Abstract
Coral reefs, especially those located near-shore, are increasingly exposed to anthropogenic, eutrophic conditions that are often chronic. Yet, corals under unperturbed conditions may frequently receive natural and usually temporary nutrient supplementation through biological sources such as fishes. We compared physiological parameters indicative of long- and short-term coral health (day and night calcification, fragment surface area, productivity, energy reserves, and tissue stoichiometry) under continuous and temporary nutrient enrichment. The symbiotic coral Acropora intermedia was grown for 7 weeks under continuously elevated (press) levels of ammonium (14 µmol L−1) and phosphate (10 µmol L−1) as separate and combined treatments, to discern the individual and interactive nutrient effects. Another treatment exposed A. intermedia twice-daily to an ammonium and phosphate pulse of the same concentrations as the press treatments to simulate natural biotic supplementation. Press exposure to elevated ammonium or phosphate produced mixed effects on physiological responses, with little interaction between the nutrients in the combined treatment. Overall, corals under press exposure transitioned resources away from calcification. However, exposure to nutrient pulses often enhanced physiological responses. Our findings indicate that while continuous nutrient enrichment may pose a threat to coral health, episodic nutrient pulses that resemble natural nutrient supplementation may significantly benefit coral health and physiology.
Similar content being viewed by others
Introduction
Coral reef ecosystems thrive in nutrient poor waters typical of many tropical coasts and oceans. The concentrations of inorganic nitrogen (N) and phosphorus (P) in coral reef waters are often amongst the lowest values recorded for aquatic systems1, 2, yet many coral reefs are typified by substantially high rates of primary productivity3. One mechanism enabling symbiotic corals to persist in these oligotrophic waters is the capacity of the coral holobiont to rapidly take up dissolved inorganic (e.g. ammonium and phosphate) and organic nutrients (e.g. amino acids) from the surrounding water4,5,6,7,8. Efficient nutrient recycling between the coral host and their dinoflagellate endosymbionts (Symbiodiniaceae)9,10,11 and other associated microbes12 further enable corals to thrive in nutrient-poor environments.
Nutrient uptake and assimilation
Inorganic N is primarily taken up by the holobiont in the form of ammonium and nitrate7, 13, 14. Nitrate must first be reduced to ammonium by the symbiont using nitrate reductase before assimilation13. While the coral host is capable of its own ammonium assimilation through the glutamine synthetase/glutamate dehydrogenase (GS/GDH) pathway, the majority of N assimilation occurs in the symbiont through the glutamine synthetase/glutamine oxoglutarate aminotransferase (GS/GOGAT) cycle10, 14, 15. Inorganic P (mainly phosphate) is assimilated primarily by the symbionts8, though low in hospite symbiont phosphate content suggests that the host controls availability16. The exact mechanisms involved in P uptake remain largely unexplored10, 17, as do the relative roles of Symbiodiniaceae and coral microbial associates in P uptake and assimilation18.
Because symbiotic corals are so finely adapted to living under nutrient-poor conditions, increases in nutrient concentrations of the surrounding seawater due to anthropogenic sources (e.g. sewage, fertilizer runoff) often disrupt host and symbiont physiology. Under natural conditions, inorganic N and P are considered limiting factors for Symbiodiniaceae growth and photosynthesis19,20,21. Addition of these nutrients to the surrounding seawater may lead to rapid nutrient uptake by the symbiont, C:N:P ratio shifts particularly within the symbiont22, increased symbiont photosynthesis, and organic carbon accumulation in both the host and the symbiont23. Nutrient enrichment therefore likely stimulates symbiont growth at the expense of the host24,25,26,27,28. Enhanced symbiont growth consumes organic carbon faster, possibly leading to lower translocation of photosynthetic carbon to the host29, 30. Extreme levels of ammonium (typically ≥ 20 µmol L−1) may directly interfere with skeletogenesis, carbohydrate metabolism, ion transport, and host-symbiont interaction5, 20, 31, 32, while imbalanced N:P ratios (usually nitrate) of available nutrients may cause stress resulting in coral bleaching, photosynthetic impairment, declining biomass, and increased mortality33,34,35.
Natural nutrient loading
Interestingly, corals naturally receive episodic pulses of nutrients several times higher than the background concentrations in the overlying waters, delivered to reefs by diurnal activities of fish and other aquatic organisms36,37,38, as well as seabirds39, 40. These nutrient influxes are dynamic and temporal, and usually reflect the diurnal migratory and feeding behaviour of fish36,37,38. The efficient nutrient uptake by corals could allow them to capitalize on episodic high nutrient influxes available in the waste products of organisms that live in close proximity37, 41, 42. Contrary to the negative effects of anthropogenic eutrophication on coral physiology, corals exposed to these temporary and natural nutrient pulses showed increased tissue thickness, CaCO3 accretion and skeletal expansion rates38, 43, 44, provided that the reef is not yet in a degraded state5, 45.
The conflicting outcomes of natural (and often temporary) vs anthropogenic (and often continuous) deposition of nutrients on coral reefs and the heterogeneous effects of inorganic N and P on coral physiology outline gaps in our knowledge regarding the influence of nutrients on symbiotic corals5, 46, 47. Firstly, it remains unclear whether elevated ammonium and phosphate are capable to interactively influence coral physiology when applied together48. Although an increasing number of studies address the effects of continuously elevated (press) levels of inorganic N and/or P on coral physiology, only few have determined their relative contribution within an orthogonal experimental design5, 20. Secondly, it is currently unknown how N and P affect coral functioning differently under continuous or temporary exposure5, 48. The present study therefore examines the effect of press elevated ammonium and phosphate under an orthogonal design on long- and short-term coral health (day and nighttime calcification, skeleton expansion, productivity, energy reserves and tissue stoichiometry) after a 7-week exposure in the symbiotic coral Acropora intermedia. Importantly, as a simulation of natural nutrient fluxes of biological origin on coral reefs, a separate treatment assessed the effects of episodic (pulse) ammonium and phosphate supplementation in comparison with no supplementation (control) and the press ammonium and phosphate treatments.
