Abstract
Magnetic resonance imaging/Ultrasound (MRI/US) fusion targeted biopsy (TB) in combination with a systematic biopsy (SB) improves cancer detection but limited data is available how to manage patients with a Prostate Imaging-Reporting and Data System (PI-RADS) ≥ 4 lesion and a negative biopsy. We evaluate the real-world management and the rate of clinically significant Prostate Cancer (csPCa) during follow-up. 1546 patients with a multi-parametric MRI (mpMRI) and a PI-RADS ≥ 3 who underwent SB and TB between January 2012 and May 2017 were retrospectively analyzed. 222 men with a PI-RADS ≥ 4 and a negative biopsy were included until 2019. For 177/222 (80%) complete follow-up data was obtained. 66/84 (78%) had an initial PI-RADS 4 and 18 (22%) a PI-RADS 5 lesion. 48% (84/177) received a repeat mpMRI; in the follow-up mpMRI, 39/84 (46%) lesions were downgraded to PI-RADS 2 and 11 (13%) to PI-RADS 3; three cases were upgraded and 28 lesions remained consistent. 18% (32/177) men underwent repeated TB and csPCa was detected in 44% (14/32). Our study presents real world data on the management of men with a negative TB biopsy. Men with a positive mpMRI and lesions with high suspicion (PI-RADS4/5) and a negative targeted biopsy should be critically reviewed and considered for repeat biopsy or strict surveillance. The optimal clinical risk assessment remains to be further evaluated.
Similar content being viewed by others
Introduction
The multi-parametric magnetic resonance imaging (mpMRI) diagnostic pathway followed by a MRI/Ultrasound (MRI/US) fusion guided targeted biospsy (TB) is becoming the new standard for men at risk of prostate cancer (PCa). The strong data from studies focusing on a primary mpMRI have led to the updated recommendation by EAU and AUA guidelines towards an indication of a mpMRI for both the primary and repeat biopsy setting1,2,3.
The combination of a TB with a SB leads to improved cancer detection. Nevertheless, about 30% of TB remain negative and the management of this subgroup currently is unclear4,5,6,7. Guidelines recommendations on this subgroup of men are currently lacking and decisions are often made individually1.
A majority of systematic biopsies in biopsy- naïve men remain negative and clinically significant (cs) PCa might be overseen8,9,10. A pre-biopsy mpMRI warrants filtering biopsy-naïve men under suspicion of PCa as a first triage test but if suspicion persists despite a negative MRI adding PSA density (PSAD) as an additional factor almost excludes the detection of csPCa if it is less than 0.15 ng/ml11,12,13.
A recent Cochrane analysis by Drost et al. further confirmed the superiority of the mpMRI pathway14. The mpMRI is more sensitive for clinically significant cancer and can avoid a biopsy in 27% of men, therefore reducing the rate of overdiagnosis of clinically insignificant PCa by 31%11. Nonetheless, a variability of mpMRI quality and published detection rates for each Prostate Imaging-Reporting and Data System (PI-RADS) score persists, hence leading to a range of PCa detection rates15. Despite the improved mpMRI pathway and a suspicious lesion on mpMRI, approximately 33–52% of men will have negative histopathology12,16.
This retrospective study evaluated the management of men with PI-RADS 4 and 5 lesions after a negative MRI/US TB in a real-world setting of a consecutive cohort.
Patients and methods
Study population
Out of a prospective database of 1546 men who underwent transrectal MRI/US TB and SB due to suspicion of prostate cancer at Charité-Universitätsmedizin Berlin between January 2012 and May 2017, we retrospectively analyzed men with a negative biopsy of PI-RADS Score ≥ 4 lesions. Follow-up data were extracted from patient files where available and by a patient questionnaire. The questions included inter alia post-biopsy patient guidance and the indication for a repeat mpMRI or biopsy. The indication was made by the treating outpatient urologist as it is the standard of care in Germany.
