that ALC and ANC at pneumonia diagnosis can serve as prognostic factors in POP patients. Differences in time-course changes of ALC and ANC between survivors and non-survivors may provide important information for future immunological research in pneumonia.
Similar content being viewed by others
Introduction
Postoperative pneumonia (POP) is a serious infectious complication associated with increased morbidity and mortality in postoperative patients1,2,3,4,5,6. Despite advances in surgical techniques, anesthesia, and perioperative care, POP remains one of the most common postoperative complications7. The mortality in patients with POP even when appropriate antimicrobials are prescribed is high1,2,3. Thus, a better understanding of the state of illness and novel management strategies are needed to improve POP outcomes.
It is important to assess individual risk factors associated with mortality to optimize treatment for patients with pneumonia, and numerous prognostic indicators have been established8,9. Among these factors, the absolute lymphocyte count (ALC) and absolute neutrophil count (ANC) in blood have been recently recognized as useful risk stratification markers for patients with pneumonia10,11,12,13. Previous studies in patients with community-acquired pneumonia (CAP) have shown that lymphopenia or failure to increase the neutrophil count at diagnosis was associated with an increased mortality risk11,13. The neutrophil to lymphocyte ratio (NLR) has also been reported as a risk factor for mortality in patients with CAP10. Furthermore, ALC and ANC can potentially provide information about individual immunological phenotypes of patients with pneumonia. Specifically, lymphopenia is a marker of impaired immunity in critically ill patients, and an increased number of circulating neutrophils usually reflects systemic inflammation14,15,16,17. Therefore, investigation of the relationship between these immune cell counts and mortality would be helpful to characterize individual host immunological phenotypes associated with poor outcomes in patients with pneumonia; however, there is a lack of evidence regarding patients with POP in the literature. POP is a second-hit infection following a surgical insult and is associated with postoperative immune dysregulation18,19. Thus, investigating the kinetics of ALC and ANC in patients with POP may highlight the immunological features that are distinct from those of CAP.
The study aim was to investigate the prognostic effects of ALC and ANC in patients with POP. Furthermore, we evaluated the time courses of ALC and ANC during hospitalization. To highlight features of POP as a second-hit infection, we comparatively analyzed patients with CAP.
Methods
Study design and participants
This study was a post-hoc analysis of a prospective observational study conducted between March 2010 and March 2017 at Nagoya University Hospital (a tertiary hospital, Nagoya, Japan). Details of the study methods have been described elsewhere20. Briefly, patients aged ≥ 20 years old with pneumonia who needed in-hospital treatment at the study institution were consecutively enrolled. Pneumonia was diagnosed according to international guidelines21,22, and was classified into the following three categories: ventilator-associated pneumonia (VAP), hospital-acquired pneumonia (HAP), and CAP, including health-care associated pneumonia (HCAP). These definitions have been described previously23. All patients received antimicrobials at the discretion of the physicians in charge. This study was approved by the Ethics Review Committee of Nagoya University School of Medicine and adhered to the Japanese Ethical Guidelines for Medical and Health Research Involving Human Subjects. The Ethics Review Committee of Nagoya University School of Medicine approved that informed consent of the participants was waived, but the opt-out method was adopted in accordance with the Japanese ethics guidelines. This study was registered with the University Hospital Medical Information Network in Japan (number, UMIN000007090).
Postoperative pneumonia was included in HAP or VAP and defined as pneumonia that developed ≤ 30 days after surgery5. Patients with POP were analyzed in this study. To highlight features of POP as a second-hit infection, we comparatively analyzed patients with CAP as a first-hit infection. The patients were excluded from analyses when they met the following criteria: had undergone surgery under topical anesthesia, had an immunocompromised disorder (human immunodeficiency virus-positive, hematological diseases, primary immunodeficiency diseases, asplenia, solid-organ transplantation, ≥ 14 days of treatment with ≥ 10 mg/day of prednisone or equivalent, cytotoxic chemotherapy, and other immunosuppressive drugs), and had missing data of ALC or ANC at pneumonia diagnosis. When patients had multiple registrations, only data from the initial admission were evaluated.
Data collection and definition of variables
Data prospectively collected at pneumonia diagnosis (day 0) included baseline demographic information, laboratory findings at diagnosis, illness severity, microbiological characteristics, and initial treatment for pneumonia. Clinical outcome data were collected 30 and 90 days after the diagnosis of pneumonia. Detailed information on the preceding surgical procedures and sequential laboratory findings, including total and differential leukocyte counts, were retrospectively collected from medical records. For patients with POP, to estimate the severity of surgical procedures, the surgery severity score was calculated using the Physiological and Operative Severity Score for enUmeration of Mortality and morbidity (POSSUM), which is a scoring system for prediction of perioperative mortality and morbidity in various types of surgery24. Details of the microbiological evaluation have been described elsewhere20. Antibiotic treatment was classified as appropriate when the identified pathogens were susceptible to the initially prescribed antibiotic(s)23.