Materials and methods
Experimental design
This experiment was performed on Heron Island, located approximately 70 km off the eastern shore of Australia on the southern Great Barrier Reef, and unaffected by terrestrial run-off49. Despite significant guano deposition by the resident seabird population39, 40, average seawater conditions around Heron Island are mostly oligotrophic (0.49–0.55 µmol L−1 ammonium and 0.13–0.26 µmol L−1 phosphate) but can temporarily exceed 20 µmol L−1 ammonium and 4.5 µmol L−1 phosphate40, 50. During this study, seawater concentrations of ammonium and phosphate averaged 0.55 and 0.32 µmol L−1 respectively across the experimental period, with respective maxima of 0.91 and 0.62 µmol L−1 (Table S1, Figure S1). Seawater nitrate and nitrite concentrations were not measured during this study, but previous studies recorded concentrations comparable to ammonium51.
A total of 150 branch tip fragments of A. intermedia were collected in August 2015 on the reef flat of Heron Island between 0.5 and 1 m water depth from 15 different adult colonies. Upon returning to the lab, fragments were selected for equal thickness and trimmed to 5 cm length and suspended upright from monofilament line in 30 L aquaria with untreated flow-through seawater. Water flow was maintained at 1 L min−1, and all aquaria were fitted with a power head (infinity 800, Clearpond, WA, Australia) for water circulation. Fragments were randomly distributed over 20 aquaria with 7 or 8 fragments per aquarium. Aquaria were shaded to allow approx. 700 µmol quanta m−2 s−1 of PAR (LI-1400, LI-COR Inc., Lincoln, Nebraska, USA) at midday, which is comparable to light intensities measured at the coral collection site. Fragments were left to recover for 6 days, after which five separate experimental treatments (n = 4 aquaria per treatment) were initiated: (1) Control; neither ammonium nor phosphate was elevated (ambient seawater concentrations). (2) Press N; ammonium was permanently elevated to 14 µmol L−1 while phosphate was kept at ambient levels. (3) Press P; phosphate was permanently elevated to 10 µmol L−1 while ammonium was kept at ambient levels. (4) Press NP; ammonium and phosphate were both permanently elevated to 14 and 10 µmol L−1 respectively. (5) Pulse NP; ammonium and phosphate were elevated twice daily (09:00 and 17:00) to 14 and 10 µmol L−1 for 30 min each time. After 30 min excess nutrients were flushed out and concentrations of both nutrients returned to ambient levels. These concentrations of ammonium and phosphate were chosen because they simulate severe nutrient pollution5, comparable to maximum values recorded for Heron Island40.
In the press treatment, the nutrient perturbation was sustained with a new equilibrium being achieved and maintained, whereas in the pulse treatment the perturbation was temporary, after which the system returned to its original equilibrium52. Press treatment conditions were created by continuously feeding a concentrated ammonium (4.7 mmol L−1) and/or phosphate (3.3 mmol L−1) solution into the aquaria. Solutions were prepared fresh daily with 0.45 µm filtered seawater (FSW) and kept in darkened 20 L sumps. Each solution was dripped into the aquaria using small pumps (Aquagarden Mako 4000, Clearpond, WA, Australia) set at 3 ml min−1. Normal untreated seawater flow into the aquaria was maintained at 1 L min−1. Untreated seawater flow and nutrient drip speeds were monitored and adjusted daily. Nutrient pulses in the pulse treatment were added twice daily at 09:00 and 17:00. During the pulse, water flow and nutrient drips (but not water mixing) to all aquaria (press and pulse) was interrupted for 30 min, after which it was resumed normally. Tubing for the nutrient drips was darkened, and nutrient sumps were completely emptied and cleaned weekly to prevent algae growth. Treatment aquaria were cleaned daily, and water samples taken from the aquaria were analysed for ammonium and phosphate concentrations. Ammonium and phosphate assays were carried out using a photometric approach after 53 (pp. 14–17 and pp. 22–25 for ammonium and phosphate respectively). Corals were kept under treatment conditions for 7 weeks before physiological measurements were done.
Metabolic parameters
Coral net photosynthesis (PNET) and dark respiration (RDARK) rates (n = 12 per treatment) were obtained from metabolic oxygen flux measurements over light–dark cycles. Corals were incubated in sealed 250 ml acrylic chambers under 700 µmol quanta m−2 s−1 (Aqua Medic Ocean Lights, Aqua Medic, Bissendorf, Germany). Chambers were filled with 0.45 µm FSW from the respective press treatments. Corals from the pulse NP treatment were incubated under untreated (ambient) seawater conditions to obtain a response more reflective of the overall treatment history. Starting seawater oxygen content was reduced to approximately 60% air saturation (60.8 ± 5.1, mean ± sd) by N2 gas bubbling, possibly also slightly affecting carbonate chemistry. Chambers were temperature controlled using a water bath (Julabo F33ME refrigerated/heating circulator, Seelbach, Germany). Oxygen concentration in the chambers was logged at 15 s intervals using a PreSens OXY-10 mini oxygen meter (PreSens, Regensburg, Germany) over 30–30 min light–dark cycles. PNET and RDARK rates were determined from the light measurements and after a 20 min dark adaptation period respectively to determine holobiont potential for remaining net photosynthetic over a 24-h period, and to estimate the amount of photosynthetic carbon potentially available for translocation to the host.
Skeletogenesis
Rates of day and night calcification (GTA) were measured using the total alkalinity (TA) anomaly technique54. Corals (n = 12 per treatment) were incubated for 1 h in temperature-controlled (Julabo F33ME refrigerated/heating circulator, Seelbach, Germany) 250 ml acrylic chambers containing 0.45 µm FSW from the respective press treatment, or untreated (ambient) seawater for the pulse NP treatment. ΔTA was measured by Gran titration after 55, and corrected for the minimal ammonium and/or phosphate fluctuations (Table S2 and Supplementary methods) in the chambers during the incubation56.
