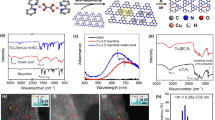
Abstract
Synthetic homogeneous system known to date performing methane to methanol conversion using O2 as terminal oxidant is unique and based on copper complex with piperazine-based ligand (Cu3L in Fig. 1) in a medium of acetonitrile. Prior work have shown that in order to achieve catalytic turnover, hydrogen peroxide is needed to regenerate the active site. We show in this paper that reaction solvent based on organic nitrile decompose concurrently with methane activation and that in the absence of either acetonitrile, Cu complex or hydrogen peroxide, the catalytic turnover does not happen. We show in this manuscript that the direct methane oxidation to methanol might have been mediated by catalytic Radziszewski oxidation between acetonitrile and H2O2. Additionally we have discovered that in the absence of methane, peroxide mediated acetonitrile decomposition also makes methanol via a background reaction which was hitherto unknown.
Similar content being viewed by others
Introduction
Methane monooxygenase serves as a model to develop biomimetic complexes to turnover methane to methanol under mild conditions. In a remarkable study, Chan et al. introduced the Cu3LO2 complex made after pMMO active site1,2,3. Mechanistic differences in the Cu3LO2 complex3 and Cu-oxo clusters in zeolites are still unresolved4,5,6. Chan et al., showed that in the absence of methane, the use of O2 and H2O2 as oxidant and reductant could lead to an abortive cycle7. The supported version of this catalyst exhibited 171 turnover number when using nearly 200 equivalents of H2O2 in the presence of acetonitrile8. Although the supported hybrid organic–inorganic Cu complex showed a high degree of selectivity, the system did not exhibit catalytic turnover using O2 as terminal oxidant. We had tried H2 + O2, Au–Pd/TiO2 system9 for generating hydrogen peroxide in situ along with Cu3L complex where we saw almost no methanol formation with water as the solvent. We had also looked at tert-Butyl hydroperoxide (TBHP) which did not succeed as a reducing agent for Cu3L regeneration. Given that the Cu3L complex does neither turnover with O2, nor in aqueous solution we sought to further probe the role of acetonitrile in this catalytic mechanism3. The Radziszewski oxidation10,11 proceeds via nucleophilic addition of hydroperoxide anion to an organic nitrile carbon to afford peroxyimidate which spontaneously undergoes a rearrangement reaction to release singlet oxygen and amide12. The evolution of reactive oxygen species from this nitrile decomposition is used for affording selective partial oxidation of a number of functional groups such as sulfoxidation13,14, epoxidation15, and Bayer–Villiger oxidation16. Peroxide and acetonitrile combination in the presence of metals catalyze the partial oxidation of alkanes17,18,19,20. Complexes of Cu(II) along with H2O2 result in producing O2 and OH. radicals via a Cu(II)/Cu(I) redox cycle21. Copper catalyzed decomposition of H2O2 is well studied through Fenton like autocatalysis22, alkaline solutions23, and Cu(I)/Cu(II) and Cu(II)/Cu(III) redox pairs24. We probed the rate of Cu(I) incorporation to form the Cu3L framework. Ligand structure controls the copper oxygen bonding type25 and substrate angle by steric effects3,26. UV–Vis data indicates counter anions27 can alter the redox properties as indicated by a shift in the charge transfer bands for bidentate ligands28 with fewer coordinating anions leading to bis(µ-oxo)dicopper(III) and more strongly coordinating anions leading to peroxodicopper(II). Spectroscopic methods are capable of determining the nature of copper oxygen bonding species29. ESI–MS analysis is used to show qualitative purity of Cu3L complex species in a solution30. There are instances of acetonitrile and hydrogen peroxide environments for C–H bond activation where solvent decomposition and generation of reactive oxygen species through acetonitrile oxidation has not been investigated31,32,33,34,35,36,37,38. Palomas et al., used multicopper complexes for methane oxidation which was rather selective towards CO239. In all instances of such complexes showing activity, there was a medium of peroxide and nitriles present1,3,7,40,41,42. Previously the role of acetonitrile as a mediator for oxygen transfer was not known. Here we further elucidate mechanism by independently assessing various organic nitrile decomposition with peroxide.