Total and differential leukocyte counts were submitted for routine clinical analysis in the laboratories of the study institution. ALC and ANC were evaluated at the following time points: day 0–1 (day 0), day 2–4 (day 3), day 5–9 (day 7), day 11–17 (day 14), day 18–24 (day 21), and day 25–31 (day 28). For patients with POP, ALC, and ANC before pneumonia diagnosis were additionally assessed (i.e., findings of the preoperative laboratory test and the first and last postoperative laboratory test before diagnosis of POP).
Outcome measures
The primary endpoint was 90-day all-cause mortality, defined as death within 90 days after pneumonia diagnosis. In addition, 90-day pneumonia-related mortality was assessed. Pneumonia-related mortality was defined as death caused by respiratory failure or multiple organ failure following POP. This cause of death was confirmed by the physician in charge and one of the research members (YM, MS, JO, TS, HK, and YS).
Statistical analysis
All statistical tests were two-tailed, and a p value of < 0.05 was considered indicative of statistical significance. Categorical data were described as frequencies in percentage, and continuous data were described as the median with interquartile range. Pearson’s chi-square test or Fisher’s exact test was used to analyze discrete variables, as appropriate, and the Mann–Whitney U-test was used to analyze continuous variables.
To assess the effects of ALC and ANC on 90-day all-cause mortality, we performed survival analysis based on ALC and ANC at the diagnosis of pneumonia. In this analysis, ALC and ANC were categorized into the following classes considering previous literatures and the upper limit of the normal range of ANC: without lymphopenia (ALC ≥ 1 × 103 cells/μL), mild lymphopenia (0.5 × 103 cells/μL ≤ ALC < 1 × 103 cells/μL), and severe lymphopenia (ALC < 0.5 × 103 cells/μL)11,16,25. ANC was categorized as follows: with neutrophilia (ANC > 7.5 × 103 cells/μL) and without neutrophilia (ANC ≤ 7.5 × 103 cells/μL). The Kaplan–Meier method was used to generate survival curves, and the log-rank test was performed for comparisons among different groups. Time to death due to any cause was estimated from the date of diagnosis of pneumonia. The Cox proportional hazards model was used to estimate the hazard ratio (HR) and 95% confidence interval (CI) of ALC and ANC for 90-day mortality. Several clinically important confounders, i.e., age, sex, severity of illness [surgery severity score and the Sequential Organ Failure Assessment (SOFA) score in patients with POP and the Pneumonia Severity Index in patients with CAP] were included in the multivariable models.
As sub-analyses, the effects of ALC and ANC on 90-day pneumonia-related mortality were assessed using the Cox proportional hazards model. Furthermore, among patients who received appropriate initial antibiotic treatment, the Kaplan–Meier survival curves were generated, and the log-rank test was performed. In addition, the effects of ALC and ANC at diagnosis of pneumonia on 90-day all-cause and pneumonia-related mortality in those patients were assessed, as well as the above main analysis. All statistical data were analyzed using SPSS Statistics software (version 26; IBM, Armonk, NY, USA).
Results
Participants and baseline characteristics
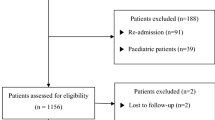
Of 2451 patients assessed for eligibility, 634 had nosocomial pneumonia and 1582 had CAP. Among these 634 and 1582 patients, 228 with POP and 1027 with CAP without immunocompromised disorders were included in the analysis (Fig. 1). In the patients with POP and CAP, 20 (8.8%) and 145 (14.1%) died within 90 days after diagnosis of pneumonia, respectively. Of the patients with POP, 12 died due to pneumonia-related death.
Table 1 shows the characteristics in patient with POP and CAP. In the patients with POP, there were no differences in age, sex, frequency of VAP, low body weight, and comorbidities between survivors and non-survivors. The median time to pneumonia diagnosis from surgery was 5 days in the survivors and non-survivors. Although the frequency of gastroenterological and lung surgery was higher in the non-survivors, the estimated surgery severity scores were comparable between survivors and non-survivors. The SOFA score and frequency of low blood pressure tended to be higher in the non-survivors. In the patients with CAP, there were significant differences in age, sex, frequency of low body weight and several comorbidities, and severity of illness between the survivors and non-survivors. Causative pathogens were identified in 155 patients with POP (68.0%) and 441 with CAP (42.9%). Among the patients with POP, Enterobacter spp. were most frequent, followed by Pseudomonas aeruginosa and Klebsiella pneumoniae. The details are shown in Supplementary Table S1. Among the POP patients with identified pathogens, appropriate initial antibiotics were administered in 88 (62.9%) of 140 survivors and 13 (86.7%) of 15 non-survivors. Less than 5% of patients received corticosteroids for treatment of pneumonia in both types of pneumonia.
