Abstract
Short-term changes in dietary intake can induce changes in gut microbiome. While various dietary polyphenols have been shown to modulate gut microflora, the acute influence of polyphenol-rich mixed spices has not been explored in a controlled setting. We investigated the effects of a single serving of mixed spices Indian curry consumption, in two separate doses, on the gut microbiome in 15 healthy, Singaporean Chinese males, with age and BMI of 23.5 ± 2.4 years and 22.9 ± 2.2 kg/m2 respectively. We found that a low-polyphenol, no spices Dose 0 Control (D0C) meal led to an increase in Bacteroides and a decrease in Bifidobacterium. In comparison to D0C, there was significant suppression of Bacteroides (p < 0.05) and an increase in Bifidobacterium (p < 0.05) with increasing doses of curry meal Dose 1 Curry (D1C) and Dose 2 Curry (D2C) containing 6 g and 12 g mixed spices respectively. Significant correlations were also found between bacterial changes and plasma phenolic acids. No differences between treatments were observed in the alpha-diversity of the gut microflora. This study has shown that a single serving of mixed spices can significantly modify/restore certain commensal microbes, particularly in people who do not regularly consume these spices.
Similar content being viewed by others
Introduction
The gut microbiome has been widely acknowledged as a dynamic and complex community of bacteria that is dependent on extrinsic and intrinsic factors, of which diet is one of the most dominant modulators1,2,3. Several previous studies have shown that dietary patterns can affect gut microbiome4,5,6,7. Numerous studies indicate fruits8,9,10 and/or vegetables11,12,13,14 as well as whole grain cereals12,15 can directly affect gut microflora composition. Besides these dietary patterns, foods and established nutrients, various ‘non-nutrient bioactive compounds’ in diet, including polyphenols, can significantly modulate gut microflora composition16,17,18. Spices that are often used as culinary ingredients for flavouring, colouring or prolonging food shelf-life19 are rich in polyphenols that are generally poorly absorbed by the upper intestine and present in relatively high concentrations in the lower gut20. Therefore, they are expected to interact and modulate the gut microbiome.
Depending on the polyphenol type, they may either have antimicrobial effects towards certain gut bacteria, whereas, stimulatory effects on other types of bacteria21,22, as well as having the ability to alter the ratios of Firmicutes/Bacteroidetes23 and Prevotella/Bacteroides24. These studies highlighted the potential modulatory role of polyphenol-rich spices on the composition of the human gut microbiome. Given that nowadays, spices are widely consumed globally, albeit on an occasional basis, it is therefore important to investigate the acute effects of spice consumption on the gut microbiome. Most previous research examined the effect of single spices or polyphenolic extracts from single spices such as curcumin on both human subjects and in animal studies18,21,25,26. However, few prior studies have examined the effects of introducing a dietary concoction of several spices in combination, which is how they are typically consumed in different curries across various cultures worldwide. To the best of our knowledge, only one recent study has attempted to test the effects of five different mixed spices in combination (cinnamon, oregano, ginger, rosemary, black pepper and cayenne pepper), in dietary doses, on gut microbiome and found that up to 26 operational taxonomic units (OTUs) were modulated as a result of the mixed-spice treatment compared with placebo27.
Therefore, we investigated the acute effects on gut microflora of a combination of 7 different spices as a secondary analysis of a recently completed study that measured various metabolic outcomes28,29. Considering that this study was undertaken in an acute setting with two separate mixed spices doses, it provided an ideal setting to investigate acute, dose-dependent changes in gut microbiome as a result of the consumption of these mixed spices. From this same study, since we have previously reported dose-dependent increases in two phenolic acids (i.e., cinnamic acid (CNA) and phenylacetic acid (PAA)) as well as increase in the total polyphenol excretion (TPE)29,30, we have also investigated the associations between these parameters as objective markers of the spices intake with the changes in the gut microbiome.
Results
Basic demographics and dietary intake 3-days prior to each session
The mean age and BMI of the participants at the first session who provided the stool samples were 23.5 ± 2.4 years and 22.9 ± 2.2 kg/m2 respectively. The reported intakes of energy and some key nutrients during the 3-day ‘run-in periods’ immediately prior to the main study day (Day 1) are shown in Table 1. There were no significant differences between the intervention sessions in any of the nutrients or energy intake.