Results
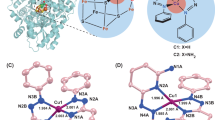
Ligand 3,3′-(1,4-diazepane-1,4-diyl)bis[1-(4-ethylpiperazine-1-yl)propan-2-ol] (7-N-Etppz) and copper incorporation into this complex is controlled by time and ligand ratio as indicated by the ESI–MS stacked plot (Supplementary Fig. 2–3). Acetonitrile attaches to the complex in nearly all of the tricopper complex species Supplementary Fig. 2–4. More tricopper species are formed with acetate and perchlorate than with tetrafluoroborate, exemplified by Supplementary Table 1. The ligand species is indicated by 441 m/z, and 503 m/z is the monocopper incorporated species. When the ratio of copper to ligand is around 3, the ligand fully incorporates into the complex (Supplementary Fig. 3). The spectroscopic characteristics are summarized in Supplementary Table 2. Characterization spectroscopically helps determine copper species as summarized in EPR Table Supplementary Table 3. Addition of hydrogen peroxide led to hyperfine coupling features in EPR hence less interactive CuII core, while the oxidized Cu3LO2 complex has broad features indicative of closely interacting CuII core. The complex with BF4 precursor does not show signal prior to oxidation since all is in CuI form. UV–Vis characterization of oxidized (Supplementary Fig. 5) and H2O2 treated (Supplementary Fig. 6) indicates closed complex and integrated clusters with Acetate and ClO4 counter anions. H2O2 addition reduces the oxide-to-CuII LMCT. Since nitrile coordinates towards copper site in the complex, it becomes susceptible to nucleophilic attack. Figure 1 and Supplementary Table 4 summarize the Radziszewski acetonitrile partial oxidation. The copper (I) 7-N-Etppz complex and H2O2 are required to produce hydrolysis products of acetonitrile (Supplementary Table 4, entries 1–5). With the copper salt and addition of hydrogen peroxide without the ligand there is some acetonitrile hydrolysis and minimal acetic acid production. Water is incapable of carrying out the hydrolysis. Among copper precursors, BF4 salt produces the most active complex in hydrolyzing MeCN (Supplementary Table 4, entries 5–7). The limiting reagent is H2O2 and the highly concentrated complex solution results in a lower TON (Supplementary Table 4, entries 8–11). The rate of addition (Table 2) and the amount of H2O2 impact the reaction exemplified by two different MeCN:H2O2 ratios (10:25 vs 57.5:1 Supplementary Table 4, entries 5,10). The cumulative turnover for MeCN hydrolysis of 1.6 was achieved with the MeCN:H2O2 molar ratio of 2 and reaction time of 24 h (Supplementary Table 4, entry 11). To examine the extent of complex activity towards hydrolyzing acetonitrile, we performed reactions with high H2O2 to acetonitrile ratio which led to significantly higher amounts of acetamide and acetic acid products (Supplementary Table 4, entry 10–11).
Figure 2 shows that initially, the rate of acetonitrile to acetamide oxidation is faster than the subsequent amide to acid hydrolysis. At a certain point the acetamide concentration decreases due to hydrolysis to acetic acid. The nitrile decomposition reaction ceases after about two hours due to complex deactivation and hydrogen peroxide degradation (Supplementary Fig. 10). Nitrile decomposition for a variety of nitrile compounds is illustrated by Table 1.
Time dependent acetamide and acetic acid production (Fig. 1) by complex 7-N-Etppz using BF4 based salta. Due to low concentration, there is moderate uncertainty in acetic acid quantification. aReaction Conditions: The complex (40 mM), H2O2 (5 mmol), acetonitrile (1.0 mL) room temperature.