Laboratory findings of lymphocytes and neutrophils at pneumonia diagnosis
Laboratory findings of lymphocytes and neutrophils at pneumonia diagnosis are summarized in Table 2. In the patients with POP, 65.8% of the total patients had lymphopenia, and non-survivors had more severe lymphopenia than survivors. ANC was lower for the non-survivors than for the survivors. Neutrophilia was found in only 40.0% of the non-survivors but in 68.3% of the survivors. The percentages of lymphocytes and neutrophils among the total leukocytes and NLR did not differ between the survivors and non-survivors. Lymphopenia was observed in 53.7% of all patients with CAP, and ALC was lower in the non-survivors than survivors. Over 60% of the survivors and non-survivors showed neutrophilia. Non-survivors had higher ANC than survivors. Compared with survivors, non-survivors had a lower percentage of ALC in the total leukocytes and higher percentages of ANC and NLR.
Effects of lymphocyte and neutrophil counts at pneumonia diagnosis on mortality
Figure 2A shows the survival curves stratified by ALC at pneumonia diagnosis. In patients with POP, there were significant differences in the survival rate among the three ALC categories (log-rank p = 0.014), and the survival rate was worse in the severe lymphopenia group than in the other two groups. In the patients with CAP, severe lymphopenia was also associated with worse 90-day survival (p = 0.016). Figure 2B graphically shows curves according to the presence of neutrophilia at the diagnosis of pneumonia. In the patients with POP, neutrophilia at pneumonia diagnosis was associated with better survival (p = 0.010). In contrast, CAP patients with neutrophilia tended to have a lower 90-day survival rate (p = 0.078).
Survival curves for 90-day survival according to lymphocyte and neutrophil counts at pneumonia diagnosis. Kaplan–Meier curves for 90-day survival in patients with postoperative pneumonia and in those with community-acquired pneumonia are shown stratified by absolute lymphocyte count (ALC) (A) and neutrophil count (ANC) (B) at the time of pneumonia diagnosis. Patients were classified according to ALC as follows: severe lymphopenia, ALC < 0.5 × 103 cells/μL; mild lymphopenia, ALC from 0.5 to 0.9 × 103 cells/μL; and without lymphopenia, ALC ≥ 1.0 × 103 cells/μL. According to ANC, patients with ANC > 7.5 × 103 cells/μL were defined as those with neutrophilia, and patients with ANC ≤ 7.5 × 103 cells/μL were defined as those without neutrophilia. The P values were calculated using the log-rank test.
To estimate the effect of ALC and ANC at pneumonia diagnosis on the all-cause mortality, we additionally performed multivariable analyses, adjusting age, sex, surgery severity score, and SOFA score as clinically important confounders. The findings are shown in Supplementary Table S2. Because of the difference in survival curves between patients with and without severe lymphopenia (Fig. 2A), we compared the effects of severe lymphopenia and no or mild lymphopenia on mortality. In the patients with POP, severe lymphopenia increased the risk of mortality [adjusted HR (aHR), 2.65; 95% CI, 1.00–7.02; p = 0.050]. Neutrophilia showed a trend toward decreased mortality risk (aHR, 0.44; 95% CI, 0.17–1.18; p = 0.103) in this model. In the patients with CAP, severe lymphopenia and neutrophilia were associated with an increased mortality risk (aHR, 1.39; 95% CI, 0.91–2.12; p = 0.129; aHR, 1.41; 95% CI, 0.99–2.01; p = 0.059, respectively) although the difference was not statistically significant.
As a sub-analysis, we assessed the effect of ALC and ANC at pneumonia diagnosis on 90-day pneumonia-related mortality. Severe lymphopenia increased the risk of mortality (aHR, 3.66; 95% CI, 1.10–12.19; p = 0.034) in the patients with POP (Supplementary Table S3). The trend of findings was similar to that of the main analysis. For patients who received appropriate initial antibiotic treatment, the survival curves stratified by ALC at pneumonia diagnosis were shown in Supplementary Fig. S1. Key trends were increased risk of mortality from severe lymphopenia and decreased risk of mortality from neutrophilia. These findings were similar to those for all patients with POP. In addition, effects of ALC and ANC at the time of pneumonia diagnosis were also similar for both the 90-day all-cause and pneumonia-related mortality outcomes (Supplementary Tables S4 and S5).