Patient meta-data was collected in accordance with the standards of reporting for MRI-TB studies (START) checklist in a prospective database17. Subgroups of this cohort were included in previous analyses17. All patients signed a written informed consent for the intervention, data acquisition, and data appraisal. The study was performed according to the Declaration of Helsinki and authorized by the Institutional Review Board of the Charité-Universitätsmedizin Berlin.
Multiparametric imaging control
All patients received a 3-T mpMRI (Magnetom Skyra; Siemens Medical Systems, Erlangen, Germany) at Charité-Universitätsmedizin Berlin. The MRI protocol always comprised multi- planar (axial and coronal) high spatial resolution T2-weighted turbo spin-echo sequences (T2w TSE), axial T1-weighted images, axial diffusion-weighted images (DWIs; measured b-values 0.400 and 800 s/mm2, calculated b-value of 1 400 s/mm2) and gadolinium-based dynamic contrast-enhanced (DCE) sequences. T2w imaging and DWIs were performed in all patients and DCE MRI in most patients. In compliance with the guidelines of the European Society of Urogenital Radiology (ESUR) the evaluation and validation of the mpMRI image data were performed or supervised by a team of experienced expert radiologists at Charité-Universitätsmedizin Berlin using PI-RADS version 2 (v2). Lesions initially rated using PI-RADS V1 were re-rated using V2 for the analysis. Experienced radiologists were defined by the consensus statement with a minimum number read of 1000 mpMRIs and a yearly read over 20018. Considering clinical routine, radiologists were not blinded to clinical data. For patients with multiple lesions, the maximal PI-RADS score was used for further analysis.
MRI/US fusion-guided TB and SB
In accordance with the EAU guidelines at the time of the biopsy, the transrectal interventions were performed under antibiotic prophylaxis with a fluoroquinolone (ciprofloxacin) by a team of experienced urologists. MRI/US fusion-guided TB of the prostate was performed first, using the high-end US machine HiVison Preirus (Hitachi Medical Systems, Tokyo, Japan) or Aplio 500 (Toshiba, Otawara, Japan) with endocavity endfire probes (11C3, Toshiba; EUP V53 W; Hitachi Medical Systems) or a biplane probe transrectal (EUP CC531, Hitachi Medical Systems), as described previously6. Per PI-RADS lesion 3 targeted biopsies were obtained. Subsequently, transrectal SB was performed with 10 cores from left/right apex, left/right lateral midgland, left/right base, left/right ventral and left/right paraurethral.
All cores were potted and documented separately and were examined and analyzed by a certain team of experienced pathologists at Charité-Universitätsmedizin Berlin.
Definition of clinically significant prostate cancer
Clinically significant prostate cancer was defined as Gleason score ≥ 3 + 4 = 7 (International Society of Urological Pathology [ISUP] grade 2). Gleason scoring was based on the highest grade detected on histological analysis.
Statistical analyses
Continuous variables were described using medians and interquartile ranges (IQR), whereas categorical variables were characterized using proportions. All descriptive analyses were performed using the Statistical Package for the Social Sciences (SPSS) software version 25 for Mac OS (SPSS Inc., IBM Company, Chicago, IL, USA).
Research involving human participants
Approval was obtained from the ethics committee of Charité University Medicine Berlin. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Results
The definitive cohort included a total number of 222 men with a PI-RADS score of 4 or 5 and a negative TB in combination with a SB. Complete Follow-up data was available for 177/222 (80%) men with a median follow-up of 45 months (IQR 35–61) (Fig. 1 and 2).
The median patient age was 66 years (IQR 59–70), PSA level was 8 ng/ml (IQR 6–13), PSAD was 0.13 ng/ml2 (IQR 0.09–0.21) and the median prostate volume was 63 ml (IQR 47–86). 81/222 (36%) of men had one prior negative biopsy, 52 (23%) had two and 39 (18%) three or more previous negative biopsies (Table 1).