Tissue composition
After the incubations, coral specimens (n = 12 per treatment) were frozen for tissue analysis (detailed in Supplementary methods). Briefly, tissue was removed from the skeleton using an airbrush and the resulting tissue mixture was vortexed and stored at -20 °C. Water-soluble host protein concentration was determined by differential absorbance at 235 and 280 nm through spectrometry (Spectra Max 2, Molecular Devices, Sunnyvale, California) according to 57. Total lipid concentration was quantified from freeze-dried samples (ScanVac CoolSafe, LaboGene, Lillerød, Denmark) after extraction in chloroform/methanol (2:1) solution and subsequent washing in 0.1 mol KCl and methanol/MQ (1:1) solutions58, 59.
Holobiont total organic carbon (TOC) content and N:P ratios were determined from freeze-dried samples (ScanVac CoolSafe, LaboGene, Lillerød, Denmark) at the Analytics Lab at the School of Agriculture and Food Sciences, The University of Queensland. Total nitrogen (TN) was analysed by combustion analysis (LECO TruSpec analyser, Michigan, USA), total phosphorus (TP) by acid digestion and inductively coupled plasma optical emission spectrometry (ICP-OES) analysis, and TOC was analysed by combustion (LECO TruSpec analyser, Michigan, USA) on acidified samples to remove carbonates. In order to meet the minimal analysis weight, samples from each tank (n = 4) were pooled and analysed at tank level.
After removal of the tissue, the coral skeletons were treated with 10% hypochlorite solution for 12 h until completely white and all remaining tissue had been removed60. Skeletons were then rinsed in fresh water and dried overnight at 60 °C. End-of-experiment skeleton surface areas of the coral fragments were assessed using the double waxing method61. Surface areas of a fragment subset (n = 20) taken at the start of the experiment were measured to confirm that initial surface areas of the fragments used in the experiment were comparable (13.66 ± 1.53 cm2; mean ± sd).
Statistical analysis
The interactive effect of ammonium and phosphate (between the control, press N, press P and press NP treatments) on photobiology, tissue lipid and protein content, and skeleton surface area responses were tested in a nested three-factorial ANOVA design (factors: Tank, Ammonium and Phosphate). In this design, Tank was nested in the interactive effect of categorical factors Ammonium and Phosphate (levels: ambient and elevated). Due to the pooling of the samples and analysis at tank level, holobiont N:P ratios and TOC content were analysed in a two-factorial ANOVA design (factors: Ammonium and Phosphate; levels: ambient and elevated). Day and Night rates of GTA were individually explored for tank effects in a preliminary analysis, and after none were detected (Table S3) these variables were further analysed using a three-factorial repeated measures design with categorical factors Ammonium and Phosphate (levels: ambient and elevated), and Time (Day and Night) as the within subject factor. The effect of continuous vs episodic nutrient loads (press vs pulse) was tested for all variables except GTA and tissue TOC content and N:P ratio, between the control, press NP and pulse NP treatments in a nested one-way ANOVA, where tank was nested in the treatments. Here, GTA was analysed in a repeated measures one-way ANOVA after it was determined in a preliminary analysis that there were no tank effects (Table S3), and tissue N:P ratio as well as TOC content were analysed at tank level in a one-way ANOVA. In all press vs pulse comparisons, significant outcomes were further explored using Tukey HSD post-hoc analyses. All analyses were tested for violations in assumptions for normality (Shapiro–Wilk test) and homogeneity of variances (Levene’s test), and the data were log or square root transformed where necessary. Analyses were performed to the α = 0.05 significance level. Heteroscedastic datasets were assessed at α = 0.01 significance level62. All statistics were performed using Statistica 13.2 (Statsoft, Tulsa, OK, USA).
Results
Skeletogenesis
Calcification rates (GTA) after seven weeks of treatment conditions showed a strong dependence on time of day (Fig. 1). During nighttime, rates of GTA fell to below zero values in the press N and press P treatments (− 0.017 and − 0.018 µmol cm−2 h−1 respectively). Overall, GTA was governed by an interactive effect between levels of ammonium, and the time of day (interactive effect Time × N; F1,44 = 10.98, p = 0.002) regardless of the level of phosphate. Elevated levels of ammonium significantly reduced daytime GTA, but had no effect on nighttime GTA.
Rates of calcification measured at the end of the experiment (GTA, n = 12) for Acropora intermedia in an experimental study done on Heron Island on the southern Great Barrier Reef. Corals were exposed for 7 weeks under an orthogonal design to continuous (press) addition of ammonium (N) and/or phosphate (P). GTA rates (mean ± SE) were measured during day (green bars) and nighttime (grey bars) in one-hour light and dark incubations respectively, and standardized to end-of-experiment tissue surface areas.
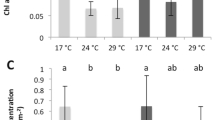
End-of-experiment skeleton surface areas of the fragments differed significantly between treatments (Fig. 2a). Largest fragment surface areas were measured for the control treatment (26.14 ± 0.63 cm2; mean ± SE). Fragment surface areas were reduced when exposed to press elevated levels of phosphate to 23.09 ± 0.74 cm2 (mean ± SE), but were unaffected by levels of ammonium (main effect P; F1,48 = 6.66, p = 0.013).
Metrics of coral physiology (mean ± SE) of Acropora intermedia fragments after 7-week exposure to elevated ammonium (N) and/or phosphate (P) under an orthogonal design. Fragment surface area (n = 16) was measured at the end of the experiment (a). Net photosynthesis (PNET) to dark respiration (RDARK) ratios (n = 12) were obtained from oxygen flux measurements (b), and total tissue protein (c) and lipid (d) concentrations (n = 12), and tissue N:P ratio (e) and organic carbon (f) content (n = 4) were measured from tissue samples collected at the end of the experiment.
Metabolic parameters and tissue composition
Elevated phosphate levels reduced PNET:RDARK ratios (Fig. 2b) irrespective of the level of ammonium (main effect P; F1,32 = 24.08, p < 0.001). These differences were caused by reduced PNET under elevated phosphate (main effect P; F1,32 = 4.24, p = 0.048), as there was no effect of treatment on the dark respiration rate.
Host protein concentration (Fig. 2c) did not differ significantly between any of the treatments. Tissue lipid content (Fig. 2d) was influenced by exposure to ammonium (main effect N; F1,32 = 11.59, p = 0.002) and phosphate (main effect P; F1,32 = 19.77, p < 0.001), but was unaffected by their interaction. Press exposure to elevated ammonium increased the tissue lipid content, whereas press exposure to phosphate decreased tissue lipid content.