Time courses of the lymphocyte and neutrophil counts and their effects on survival
Figure 3 shows the temporal changes in ALC and ANC between the survivors and non-survivors. The ALC findings are shown in Fig. 3A. Among the patients with POP, the non-survivors had slightly lower lymphocyte counts before surgery; the lymphocyte counts of both the survivors and non-survivors declined after surgery. Although most patients in both patient groups showed lymphopenia at pneumonia diagnosis, the ALC level remained higher in the survivors than in the non-survivors. After the diagnosis of pneumonia, the ALC of the survivors improved over time whereas that of the non-survivors declined for 3 days after the pneumonia diagnosis and failed to improve thereafter. Among the patients with CAP, the non-survivors showed persistent lymphopenia after the diagnosis; however, the degree of progressive lymphopenia was less severe than in the patients with POP.
Time courses of absolute lymphocyte and neutrophil counts between survivors and non-survivors. Abbreviation: Preop = Preoperative day, Postop = Postoperative day.Time courses of absolute lymphocyte count (ALC) (A) and absolute neutrophil count (ANC) (B) between 90-day survivors and non-survivors in postoperative and community-acquired pneumonia are shown. Patients with community-acquired pneumonia included eligible patients without immunocompromised disorders (n = 1027). The dates of the preoperative laboratory test and the first and last postoperative laboratory test before pneumonia diagnosis are expressed as preoperative day, postoperative day 1, and postoperative day 2, respectively. The date of pneumonia diagnosis is described as day 0. The data at each time point are presented as the median and interquartile range. Dotted lines indicate the lower limit of the normal of ALC (1.0 × 103 cells/μL) and the upper limit of the normal of ANC (7.5 × 103 cells/μL). *P value < 0.05 for the comparison of cell counts between survivors and non-survivors at each time point.
The time courses of ANC related to mortality differed between POP and CAP (Fig. 3B). In the patients with POP, the ANC of the survivors and non-survivors increased after surgery. However, the ANC changes after pneumonia diagnosis differed between the survivors and non-survivors. At the pneumonia diagnosis, most of the survivors showed neutrophilia, which rapidly declined after the diagnosis. In contrast, in the non-survivors, ANC failed to increase at the pneumonia diagnosis and showed a delayed increase after the diagnosis. Among the patients with CAP, neutrophilia at diagnosis was more severe and decline in ANC was slower after the diagnosis in the non-survivors than in the survivors.
We also assessed time-course changes of ALC and ANC between survivors and non-survivors with POP who received appropriate initial antibiotics (Supplementary Fig. S2). The trend of findings was almost identical with that for all patients with POP.
Discussion
In this study, we evaluated the associations between mortality and status of leukocyte phenotypes (ALC and ANC at pneumonia diagnosis and their time-course changes) in patients with POP and examined the differences in those findings between POP and CAP. The main findings of our study were as follows: (1) lymphopenia and neutrophilia at pneumonia diagnosis provided prognostic information in patients with POP; (2) prolonged lymphopenia and delayed increase in neutrophils were characteristic time-course changes of the non-survivors in the patients with POP; and (3) the prognostic effects and time-course changes of ALC and ANC between the survivors and non-survivors in POP showed different trends from those in CAP.
Previous studies have reported that blood lymphocyte and neutrophil counts in critically ill patients reflect the balance of pro- and anti-inflammatory host immune responses16,26,27. Host responses to infections have been well-investigated in sepsis17,28. However, evidence regarding pneumonia, except in coronavirus disease 19 (COVID-19), is limited. Host responses to infections differ among infection sites29,30. Thus, the findings of sepsis, including various infection types, may not be applied to all patients with pneumonia. In the research on pneumonia, investigation of the differences in host immune responses between HAP and CAP is an important topic because levels of immune impairment following acute or chronic illnesses can be higher in HAP than in CAP. Van Vught et al. compared plasma biomarkers and blood gene expression profiles between HAP and CAP and could not detect any significant differences in host responses31. In most patients in their study, HAP developed after medical admission, and the patients had various underlying illnesses. Thus, the generalizability of their results based on different patient populations is unclear. We hypothesized that pneumonia following a surgical insult may provide representative information of host immune responses as a second-hit infection. Thus, we compared the characteristics of POP and CAP in which pneumonia was a first-hit infection.