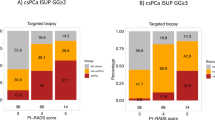
The distribution of the lesion as prior to the initial TB at was PI-RADS 4 in 182/222 (82%) and PI-RADS 5 in 40/222 (18%) patients and 145/177 (82%) PI-RADS 4 lesions and 32/177 (18%) PI-RADS 5 lesion for men with follow-up. A repeat mpMRI (after the initial negative TB) was performed in 84/177 (47%) men within a median time of 28 months (IQR 16–34) and led to a repeat TB in 24/177 (14%) cases. On follow-up 93/177 (53%) patients had no new mpMRI; 8 (5%) received a follow-up TB nonetheless due to a rising PSA and/ or a suspicion in the digital rectal examination (DRE) (Fig. 2).
The initial PI-RADS rating changed from 66/84 (78%) PI-RADS 4 and 18 (22%) PI-RADS 5 lesions to 39 (48%) PI-RADS 2 lesions, 11 (13%) PI-RADS 3 lesions, 19 (23%) PI-RADS 4 lesions and 12 (15%) PI-RADS 5 in the follow-up mpMRI (Fig. 3). Overall, a PI-RADS downgrading occurred in 50 (60%) cases and an upgrading in three (4%) cases. A median of 1 (IQR 1–2) lesion per patient was found in mpMRI. The median lesion size was 12 mm (IQR 9–20) (Table 1).
The indication for a follow-up mpMRI was justified by either a rising PSA value (median rise 3.2 ng/ml (IQR 3.9 (1.6–5.4)), a new suspicion in the DRE or a combination of both. Within the subgroup of men with repeated mpMRIs, there were 19/84 (23%) men with more than one additional mpMRI due to a change in the clinical parameter (rising PSA and/or new suspicion in DRE).
60/84 (71%) men received a new MRI but no additional biopsy because of a downgrading of the lesion or a constant finding either in MRI and/ or because of constant clinical parameters.
In total, 32/177 (18%) men had a follow-up transrectal TB within a median of 14 months (IQR 9–26) from the initial TB at our institution (Fig. 1). The PI-RADS scores were PI-RADS 4 in 11 cases and PI-RADS 5 in 10 cases (Table 2). The overall detection rate was 66% (21/32) and csPCA was detected in 14/32 (44%) men. This represents 12% (21/177) and 8% (14/177) of the whole cohort (Fig. 2). Please see Fig. 3 for a summary of the initial MRI, the follow-up and the related biopsy results.
The ISUP Group Grade distribution of the follow-up TB was as follows: 7/21 (33%) cases of ISUP 1, 3 (14%) cases of ISUP 2, 2 (10%) men of ISUP 3, 5 (24%) men of ISUP 4 and 4 (19%) with an ISUP 5. The correlation of the ISUP Grade Group and the PI-RADS ratings are likewise shown in Table 2. The chosen treatment is shown in Supplemental 1. After a radical prostatectomy the biopsy GS was upgraded in 15% and downgraded in 31% of the cases (Supplemental 2).
The underlying reasons for no further biopsies were available in 110/177 (62%) men (Supplemental 3). In 71% (78/110) no further biopsy was warranted due to stable clinical parameters such as PSA and DRE. 12% (13/110) of men had left urological care and 7% (8/110) patients did not agree to a new biopsy despite an indication. In 79% (85/110) of the patients, the decision for no further biopsy was made by the treating outpatient urologist and in 21% (23/110) men the decision against a biopsy was based on their own opinion.
Discussion
Despite the positive impact of the mpMRI on selected men at risk for csPCa not all men with a PI-RADS 4 or 5 lesion have cancer detected in prostate biopsy and their follow-up remains unclear. Currently, there is little data on the real-life management of men after a negative TB approach.
