Abstract
Long-term use of urate-lowering therapies (ULT) may reduce inflammaging and thus prevent cognitive decline during aging. This article examined the association between long-term use of ULT and cognitive decline among community-dwelling older adults with spontaneous memory complaints. We performed a secondary observational analysis using data of 1673 participants ≥ 70 years old from the Multidomain Alzheimer Preventive Trial (MAPT Study), a randomized controlled trial assessing the effect of a multidomain intervention, the administration of polyunsaturated fatty acids (PUFA), both, or placebo on cognitive decline. We compared cognitive decline during the 5-year follow-up between three groups according to ULT (i.e. allopurinol and febuxostat) use: participants treated with ULT during at least 75% of the study period (PT ≥ 75; n = 51), less than 75% (PT < 75; n = 31), and non-treated participants (PNT; n = 1591). Cognitive function (measured by a composite score) was assessed at baseline, 6 months and every year for 5 years. Linear mixed models were performed and results were adjusted for age, sex, body mass index (BMI), diagnosis of arterial hypertension or diabetes, baseline composite cognitive score, and MAPT intervention groups. After the 5-year follow-up, only non-treated participants presented a significant decline in the cognitive composite score (mean change − 0.173, 95%CI − 0.212 to − 0.135; p < 0.0001). However, there were no differences in change of the composite cognitive score between groups (adjusted between-group difference for PT ≥ 75 vs. PNT: 0.144, 95%CI − 0.075 to 0.363, p = 0.196; PT < 75 vs. PNT: 0.103, 95%CI − 0.148 to 0.353, p = 0.421). Use of ULT was not associated with reduced cognitive decline over a 5-year follow-up among community-dwelling older adults at risk of dementia.
Similar content being viewed by others
Introduction
Cognitive decline is a rising issue as the population of older adults keeps increasing. Inflammaging, defined as a chronic, sterile and low-grade inflammation1, can lead to age-related diseases. Neuroinflammation involving oxidative stress has been included in the inflammaging process and is reported to play a major role2 on cognitive impairment.
Though the hypothesis has been raised that hyperuricemia or gout may be protective against dementia, Alzheimer’s disease (AD) and cognitive decline3,4, results were mainly issued from cross-sectional studies and are somewhat conflicting5. It was also reported in a large cohort that gout was independently associated with a 15% higher risk of incident dementia among older adults6.
Publications have reported that urate-lowering therapies (ULT) may decrease the systemic inflammation and reduce the production of oxidative species7,8. ULT may also reverse endothelial dysfunction and thus have cardioprotective benefits. Neuroprotective effects of ULT both in animal studies9 and human cohorts10 have been reported. Singh et al.11 reported a dose-related reduction in the risk of dementia among people older than 65 years treated with ULT.
These studies remain scarce and controversial. Some of their limitations were the lack of evaluation criteria of cognitive decline12,13,14,15 and short-term follow-ups. Furthermore, analyses were based on general data of the health administrative database. In addition, participants investigated in these studies were not at higher risk of cognitive decline or AD, thus limiting the possibility of studying the link between cognitive decline and ULT.
The aim of the present study was to investigate the association between long-term ULT and cognitive decline in a sample of community-dwelling older adults with subjective memory complaints over a five-year follow-up.
Materials and methods
Study population
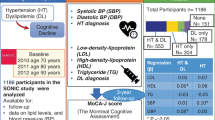
To evaluate a potential association between ULT and cognitive decline, we used data from the Multidomain Alzheimer Preventive Trial (MAPT) Study. The design of MAPT has been previously described in details elsewhere16. Briefly, the MAPT study is a randomized clinical trial performed in 13 centers in France and Monaco assessing cognitive outcomes in patients at risk of cognitive decline, including subjects older than 70 years with the following criteria: spontaneous memory complaint, limitations in one Instrumental Activity of Daily Living (IADL) or slow gait speed (≤ 0.8 m/s). Participants were assigned to different interventions for 3 years, and then followed for additional 2 years. Interventions comprised supplementation with omega 3 polyunsaturated fatty acids, multidomain intervention (covering cognitive training, physical activity and nutrition counselling), or both. These three groups were compared to a placebo group. Follow-up visits were scheduled at 6 and 12 months and then every year to assess outcomes, diseases and corresponding treatments. Participants were invited to come with all their treatment orders.
