Abstract
To evaluate overall survival and locoregional recurrence between patients with invasive breast tumours and sentinel node metastasis undergoing sentinel lymph node dissection (SLND) alone and those undergoing complete axillary lymph node dissection (ALND). In this retrospective cohort study, we reviewed the medical records of patients with invasive breast carcinoma who underwent lumpectomy at a public university hospital in Brazil between 2008 and 2018. We evaluated the overall survival and the locoregional recurrence using Kaplan–Meier and Cox regression analyses, respectively. Overall, 97 participants who underwent lumpectomy were enroled; 41 in the ALND group, and 56 in the SLND group, according to Z0011 criteria. Only 17% of the patients in the ALND group had an additional biopsy-proven axillary disease, and 83% were treated with complete dissection unnecessarily. The 5-year survival rates were 80.1% and 87.5% for SLND and ALND, respectively (p = 0.376). Locoregional recurrence was rare (1.7% and 7.3% in the SLND and ALND, respectively; p = 0.3075). Overall survival and locoregional recurrence were similar between the two groups. The de-escalation of ALND to SLND in women with metastasis in the sentinel lymph node treated with conservative surgery and radiotherapy that meet the Z0011 criteria is feasible even in developing countries.
Similar content being viewed by others
Introduction
Breast cancer is the most common type of cancer worldwide and is responsible for 15% of cancer-related deaths among women1. Sociodemographic index levels are significant determinants of breast cancer incidence and mortality. In low- and middle-income countries, the incidence rate of breast cancer is low2. However, breast cancer mortality remains high due to limitations in early diagnosis and treatment options3,4.
In recent decades, the standard axillary management of early-stage breast cancer has changed dramatically. Axillary lymph node dissection (ALND) has been replaced by sentinel lymph node dissection (SLND) for the treatment of clinically negative lymph node breast cancer5. The American College of Surgeons Oncology Group (ACOSOG) Z0011 was a milestone in the surgical treatment of the axilla in patients with early breast cancer, and it significantly contributed to reducing the extent of breast surgery6,7,8,9,10,11. Consequently, the omission of ALND resulted in reduced morbidity and improved quality of life12,13,14,15.
ACOSOG Z0011 was a randomized, and non-inferiority clinical trial on women with invasive breast tumours measuring up to 5 cm with clinically negative axilla and up to two metastatic sentinel lymph nodes (SLNs). Patients were treated with lumpectomy, breast radiotherapy, and systemic adjuvant therapy. This study showed that completion of ALND neither significantly improved overall survival or disease-free survival nor did it reduce locoregional recurrence in these patients15,16. The ACOSOG Z0011 data were updated with a 10-year follow-up, and the results confirmed the evidence, leading to fundamental changes in the surgical management of the axilla17,18.
Other studies have corroborated the results of the Z0011trial. NSABP B4 found no benefit with resection of positive occult lymph nodes at the time of surgery19. IBCSG 23-01 also evaluated patients with minimal lymph node involvement and showed that ALND could be avoided20. The AMAROS trial confirmed that axilla treatment (surgery or radiotherapy) in patients with metastatic axillary SLNs provides comparable axillary control with no significant differences in overall survival between treatment groups. Additionally, axillary radiotherapy can be used as an alternative to ALND in patients with metastasis in the SLN, which fulfils the ACOSOG Z0011 exclusion criteria21.
However, several shortcomings were identified in the Z0011 trial, such as unmet accrual goal in enrollment, absence of standard testing for human epidermal growth factor receptor 2 (HER2), doubts about the radiotherapy fields, and applicability to other patient populations22.
In Brazil, the conservative approach to the axilla in patients with positive SLN biopsies has been challenged because survival after breast cancer remains poor, as in other developing countries; this is probably due to diagnosis of the disease at more advanced stages23,24. In addition, there is resistance in some cancer centres to adhere to the new surgical approach. These facts motivated this study.
This study aimed to validate the applicability of the Z0011 trial approach by evaluating the overall survival and locoregional recurrence in patients with invasive breast cancer who underwent either SLND as indicated by biopsy or complete ALND.
Materials and methods
Study design, setting, and ethics
This was a retrospective cohort study based on medical records evaluating the survival in consecutive patients with primary invasive carcinoma of the breast who underwent conservative surgery at a public university hospital in Brazil (Hospital São Paulo, Universidade Federal de São Paulo, UNIFESP) between February 2008 and December 2018. We evaluated overall survival and locoregional recurrence in patients who underwent either SLND or ALND.