Kinnaird et al. support that a follow-up TB is necessary if suspicious lesions are seen on the follow-up MRI as 54% of the patients had csPCa with a PI-RADS 5 lesion in their cohort, which is in line with our findings19. Hence, mpMRI works as a triage-test in biopsy-naïve men and is further highly reliable in excluding significant disease in men with previous negative biopsy11,20. Panebianco et al. underline that as 1827 (75% of 2422 in total) patients with positive mpMRI after an initial negative were diagnosed with PCa and 1103 (46%) had csPCa, respectively21. At the same time, a MRI may indicate false-positive findings (e.g. PROMIS: positive predictive value 51%, specificity 41%), so that a biopsy may be unnecessary11,22.
TB surpasses the SB in terms of detection rate in ISUP Grade > 2 in the repeat-biopsy setting2. In our follow-up biopsy setting the cancer detection rate (CDR) in total was 66% (21/32) whereas in a primary setting the CDR for a targeted biopsy alone and for TB in combination with SB was 63% and 74% in our institution in a similar cohort, respectively6.
There is little data or guidance on how to manage these men especially if a PI-RADS ≥ 4 lesion was biopsy-negative. Ullrich et al. recommend a re-biopsy of a PI-RADS 4 lesion in the peripheral zone when there has been a negative biopsy before but suggest a follow-up for negatively biopsied transition zone lesions23. The updated German guidelines have added a recommendation that no further imaging or biopsy is indicated after a negative TB if clinical parameters are stable22.
Additionally, one needs to strongly consider a reevaluation of the MRI and technical issues of the TB itself may have led to false-negative biopsy results. With a broader use of the mpMRI, the known pitfalls such as reader inter-variability, imaging quality and missed TB may lead to an increase of men with negative TB and the need of standardized patient management24. As we found a high rate of downgrading (60% (50/84)), a “reference” radiology might give further guidance. One explanation may be prostatic inflammations which mimic cancer lesions on PI-RADS, which can disappear on further follow-up23,25,26.
Concordant to our findings Meng et al. established that 88/497 (18%) of patients with a PIRADS 4 or 5 lesion prior to initial biopsy had no cancer in TB27 due to a PI-RADS downgrading in 73% of men on repeat MRI27. The prospective study by Meng27 over 4 years included 497 men with a negative biopsy out of a cohort of 1595 men (overall 31% of men), whereas in our consecutive cohort only 14% (222/1546) of PI-RADS 4/5 cases presented with a negative biopsy result. This finding may be explained by our single center experience with a high primary detection rate6.
Persistent PI-RADS 4/5 lesions are at high risk of missed PCa, but false positive findings still exist as e.g. PCa in the transitional zone is still challenging23,27,28. Prostate deformation, patient movement, mismatch in imaging may be other reasons for failure in TB24.
With a follow-up of 80% and its prospective documentation this study needs further long-term data and potential data of the PRECSION trial will give further insight into risk assessment of patients with a negative biopsy, either SB or TB12. The limitations of our study are its retrospective nature and the lack of histological proof of absence of cancer in all men. A further limitation is, that the analysis presents the real-world follow up data and clinical decision were not based on a standard follow-up protocol and may therefore underestimate the absence on csPCa during follow-up.
Conclusion
Our study presents real world data on the management of men with a negative TB biopsy. Men with a positive mpMRI and lesions with high suspicion (PI-RADS4/5) and a negative targeted biopsy should be critically reviewed and considered for repeat biopsy or strict surveillance. The optimal clinical risk assessment remains to be further evaluated.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Rosenkrantz, A. B. et al. prostate magnetic resonance imaging and magnetic resonance imaging targeted biopsy in patients with a prior negative biopsy: A consensus statement by AUA and SAR. J. Urol. 196, 1613–1618. https://doi.org/10.1016/j.juro.2016.06.079 (2016).
Mottet, N. et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: Screening, diagnosis, and local treatment with curative intent. Eur. Urol. 71, 618–629. https://doi.org/10.1016/j.eururo.2016.08.003 (2017).
Bjurlin, M. A. et al. Update of the AUA policy statement on the use of multiparametric magnetic resonance imaging in the diagnosis, staging and management of prostate cancer. J. Urol. https://doi.org/10.1097/JU.0000000000000617 (2019).