Substrate scope analysis was performed with high nitrile concentration and limited amounts of hydrogen peroxide. The trichloroacetonitrile and fluoroacetonitrile led to highest turnovers of nitrile to amide (Table 1, entries 1–2). Due to the presence of electron withdrawing groups, the nitrile group is more vulnerable to nucleophilic approach. The conversions were calculated based on hydrogen peroxide transformation to hydrolysis products since that was the limiting reagent. Electron donating groups such as aryl and vinyl prevent the hydrolysis (Table 1, entry 3–4). Complex is more active towards malononitrile compared to acetonitrile, due to the presence of more electron withdrawing effect of CH2CN compared to H (Table 1, entry 5, 7). Greater H2O2 conversion and TON are indicative of higher catalytic activity of malononitrile substrate compared to acetonitrile (Table 1, entries 5, 7). The further the nitrile groups are apart, the less they affect one another in the case of adiponitrile (Table 1, entry 6). Methanol production is summarized in Table 2. Hydrogen peroxide is better utilized when added slowly due to controlled degradation rate. When urea adduct of hydrogen peroxide was used, higher activity was achieved due to slow dissolution process of H2O2 into the medium (Table 2, entry 1–2). When added continuously with a rate close to acetonitrile hydrolysis, the complex activity is increased towards methanol production and acetonitrile hydrolysis (Table 2, entries 3–4). Bringing down the addition rate does not improve the activity except slightly (Table 2, entry 5).
Discussion
ESI–MS results imply the existence of a variety of complex species. A more prevalent presence of di and tricopper species is observed with perchlorate and acetate precursor compared to the tetrafluoroborate salt. This observation implies an interaction between the complex and counter anions43. Electronic absorbance spectra exhibits a 265–300 nm peak which is assigned to “oxide”-to-CuII LMCT41. The absorbance is more prominent for ClO4 and acetate based species where the tricopper species are more abundant. The considerably larger rate of H2O2 decomposition compared to the rate of acetamide production suggests that only about 10% of H2O2 is being utilized in the reaction. We have directionally showed by doing slow addition one can improve the overall TON for methanol. Isotope labeled experiment in presence of 18O2 showed some 18O incorporation in the acetamide compared to when complex is activated by natural isotope O2. Experiment with 18O labeled H2O2 also showed incorporation of 18O in the acetamide (Supplementary Fig. 11). This could be due to the stoichiometric reaction of Cu3LO2 making methanol. Addition of unlabelled H2O2 takes over in the subsequent steps of catalytic turn overs. We determined that produced methanol is at least partly supplied by solvent when acetonitrile was used. Reaction in absence of methane led to some methanol production and 13C labeled acetonitrile used as the solvent led to 13C methanol (Supplementary Fig. 12). We have observed exclusive formation of Cl3CONH2 and FCH2CONH2 instead of any chlorinated or fluorinated alcohol when trichloroacetonitrile or fluoroacetonitrile were used as solvent.
As a part of interrogating the reaction mechanism, we have studied the catalyst decomposition pathway. This aspect of the chemistry was not probed previously, and we recognize this issue as a key problem for this catalytic system. The active life-time of the complex is affected by a Cu leaching mechanism that eventually makes Cu(NH3)x species. We believe ammonia evolution occurs through amide hydrolysis. Evidence of ammonia, water, and acetonitrile coordinated copper is present in the DART-TOF spectra in the range of 80–98 m/z (Supplementary Fig. 13) and ESI–MS (Supplementary Fig. 14). In the example of trichloroacetonitrile where the TON is high, there was no carboxylic acid detected in the product. This implies that subsequent hydrolysis of amide to acid is correlated with deactivation of the complex. However, due to poor solubility of complex (and possibly methane) into the solvent the TON for methane to methanol conversion is only modestly improved. Overall, we find that acetonitrile is not an innocent solvent in the peroxide mediated catalytic partial oxidation of methane to methanol using the tricopper complex. For the 7-N-Etppz copper complex to form tricopper species, the role of the counter anion needs to be considered since by changing this species, different spectroscopic and activity results are obtained. The extent of nitrile decomposition is controlled by the peroxide amount and the rate of addition as well as electron withdrawing groups attached to the nitrile. At least 90% conversion of peroxide (TON of 25) to amide is achieved in the case of trichloroacetonitrile and a TON of 1.6 was observed for the case of acetonitrile conversion to acetamide and acetic acid. When used as a medium for methane to methanol conversion, chlorinated acetonitrile led to a slightly higher TON of complex activity partly due to faster reactive oxygen species generation but was hindered by the lack of solubility of the complex in the medium.