Blood lymphocyte and neutrophil counts have been increasingly recognized as important risk factors for mortality in patients with pneumonia10,11,12,13. In patients with CAP, Bermejo–Martin et al. demonstrated that lymphopenia (< 724 cells/μL) was independently associated with 30-day mortality in their multicenter observational study11. Several studies have shown that lymphopenia is associated with worse outcomes in patients with COVID-1932,33. In particular, patients with COVID-19 and severe lymphopenia (< 500 cells/μL) were at a high risk of death34,35. The neutrophil count has also been proposed as a prognostic factor in patients with severe CAP and septic shock13. However, to the best of our knowledge, the prognostic effects of ALC and ANC in patients with POP have never been evaluated. In our study, severe lymphopenia at diagnosis of pneumonia increased the mortality risk, whereas neutrophilia was associated with improved survival in patients with POP, even after adjustment for severity of surgery and organ failure. Similar trends of these findings were observed when the analysis was performed in patients with POP who received appropriate initial antibiotics and when the endpoint was changed from all-cause mortality to pneumonia-related mortality. In the analysis of patients with CAP, severe lymphopenia was also associated with an increased mortality risk; however, neutrophilia had a negative effect on survival. The findings in patients with CAP in our study were consistent with those in previous studies10,11. Thus, our findings suggest that decreased ALC and non-increasing ANC at diagnosis of pneumonia could be prognostic factors in patients with POP, and their effects on mortality differ from those in patients with CAP.
Consecutive biomarker measurement is useful to improve accuracy for the prediction of mortality risk in patients with pneumonia. Previous studies of patients with CAP showed that serial assessment of C-reactive protein, procalcitonin, or mid regional pro-atrial natriuretic peptide was useful for predicting complicated outcomes36,37,38. In our study, ALC declined within 3 days after pneumonia diagnosis and failed to improve over time in non-survivors of POP and CAP. However, the ALC of survivors recovered in both types of pneumonia. These findings were compatible with those of a previous study involving patients with sepsis in which persistent lymphopenia on the fourth day following diagnosis was more important than initial lymphopenia for prediction of mortality risk15. Our findings suggest that lymphocyte depletion after pneumonia diagnosis has a significant effect on mortality in patients with pneumonia and that development of therapeutic approach to prevent and improve progressive lymphopenia is a key strategy for improving their mortality.
Understanding the biological mechanisms underlying our findings can help to elucidate the pathogenesis of pneumonia and develop future treatment strategies for improving patient outcomes. Our finding that a higher proportion of appropriate initial antibiotic treatment for patients with POP who were non-survivors versus survivors suggests the importance of development of new therapeutic strategies other than antibiotic treatment. Bone marrow failure or massive migration of immune cells to lungs could explain the fluctuations in ALC or ANC39,40. However, there are several other possible mechanisms. In our study, prolonged lymphopenia was observed in the non-survivors. One possible explanation for this phenomenon is lymphocyte apoptosis. Several studies have shown that infection or surgery can induce extensive apoptosis of lymphocytes as anti-inflammatory processes15,17,19,28. Profound lymphocyte depletion in patients with POP may be related to recurrent lymphocyte apoptosis induced by the double hits of surgery and pneumonia. In patients with POP who did not survive, the lack of an increase in the neutrophil count at pneumonia diagnosis was a characteristic finding. We speculate that this finding may reflect excessive anti-inflammatory responses induced by surgical procedures. After acute inflammation, anti-inflammatory cytokines or phagocytosis of apoptotic neutrophils can reduce recruitment or production of neutrophils as counter-regulatory mechanisms to compensate for inflammation41,42. Based on these hypotheses, our study findings imply that the higher levels of immunosuppression and blunt pro-inflammatory responses are characteristic immunological phenomena of non-survivors in patients with POP. As a reference, previous animal studies revealed that pro-inflammatory cytokine production after pneumonia onset was reduced in the two-hit infection model (sepsis followed by P. aeruginosa pneumonia), compared with that in the one-hit model (P. aeruginosa pneumonia only)43,44. In contrast, pro-inflammatory responses may be associated with an increased mortality risk in patients with CAP. Therefore, immunotherapies that modulate host immune responses can be a good therapeutic option for both POP and CAP. However, different strategies may be needed for these two types of pneumonia17,28. Further studies are warranted to verify these hypotheses and to investigate optimal biomarkers to accurately characterize the immune status of patients with POP, as in recent research for COVID-1945.
There were several limitations in this study. First, this was a single-center study, so our findings may not be generalizable to other institutions. Second, our analyses could not fully consider therapeutic factors, including the overuse of broad-spectrum antibiotics, that might affect ALC and ANC kinetics. Third, we could not assess pathogen-specific characteristics because of an insufficiently large sample. Therefore, our findings should be validated in large-scale prospective studies by different research groups. However, we believe that our results provide a basis for future investigations, including immunological research to improve outcomes of patients with pneumonia.