Borkowetz, A. et al. Evaluation of prostate imaging reporting and data system classification in the prediction of tumor aggressiveness in targeted magnetic resonance imaging/ultrasound-fusion biopsy. Urol. Int. 99, 177–185. https://doi.org/10.1159/000477263 (2017).
Cash, H. et al. The detection of significant prostate cancer is correlated with the prostate imaging reporting and data system (PI-RADS) in MRI/transrectal ultrasound fusion biopsy. World J. Urol. 34, 525–532. https://doi.org/10.1007/s00345-015-1671-8 (2016).
Hofbauer, S. L. et al. Validation of prostate imaging reporting and data system version 2 for the detection of prostate cancer. J. Urol. 200, 767–773. https://doi.org/10.1016/j.juro.2018.05.003 (2018).
Radtke, J. P. et al. Comparative analysis of transperineal template saturation prostate biopsy versus magnetic resonance imaging targeted biopsy with magnetic resonance imaging-ultrasound fusion guidance. J. Urol. 193, 87–94. https://doi.org/10.1016/j.juro.2014.07.098 (2015).
Gore, J. L. et al. Optimal combinations of systematic sextant and laterally directed biopsies for the detection of prostate cancer. J. Urol. 165, 1554–1559 (2001).
Shinohara, K., Nguyen, H. & Masic, S. Management of an increasing prostate-specific antigen level after negative prostate biopsy. Urol. Clin. North Am. 41, 327–338. https://doi.org/10.1016/j.ucl.2014.01.010 (2014).
Sayyid, R. K. et al. Population-based outcomes of men with a single negative prostate biopsy: Importance of continued follow-up among older patients. Urol. Oncol. 37, 298 e19-298 e27 (2019).
Ahmed, H. U. et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): A paired validating confirmatory study. Lancet 389, 815–822. https://doi.org/10.1016/S0140-6736(16)32401-1 (2017).
Kasivisvanathan, V. et al. MRI-targeted or standard biopsy for prostate-cancer diagnosis. N. Engl. J. Med. 378, 1767–1777. https://doi.org/10.1056/NEJMoa1801993 (2018).
Pagniez, M. A. et al. Predictive factors of missed clinically significant prostate cancers in men with negative magnetic resonance imaging: A systematic review and meta-analysis. J. Urol. 204, 24–32. https://doi.org/10.1097/JU.0000000000000757 (2020).
Drost, F. H. et al. Prostate magnetic resonance imaging, with or without magnetic resonance imaging-targeted biopsy, and systematic biopsy for detecting prostate cancer: A cochrane systematic review and meta-analysis. Eur. Urol. https://doi.org/10.1016/j.eururo.2019.06.023 (2019).
Mehralivand, S. et al. Prospective evaluation of PI-RADS version 2 using the international society of urological pathology prostate cancer grade group system. J. Urol. 198, 583–590. https://doi.org/10.1016/j.juro.2017.03.131 (2017).
Maxeiner, A. et al. Primary magnetic resonance imaging/ultrasonography fusion-guided biopsy of the prostate. BJU Int. 122, 211–218. https://doi.org/10.1111/bju.14212 (2018).
Moore, C. M. et al. Standards of reporting for MRI-targeted biopsy studies (START) of the prostate: Recommendations from an international working group. Eur. Urol. 64, 544–552. https://doi.org/10.1016/j.eururo.2013.03.030 (2013).
de Rooij, M. et al. ESUR/ESUI consensus statements on multi-parametric MRI for the detection of clinically significant prostate cancer: Quality requirements for image acquisition, interpretation and radiologists’ training. Eur. Radiol. 30, 5404–5416 (2020).
Kinnaird, A. et al. Risk of prostate cancer after a negative magnetic resonance imaging guided biopsy. J. Urol. https://doi.org/10.1097/JU.0000000000001232 (2020).
Schouten, M. G. et al. Why and where do we miss significant prostate cancer with multi-parametric magnetic resonance imaging followed by magnetic resonance-guided and transrectal ultrasound-guided biopsy in biopsy-naive men?. Eur. Urol. 71, 896–903. https://doi.org/10.1016/j.eururo.2016.12.006 (2017).