This project was approved by the institutional review board (Comitê de Ética em Pesquisa da Universidade Federal de São Paulo—UNIFESP) under number (1.727.717/2016). Informed consent was waived for this observational study, patients signed informed consents for each procedure individually and the study did not pose any additional risk or discomfort for patients. Anonymity was guaranteed. This study is reported according to the STROBE Statement (STrengthening the Reporting of Observational studies in Epidemiology) reporting guideline and is in accordance with the Declaration of Helsinki.
Participants, sources of data, and treatments received
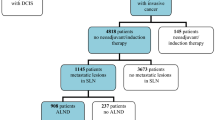
We reviewed an electronic database of the surgical ward records to identify patients (only women) who underwent surgery during the study period for a primary breast tumour measuring up to 5 cm. We excluded patients who underwent a mastectomy, complete axillary lymph node resection without a previous SLN biopsy, and those who received neoadjuvant therapy. We considered only women who underwent conservative breast surgery and SLN biopsy. Next, we excluded patients who had negative sentinel lymph node biopsy test findings. Therefore, only those who underwent conservative surgery (lumpectomy) and had positive sentinel lymph nodes were finally included. The axillae of these women were clinically negative (N0).
We divided the participants into two groups: the ALND group, which included patients who underwent complete ALND, and the SLND group, which included those who underwent dissection of only the axillary nodes indicated by the SLN biopsy. Figure 1 shows the treatment protocols for patients at our hospital before and after the publication of the Z0011 trial. All patients in the SLND group met the Z0011 criteria and ALND can be omitted. The Z0011 criteria included women with clinical T1-T2 invasive breast cancer, no palpable adenopathy, and 1 to 2 SLNs containing metastases, who underwent lumpectomy and tangential whole-breast irradiation15.
The only Z0011 trial criterion that was not considered at our hospital was the presence of extracapsular extension identified as focal or smaller than 2 mm. These patients were treated with SLND. Patients with Z0011 exclusion criteria were treated with ALND.
During the study period, ultrasonography was not routinely performed at our hospital. Sentinel lymph node biopsy was performed using patent blue dye, while some patients were treated using a combined technique with technetium-99m. Lymphoscintigraphy was not routinely performed because of the lack of resources at our hospital. Intraoperative assessment of SLNs could be performed during surgery at the surgeon's discretion.
Study endpoint, variables, and sample size calculation
We compared survival between the groups. The primary endpoint was overall survival, which was defined as the time between surgery and death for any cause. Secondary endpoints were locoregional recurrence, which was determined by the return of the disease either in the breast, ipsilateral lymph nodes in the axilla, or in the internal supraclavicular, infraclavicular, or thoracic (mammary) chains and survival after the recurrence of the disease, which was considered as the time between surgery and the onset of locoregional recurrence.
We also compared sociodemographic and clinical variables such as age group (younger than 50 years or aged 51 years or older), race, educational level, histological diagnosis and grade, angiolymphatic invasion, hormonal receptors, HER-2 positivity, Ki-67, tumor size, number of positive lymph nodes, and therapy between the groups.
The sample size was calculated to detect the non-inferiority of risks (hazard ratio) with a power of 85% and a significance level of 5%. We admitted a non-inferiority margin of 0.30 for the risks and a follow-up of 5 years, in which we assumed an approximately exponential distribution. A minimum sample size of 43 patients for each group (total, 86 patients) was determined. We used PASS 14 software (Power Analysis and Sample Size System, NCSS) for this calculation.
Statistical analysis
We analyzed the data descriptively. For categorical variables, we present absolute and relative frequencies and, for numerical variables, summary measures (mean, quartiles, minimum, maximum, and standard deviation).
We verified associations between two categorical variables using a Chi-square test or in cases of small samples with a Fisher's exact test. We compared two means using a student’s t-test for independent samples, with the assumption of normality verified using a Kolmogorov–Smirnov test.
The survival analyses in this study evaluated the time until the occurrence of death or recurrence, considering censorship (cases that did not experience the event during the analysis period). Initially, we analyzed survival functions separately for each predictor variable (univariate analysis). Kaplan–Meier models were used for categorical variables. We estimated survival functions for each level of these variables, and then compared them using a log-rank test (Mantel–Cox).
Additionally, we adjusted univariate Cox regression models for all the predictor variables considered, and a multivariate Cox model was adjusted for significant characteristics identified in the univariate models (backward method). A Cox's model assumes the existence of proportional risks, which was verified via a test based on Schoenfeld residuals.