In summary, no statistically significant difference has been noted in the evolution of the main primary outcome (a composite cognitive score) between the four groups at 3 years, after controlling for multiple comparisons17.
From the total of 1,679 participants originally analyzed in the MAPT Study, those with information on ULT were included in the present study (n = 1673).
ULT data collection
The use of ULT (Anatomic Therapeutic Chemical code: M04AA01 for allopurinol or M04AA03 for febuxostat) were recorded at each visit. Following the 2016 EULAR recommendations for the management of gout18, allopurinol, a xanthine-oxidase (XO) inhibitor19, is the first line therapy for chronic gout and febuxostat (which is also a XO-inhibitor) should be used in second intention. Patients treated (PT) by uricosuric or uricolytic drugs or treated with colchicine were not included in the treated groups, given the different mechanism of action of these drugs20 (uricosuric drugs including benzbromarone, sulfinpyrazone, and probenecid block renal tubular urate reabsorption21 and are second-line therapies for gout).
Three different groups were defined according to the use of urate-lowering medication, as follows: participants treated with ULT during at least 75% (PT ≥ 75) of the study 5-year period (n = 51), participants treated with ULT for less than 75% (PT < 75) of the study (n = 31), and participants never treated (PNT) with ULT during the study (n = 1.591). Among those treated with ULT, 14 subjects had no treatment start date; we then considered that these subjects were treated since baseline or earlier.
Outcome measure
Cognitive function was assessed by a composite cognitive score combining the mean Z-score of four tests exploring: (i) episodic memory (Free and Cued Selective Reminding test: sum of the free and total (free + cued) recalls), (ii) orientation (10 orientation items from the Mini Mental State Examination—MMSE test), and (iii) executive function (WAIS Digit Symbol Substitution Test, and Category Fluency Test).
Cognitive assessments were performed at inclusion, every six months during the first year and every year until the fifth year of follow-up.
Confounding variables
To take into account potential confounding factors, the following variables were considered: age, sex, body mass index (BMI = weight (kg)/height (m)2), diagnosis of arterial hypertension or diabetes, physical activity (minutes per week) and allocation to MAPT intervention groups. History of smoking and alcohol intake was not included in the analysis because data were available for less than half of the participants22.
Statistical analysis
To describe the population, means and standard deviation for quantitative variables, and frequencies and percentage for qualitative variables were used. To compare the characteristics at baseline between the three urate-lowering treatment groups (PT ≥ 75, PT < 75 and PNT), the Chi-square test or Fisher's exact tests (in the case of expected frequencies < 5) were used for the qualitative variables, and Fisher test or the non-parametric Kruskal–Wallis test were used for quantitative variables.
To study the change of the composite cognitive score over time according to the ULT groups, mixed linear models were performed to take into account the correlated structure of the data (intra-center and intra-subject correlation, with subjects nested into the center). We included as random effects a random center intercept, a random subject intercept, and a random subject slope.
This model was performed without adjustment with the following fixed effects: time, ULT groups and time × group interaction. An adjusted model was then performed by adding the above-mentioned confounding variables as fixed effects. The statistically significant difference at baseline in the composite cognitive score according to the treatment group was also considered in the mixed model. Time was treated as a continuous variable, with a cubic trajectory since the terms time2 and time3 were significant.
All analyses were performed using SAS software version 9.4 (SAS Institute Inc., Cary, NC), and results were considered significant if p < 0.05.
Ethical approval and consent to participate
All participants were recruited by the investigating physicians, after obtaining their written informed consent. The trial protocol was approved by the French Ethical Committee located in Toulouse, France (CPP SOOM II) and was authorized by the French Health Authority in 2007. All methods were performed in accordance with the relevant guidelines and regulations.
Consent for publication
The publication was approved by the MAPT/DSA group.