Panebianco, V. et al. Negative multiparametric magnetic resonance imaging for prostate cancer: What’s next?. Eur. Urol. 74, 48–54. https://doi.org/10.1016/j.eururo.2018.03.007 (2018).
Interdisziplinäre Leitlinie der Qualität S3 zur Früherkennung, Diagnose und Therapie der verschiedenen Stadien des Prostatakarzinoms, Langversion 5.1, 2019, AWMF Registernummer: 043/022OL. In: Leitlinienprogramm Onkologie (Deutsche Krebsgesellschaft, Deutsche Krebshilfe, AWMF). Available: http://www.leitlinienprogramm-onkologie.de/leitlinien/prostatakarzinom/
Ullrich, T. et al. Analysis of PI-RADS 4 cases: Management recommendations for negatively biopsied patients. Eur. J. Radiol. 113, 1–6. https://doi.org/10.1016/j.ejrad.2019.01.030 (2019).
Cash, H. et al. Prostate cancer detection on transrectal ultrasonography-guided random biopsy despite negative real-time magnetic resonance imaging/ultrasonography fusion-guided targeted biopsy: Reasons for targeted biopsy failure. BJU Int. 118, 35–43. https://doi.org/10.1111/bju.13327 (2016).
Pepe, P. & Pennisi, M. Negative biopsy histology in men with PI-RADS score 5 in daily clinical practice: Incidence of granulomatous prostatitis. Clin. Genitourin. Cancer. https://doi.org/10.1016/j.clgc.2020.04.001 (2020).
Twum-Ampofo, J., Ceraolo, C., Gusev, A. & Feldman, A. S. Detection of clinically significant prostate cancer after negative fusion and systematic biopsy. J. Clin. Oncol. 38, 288–288. https://doi.org/10.1200/JCO.2020.38.6_suppl.288 (2020).
Meng, X. et al. Follow-up of men with PI-RADS 4 or 5 abnormality on prostate MRI and nonmalignant pathologic findings on initial targeted prostate biopsy. J. Urol. https://doi.org/10.1097/JU.0000000000001424 (2020).
Ploussard, G. et al. Risk of repeat biopsy and prostate cancer detection after an initial extended negative biopsy: Longitudinal follow-up from a prospective trial. BJU Int. 111, 988–996. https://doi.org/10.1111/j.1464-410X.2012.11607.x (2013).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
H.C., K.K.; Designed the research study. M.R., A.M., K.G., B.K., K.K., M.R., SL.H., R.H.; Acquired the data. M.R., K.K., H.C.; Analyzed the data. K.K., M.R., H.C.; Wrote the paper. SL.H., LE.W., P.A., JJ.W., M.S., T.S., F.F., H.C.; Critically revised the manuscript. H.C.; Supervised the study.
Corresponding author
Ethics declarations
Competing interests
H. Cash reports receiving honoraria as a speaker and advisor for Exact Imaging. The other authors have no disclosure to declare.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kornienko, K., Reuter, M., Maxeiner, A. et al. Follow-up of men with a PI-RADS 4/5 lesion after negative MRI/Ultrasound fusion biopsy. Sci Rep 12, 13603 (2022). https://doi.org/10.1038/s41598-022-17260-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-17260-6
This article is cited by
-
PI-RADS v2.1 evaluation of prostate “nodule in nodule” variants: clinical, imaging, and pathological features
Insights into Imaging (2024)
-
Expect the unexpected: investigating discordant prostate MRI and biopsy results
European Radiology (2024)
-
Patient-related characteristics predict prostate cancers in men with PI-RADS 4–5 to further optimize the diagnostic performance of MRI
Abdominal Radiology (2023)
-
The impact of a second MRI and re-biopsy in patients with initial negative mpMRI-targeted and systematic biopsy for PIRADS ≥ 3 lesions
World Journal of Urology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.