The N:P ratio of the holobiont (Fig. 2e) was not influenced by exposure to ammonium and/or phosphate, but tissue TOC content (Fig. 2f) was governed by an interactive effect of ammonium and phosphate (interactive effect N × P; F1,12 = 26.55, p < 0.001). Exposure to phosphate resulted in increased tissue TOC content under elevated levels of ammonium, but did not influence tissue TOC content under ambient ammonium levels.
Press–pulse comparison
The results of the comparison between permanent (press), episodic (pulse) and no (control) exposure to elevated levels of ammonium and phosphate are summarized in Table 1. The rate of coral CaCO3 accretion (GTA) at the end of the experiment was significantly lower in all treatments under nighttime compared to daytime conditions (F1,9 = 51.06, p < 0.001), and also differed between treatments (F2,9 = 6.06, p = 0.022). Exposure to press levels of ammonium and phosphate resulted in reduced GTA compared to both the control (Tukey HSD p = 0.031) and the pulse NP treatments (Tukey HSD p = 0.041), whereas no differences in GTA were observed between the control and pulse NP treatments (Tukey HSD p = 0.982). Under press concentrations of ammonium and phosphate, fragment surface areas were significantly reduced compared to the control (Tukey HSD p = 0.046) and the pulse NP (Tukey HSD p < 0.001) treatments, while no differences in fragment surface areas were observed between the control and the pulse NP treatments (Tukey HSD p = 0.182). No significant differences in PNET:RDARK ratios, host protein concentration and tissue N:P ratio were found between the control, press NP and pulse NP treatments. Tissue lipid content differed between the press and pulse treatments (F2,24 = 9.048, p = 0.001). Lipid concentrations were increased under the pulse NP treatments compared to the control (Tukey HSD p = 0.009) and press NP treatments (Tukey HSD p < 0.002). Pulses of ammonium and phosphate also significantly influenced tissue TOC content (F2,9 = 333.3, p < 0.001). Tissue TOC content was higher in corals in the pulse treatment compared to the control (Tukey HSD p < 0.001) and the press treatment (Tukey HSD p < 0.001). Additionally, TOC content was elevated in the press treatment over the control treatment (Tukey HSD p < 0.001).
Discussion
This study reveals that perturbations to local nutrient levels can significantly affect key aspects of the physiology of Acropora intermedia, a common reef-building coral on the Great Barrier Reef. Inorganic nutrient levels are increasing in coastal waters where many coral reefs grow63. Understanding how local pressures influence corals and their symbionts is timely and important, particularly in the context of a rapidly changing global environment. Our results demonstrate (1) how multiple nutrients do not interact to produce synergistic or antagonistic outcomes, and (2) how A. intermedia benefits from episodic pulses of concentrated nutrients. Together, these findings highlight the importance of considering nutrient form, stoichiometry and origin on coral reefs. Overall, under permanently elevated ammonium and phosphate A. intermedia allocated resources towards tissue growth at the expense of skeletogenesis (Fig. 3).
Conceptual summary depicting changes or trade-offs in survival strategy of Acropora intermedia under different treatments of elevated ammonium (N) and/or phosphate (P), as permanent (press) or temporary (pulse) elevations. To create this plot, the average performance for each physiological parameter (calcification, skeleton expansion, tissue protein, productivity, or stored energy reserves) in a treatment is calculated and represented as a fraction of the respective parameter for the control treatment, which is always fixed at 1 (red line). The three main strategies of long-term colony viability are represented on the colored circle: growth (blue), competition (orange), and reproduction (green). Here, growth is defined as the combined capacity to amass skeleton and energy reserves, competition as the capacity to occupy or consolidate physical space on the reef, and reproduction as the capacity to produce offspring. Calcification was calculated as the 24 h-average of day and nighttime calcification based on a 12–12 h day–night cycle.
Individual and combined effects of press elevated ammonium and phosphate
Press elevated levels of ammonium or phosphate induced contrasting physiological responses in A. intermedia. Elevated levels of ammonium led to a substantial decrease in CaCO3 accretion rates. Primary productivity and tissue lipid concentration were unaffected. By contrast, elevated phosphate did not affect net CaCO3 accretion rates, but it reduced skeletal surface area expansion, a measure of the coral’s potential to expand and occupy new territory. Additionally, elevated phosphate reduced primary productivity rates and tissue lipid concentrations per surface area. We found that ammonium and phosphate rarely produced interactive effects when applied together, and most physiological parameters assessed in this study were individually influenced by one of the two nutrients.
Calcification rates (GTA) of A. intermedia under daytime conditions decreased after 7 weeks of exposure to press elevated ammonium concentrations of 14 µmol L−1. This is consistent with previous studies which found reduced calcification under high concentrations (10–109 µmol L−1) of ammonium20, 29, 32, and nitrate25, 26, 64. Other studies found increased skeletal growth for several coral species reared under mild concentrations of 2 and 5 µmol L−1 of ammonium and nitrate respectively65, or heterogenous effects under 11.3–36.2 µmol L−1 ammonium5. Similarly, studies on coral calcification (mainly linear expansion rates and buoyant weight measurements) under elevated phosphate have reported mixed effects for different coral species, describing both enhancement5, 66, 67 and reduction5, 20, 29, 32 under phosphate concentrations ranging from 0.5 to 13 µmol L−1. Skeletal surface area increase in the present study was reduced under elevated phosphate concentrations (10 µmol L−1), despite no observed changes in net CaCO3 accretion. This indicates a shift towards higher skeletal density and lower skeletal expansion, thus reducing the competitive ability of A. intermedia to occupy new territory, while gaining improved tolerance to physical damage and fragmentation68. Such shifts are notably beneficial in upwelling areas characterized by high nutrients but also intensified wave energy69.