Locoregional recurrence rates were compared with the Fisher’s exact test. All statistical tests were two-tailed, and a significance level of 5% was used. Statistical analyses were performed using the statistical software Statistical Package for the Social Sciences (SPSS) 20.0 and STATA 12.
Ethics approval and consent to participate
The Institutional Review Board of Universidade Federal de São Paulo, number 1.727.717/2016, approved this project. All the procedures of the study were in accordance with the ethical standards of the Helsinki declaration. Patients were informed about the study objectives and agreed to have their clinical data used in the research without any incentive, signing an informed consent form.
Results
Patient characteristics
During the study period, 1009 patients underwent surgery for breast cancer at our hospital. After applying the exclusion criteria, we identified 97 patients with positive lymph nodes who underwent conservative breast surgery. These patients were diagnosed with invasive breast carcinoma, with tumours of 2 cm or less (T1) or 2.1 cm to 5 cm (T2), all with clinically negative axilla (N0). There were 41 and 56 patients in the ALND and SLND groups, respectively (Fig. 2).
The mean age was similar between the groups (Table 1). The mean tumour size was 1.7 cm, and it was significantly larger in the ALND group compared with that in the SLND group (p = 0.048). The number of positive lymph nodes was significantly higher in the ALND group (median, 2) than in the SLND group (median, 1). Only one lymph node was positive in 91% of the cases. In the SLND group, all patients had one or two positive lymph nodes. In the ALND group, only 17% of patients had an additional axillary disease (as shown by biopsy), suggesting that the complete dissection performed in 83% of patients was unnecessary. The ALND group had a significantly higher number of total lymph nodes removed (median, 14) than the SLND group did (median, 2). The ALND group also had a significantly longer follow-up time than the SLND group did (median, 5.3 vs. 3.5 years).
There was no significant difference in race, educational level, histological diagnosis and grade, hormonal receptors, HER-2 positivity, tumour size (according to TNM pathological staging), and angiolymphatic invasion between the groups (Table 2). However, the ALND group had a significantly higher number of positive lymph nodes and macrometastases (p < 0.001 for both), as well as higher axillary involvement, as seen by the higher frequency of pN1, pN2, and pN3 cases (p < 0.001). In contrast, the SLND group had less extracapsular extension (p < 0.001). Adjuvant chemotherapy was more frequent in the ALND group (p = 0.024). Furthermore, the types of radiotherapy and hormonal therapy performed were similar between the groups.
Radiation oncologists at our institution used nomograms to predict the likelihood of metastasis in non-SLNs, and this aided in clinical decision-making. The most commonly used nomograms were from the Memorial Sloan-Kettering Cancer Center25 and MD Anderson Cancer Center26. Patients who had greater than 30% risk of additional lymph node involvement were treated with drainage radiotherapy27 although it is not recommended in the ACOSOG Z0011 protocol. We identified 12.5% and 4.9% (p = 0.12) cases that received drainage radiotherapy in the SLND alone and ALND groups, respectively.
There was a significant reduction in the intraoperative assessment of SLNs after the publication of the ACOSOG Z0011 clinical trial and its adoption as a guideline at our hospital. Of the 415 patients evaluated, 90.2% (46 of 51 patients operated) were subjected to the exam before publication and 30.8% (112 of 364) after. The rate of patients undergoing a second surgery as a result of the anatomopathological result was 3.8%, and the main indications for the second surgical procedure were the presence of three or more positive lymph nodes and gross extranodal disease.
Overall survival
The mean overall survival was 9.18 years (95% confidence interval [CI], 8.47–9.90). There was no difference in survival between the two groups; the 5-year overall survival was 80.1% and 87.5% in the SLND and ALND groups (p = 0.376), respectively (Fig. 3). Only hormonal therapy had a significant effect on survival; patients with positive hormone receptors status lived longer (see Supplementary Table S1). Patients with negative receptors for estrogen (ER) and positive receptor for progesterone (PR) had shorter survival, but only two patients were classified as such; therefore, they were not considered (Table 1). All patients positive for HER2 received targeted therapy, and there was no difference in survival between patients with positive and negative HER2 status. In the survival model, only hormonal therapy had a significant effect (p = 0.018, Table 3), and patients receiving this therapy had a 78% lower risk of death. As in the previous analysis, the two patients who were ER- and PR + were not considered.