Methods
Materials
All chemicals were purchased from Sigma-Aldrich unless mentioned otherwise. Solvents (Methanol and Acetonitrile) were first subject to continuous bed drying by solvent dispenser system under argon. They were subject to a freeze pump thaw procedure to remove residual oxygen content and finally were subject to 48 h of static drying by 3 A molecular sieves under argon and subsequent filtration prior to use. The precursors and solvents were stored under argon atmosphere.
7-N-Etppz ligand synthesis
The ligand was synthesized according to procedures introduced by Chan et al3. 15 mL of a 1.3 M solution of epichlorohydrin in methanol was added slowly to a 0.33 M solution of homopiperazine while stirred at − 5 °C. After 72 h the solution was briefly subject to rotary evaporation at room temperature to remove most of the solvent. 4.3 g of the resulting compound was added into a solution with 15 mL of acetonitrile, 4.15 g of K2CO3 and 3.5 g of 1-ethylpiperazine and heated to 80 °C for 48 h under argon atmosphere. The solution was then filtered and dried via a rotary evaporator at room temperature and stored under argon atmosphere. The H-NMR, and C-NMR characterization of the resulting ligand is provided in the SI. The NMR spectra (Supplementary Fig. 1) is consistent with previously reported NMR of the ligand. The ESI–MS characterization of ligand showed the major molecular ion peak at m/z 441.
Copper complex preparations
In a typical run for a 13 mM solution of complex in acetonitrile, 18 mg of 7-N-Etppz ligand and 36 mg of tetrakis(acetonitrile)copper(I) tetrafluoroborate were added to 3 mL of acetonitrile and stirred for desired amount of time prior to use.
Acetonitrile to acetic acid and acetamide transformation
In a typical run after the complex preparation within glovebox in a biotage sealed cap vial, up to 0.1 mL of a 35% (w/w) H2O2 solution was added to the solution and left stirring while taking samples at desired time intervals. The reaction is complete within the first 2 h.
Continuous hydrogen peroxide flow for methane to methanol conversion
In a typical run 0.01 mol per min of hydrogen peroxide was added to a 35 mL glass bottle with 3 mL of acetonitrile, catalyst concentration of 13 mM, and methane pressure of slightly above 1 atm.
Characterizations
1H and 13C NMR were performed with CDCl3 with a Bruker AVANCE 300 MHz. Electrospray Ionization Mass Spectrometry (ESI–MS) characterization of the complex was done by diluting complex solutions 1000 times from the original 13 mM to reach acceptable limits for use of a triple quadropole Quattro-II (Waters) instrument for low resolution analysis. For high resolution analysis a QStar Elite (AB sciex) instrument was used. Electron Paramagnetic Resonance (EPR) analysis was carried out on a Bruker instrument at 110 K, with a microwave frequency of 9.39 GHz and a modulation amplitude of 10 G. Ultraviolet–visible spectra of samples were obtained with a Shimadzu UV-2450 ultraviolet–visible spectrophotometer. Samples were exposed to air before UV–Vis spectrometer measurements.