Despite the highest PNET:RDARK ratios measured in the press N treatment, productivity was not significantly stimulated under elevated N. Previous studies report up to 5 times higher symbiont densities, and 150% increase in primary productivity under elevated ammonium concentrations20, 24, 47, identifying nitrogen availability as a limiting factor for productivity alongside bicarbonate21, 70. Possible benefits of increased nitrogen availability could have been balanced by increased symbiont competition for other resources27, 30. Similarly, productivity decreased under press phosphate addition, possibly due to higher symbiont densities and increased self-shading24, 71. Excess photosynthetic carbon could have been stored as somatic lipid reserves58, explaining elevated tissue lipid but not protein content under increased ammonium. Alternatively, excess autotrophic carbon could have been allocated towards other processes not assessed here such as increased host mucus production72 or symbiont population16, 47. There is growing evidence that symbiont growth reduces carbon translocation to the host, and thereby influences host processes such as skeletogenesis16, 28,29,30. Strong competition for DIC between a larger symbiont population (for photosynthesis) and the host (for calcification) could explain reduced coral skeletal expansion and CaCO3 accretion rates under press elevated nutrients.
The accumulation of organic carbon in the holobiont tissue was unchanged by the addition of ammonium or phosphate individually but increased when the two were added as a combined treatment. This suggests co-limitation of these two nutrients to biomass production in the holobiont23, 66, 73 under ambient conditions on Heron Island. Tissue TOC and lipid content was further increased under the pulse NP treatment, where the highest tissue TOC and lipid content were recorded. Interestingly, holobiont N:P ratios were unaffected by the addition of ammonium and/or phosphate, neither as press nor pulse treatments, despite the imbalanced N:P ratio of the seawater (Table S1). This suggests no differential increase in the assimilation of these nutrients. Muller-Parker et al. (1994a) reported N enrichment in Symbiodiniaceae but not in the host after 8 weeks exposure to 20–50 µmol L−1 ammonium, further supporting that nutrients are primarily utilized by Symbiodiniaceae, and that nutrient enrichment benefits the symbiont rather than the host. The uptake of ammonium or phosphate and subsequent shift in holobiont N:P ratio may have been diluted here, given that N and P are predominantly assimilated by Symbiodiniaceae and the microbial community of the holobiont74, 75, which occupy a relatively small fraction of the holobiont mass22, and have limited nutrient storage capabilities76.
Benefits of episodic nutrient addition
Concentrations of inorganic N and P in seawater overlying coral reefs are generally low, and nutrient enrichment has often been negatively associated with coral growth5, 26, 48. Interestingly, fish-derived nutrients are known to improve coral physiology37, 38, 43, 44, yet the underlying stoichiometry and physiological mechanisms are not yet investigated48. Shantz and Burkepile (2014) concluded that anthropogenic nutrient enrichment tended to negatively influence overall coral performance while biologically mediated nutrients generally had positive effects, and hypothesized that the pulsed nature of natural nutrient enrichment could be responsible. Corals under unperturbed conditions are naturally accustomed to high concentration nutrient pulses37, 38, 77, but the daily migratory and feeding behaviour of reef organisms mean that these nutrients fluctuate considerably37, 38, simulated by our pulse treatment. We provide further evidence that short, strong nutrient pulses benefit A. intermedia compared to continuously elevated nutrients, and for some parameters even compared to unmanipulated control conditions. Rates of day and nighttime GTA, PNET:RDARK, fragment surface area, and holobiont tissue TOC and lipid content were all distinctly higher in the pulse NP treatment compared to the press NP treatment, while lipid concentration, holobiont TOC content, night-time GTA and fragment surface area even increased compared to the control treatment.
Pulsed addition of anthropogenic nutrients has previously been found to positively influence coral communities5 as well as other aquatic ecosystems such as seagrass beds and macroalgae communities78, 79. Our results indicate that the brevity of the pulse possibly plays a key role in the coral’s ability to manage high nutrient concentrations. Non-linearity between continuous nutrient exposure and coral calcification has previously been shown, suggesting that low nutrient doses (i.e. nutrient pulse in our study) enhanced calcification while high doses (i.e. nutrient press) reduced calcification80. Rapid uptake and assimilation of phosphate and ammonium by Symbiodiniaceae81, oxidation of ammonium to nitrite and nitrate by the coral microbial associates74, 75, or direct host ammonium assimilation through the GS/GDH pathway82 all potentially mitigated nutrient impact during short pulses, allowing corals to profit. Overall, fragments that received nutrient pulses had improved growth (both as CaCO3 accretion and skeleton expansion) and energy reserves, without trade-offs in other fields such as productivity. On the other hand, corals under press elevated N and/or P were able to consolidate their productivity, tissue protein and lipid stores, but at the expense of much of their skeletal growth (Fig. 3). Such trade-offs could result in immediate changes to the competitive balance within reef communities and compromise long-term coral viability34, 83, 84.
Conclusions
Inorganic nutrients remain important factors in influencing physiology in coral-dinoflagellate symbioses. The present study provides insights into the stoichiometry of two of the most important inorganic nutrients, ammonium and phosphate, and their individual and combined influence on parameters of skeletogenesis, photobiology and tissue properties in A. intermedia. At the same time, ammonium and phosphate concentrations that would normally compromise coral health if maintained permanently, are shown to benefit corals when administered in shorter pulses. Together, these results underline the complex and context-specific relationship between coral holobiont physiology and elevated nutrients.
References
Schaffelke, B., Carleton, J., Skuza, M., Zagorskis, I. & Furnas, M. J. Water quality in the inshore Great Barrier Reef lagoon: implications for long-term monitoring and management. Mar. Pollut. Bull. 65, 249–260 (2012).
Kleypas, J. A., McManus, J. W. & Meñez, L. A. B. Environmental limits to coral reef development: Where do we draw the line?. Am. Zool. 39, 146–159 (1999).
Barnes, D. J. & Devereux, M. J. Productivity and calcification on a coral reef: A survey using pH and oxygen electrode techniques. J. Exp. Mar. Biol. Ecol. 79, 213–231 (1984).
Hoegh-Guldberg, O. & Williamson, J. Availability of two forms of dissolved nitrogen to the coral Pocillopora damicornis and its symbiotic zooxanthellae. Mar. Biol. 133, 561–570 (1999).
Koop, K. et al. ENCORE: The effect of nutrient enrichment on coral reefs. Synthesis of results and conclusions. Mar. Pollut. Bull. 42, 91–120 (2001).