Results
Among the 1673 participants of the study, 1591 were not treated with ULT (PNT, 95%), whereas 82 were treated with ULT, with the following distribution: 31 in the group PT < 75 (2%) and 51 in the group PT ≥ 75 (3%). Baseline characteristics of the groups are reported in Table 1. In summary, both PT < 75 and PT ≥ 75 groups presented higher BMI, were older, more often hypertensive and more frequently male than the PNT group. Diabetes was significantly more prevalent in the PT ≥ 75 group compared to the other groups. The composite cognitive score at baseline were statistically different among the groups (0.00, SD = 0.67 for PNT; 0.30, SD = 0.56 for PT < 75; − 0.12, SD = 0.76 for PT ≥ 75; p = 0.019). Other parameters such as the inclusion criteria, allocation to MAPT intervention group, level of education, physical activity or the presence of the apolipoprotein E4 (APOE4) mutation, were not differently distributed between the groups.
After the 5-year follow-up, only the PNT group presented a significant decline in the composite cognitive score (mean change − 0.173, 95%CI − 0.212 to − 0.135, p < 0.0001) (Fig. 1 and Table 2).
Evolution of the cognitive composite score (Z-Score) in the groups with participants not treated (PNT) with urate-lowering therapies (ULT), participants treated with ULT less than 75% of the study follow-up (PT < 75) and participants treated with ULT more than 75% of the study follow-up (PT ≥ 75) during a 5-year follow-up, among the participants of the MAPT study. PNT: patients not treated with urate-lowering therapies. PT < 75 : participants treated with urate-lowering therapies less than 75% of the study follow-up. PT ≥ 75 : participants treated with urate-lowering therapies more than 75% of the study follow-up.
However, no differences were observed when comparing evolution of the composite cognitive score between the treated groups and the PNT group (PT ≥ 75 vs. PNT: 0.085, 95%CI − 0.130 to 0.300, p = 0.437; PT < 75 vs. PNT: 0.018, 95%CI − 0.235 to 0.271, p = 0.889). Results remained similar in the adjusted models (PT ≥ 75 vs. PNT: 0.144, 95%CI − 0.075 to 0.363, p = 0.196; PT < 75 vs. PNT: 0.103, 95%CI − 0.148 to 0.353, p = 0.421) (Table 2).
Discussion
Our study evaluated the evolution of a composite cognitive score in a long follow-up of 5 years between participants treated with ULT and participants not treated with ULT, in a sample of community-dwelling older adults at risk of cognitive decline. To our knowledge, this is the first study assessing the long-term association of ULT with cognitive function in a population at risk of cognitive decline. No statistically significant differences in the evolution of a composite cognitive score was observed between the participants treated with ULT (whether they were treated during a long period of time or not) and the participants not treated with ULT.
Hyperuricemia is associated with several co-morbidities such as chronic kidney disease23, cardiovascular heart disease, arterial hypertension24, obesity and diabetes25. An important factor that may link hyperuricemia with these diseases is its deleterious effect on small vessels26. Despite having extracellular antioxidant properties, the urate acid induces endothelial dysfunction inside the cell27, and lowering uric acid concentrations has been reported to reduce cardiovascular events in patients with chronic kidney disease28. Our research are in line with a recent large systematic review and meta-analysis involving 16,000 participants, that found no significant association between serum urate levels and cognition29.
Both allopurinol and febuxostat can cross the blood–brain barrier (with a probability of 99% and 79%, respectively30, calculated with the chemical absorption, distribution, metabolism, excretion, and toxicity—ADMET features of these drugs31) and can directly interfere with intracerebral metabolism and neuronal cell32. Allopurinol was reported to reduce oxidative stress and proinflammatory molecules in the vessels33, thus reducing vascular damages in the brain. This neuroprotective effect has been reported to be related to an inhibition of the nitrosative stress and an attenuation of microglia infiltration and astrocytes reactivation in a mouse model of cortical microinfarction9. The potential benefits of the neuroprotective effect of allopurinol have been currently investigated in a phase III clinical trial in the context of hypoxic-ischemic brain injury in neonatology10. Cerebral microinfarcts are very frequent in patients with mild cognitive decline34, vascular dementia and AD35. Our research, however, did not show that ULT was associated with a protection of cognitive function (i.e., slower cognitive decline).