The multivariate Cox regression model included the following as predictor variables in the initial model: the intervention group (regardless of their significance), hormonal therapy (significant at 5% in the Cox univariate model), tumour size, the number of positive lymph nodes, the number of resected lymph nodes, capsular extension, the size of axillary metastasis, and adjuvant chemotherapy (different characteristics by intervention group) (Table 4). In this analysis, there was no difference in survival between the groups (p = 0.536) (Table 4). However, the risk of death was 91% lower in patients undergoing hormonal therapy in the final model (p = 0.005) and 86% lower in those treated with adjuvant chemotherapy (p = 0.027). Schoenfeld residuals testing showed that the hazards were proportional in the initial model (p = 0.453) and at the end (p = 0.194), indicating the absence of violation of this assumption.
Locoregional recurrence
Locoregional recurrence was a rare event, with only four patients having disease recurrence: 7.3% of patients in the ALND group and 1.7% in the SLND group (p = 0.3075). Recurrence occurred in all cases within 18 months of follow-up. Survival after locoregional recurrence was 10.1 years (95%CI 9.62–10.40), and there was no statistically significant difference between the groups (p = 0.196) (see Supplementary Table S2). Figure 4 shows survival after locoregional recurrence per group.
Discussion
This study showed that completion of ALND did not improve overall survival or locoregional recurrence in patients with invasive breast cancer and sentinel node metastasis. The similar overall survival between the two groups provides evidence that ALND is unnecessary in patients with up to two metastatic SLNs treated with conservative surgery and radiotherapy. This finding suggests that even in countries such as Brazil, where the overall survival in patients with breast cancer is lower than that in patients in developed nations23, conservative surgical treatment of the axilla, in patients who meet Z0011 criteria, is possible. This result corroborates the data from the ACOSOG Z0011 trial15.
Patients undergoing SLND alone had a similar survival rate. However, in our study, patients in the SLND group received adjuvant chemotherapy less frequently (75% vs. 92.7% in SLND alone and ALND, respectively, p = 0.024, Table 1). Additionally, they were spared adjuvant chemotherapy because of comorbidities, and these data were not evaluated in the study.
The tendency towards reducing the use of intraoperative lymph node evaluation at our hospital following the publication of the ACOSOG Z0011 trial was similar to that in other studies6,7,28,29. Intraoperative assessment of the SLN can be associated with a shorter average time of surgery6, a reduction in perioperative costs30,31, and a significant increase in the proportion of patients in whom complete dissection can be avoided32. According to van der Noordaa et al., intraoperative assessments of the SLN should be performed only in patients with a restricted indication for lymph node dissection in the presence of metastasis in SLN biopsy33. Thus, the intraoperative assessment of the SLN is not necessary for patients who meet the ACOSOG Z0011 criteria, and the surgical re-approach resulting from the definitive anatomopathological result of the axilla is rare (3.8%). This is an important finding in our study that can promote the practice of avoiding the intraoperative assessment of SLNs.
Locoregional recurrence was rare, and the rate was similar between the groups. Furthermore, we believe that our follow-up duration was adequate and was suitable for measuring recurrence. Long-term follow-up data from the NSABP trial19 showed that recurrence usually occurred early, at 14.8 months on average. In the ACOSOG Z0011 trial18, recurrences occurred in 3.1 years, a bit shorter than our average follow-up time of 3.7 years.
The SLND group included postmenopausal women with small tumours (pT1), positive hormone receptors status, and small axillary involvement (35.7% with micrometastasis in the SLN biopsy). These characteristics were similar to those of the same arm in the Z0011 trial15,18. However, in the Z0011 study15, the arm undergoing complete axillary dissection also had a high prevalence of micrometastases (37.5%), in contrast to that noted in our study.
The ACOSOG Z0011 trial criteria can be considered for patients with HER2 overexpression, triple-negative tumours, and those aged below 50 years. Chung et al. reported no benefit in performing ALND in this subgroup34. In our study, the groups were homogeneous in terms of these three variables. The underrepresentation of this group in the ACOSOG Z0011 trial may be due to the local demographic characteristics of patients with breast carcinoma. Nevertheless, it was assumed that the distribution of HER2-positive tumours was balanced between the two arms of the trial.
Several studies around the world have identified increasing acceptance of the Z0011 results and a change in clinical practice in relation to the standard treatment of axillary lymph nodes in patients with breast cancer35,36,37,38.
A meta-analysis comparing SLND/radiotherapy only with ALND in early-stage breast cancer with limited sentinel node metastasis estimated that overall survival and disease-free survival were higher in the SLND group than in the ALND group, and a greater axillary recurrence was observed in the SLND/radiotherapy group. In conclusion, the omission of ALND in patients with one or two positive SLNs is indicated39.