Conclusions
In patients with POP, ALC and ANC at pneumonia diagnosis was useful for stratifying mortality risks. In addition, the time-course of ALC and ANC differed between survivors and non-survivors in patients with POP. Prolonged lymphopenia and a delayed increase in neutrophils were characteristic findings associated with mortality. These findings showed different trends from those in patients with CAP, suggesting that patients with POP have immunological features distinct from those of patients with CAP. Future studies are warranted to validate the prognostic significance of ALC and ANC and to elucidate the biological mechanisms of our findings for the development of future immunomodulatory therapies.
Data availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ALC:
-
Absolute lymphocyte count
- ANC:
-
Absolute neutrophil count
- CAP:
-
Community-acquired pneumonia
- CI:
-
Confidence interval
- COVID-19:
-
Coronavirus disease 19
- HAP:
-
Hospital-acquired pneumonia
- HCAP:
-
Health-care associated pneumonia
- HR:
-
Hazard ratio
- NLR:
-
Neutrophil to lymphocyte ratio
- POP:
-
Postoperative pneumonia
- SOFA:
-
Sequential organ failure assessment
- VAP:
-
Ventilator-associated pneumonia
References
Allou, N. et al. Postoperative pneumonia following cardiac surgery in non-ventilated patients versus mechanically ventilated patients: Is there any difference?. Crit. Care 19, 116. https://doi.org/10.1186/s13054-015-0845-5 (2015).
Dupont, H. et al. Outcome of postoperative pneumonia in the Eole study. Intens. Care Med. 29, 179–188. https://doi.org/10.1007/s00134-002-1603-0 (2003).
Fujita, T., Ishida, Y. & Yanaga, K. Impact of appropriateness of initial antibiotic therapy on outcome of postoperative pneumonia. Langenbecks Arch. Surg. 393, 487–491. https://doi.org/10.1007/s00423-007-0271-5 (2008).
Kazaure, H. S., Martin, M., Yoon, J. K. & Wren, S. M. Long-term results of a postoperative pneumonia prevention program for the inpatient surgical ward. JAMA Surg. 149, 914–918. https://doi.org/10.1001/jamasurg.2014.1216 (2014).
Simonsen, D. F., Sogaard, M., Bozi, I., Horsburgh, C. R. & Thomsen, R. W. Risk factors for postoperative pneumonia after lung cancer surgery and impact of pneumonia on survival. Respir. Med. 109, 1340–1346. https://doi.org/10.1016/j.rmed.2015.07.008 (2015).
Thompson, D. A., Makary, M. A., Dorman, T. & Pronovost, P. J. Clinical and economic outcomes of hospital acquired pneumonia in intra-abdominal surgery patients. Ann. Surg. 243, 547–552. https://doi.org/10.1097/01.sla.0000207097.38963.3b (2006).
Eappen, S. et al. Relationship between occurrence of surgical complications and hospital finances. JAMA 309, 1599–1606. https://doi.org/10.1001/jama.2013.2773 (2013).
Lim, W. S. et al. Defining community acquired pneumonia severity on presentation to hospital: An international derivation and validation study. Thorax 58, 377–382 (2003).
Fine, M. J. et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N. Engl. J. Med. 336, 243–250 (1997).
de Jager, C. P. et al. The neutrophil-lymphocyte count ratio in patients with community-acquired pneumonia. PLoS ONE 7, e46561. https://doi.org/10.1371/journal.pone.0046561 (2012).
Bermejo-Martin, J. F. et al. Lymphopenic community acquired pneumonia (L-CAP), an immunological phenotype associated with higher risk of mortality. EBioMedicine 24, 231–236. https://doi.org/10.1016/j.ebiom.2017.09.023 (2017).
Ceccato, A. et al. Lymphocytopenia as a predictor of mortality in patients with ICU-acquired pneumonia. J. Clin. Med. https://doi.org/10.3390/jcm8060843 (2019).
Guell, E. et al. Impact of lymphocyte and enutrophil counts on mortality risk in severe community-acquired pneumonia with or without septic shock. J. Clin. Med. https://doi.org/10.3390/jcm8050754 (2019).
Lee, A., Whyte, M. K. & Haslett, C. Inhibition of apoptosis and prolongation of neutrophil functional longevity by inflammatory mediators. J. Leukoc. Biol. 54, 283–288 (1993).
Drewry, A. M. et al. Persistent lymphopenia after diagnosis of sepsis predicts mortality. Shock 42, 383–391. https://doi.org/10.1097/SHK.0000000000000234 (2014).
Chung, K. P. et al. Severe lymphopenia is associated with elevated plasma interleukin-15 levels and increased mortality during severe sepsis. Shock 43, 569–575. https://doi.org/10.1097/SHK.0000000000000347 (2015).