Our study has several strengths. Our selected population reported subjective memory complaints, a well-known condition associated with higher risk of cognitive decline36. Another strength of our study is the use of a composite cognitive score based on multiple cognitive tests to evaluate cognitive functions, which has been repeatedly validated37. Moreover, the measures of cognitive function have been repeated at several time-points, helping us follow the different cognitive trajectories more accurately. On the other hand, some limitations should be addressed. First, this is a post-hoc observational study using data from a RCT that was not designed to test the effects of ULT on cognition. Second, the small number of subjects in the PT < 75 and PT ≥ 75 groups may have limited the power of the statistical analysis. Third, serum uric acid levels were not available, what impeded us of investigating their interaction with ULT. Fourth, dose–response associations could not be explored since ULT dosing was not available. Finally, although based in our clinical experience, the 75% cut-off for ULT taking was arbitrary; other cut-offs should be tested in future works.
It would be interesting to wonder what practical implications should be drawn if our study had shown significant results and ULT be considered a treatment to slowing cognitive decline. ULT require few clinical and biological controls, thus their use is considered easy to handle38, not to mention their advantageous cost-effectiveness39. Moreover, uricemia often stays above the recommended levels in gouty patients treated with ULT40. Pursuing a dose-escalation of this therapy could lead to dwindle oxidative stress, as Singh et al. pointed out a dose-related effect of ULT on the onset of dementia in their study11. Eventually, as dementia is a slow-developing process, longer durations of ULT administration (as seen in chronic gout treatment) should be necessary to observe effective changes in cognitive functions.
In conclusion, this study did not find significant associations between ULT and changes in cognitive function over time in a population of older adults at risk of cognitive decline. Given the small fraction of people under ULT, larger observational prospective studies are needed to examine the associations between ULT and cognition over time. Randomized controlled trials investigating the effects of ULT on the cognitive function of patients might shed light on this topic.
Data availability
Pr Yves Rolland and Pr Philipe Barreto, CERPOP, UMR1295, unité mixte INSERM—Université Toulouse III Paul Sabatier have the full access to the MAPT database. The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
Abbreviations
- AD:
-
Alzheimer’s Disease
- ADMET:
-
Absorption, Distribution, Metabolism, Excretion, and Toxicity
- APOE4:
-
Apolipoprotein E4
- BMI:
-
Body Mass Index
- IADL:
-
Instrumental Activity of Daily Living
- MAPT:
-
Multidomain Alzheimer Preventive Trial
- MMSE:
-
Mini Mental Status Evaluation
- PNT:
-
Participants Not Treated
- PT < 75:
-
Participants Treated with ULT for less than 75% of the study
- PT ≥ 75:
-
Participants Treated with ULT during at least 75% of the study
- SD:
-
Standard Deviation
- ULT:
-
Urate-Lowering Therapies
References
Franceschi, C., Garagnani, P., Parini, P., Giuliani, C. & Santoro, A. Inflammaging: A new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 14, 576–590 (2018).
Butterfield, D. A. & Halliwell, B. Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat. Rev. Neurosci. 20, 148–160 (2019).
Tuven, B., Soysal, P., Unutmaz, G., Kaya, D. & Isik, A. T. Uric acid may be protective against cognitive impairment in older adults, but only in those without cardiovascular risk factors. Exp. Gerontol. 89, 15–19 (2017).
Lu, N. et al. Gout and the risk of Alzheimer’s disease: a population-based, BMI-matched cohort study. Ann. Rheum. Dis. 75, 547–551 (2016).
Latourte, A., Bardin, T. & Richette, P. Uric acid and cognitive decline: A double-edge sword?. Curr. Opin. Rheumatol. 30, 183–187 (2018).
Singh, J. A. & Cleveland, J. D. Gout and dementia in the elderly: a cohort study of Medicare claims. BMC Geriatr 18, (2018).