Another meta-analysis of real-world cases evaluating the effects of SLND alone in patients with early-stage breast cancer and one or two positive SLN metastases in the post-Z011 era showed equivalent survival and recurrence outcomes between those undergoing SNLD alone and those undergoing ALND, which demonstrates that SLND alone was safe40. However, this shift in clinical practice should not occur in patients with residual lymph node disease following neoadjuvant chemotherapy41. All these studies included patients who were treated with systemic adjuvant therapy.
Complete ALND might be an overtreatment for many patients with capsular extravasation in the dissected SLNs. The Z0011 trial excluded patients with gross capsular extravasation and did not analyse the effect of microscopic capsular extravasation on recurrence or survival, making the management of these patients uncertain15,18. The extension of capsular extravasation is directly associated with the burden of axillary disease42. However, the rates of local, regional, or distant recurrence and mortality were similar between patients with and without capsular extravasation of ≤ 2 mm43, and regional recurrence was rare and was similar to that in patients without capsular extravasation even in the absence of nodal radiotherapy. Capsular extravasation is not the only reason for complete ALND44. In our study, we identified five patients with capsular extravasation of ≤ 2 mm who were treated with SLND alone, avoiding the morbidity associated with complete axillary resection. However, these patients received regional radiotherapy at our hospital.
We acknowledge that translating the Z0011 results into clinical practice is complicated due to inconsistent use of radiotherapy fields in their study. In a prospective study of 793 patients with SLN metastasis, using the ACOSOG Z0011 eligibility criteria resulted in the avoidance of ALND in 84% of patients, and the 5-year cumulative regional recurrence rate was 1%, which did not differ between radiotherapy fields. The authors concluded that even without the routine use of nodal radiotherapy, complete dissection could be avoided with excellent regional control45. Hopefully, we will have answers about the real influence of radiotherapy in regional control with the results of the ongoing trials46,47,48,49.
This was a retrospective study based on the medical records; thus, we were unable to evaluate costs and surgical times after the change in the clinical approach in our hospital after the publication of the ACOSOG Z0011 study. Studies that evaluated cost reduction associated with the elimination of complete axillary dissection30,31, did not consider the risk of surgical re-approach due to the presence of more than two SLNs with macrometastasis or capsular extravasation. The cost of a second surgery remains to be evaluated. Even the ACOSOG Z0011 trial did not report the rate of surgical re-approach in the group subjected to SLND alone. The rate of surgical re-approach in this study was very low and answered this question. In addition, this was the first study in our country to address the findings of Z0011 implementation, which was important to encourage conservative surgical treatment of the axilla in our country and other developing countries, with the aim of disseminating this practice and benefiting patients.
The preliminary internal evaluation of the results of this study prompted major changes in our hospital’s clinical approach, with more conservative surgeries being performed and the elimination of ultrasonography, the findings of which would often cause patients to undergo radical lymphadenectomy in the absence of SLN biopsy results in the past.
Conclusions
The overall survival and locoregional recurrence in patients with metastatic axillary SLNs treated with SLND were similar to those in patients treated with complete ALND. The elimination of routine axillary lymphonodectomy and the implementation of SLND in women who met the ACOSOG Z0011 criteria at our hospital benefited the patients who could be treated with less aggressive surgery. The de-escalation of ALND to SLND in women with up to two metastases in the SLN treated with conservative surgery and radiotherapy is possible. Our study showed that the ACOSOG Z0011 recommendation is feasible even in developing countries.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries [published correction appears in CA Cancer J Clin. 2020 Jul; 70(4):313]. CA Cancer J. Clin. 68, 394–424. https://doi.org/10.3322/caac.21492 (2018).
Hu, K. et al. Global patterns and trends in the breast cancer incidence and mortality according to sociodemographic indices: An observational study based on the global burden of diseases. BMJ Open 9, e028461. https://doi.org/10.1136/bmjopen-2018-028461 (2019).
Heer, E. et al. Global burden and trends in premenopausal and postmenopausal breast cancer: A population-based study. Lancet Glob. Health. 8, e1027–e1037. https://doi.org/10.1016/S2214-109X(20)30215-1 (2020).
Dreyer, M. S., Nattinger, A. B., McGinley, E. L. & Pezzin, L. E. Socioeconomic status and breast cancer treatment. Breast Cancer Res. Treat. 167, 1–8. https://doi.org/10.1007/s10549-017-4490-3 (2018).