Changes in microbiome composition
There were no significant differences in the relative abundances of the individual bacterial genera at baseline (Day 0) or at Day 2 as calculated using Friedman’s test with Dunn’s multiple comparison as shown in Fig. 1 and Supplementary Tables S1 and S2. The alpha diversity of each spices intervention is shown in Fig. 2. No significant differences were found between the various intervention sessions for both the Chao 1 and Shannon diversity indices.
Comparison of the relative abundance of bacterial genera for the three different doses of spices intervention at Day 0 (baseline) (a-c) and Day 2 (d-f) for D0C, D1C and D2C respectively. ‘Other’ was categorised as less than 1% of bacterial OTUs among the population. Bacteria at baseline or at Day 2 were not significant different between intervention sessions. D0C = Meal with no spices intervention, D1C = Meal with low (6 g) mixed spices intervention, D2C = Meal with high (12 g) mixed spices intervention, UG unknown genus, UF unknown family. n = 15 each intervention.
Alpha diversity of the three different doses of spices intervention (a) Chao 1’s and (b) Shannon’s indexes were measured and described at Y axis. No significant difference between spices intervention were found by Friedman’s test with Dunn’s multiple comparison within Day 0 or Day 2. The types and days of intervention were drawn as different symbols and colours. + shows mean. D0C = Meal with no spices intervention, D1C = Meal with low (6 g) mixed spices intervention, D2C = Meal with high (12 g) mixed spices intervention. No of samples were in the parenthesis.
However, the spice intervention led to significant changes in the relative abundances of two bacterial genera. The changes from Day 0 to Day 2 in the relative abundance of the various bacterial genus (Operational Taxonomic Units: OTUs) during each of the three intervention sessions are shown as a heatmap in Fig. 3.
Heatmap of medians relative abundance changes of the major bacterial genera after two days of intervention for the three doses of mixed spices intervention. ‘Other’ was categorised as less than 1% of bacterial OTUs among the population. The relative abundance changes (Day 2 minus Day 0) in bacteria between various doses were analysed using Friedman’s test with Dunn’s multiple comparison and described as * adjusted p < 0.05.
As can be seen in the heatmap (Fig. 3), compared with the control, the relative abundance of Bacteroides was significantly reduced with increasing spice doses (adjusted p < 0.05 for both D0C vs D1C and D0C vs D2C). This was mainly driven by the fact that there was an increase in Bacteroides relative abundance changes from Day 0 to Day 2 during the D0C session, whereas the Bacteroides relative abundance from Day 0 to Day 2 either remained the same or decreased slightly during D1C and D2C sessions. On the contrary, the relative abundances of Bifidobacterium (adjusted p < 0.05 for D0C vs D2C) increased significantly with spice intake as compared with control. This was also driven by the fact that there was a decrease in Bifidobacterium relative abundance changes from Day 0 to Day 2 during the D0C session, whereas the Bifidobacterium relative abundance from Day 0 to Day 2 either remained the same or increased slightly during D1C and D2C sessions. The extent and the variabilities in the relative abundances in these two genera during the various intervention sessions at Day 0 and 2 are shown in Fig. 4 as median and inter-quartile range (IQR). The paired individual changes in relative abundances between two time points (Day 0 to Day 2) for three spices doses in these two genera is shown in Fig. 5.
Relative abundance distribution of (a) Bacteroides and (b) Bifidobacterium at Day 0 and Day 2 for three doses of mixed spices intervention. No bacteria were significantly different between three different doses of spices intervention analysed by Friedman’s test with Dunn’s multiple comparison. D0C = Meal with no spices intervention, D1C = Meal with low (6 g) mixed spices intervention, D2C = Meal with high (12 g) mixed spices intervention. UG unknown genus, UF unknown family. No of samples (each intervention) was 15 and described in the parenthesis.
Paired individual changes in relative abundance between two time points (Day 0 to Day 2) for three spices doses (a) Bacteroides and (b) Bifidobacterium. Wilcoxon matched-pairs signed rank test was applied and significant pairs were described as * p < 0.05. D0C = Meal with no spices intervention, D1C = Meal with low (6 g) mixed spices intervention, D2C = Meal with high (12 g) mixed spices intervention. n = 15 each intervention. Dashed line indicates median change.