Grover, R., Maguer, J.-F., Reynaud-Vaganay, S. & Ferrier-Pagès, C. Uptake of ammonium by the scleractinian coral Stylophora pistillata : effect of feeding, light, and ammonium concentrations. Limnol. Oceanogr. 47, 782–790 (2002).
Grover, R., Maguer, J.-F., Allemand, D. & Ferrier-Pagès, C. Uptake of dissolved free amino acids by the scleractinian coral Stylophora pistillata. J. Exp. Biol. 211, 860–865 (2008).
Godinot, C., Ferrier-Pagés, C. & Grover, R. Control of phosphate uptake by zooxanthellae and host cells in the scleractinian coral Stylophora pistillata. Limnol. Oceanogr. 54, 1627–1633 (2009).
Wang, J. & Douglas, A. Nitrogen recycling or nitrogen conservation in an alga-invertebrate symbiosis?. J. Exp. Biol. 201, 2445–2453 (1998).
Davy, S. K., Allemand, D. & Weis, V. M. Cell biology of cnidarian-dinoflagellate symbiosis. Microbiol. Mol. Biol. Rev. 76, 229–261 (2012).
Tanaka, Y., Suzuki, A. & Sakai, K. The stoichiometry of coral-dinoflagellate symbiosis: carbon and nitrogen cycles are balanced in the recycling and double translocation system. ISME J. https://doi.org/10.1038/s41396-017-0019-3 (2018).
Lesser, M. P. et al. Nitrogen fixation by symbiotic cyanobacteria provides a source of nitrogen for the scleractinian coral Montastraea cavernosa. Mar. Ecol. Prog. Ser. 346, 143–152 (2007).
Miller, D. J. & Yellowlees, D. Inorganic nitrogen uptake by symbiotic marine cnidarians: a critical review. Proc. R. Soc. B Biol. Sci. 237, 109–125 (1989).
Pernice, M. et al. A single-cell view of ammonium assimilation in coral–dinoflagellate symbiosis. ISME J. 6, 1314–1324 (2012).
Yellowlees, D., Rees, T. A. V. & Leggat, W. Metabolic interactions between algal symbionts and invertebrate hosts. Plant, Cell Environ. 31, 679–694 (2008).
Krueger, T. et al. Intracellular competition for nitrogen controls dinoflagellate population density in corals. Proc. R. Soc. B Biol. Sci. 287, 20200049 (2020).
Godinot, C., Houlbrèque, F., Grover, R., Ferrier-Pagès, C. & Larsen, A. Coral uptake of inorganic phosphorus and nitrogen negatively affected by simultaneous changes in temperature and pH. PLoS ONE 6, e25024 (2011).
Ferrier-Pagès, C., Godinot, C., D’Angelo, C., Wiedenmann, J. & Grover, R. Phosphorus metabolism of reef organisms with algal symbionts. Ecol. Monogr. 86, 262–277 (2016).
Snidvongs, A. & Kinzie, R. A. Effects of nitrogen and phosphorus enrichement on in vivo symbiotic zooxanthellae of Pocillopora damicornis. Mar. Biol. 118, 705–711 (1994).
Ferrier-Pagès, C., Gattuso, J. P., Dallot, S. & Jaubert, J. Effect of nutrient enrichment on growth and photosynthesis of the zooxanthellate coral Stylophora pistillata. Coral Reefs 19, 103–113 (2000).
Roberty, S., Béraud, E., Grover, R. & Ferrier-Pagès, C. Coral productivity is co-limited by bicarbonate and ammonium availability. Microorganisms 8, 640 (2020).
Muller-Parker, G., Cook, C. B. & D’elia, C. F. Elemental composition of the coral Pocillopora damicornis exposed to elevated seawater ammonium. Pac. Sci. 48, 234–246 (1994).
Muller-Parker, G., McCloskey, L., Hoegh-Guldberg, O. & McAuley, P. Effect of ammonium enrichment on animal and algal biomass of the coral Pocillopora damicornis. Pac. Sci. 48, 273–283 (1994).
Dubinsky, Z. et al. The effect of external nutrient resources on the optical properties and photosynthetic efficiency of Stylophora pistillata. Proc. R. Soc. B Biol. Sci. 239, 231–246 (1990).
Marubini, F. & Davies, P. S. Nitrate increases zooxanthellae population density and reduces skeletogenesis in corals. Mar. Biol. 127, 319–328 (1996).
Silbiger, N. J. et al. Nutrient pollution disrupts key ecosystem functions on coral reefs. Proc. R. Soc. B Biol. Sci. 285, 20172718 (2018).
Morris, L. A., Voolstra, C. R., Quigley, K. M., Bourne, D. G. & Bay, L. K. Nutrient availability and metabolism affect the stability of coral-symbiodiniaceae symbioses. Trends Microbiol. 27, 678–689 (2019).
Baker, D. M., Freeman, C. J., Wong, J. C. Y., Fogel, M. L. & Knowlton, N. Climate change promotes parasitism in a coral symbiosis. ISME J. 12, 921–930 (2018).
Stambler, N., Popper, N., Dubinsky, Z. & Stimson, J. Effects of nutrient enrichment and water motion on the coral Pocillopora damicornis. Pac. Sci. 45, 299–307 (1991).
Rädecker, N. et al. Heat stress destabilizes symbiotic nutrient cycling in corals. Proc. Natl. Acad. Sci. U. S. A. 118, e2022653118 (2021).
Bassim, K. & Sammarco, P. Effects of temperature and ammonium on larval development and survivorship in a scleractinian coral (Diploria strigosa). Mar. Biol. 142, 241–252 (2003).
Langdon, C. & Atkinson, M. J. Effect of elevated pCO2 on photosynthesis and calcification of corals and interactions with seasonal change in temperature/irradiance and nutrient enrichment. J. Geophys. Res. 110, C09S07 (2005).
Rosset, S., Wiedenmann, J., Reed, A. J. & D’Angelo, C. Phosphate deficiency promotes coral bleaching and is reflected by the ultrastructure of symbiotic dinoflagellates. Mar. Pollut. Bull. 118, 180–187 (2017).