Giuliano, A. E. et al. Prospective observational study of sentinel lymphadenectomy without further axillary dissection in patients with sentinel node-negative breast cancer [published correction appears in J Clin Oncol 2000 Nov 15;18(22):3877]. J. Clin. Oncol. 18, 2553–2559. https://doi.org/10.1200/JCO.2000.18.13.2553 (2000).
Caudle, A. S. et al. American College of Surgeons Oncology Group (ACOSOG) Z0011: Impact on surgeon practice patterns. Ann. Surg. Oncol. 19, 3144–3151. https://doi.org/10.1245/s10434-012-2531-z (2012).
Weiss, A. et al. Expanding implementation of ACOSOG Z0011 in surgeon practice. Clin. Breast Cancer. 18, 276–281. https://doi.org/10.1016/j.clbc.2017.10.007 (2018).
Robinson, K. A., Pockaj, B. A., Wasif, N., Kaufman, K. & Gray, R. J. Have the American College of Surgeons Oncology Group Z0011 trial results influenced the number of lymph nodes removed during sentinel lymph node dissection?. Am. J. Surg. 208, 1060–1064. https://doi.org/10.1016/j.amjsurg.2014.08.009 (2014).
Yao, K. et al. Impact of the American College of Surgeons Oncology Group Z0011 randomized trial on the number of axillary nodes removed for patients with early-stage breast cancer. J. Am. Coll. Surg. 221, 71–81. https://doi.org/10.1016/j.jamcollsurg.2015.02.035 (2015).
Mann, J. M., Wu, X., Christos, P. & Nagar, H. The state of surgical axillary management and adjuvant radiotherapy for early-stage invasive breast cancer in the modern era. Clin. Breast Cancer. 18, e477–e493. https://doi.org/10.1016/j.clbc.2017.09.001 (2018).
Tsao, M. W. et al. A population-based study of the effects of a regional guideline for completion axillary lymph node dissection on axillary surgery in patients with breast cancer. Ann. Surg. Oncol. 23, 3354–3364. https://doi.org/10.1245/s10434-016-5310-4 (2016).
Lucci, A. et al. Surgical complications associated with sentinel lymph node dissection (SLND) plus axillary lymph node dissection compared with SLND alone in the American College of Surgeons Oncology Group Trial Z0011. J. Clin. Oncol. 25, 3657–3663. https://doi.org/10.1200/JCO.2006.07.4062 (2007).
Mansel, R. E. et al. Randomized multicenter trial of sentinel node biopsy versus standard axillary treatment in operable breast cancer: the ALMANAC Trial [published correction appears in J Natl Cancer Inst. Jun 21;98(12):876]. J. Natl. Cancer Inst. 98, 599–609. https://doi.org/10.1093/jnci/djj158 (2006).
Ashikaga, T. et al. Morbidity results from the NSABP B-32 trial comparing sentinel lymph node dissection versus axillary dissection. J. Surg. Oncol. 102, 111–118. https://doi.org/10.1002/jso.21535 (2010).
Giuliano, A. E. et al. Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: A randomized clinical trial. JAMA 305, 569–575. https://doi.org/10.1001/jama.2011.90 (2011).
Giuliano, A. E. et al. Locoregional recurrence after sentinel lymph node dissection with or without axillary dissection in patients with sentinel lymph node metastases: The American College of Surgeons Oncology Group Z0011 randomized trial. Ann. Surg. 252, 426–433. https://doi.org/10.1097/SLA.0b013e3181f08f32 (2010).
Giuliano, A. E. et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis: The ACOSOG Z0011 (Alliance) randomized clinical trial. JAMA 318, 918–926. https://doi.org/10.1001/jama.2017.11470 (2017).
Giuliano, A. E. et al. Locoregional recurrence after sentinel lymph node dissection with or without axillary dissection in patients with sentinel lymph node metastases: long-term follow-up from the American College of Surgeons Oncology Group (Alliance) ACOSOG Z0011 randomized trial. Ann. Surg. 264, 413–420. https://doi.org/10.1097/SLA.0000000000001863 (2016).
Fisher, B. et al. Twenty-five-year follow-up of a randomized trial comparing radical mastectomy, total mastectomy, and total mastectomy followed by irradiation. N. Engl. J. Med. 347, 567–575. https://doi.org/10.1056/NEJMoa020128 (2002).
Galimberti, V. et al. Axillary dissection versus no axillary dissection in patients with sentinel-node micrometastases (IBCSG 23–01): a phase 3 randomised controlled trial [published correction appears in Lancet Oncol. Jun; 14(7):e254]. Lancet Oncol. 14, 297–305. https://doi.org/10.1016/S1470-2045(13)70035-4 (2013).