References
Chen, P. P. Y., Yang, R. B. G., Lee, J. C. M. & Chan, S. I. Facile O-atom insertion into C–C and C–H bonds by a trinuclear copper complex designed to harness a singlet oxene. Proc. Natl. Acad. Sci. U. S. A. 104, 14570–14575 (2007).
Chen, Y. H., Wu, C. Q., Sung, P. H., Chan, S. I. & Chen, P. P. Y. Turnover of a methane oxidation tricopper cluster catalyst: Implications for the mechanism of the particulate methane monooxygenase (pMMO). ChemCatChem 12, 3088–3096 (2020).
Chan, S. et al. Efficient oxidation of methane to methanol by dioxygen mediated by tricopper clusters. Angew. Chemie 52, 3731–3735 (2013).
Narsimhan, K., Iyoki, K., Dinh, K. & Román-Leshkov, Y. Catalytic oxidation of methane into methanol over copper-exchanged zeolites with oxygen at low temperature. ACS Cent. Sci. 2, 424–429 (2016).
Wulfers, M. J., Teketel, S., Ipek, B. & Lobo, R. F. Conversion of methane to methanol on copper-containing small-pore zeolites and zeotypes. Chem. Commun. 51, 4447–4450 (2015).
Sushkevich, V. L., Palagin, D., Ranocchiari, M. & van Bokhoven, J. A. Selective anaerobic oxidation of methane enables direct synthesis of methanol. Science (80-). 356, 523–527 (2017).
Liu, C.-C., Mou, C.-Y., Yu, S.S.-F. & Chan, S. I. Heterogeneous formulation of the tricopper complex for efficient catalytic conversion of methane into methanol at ambient temperature and pressure. Energy Environ. Sci. 9, 1361–1374 (2016).
Liu, C.-C., Ramu, R., Chan, S. I., Mou, C.-Y. & Yu, S.S.-F. Chemistry in confined space: A strategy for selective oxidation of hydrocarbons with high catalytic efficiencies and conversion yields under ambient conditions. Catal. Sci. Technol. 6, 7623–7630 (2016).
Luo, Z. et al. TiO 2 Supported gold–palladium catalyst for effective syngas production from methane partial oxidation. Appl. Catal. A Gen. 554, 54–63 (2018).
Dotsenko, V. V., Krivokolysko, S. G., Litvinov, V. P. & Gutov, A. V. The radziszewski oxidation of (E)-3-aryl-2-(thiazol-2-yl)acrylonitriles: A convenient diastereoselective synthesis of (2S,3S)-3-aryl-2-(thiazol-2-yl)oxirane-2-carboxamides. Dokl. Chem. 412, 29–32 (2007).
Dotsenko, V. V., Krivokolysko, S. G. & Litvinov, V. P. The Radziszewski oxidation of cycloalkylidene-α-(thiazol- 2-yl)acetonitriles: A new approach toward spirooxiranes. J. Heterocycl. Chem. 48, 162–167 (2011).
Brauer, H., Eilers, B. & Lange, A. Formation of singlet molecular oxygen by the Radziszewski reaction between acetonitrile and hydrogen peroxide in the absence and presence of ketones. J. Chem. Soc. Perkin 2, 1288–1295 (2002).
Bethell, D. et al. Reactivity and selectivity in the oxidation of aryl methyl sulfides and sulfoxides by hydrogen peroxide mediated by acetonitrile. J. Chem. Soc. Perkin Trans. 2, 2161 (1993).
Plant, H. L. & Peterson, G. S. Oxidation of alpha-alkylated, benzyl-substituted 2-thiopyridine 1-oxides. (1981).
Payne, G. B., Deming, P. H. & Williams, P. H. Reactions of hydrogen peroxide. VII. Alkali-catalyzed epoxidation and oxidation using a nitrile as co-reactant. J. Org. Chem. 26, 659–663 (1961).