Rubio, I. et al. Current gaps in sepsis immunology: New opportunities for translational research. Lancet Infect. Dis. https://doi.org/10.1016/s1473-3099(19)30567-5 (2019).
Cui, P. & Fang, X. Pathogenesis of infection in surgical patients. Curr. Opin. Crit. Care 21, 343–350. https://doi.org/10.1097/MCC.0000000000000227 (2015).
Torrance, H. D., Pearse, R. M. & O’Dwyer, M. J. Does major surgery induce immune suppression and increase the risk of postoperative infection?. Curr. Opin. Anaesthesiol. 29, 376–383. https://doi.org/10.1097/ACO.0000000000000331 (2016).
Sano, M. et al. Risk factors for antibiotic resistance in hospital-acquired and ventilator-associated pneumonia. J. Infect. Chemother. https://doi.org/10.1016/j.jiac.2022.02.012 (2022).
Kalil, A. C. et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the infectious diseases society of America and the American thoracic society. Clin. Infect. Dis. 63, e61–e111. https://doi.org/10.1093/cid/ciw353 (2016).
Metlay, J. P. et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American thoracic society and infectious diseases society of America. Am. J. Respir. Crit. Care Med. 200, e45–e67. https://doi.org/10.1164/rccm.201908-1581ST (2019).
Shindo, Y. et al. Risk factors for drug-resistant pathogens in community-acquired and healthcare-associated pneumonia. Am. J. Respir. Crit. Care Med. 188, 985–995. https://doi.org/10.1164/rccm.201301-0079OC (2013).
Copeland, G. P., Jones, D. & Walters, M. POSSUM: A scoring system for surgical audit. Br. J. Surg. 78, 355–360 (1991).
Bermejo-Martin, J. F. et al. Circulating neutrophil counts and mortality in septic shock. Crit. Care 18, 407. https://doi.org/10.1186/cc13728 (2014).
Reddy, R. C. & Standiford, T. J. Effects of sepsis on neutrophil chemotaxis. Curr. Opin. Hematol. 17, 18–24. https://doi.org/10.1097/MOH.0b013e32833338f3 (2010).
Chang, K. et al. Targeting the programmed cell death 1: Programmed cell death ligand 1 pathway reverses T cell exhaustion in patients with sepsis. Crit. Care 18, R3. https://doi.org/10.1186/cc13176 (2014).
Peters van Ton, A. M., Kox, M., Abdo, W. F. & Pickkers, P. Precision Immunotherapy for Sepsis. Front. Immunol. 9, 1926. https://doi.org/10.3389/fimmu.2018.01926 (2018).
Pelekanou, A. et al. Decrease of CD4-lymphocytes and apoptosis of CD14-monocytes are characteristic alterations in sepsis caused by ventilator-associated pneumonia: Results from an observational study. Crit. Care 13, R172. https://doi.org/10.1186/cc8148 (2009).
Gogos, C. et al. Early alterations of the innate and adaptive immune statuses in sepsis according to the type of underlying infection. Crit. Care 14, R96. https://doi.org/10.1186/cc9031 (2010).
van Vught, L. A. et al. Comparative analysis of the host response to community-acquired and hospital-acquired pneumonia in critically Ill patients. Am. J. Respir. Crit. Care Med. 194, 1366–1374. https://doi.org/10.1164/rccm.201602-0368OC (2016).
Qin, C. et al. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin. Infect. Dis. 71, 762–768. https://doi.org/10.1093/cid/ciaa248 (2020).
Zhou, F. et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 395, 1054–1062. https://doi.org/10.1016/S0140-6736(20)30566-3 (2020).
Martínez-Urbistondo, M. et al. Severe lymphopenia as a predictor of COVID-19 mortality in immunosuppressed patients. J. Clin. Med. https://doi.org/10.3390/jcm10163595 (2021).
Lee, J., Park, S. S., Kim, T. Y., Lee, D. G. & Kim, D. W. Lymphopenia as a biological predictor of outcomes in COVID-19 patients: A nationwide cohort study. Cancers (Basel) https://doi.org/10.3390/cancers13030471 (2021).
Ito, A., Ishida, T., Tachibana, H., Ito, Y. & Takaiwa, T. Serial procalcitonin levels for predicting prognosis in community-acquired pneumonia. Respirology 21, 1459–1464. https://doi.org/10.1111/resp.12846 (2016).
Andersen, S. B. et al. Failure of CRP decline within three days of hospitalization is associated with poor prognosis of Community-acquired Pneumonia. Infect. Dis. 49, 251–260. https://doi.org/10.1080/23744235.2016.1253860 (2017).