Donker, M. et al. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981-22023 AMAROS): A randomised, multicentre, open-label, phase 3 non-inferiority trial. Lancet Oncol. 15, 1303–1310. https://doi.org/10.1016/S1470-2045(14)70460-7 (2014).
Gatzemeier, W. & Mann, G. B. Which sentinel lymph-node (SLN) positive breast cancer patient needs an axillary lymph-node dissection (ALND)-ACOSOG Z0011 results and beyond. Breast 22, 211–216. https://doi.org/10.1016/j.breast.2013.02.001 (2013).
Simon, S. D. et al. Characteristics and prognosis of stage I-III breast cancer subtypes in Brazil: The AMAZONA retrospective cohort study. Breast 44, 113–119. https://doi.org/10.1016/j.breast.2019.01.008 (2019).
Allemani, C. et al. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): Analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 391, 1023–1075. https://doi.org/10.1016/S0140-6736(17)33326-3 (2018).
Van Zee, K. J. et al. A nomogram for predicting the likelihood of additional nodal metastases in breast cancer patients with a positive sentinel node biopsy. Ann. Surg. Oncol. 10, 1140–1151. https://doi.org/10.1245/aso.2003.03.015 (2003).
Mittendorf, E. A. et al. Incorporation of sentinel lymph node metastasis size into a nomogram predicting nonsentinel lymph node involvement in breast cancer patients with a positive sentinel lymph node. Ann. Surg. 255, 109–115. https://doi.org/10.1097/SLA.0b013e318238f461 (2012).
Haffty, B. G., Hunt, K. K., Harris, J. R. & Buchholz, T. A. Positive sentinel nodes without axillary dissection: Implications for the radiation oncologist. J. Clin. Oncol. 29, 4479–4481. https://doi.org/10.1200/JCO.2011.36.1667 (2011).
Bishop, J. A., Sun, J., Ajkay, N. & Sanders, M. A. Decline in frozen section diagnosis for axillary sentinel lymph nodes as a result of the American College of Surgeons Oncology Group Z0011 trial. Arch. Pathol. Lab Med. 140, 830–835. https://doi.org/10.5858/arpa.2015-0296-OA (2016).
Jorns, J. M. & Kidwell, K. M. Sentinel lymph node frozen-section utilization declines after publication of American College of Surgeons Oncology Group Z0011 trial results with no change in subsequent surgery for axillary lymph node dissection. Am. J. Clin. Pathol. 146, 57–66. https://doi.org/10.1093/ajcp/aqw078 (2016).
Fillion, M. M. et al. Healthcare costs reduced after incorporating the results of the American College of Surgeons Oncology Group Z0011 trial into clinical practice. Breast J. 23, 275–281. https://doi.org/10.1111/tbj.12728 (2017).
Camp, M. S. et al. Application of ACOSOG Z0011 criteria reduces perioperative costs. Ann. Surg. Oncol. 20, 836–841. https://doi.org/10.1245/s10434-012-2664-0 (2013).
Nowikiewicz, T. et al. The current application of ACOSOG Z0011 trial results: Is further implementation of sentinel lymph node intra-operative histopathological examination mandatory in breast cancer patients—A single-centre analysis. Neoplasma 65, 449–454. https://doi.org/10.4149/neo_2018_170321N202 (2018).
van der Noordaa, M. E. M., Vrancken Peeters, M. T. F. D. & Rutgers, E. J. T. The intraoperative assessment of sentinel nodes—Standards and controversies. Breast 34, S64–S69. https://doi.org/10.1016/j.breast.2017.06.031 (2017).
Chung, A., Gangim, A., Mirocha, J. & Giuliano, A. Applicability of the ACOSOG Z0011 criteria in women with high-risk node-positive breast cancer undergoing breast conserving surgery. Ann. Surg. Oncol. 22, 1128–1132. https://doi.org/10.1245/s10434-014-4090-y (2015).
Kittaka, N. et al. A prospective feasibility study applying the ACOSOG Z0011 criteria to Japanese patients with early breast cancer undergoing breast-conserving surgery. Int. J. Clin. Oncol. 23, 860–866. https://doi.org/10.1007/s10147-018-1297-0 (2018).
Morigi, C. et al. Feasibility and surgical impact of Z0011 trial criteria in a single-Institution practice. Breast J. 26, 1330–1336. https://doi.org/10.1111/tbj.13851 (2020).