Lapointe, B. E., Brewton, R. A., Herren, L. W., Porter, J. W. & Hu, C. Nitrogen enrichment, altered stoichiometry, and coral reef decline at Looe Key, Florida Keys, USA: a 3-decade study. Mar. Biol. 166, 108 (2019).
Wiedenmann, J. et al. Nutrient enrichment can increase the susceptibility of reef corals to bleaching. Nat. Clim. Chang. 3, 160–164 (2013).
Meyer, J. L. & Schultz, E. T. Migrating haemulid fishes as a source of nutrients and organic matter on coral reefs1. Limnol. Oceanogr. 30, 146–156 (1985).
Holbrook, S. J., Brooks, A. J., Schmitt, R. J. & Stewart, H. L. Effects of sheltering fish on growth of their host corals. Mar. Biol. 155, 521–530 (2008).
Shantz, A. A., Ladd, M. C., Schrack, E. & Burkepile, D. E. Fish-derived nutrient hotspots shape coral reef benthic communities. Ecol. Appl. 25, 2142–2152 (2015).
Schmidt, S., Dennison, W. C., Moss, G. J. & Stewart, G. R. Nitrogen ecophysiology of Heron Island, a subtropical coral cay of the Great Barrier Reef, Australia. Funct. Plant Biol. 31, 517–528 (2004).
Staunton Smith, J. & Johnson, C. R. Nutrient inputs from seabirds and humans on a populated coral cay. Mar. Ecol. Prog. Ser. 124, 189–200 (1995).
Ezzat, L. et al. Nutrient starvation impairs the trophic plasticity of reef-building corals under ocean warming. Funct. Ecol. 33, 643–653 (2019).
Ezzat, L., Maguer, J. F., Grover, R. & Ferrier-Pagès, C. Limited phosphorus availability is the Achilles heel of tropical reef corals in a warming ocean. Sci. Rep. 6, 1–11 (2016).
Meyer, J. L. & Schultz, E. T. Tissue condition and growth rate of corals associated with schooling fish1. Limnol. Oceanogr. 30, 157–166 (1985).
Liberman, T., Genin, A. & Loya, Y. Effects on growth and reproduction of the coral Stylophora pistillata by the mutualistic damselfish Dascyllus marginatus. Mar. Biol. 121, 741–746 (1995).
Burkepile, D. E. et al. Nutrient supply from fishes facilitates macroalgae and suppresses corals in a Caribbean coral reef ecosystem. Sci. Rep. 3, 1493 (2013).
Burkepile, D. E. et al. Nitrogen identity drives differential impacts of nutrients on coral bleaching and mortality. Ecosystems https://doi.org/10.1007/s10021-019-00433-2 (2019).
Ezzat, L., Maguer, J. F., Grover, R. & Ferrier-Pagés, C. New insights into carbon acquisition and exchanges within the coral–dinoflagellate symbiosis under NH4+ and NO3− supply. Proc. R. Soc. B Biol. Sci. 282, 20150610 (2015).
Shantz, A. A. & Burkepile, D. E. Context-dependent effects of nutrient loading on the coral–algal mutualism. Ecology 95, 1995–2005 (2014).
Devlin, M. J. & Brodie, J. Terrestrial discharge into the Great Barrier Reef Lagoon: nutrient behavior in coastal waters. Mar. Pollut. Bull. 51, 9–22 (2005).
Bender, D., Diaz-Pulido, G. & Dove, S. The impact of CO 2 emission scenarios and nutrient enrichment on a common coral reef macroalga is modified by temporal effects. J. Phycol. 50, 203–215 (2014).
Wild, C., Woyt, H. & Huettel, M. Influence of coral mucus on nutrient fluxes in carbonate sands. Mar. Ecol. Prog. Ser. 287, 87–98 (2005).
Bender, E. A., Case, T. J. & Gilpin, M. E. Perturbation experiments in community ecology: theory and practice. Ecology 65, 1–13 (1984).
Parsons, T. R., Maita, Y. & Lalli, C. M. A Manual of Chemical and Biological Methods for Seawater Analysis (Pergamon Press, 1984).
Chisholm, J. R. M. & Gattuso, J.-P. Validation of the alkalinity anomaly technique for investigating calcification of photosynthesis in coral reef communities. Limnol. Oceanogr. 36, 1232–1239 (1991).
Dickson, A. G., Afghan, J. D. & Anderson, G. C. Reference materials for oceanic CO2 analysis: a method for the certification of total alkalinity. Mar. Chem. 80, 185–197 (2003).
Maier, C., Watremez, P., Taviani, M., Weinbauer, M. G. & Gattuso, J. P. Calcification rates and the effect of ocean acidification on Mediterranean cold-water corals. Proc. R. Soc. B Biol. Sci. 279, 1716–1723 (2012).
Whitaker, J. R. & Granum, P. E. An absolute method for protein determination based on difference in absorbance at 235 and 280 nm. Anal. Biochem. 109, 156–159 (1980).
Dunn, S. R., Thomas, M. C., Nette, G. W., Dove, S. G. & Blackburn, S. A lipidomic approach to understanding free fatty acid lipogenesis derived from dissolved inorganic carbon within Cnidarian-Dinoflagellate symbiosis. PLoS ONE 7, e46801 (2012).
van der Zande, R. M. et al. Paradise lost: end-of-century warming and acidification under business-as-usual emissions have severe consequences for symbiotic corals. Glob. Change Biol. 26, 2203–2219 (2020).
Gaffey, S. J. & Bronnimann, C. E. Effects of bleaching on organic and mineral phases in biogenic carbonates. J. Sediment. Res. 63, 752–754 (1993).
Veal, C. J., Carmi, M., Fine, M. & Hoegh-Guldberg, O. Increasing the accuracy of surface area estimation using single wax dipping of coral fragments. Coral Reefs 29, 893–897 (2010).
Underwood, A. Experiments in Ecology: Their Logical Design and Interpretation Using Analysis of Variance (Cambridge University Press, 1997). .
Wooldridge, S., Brodie, J. & Furnas, M. Exposure of inner-shelf reefs to nutrient enriched runoff entering the Great Barrier Reef Lagoon: post-European changes and the design of water quality targets. Mar. Pollut. Bull. 52, 1467–1479 (2006).