Bradley, T. D., Dragan, A. & Tomkinson, N. C. O. Baeyer–Villiger oxidation under Payne epoxidation conditions. Tetrahedron 71, 8155–8161 (2015).
Shulpin, G. B. & Nizova, G. V. Formation of alkyl peroxides in oxidation of alkanes by H2O2 catalyzed by transition metal complexes. React. Kinet. Catal. Lett. 48, 333–338 (1992).
Bonon, A. J. et al. Oxidation of alkanes and olefins with hydrogen peroxide in acetonitrile solution catalyzed by a mesoporous titanium-silicate Ti-MMM-2. Appl. Catal. A Gen. 365, 96–104 (2009).
Kirillova, M. V. et al. Participation of oligovanadates in alkane oxidation with H2O2 catalyzed by vanadate anion in acidified acetonitrile: Kinetic and DFT studies. ACS Catal. 1, 1511–1520 (2011).
Shul’pin, G. B., Nizova, G. V., Kozlov, Y. N., Gonzalez Cuervo, L. & Süss-Fink, G. Hydrogen peroxide oxygenation of alkanes including methane and ethane catalyzed by iron complexes in acetonitrile. Adv. Synth. Catal. 346, 317–332 (2004).
Lázaro-Martínez, J. M., Lombardo Lupano, L. V., Piehl, L. L., Rodríguez-Castellón, E. & Campo Dall’ Orto, V. New insights about the selectivity in the activation of hydrogen peroxide by cobalt or copper hydrogel heterogeneous catalysts in the generation of reactive oxygen species. J. Phys. Chem. C 120, 29332–29347 (2016).
Naqvi, K. R., Marsh, J. & Chechik, V. Formation of self-inhibiting copper(ii) nanoparticles in an autocatalytic Fenton-like reaction. Dalt. Trans. 43, 4745 (2014).
Luo, Y., Kustin, K. & Epstein, I. Kinetics and mechanism of H2O2 decomposition catalyzed by Cu2+ in alkaline solution. Inorg. Chem. 27, 2489–2496 (1988).
Jezowska-Bojczuk, M. et al. Molecular mechanism of hydrogen peroxide conversion and activation by Cu(II)-amikacin complexes. Chem. Res. Toxicol. 14, 1353–1362 (2001).
Taki, M. et al. Fine-tuning of copper(I)-dioxygen reactivity by 2-(2-Pyridyl)ethylamine bidentate ligands. J. Am. Chem. Soc. 124, 6367–6377 (2002).
Chan, S. I. et al. Efficient catalytic oxidation of hydrocarbons mediated by tricopper clusters under mild conditions. J. Catal. 293, 186–194 (2012).
Tse, E. C. M., Schilter, D., Gray, D. L., Rauchfuss, T. B. & Gewirth, A. A. Multicopper models for the laccase active site: Effect of nuclearity on electrocatalytic oxygen reduction. Inorg. Chem. 53, 8505–8516 (2014).
Mirica, L. M., Ottenwaelder, X. & Stack, T. D. P. Structure and spectroscopy of copper−dioxygen complexes. Chem. Rev. 104, 1013–1046 (2004).
Elwell, C. E. et al. Copper-oxygen complexes revisited: structures, spectroscopy, and reactivity. Chem. Rev. 117, 2059–2107 (2017).
Son, J.-H. & Casey, W. H. A decatungstate-type polyoxoniobate with centered manganese: [H2Mn(IV)Nb10O32]8-as a soluble tetramethylammonium salt. Dalton Trans. 42, 13339–13342 (2013).
You, X. et al. Synthesis of two copper clusters and their catalysis towards the oxidation of benzene into phenol. RSC Adv. 4, 61790–61798 (2014).
Bianchi, D., Bertoli, M., Tassinari, R., Ricci, M. & Vignola, R. Direct synthesis of phenols by iron-catalyzed biphasic oxidation of aromatic hydrocarbons with hydrogen peroxide. J. Mol. Catal. A Chem. 200, 111–116 (2003).