Moorhouse, P. C., Grootveld, M., Halliwell, B., Quinlan, J. G. & Gutteridge, J. M. Allopurinol and oxypurinol are hydroxyl radical scavengers. FEBS Lett 213, 23–28 (1987).
Fukui, T. et al. Effects of Febuxostat on Oxidative Stress. Clin. Ther. 37, 1396–1401 (2015).
Zhang, Q. et al. Allopurinol protects against ischemic insults in a mouse model of cortical microinfarction. Brain Res. 1622, 361–367 (2015).
Maiwald, C. A. et al. Effect of allopurinol in addition to hypothermia treatment in neonates for hypoxic-ischemic brain injury on neurocognitive outcome (ALBINO): Study protocol of a blinded randomized placebo-controlled parallel group multicenter trial for superiority (phase III). BMC Pediatr. 19, 210 (2019).
Singh, J. A. & Cleveland, J. D. Comparative effectiveness of allopurinol versus febuxostat for preventing incident dementia in older adults: A propensity-matched analysis. Arthritis Res. Ther. 20, 167 (2018).
Latourte, A. et al. Uric acid and incident dementia over 12 years of follow-up: A population-based cohort study. Ann. Rheum. Dis. 77, 328–335 (2018).
Scheepers, L. E. J. M. et al. Urate and risk of Alzheimer’s disease and vascular dementia: A population-based study. Alzheimers Dement 15, 754–763 (2019).
Engel, B. et al. Hyperuricemia and dementia—a case-control study. BMC Neurol 18, (2018).
Erkinjuntti, T., Ostbye, T., Steenhuis, R. & Hachinski, V. The effect of different diagnostic criteria on the prevalence of dementia. N. Engl. J. Med. 337, 1667–1674 (1997).
Vellas, B. et al. Mapt study: A multidomain approach for preventing alzheimer’s disease: Design and baseline data. J. Prev. Alzheimer’s Disease 1, 13–22 (2014).
Andrieu, S. et al. Effect of long-term omega 3 polyunsaturated fatty acid supplementation with or without multidomain intervention on cognitive function in elderly adults with memory complaints (MAPT): a randomised, placebo-controlled trial. Lancet Neurol 16, 377–389 (2017).
Richette, P. et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann. Rheum. Dis. 76, 29–42 (2017).
Pacher, P., Nivorozhkin, A. & Szabó, C. Therapeutic Effects of Xanthine Oxidase Inhibitors: Renaissance half a century after the discovery of Allopurinol. Pharmacol Rev. 58, 87–114 (2006).
Slobodnick, A., Shah, B., Krasnokutsky, S. & Pillinger, M. H. Update on colchicine, 2017. Rheumatology (Oxford) 57, i4–i11 (2018).
Kippen, I., Whitehouse, M. W. & Klinenberg, J. R. Pharmacology of uricosuric drugs. Ann. Rheum Dis. 33, 391–396 (1974).
Raffin, J. et al. Associations between physical activity, blood-based biomarkers of neurodegeneration, and cognition in healthy older adults: the MAPT study. J. Gerontol A Biol. Sci. Med. Sci. 76, 1382–1390 (2021).
Johnson, R. J. et al. Uric acid and chronic kidney disease: Which is chasing which?. Nephrol Dial Transplant 28, 2221–2228 (2013).
Richette, P. et al. Improving cardiovascular and renal outcomes in gout: What should we target?. Nat. Rev. Rheumatol 10, 654–661 (2014).
Sluijs, I. et al. A Mendelian randomization study of circulating uric acid and type 2 diabetes. Diabetes 64, 3028–3036 (2015).
Masi, S. et al. The complex relationship between serum uric acid, endothelial function and small vessel remodeling in Humans. J. Clin. Med. 9, E2027 (2020).
Khosla, U. M. et al. Hyperuricemia induces endothelial dysfunction. Kidney Int. 67, 1739–1742 (2005).
Goicoechea, M. et al. Effect of allopurinol in chronic kidney disease progression and cardiovascular risk. Clin. J. Am. Soc. Nephrol 5, 1388–1393 (2010).