Ferrier-Pagès, C., Schoelzke, V., Jaubert, J., Muscatine, L. & Hoegh-Guldberg, O. Response of a scleractinian coral, Stylophora pistillata, to iron and nitrate enrichment. J. Exp. Mar. Bio. Ecol. 259, 249–261 (2001).
Atkinson, M. J., Carlson, B. & Crow, G. L. Coral growth in high-nutrient, low-pH seawater: a case study of corals cultured at the Waikiki Aquarium, Honolulu, Hawaii. Coral Reefs 14, 215–223 (1995).
Godinot, C., Ferrier-Pagès, C., Montagna, P. & Grover, R. Tissue and skeletal changes in the scleractinian coral Stylophora pistillata Esper 1797 under phosphate enrichment. J. Exp. Mar. Biol. Ecol. 409, 200–207 (2011).
Dunn, J. G., Sammarco, P. W. & LaFleur, G. Effects of phosphate on growth and skeletal density in the scleractinian coral Acropora muricata: a controlled experimental approach. J. Exp. Mar. Biol. Ecol. 411, 34–44 (2012).
Marshall, P. A. Skeletal damage in reef corals: relating resistance to colony morphology. Mar. Ecol. Prog. Ser. 200, 177–189 (2000).
Andrews, J. C. & Gentien, P. Upwelling as a source of nutrients for the Great Barrier Reef ecosystems: A solution to Darwin’s question?. Mar. Ecol. Prog. Ser. 8, 257–269 (1982).
Marubini, F. & Thake, B. Bicarbonate addition promotes coral growth. Limnol. Oceanogr. 44, 716–720 (1999).
Hoegh-Guldberg, O. & Smith, G. J. The effect of sudden changes in temperature, light and salinity on the population density and export of zooxanthellae from the reef corals Stylophora pistillata Esper and Seriatopora hystrix Dana. J. Exp. Mar. Bio. Ecol. 129, 279–303 (1989).
Quinlan, Z. A. et al. Fluorescent organic exudates of corals and algae in tropical reefs are compositionally distinct and increase with nutrient enrichment. Limnol. Oceanogr. Lett. 3, 331–340 (2018).
Tanaka, Y., Grottoli, A., Matsui, Y., Suzuki, A. & Sakai, K. Effects of nitrate and phosphate availability on the tissues and carbonate skeleton of scleractinian corals. Mar. Ecol. Prog. Ser. 570, 101–112 (2017).
Siboni, N., Ben-Dov, E., Sivan, A. & Kushmaro, A. Global distribution and diversity of coral-associated Archaea and their possible role in the coral holobiont nitrogen cycle. Environ. Microbiol. 10, 2979–2990 (2008).
Rädecker, N., Pogoreutz, C., Voolstra, C. R., Wiedenmann, J. & Wild, C. Nitrogen cycling in corals: the key to understanding holobiont functioning?. Trends Microbiol. 23, 490–497 (2015).
Kopp, C. et al. Highly dynamic cellular-level response of symbiotic coral to a sudden increase in environmental nitrogen. MBio 4, e00052-e113 (2013).
Meyer, J. L., Schultz, E. T. & Helfman, G. S. Fish schools: an asset to corals. Science 220, 1047–1049 (1983).
Fong, C. R. & Fong, P. Nutrient fluctuations in marine systems: press versus pulse nutrient subsidies affect producer competition and diversity in estuaries and coral reefs. Estuaries Coasts 41, 421–429 (2018).
Allgeier, J. E., Burkepile, D. E. & Layman, C. A. Animal pee in the sea: consumer-mediated nutrient dynamics in the world’s changing oceans. Glob. Change Biol. 23, 2166–2178 (2017).
Gil, M. A. Unity through nonlinearity: a unimodal coral–nutrient interaction. Ecology 94, 1871–1877 (2013).
McAuley, P. J. & Smith, V. J. Effect of diel photoperiod on nitrogen metabolism of cultured and symbiotic zooxanthellae. Mar. Biol. 123, 145–152 (1995).
Bruggeman, F. J., Boogerd, F. C. & Westerhoff, H. V. The multifarious short-term regulation of ammonium assimilation of Escherichia coli: dissection using an in silico replica. FEBS J. 272, 1965–1985 (2005).
D’Angelo, C. & Wiedenmann, J. Impacts of nutrient enrichment on coral reefs: new perspectives and implications for coastal management and reef survival. Curr. Opin. Environ. Sustain. 7, 82–93 (2014).
Vega Thurber, R. L. et al. Chronic nutrient enrichment increases prevalence and severity of coral disease and bleaching. Glob. Change Biol. 20, 544–554 (2014).
Acknowledgements
This research was funded by the Australian Research Council (ARC) and the NOAA Coral Reef Watch, an ARC Laureate grant FL120100066 (OHG) and an ARC Centre of Excellence for Coral Reef Studies Grant CE0561435 (SD and OHG), a University of Queensland International Scholarship UQI (RZ) and a Holsworth Wildlife Research Endowment grant by Equity Trustees Charitable Foundation (RZ). We would like to thank the HIRS staff, A. Chai and G. Bernal-Carillo for in-field assistance. We are grateful to A. Kubicek and E. Bayraktarov for advice, M. Achlatis for feedback, and D. Appleton from the School of Agriculture and Food Sciences at The University of Queensland for the elemental ratio analyses. All research was conducted under permit no. G14/37212.1 issued by the GBR Marine Park Authority.
Author information
Authors and Affiliations
Contributions
R.Z., S.D. and O.H.G. conceived and designed the study, with advice from D.B.C. R.Z. and Y.M. performed the experiment. Data were analyzed by R.Z., Y.M. and S.D., and D.B.C. and O.H.G. contributed to data interpretation. R.Z. wrote the manuscript with all co-authors contributing to its final form.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
van der Zande, R.M., Mulders, Y.R., Bender-Champ, D. et al. Asymmetric physiological response of a reef-building coral to pulsed versus continuous addition of inorganic nutrients. Sci Rep 11, 13165 (2021). https://doi.org/10.1038/s41598-021-92276-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-92276-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.