Jung, J. et al. Validating the ACOSOG Z0011 trial result: A population-based study using the SEER database. Cancers (Basel) 12, 950. https://doi.org/10.3390/cancers12040950 (2020).
Tseng, J. et al. Changes in utilization of axillary dissection in women with invasive breast cancer and sentinel node metastasis after the ACOSOG Z0011 trial. Breast J. 27, 216–221. https://doi.org/10.1111/tbj.14191 (2021).
Peristeri, D. V. & Harissis, H. V. Axillary lymph node dissection vs sentinel biopsy only among women with early-stage breast cancer and sentinel node metastasis: A systematic review and meta-analysis. Breast J. 27, 158–164. https://doi.org/10.1111/tbj.14140 (2021).
Huang, T. W., Su, C. M. & Tam, K. W. Axillary management in women with early breast cancer and limited sentinel node metastasis: a systematic review and metaanalysis of real-world evidence in the post-ACOSOG Z0011 era [published correction appears in Ann Surg Oncol. 2020 Aug 10]. Ann. Surg. Oncol. 28, 920–929. https://doi.org/10.1245/s10434-020-08923-7 (2021).
Almahariq, M. F. et al. Omission of axillary lymph node dissection is associated with inferior survival in breast cancer patients with residual N1 nodal disease following neoadjuvant chemotherapy. Ann. Surg. Oncol. 28, 930–940. https://doi.org/10.1245/s10434-020-08928-2 (2021).
Gooch, J. et al. The extent of extracapsular extension may influence the need for axillary lymph node dissection in patients with T1–T2 breast cancer. Ann. Surg. Oncol. 21, 2897–2903. https://doi.org/10.1245/s10434-014-3752-0 (2014).
Choi, A. H. et al. Size of extranodal extension on sentinel lymph node dissection in the American College of Surgeons Oncology Group Z0011 Trial Era. JAMA Surg. 150, 1141–1148. https://doi.org/10.1001/jamasurg.2015.1687 (2015).
Barrio, A. V. et al. Microscopic extracapsular extension in sentinel lymph nodes does not mandate axillary dissection in Z0011-eligible patients. Ann. Surg. Oncol. 27, 1617–1624. https://doi.org/10.1245/s10434-019-08104-1 (2020).
Morrow, M. et al. Axillary dissection and nodal irradiation can be avoided for most node-positive Z0011-eligible breast cancers: A prospective validation study of 793 patients. Ann. Surg. 266, 457–462. https://doi.org/10.1097/SLA.0000000000002354 (2017).
Goyal, A. & Dodwell, D. POSNOC: A randomised trial looking at axillary treatment in women with one or two sentinel nodes with macrometastases. Clin. Oncol. (R. Coll. Radiol.). 27, 692–695. https://doi.org/10.1016/j.clon.2015.07.005 (2015).
de Boniface, J. et al. Survival and axillary recurrence following sentinel node-positive breast cancer without completion axillary lymph node dissection: The randomized controlled SENOMAC trial. BMC Cancer 17, 379. https://doi.org/10.1186/s12885-017-3361-y (2017).
Houvenaeghel, G. et al. Overview of the pathological results and treatment characteristics in the first 1000 patients randomized in the SERC trial: Axillary dissection versus no axillary dissection in patients with involved sentinel node. BMC Cancer 18, 1153. https://doi.org/10.1186/s12885-018-5053-7 (2018).
Algara López, M. et al. OPTimizing Irradiation through Molecular Assessment of Lymph node (OPTIMAL): A randomized open label trial. Radiat. Oncol. 15, 229. https://doi.org/10.1186/s13014-020-01672-7 (2020).
Acknowledgements
We are extremely grateful to Prof. Dr. Armando Giuliano for his review of and suggestions regarding this manuscript.
Funding
The first author (VMS) was supported by a doctoral scholarship from Conselho Nacional de Desenvolvimento Científico e Tecnológico. No other funding was received for this study.
Author information
Authors and Affiliations
Contributions
V.M.S. designed the study, performed the experiments, analyzed the data, and wrote the manuscript; S.E. revised the manuscript; G.F. revised the manuscript; S.E.B. revised the manuscript; A.C.P.N. designed the study, analyzed the data, and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sanvido, V.M., Elias, S., Facina, G. et al. Survival and recurrence with or without axillary dissection in patients with invasive breast cancer and sentinel node metastasis. Sci Rep 11, 19893 (2021). https://doi.org/10.1038/s41598-021-99359-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-99359-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.