Lacoma, A. et al. Usefulness of consecutive biomarkers measurement in the management of community-acquired pneumonia. Eur. J. Clin. Microbiol. Infect. Dis. 31, 825–833. https://doi.org/10.1007/s10096-011-1381-0 (2012).
Shoup, M. et al. Mechanisms of neutropenia involving myeloid maturation arrest in burn sepsis. Ann. Surg. 228, 112–122. https://doi.org/10.1097/00000658-199807000-00017 (1998).
Jafarzadeh, A., Jafarzadeh, S., Nozari, P., Mokhtari, P. & Nemati, M. Lymphopenia an important immunological abnormality in patients with COVID-19: Possible mechanisms. Scand. J. Immunol. 93, e12967. https://doi.org/10.1111/sji.12967 (2021).
Keel, M. et al. Interleukin-10 counterregulates proinflammatory cytokine-induced inhibition of neutrophil apoptosis during severe sepsis. Blood 90, 3356–3363 (1997).
Stark, M. A. et al. Phagocytosis of apoptotic neutrophils regulates granulopoiesis via IL-23 and IL-17. Immunity 22, 285–294. https://doi.org/10.1016/j.immuni.2005.01.011 (2005).
Muenzer, J. T. et al. Characterization and modulation of the immunosuppressive phase of sepsis. Infect. Immun. 78, 1582–1592. https://doi.org/10.1128/IAI.01213-09 (2010).
Shindo, Y. et al. Interleukin 7 immunotherapy improves host immunity and survival in a two-hit model of Pseudomonas aeruginosa pneumonia. J. Leukoc. Biol. 101, 543–554. https://doi.org/10.1189/jlb.4A1215-581R (2017).
van de Veerdonk, F. L. et al. A guide to immunotherapy for COVID-19. Nat. Med. https://doi.org/10.1038/s41591-021-01643-9 (2022).
Acknowledgements
We thank Drs. Daisuke Kobayashi, Ryota Ito, Mitsuru Odate, Hiroshi Morioka, Yuka Tomita, and Nancy Thabet for data acquisition and their comments on results. We are indebted to the clinical research coordinators (Kyoko Kazeto and Sumiyo Tanaka), laboratory staff (Yukari Osada, Teruko Okura, Nami Shimaoka, Toshi Nada, Mariko Mochizuki, Miho Saito, and Nobuya Sakagami), and all health-care professionals who participated in data collection. We would like to thank Enago (www.enago.jp) for the English language review.
Funding
This work was partially supported by the Japan Society for the Promotion of Science KAKENHI (Grant Number 20K08517).
Author information
Authors and Affiliations
Contributions
All authors meet the ICMJE authorship criteria. Y.S., M.S., Y.M., and Y.H. designed this study. Y.M., Y.S., H.K., T.S., J.O., M.S., and M.I. participated in data acquisition. Y.M., Y.S., K.T., and S.M. created the statistical analysis plan, which was reviewed by all authors. Y.M., Y.S., H.K., T.S., J.O., and M.S. contributed to data interpretation. T.Y. and Y.H. contributed to study supervision. Y.M. wrote the initial draft of the manuscript. Y.S., K.T., S.M., and Y.H. contributed to the critical revision of the manuscript for important intellectual content. All authors approved the final draft.
Corresponding author
Ethics declarations
Competing interests
The following information describes relevant financial activities outside of the submitted work. YS reports personal fees (payment for lectures, including service on speakers bureaus) from KYORIN Pharmaceutical Co., Ltd., AstraZeneca K.K., DAIICHI SANKYO COMPANY, LIMITED, Nippon Boehringer Ingelheim Co., Ltd., GlaxoSmithKline plc, and Gilead Sciences Inc. and participates as a member of the case adjudication committee of GlaxoSmithKline Biologicals SA. TY reports grants and personal fees (payment for lectures, including service on speakers bureaus) from Shionogi & Co., Ltd., Dainippon Sumitomo Pharma Co., Ltd., and MSD K.K. SM reports personal fees (payment for consultations in other studies) from Takeda Pharmaceutical Co., Ltd. YH reports grants and personal fees (payment for lectures, including service on speakers bureaus) from Chugai Pharmaceutical Co., Ltd., MSD K.K., GlaxoSmithKline plc, KYORIN Pharmaceutical Co., Ltd., Pfizer Japan Inc., Meiji Seika Pharma Co, Ltd., Sanofi K.K., and Daiichi Sankyo, Inc. All other authors have nothing to declare. The funders had no roles in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Murakami, Y., Shindo, Y., Sano, M. et al. Effects of lymphocyte and neutrophil counts and their time courses on mortality in patients with postoperative pneumonia. Sci Rep 12, 14564 (2022). https://doi.org/10.1038/s41598-022-18794-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-18794-5
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.