Khan, A. A., Quinn, T. J., Hewitt, J., Fan, Y. & Dawson, J. Serum uric acid level and association with cognitive impairment and dementia: systematic review and meta-analysis. Age (Dordr.) 38, 16 (2016).
Wishart, D. S. et al. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 46, D1074–D1082 (2018).
Lagorce, D., Douguet, D., Miteva, M. A. & Villoutreix, B. O. Computational analysis of calculated physicochemical and ADMET properties of protein-protein interaction inhibitors. Sci. Rep. 7, (2017).
Scherrmann, J. M. Drug delivery to brain via the blood-brain barrier. Vascul. Pharmacol. 38, 349–354 (2002).
Muir, S. W. et al. Allopurinol use yields potentially beneficial effects on inflammatory indices in those with recent ischemic stroke: A randomized, double-blind, placebo-controlled trial. Stroke 39, 3303–3307 (2008).
Sonnen, J. A. et al. Pathological correlates of dementia in a longitudinal, population-based sample of aging. Ann. Neurol 62, 406–413 (2007).
Haglund, M., Passant, U., Sjöbeck, M., Ghebremedhin, E. & Englund, E. Cerebral amyloid angiopathy and cortical microinfarcts as putative substrates of vascular dementia. Int. J. Geriatr. Psychiatry 21, 681–687 (2006).
Gifford, K. A. et al. The source of cognitive complaints predicts diagnostic conversion differentially among nondemented older adults. Alzheimers Dement 10, 319–327 (2014).
Donohue, M. C. et al. The preclinical Alzheimer cognitive composite: Measuring amyloid-related decline. JAMA Neurol 71, 961–970 (2014).
Stamp, L. K. et al. Using allopurinol above the dose based on creatinine clearance is effective and safe in patients with chronic gout, including those with renal impairment. Arthritis Rheum 63, 412–421 (2011).
Jutkowitz, E., Choi, H. K., Pizzi, L. T. & Kuntz, K. M. Cost-effectiveness of allopurinol and febuxostat for the management of gout. Ann. Intern. Med. 161, 617–626 (2014).
Jeyaruban, A., Larkins, S. & Soden, M. Management of gout in general practice–a systematic review. Clin. Rheumatol 34, 9–16 (2015).
Funding
The MAPT study was supported by grants from the French Ministry of Health (PHRC 2008, 2009), University Hospital Center of Toulouse/Gérontopôle, Pierre Fabre Research Institute (manufacturer of the omega-3 supplement), ExonHit Therapeutics (biological sample collection) and Avid Radiopharmaceuticals (PET-amyloid measurement). The promotion of this study was supported by the University Hospital Center of Toulouse. The data sharing activity was supported by the Association Monegasque pour la Recherche sur la maladie d’Alzheimer (AMPA) and the UMR 1027 Unit INSERM-University of Toulouse III. The present work was performed in the context of the Inspire Program, a research platform supported by grants from the Region Occitanie/Pyrénées-Méditerranée (Reference number: 1901175) and the European Regional Development Fund (ERDF) (Project number: MP0022856) and received funds from Alzheimer Prevention in Occitania and Catalonia (APOC Chair of Excellence—Inspire Program).
Author information
Authors and Affiliations
Consortia
Contributions
L.M.B. designed and conceptualized the research, interpreted the data and drafted the manuscript. K.V.G. and P.S.B. interpreted the data and revised the draft critically for intellectual content. C.C. performed statistical analyses, interpreted the data and revised the draft critically for intellectual content. Y.R. designed and conceptualized the research, interpreted the data and revised the draft critically for intellectual content. All authors have read and approved the final manuscript submitted for publication.
Corresponding author
Ethics declarations
Competing interests
JANSSEN-CILAG (Luc Molet-Benhamou). The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Molet-Benhamou, L., Giudici, K.V., de Souto Barreto, P. et al. Association between urate-lowering therapies and cognitive decline in community-dwelling older adults. Sci Rep 12, 15299 (2022). https://doi.org/10.1038/s41598-022-17808-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-17808-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.