Fernandes, R. R., Kirillova, M. V., da Silva, J. A. L., Fraústo da Silva, J. J. R. & Pombeiro, A. J. L. Oxidations of cycloalkanes and benzene by hydrogen peroxide catalyzed by an FeIIIN2S2 centre. Appl. Catal. A Gen. 353, 107–112 (2009).
Du, Y., Xiong, Y., Li, J. & Yang, X. Selective oxidation of cyclohexane with hydrogen peroxide in the presence of copper pyrophosphate. J. Mol. Catal. A Chem. 298, 12–16 (2009).
Shul’pina, L. S., Takaki, K., Strelkova, T. V. & Shul’pin, G. B. Benzene oxidation with hydrogen peroxide catalyzed by soluble and heterogenized copper compounds. Pet. Chem. 48, 219–222 (2008).
Fernandes, T. A. et al. Copper(II) coordination polymers self-assembled from aminoalcohols and pyromellitic acid: Highly active precatalysts for the mild water-promoted oxidation of alkanes. Inorg. Chem. 55, 125–135 (2016).
Dias, S. S. P., Kirillova, M. V., Andre, V., Klak, J. & Kirillov, A. M. New tricopper(ii) cores self-assembled from aminoalcohol biobuffers and homophthalic acid: Synthesis, structural and topological features, magnetic properties and mild catalytic oxidation of cyclic and linear C5 ?C8 alkanes. Inorg. Chem. Front. 2, 525–537 (2015).
Kirillova, M. V. et al. Remarkably fast oxidation of alkanes by hydrogen peroxide catalyzed by a tetracopper(II) triethanolaminate complex: Promoting effects of acid co-catalysts and water, kinetic and mechanistic features. J. Catal. 268, 26–38 (2009).
Palomas, D. et al. Re-evaluating selectivity as a determining factor in peroxidative methane oxidation by multimetallic copper complexes. Catal. Sci. Technol. 5, 4108–4115 (2015).
Nagababu, P., Yu, S.S.-F., Maji, S., Ramu, R. & Chan, S. I. Developing an efficient catalyst for controlled oxidation of small alkanes under ambient conditions. Catal. Sci. Technol. 4, 930–935 (2014).
Maji, S. et al. Dioxygen activation of a trinuclear CuICuICuI cluster capable of mediating facile oxidation of organic substrates: Competition between O-atom transfer and abortive intercomplex reduction. Chem. A Eur. J. 18, 3955–3968 (2012).
Nagababu, P. et al. Efficient room-temperature oxidation of hydrocarbons mediated by tricopper cluster complexes with different ligands. Adv. Synth. Catal. 354, 3275–3282 (2012).
Grenthe, I., Paoletti, P., Sandstroem, M. & Glikberg, S. Thermochromism in copper(II) complexes. Structures of the red and blue-violet forms of bis(N,N-diethylethylenediamine)copper(II) perchlorate and the nonthermochromic violet bis(N-ethylethylenediamine)copper(II) perchlorate. Inorg. Chem. 18, 2687–2692 (1979).
Funding
Funding was provided by ExxonMobil Research and Engineering Company. We acknowledge the US Department of Energy, Office of Basic Energy Sciences, Division of Chemical, Biological and Geological Sciences under grant DE-FG02-86ER13622 for partial support of this research.
Author information
Authors and Affiliations
Contributions
P.N., S.L.S. and E.M. designed the experiments, E.M., T.J., D.R., H.K., C.K. performed experiments and analyzed the data, E.M., S.L.S. and P.N. wrote the manuscript. All co-authors have read the manuscript and agreed to the final version for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moharreri, E., Jafari, T., Rathnayake, D. et al. Role of catalytic nitrile decomposition in tricopper complex mediated direct partial oxidation of methane to methanol. Sci Rep 11, 19175 (2021). https://doi.org/10.1038/s41598-021-98721-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-98721-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.