Figure 6 shows the changes in relative abundances of (a) Bacteroides and (b) Bifidobacterium in relation to their absolute relative abundance at baseline (Day 0) for all three doses. We found a significant correlation (R2 = 0.284, p = 0.041) for D2C in Bacteroides but, no such correlation for D0C. On the other hand, those with low Bifidobacterium relative abundance at baseline had the greatest changes in relative abundances for D0C (R2 = 0.577, p = 0.001) and D2C (R2 = 0.433, p = 0.008).
Changes in relative abundances of (a) Bacteroides and (b) Bifidobacterium in relation to baseline relative abundance for the three doses. Simple linear regression was applied and R2 and p values were described for the group that had *p < 0.05. The relative abundance of bacteria at Day 0 and relative abundance changes were represented at X and Y axes, respectively. The colours represent the respective spices intervention session. D0C = Meal with no spices intervention, D1C = Meal with low (6 g) mixed spices intervention, D2C = Meal with high (12 g) mixed spices intervention. No of samples (each intervention) was 15.
Association between changes of bacterial genus and two plasma phenolic acids and changes urinary total polyphenol excretion in 24 h
As shown in Table 2, when we pooled the data across all intervention time points, we found significant positive associations between plasma concentration (on Day 1) of CNA and Bifidobacterium whereas negative associations between plasma CNA and Bacteroides, Ruminococcus and Oscillospira. Similarly, plasma PAA concentration was also negatively associated with Bacteroides, Lachnospira and Phascolarctobacterium. On the contrary, only Coprococcus, had a significant negative association with changes in 24 h urinary total polyphenol excretion (Day 1–Day 0).
Discussion
While polyphenolic compounds in diet are known to modulate gut microbiome16,17,18, metabolism of dietary polyphenols also rely on the gut microbiome to exert their biological effects in humans, particularly for their breakdown into secondary metabolites and subsequent absorption31. This bi-directional association between dietary polyphenols and the gut microbiome is integral to the host-gut bacteria symbiosis. The present study is one of the few studies in humans investigating the influence of polyphenol-rich mixed spices, contained within curry meals, in two separate doses on acute changes in gut microflora. To the best of our knowledge, there has been only one other similar previous study undertaken with mixed spices in humans27. While this previous study by Lu et al. explored the effects of mixed spices consumption (5 g/day) over a longer time frame (2 weeks), our study showed that even a single meal containing mixed spices can modify/restore gut microflora within a period of 24–48 h. Moreover, our study had a rigorous study design of avoiding polyphenol-rich foods 3-days prior to each intervention session (i.e., during run-in period) and the rest of the meals during the measurement days apart from the intervention (D0C, D1C, and D2C) test meals were standardized and provided. Given that our study was undertaken in a Chinese population who otherwise consumed Indian mixed spices less frequently, it minimised any residual confounding due to prior habituation of mixed spices intake. It appears from our results that a continuous adherence of a low polyphenol diet during the D0C (control) session led to an increase in the relative abundance of Bacteroides and a decrease in the relative abundance of Bifidobacterium between Days 0 to 2 of the intervention, whereas the re-introduction of polyphenol rich spices at the two doses of curry somewhat restored/reversed this trend within this same time frame. The rapid time frame of this change in gut microbiome as a result of a single bout of curry, at 2 separate doses, is not surprising given that previous studies have also shown alterations in gut microflora within a 24-h time scale32,33,34.
Prior to their human trial, Lu et al. undertook a separate study in vitro which showed prebiotic potential, altering greater than 120 bacterial species, including the growth promotion of certain probiotics such as Bifidobacterium spp. and inhibition of growth of pathogenic bacteria such as Clostridium spp.35. In their human trial, the same authors found that the mixed spices affected 26 OTUs, including growth promotions of Bifidobacteria spp., Lactobacilli spp., Bacteroides spp. and inhibition of Clostridia spp.27. In our trial, while Bifidobacterium also increased in the highest dose of mixed spices (D2C) vs control (D0C), there were significant reductions in Bacteroides (in both D0C and D2C vs D0C) populations with increasing mixed spices dose. The variations in the findings between the two studies may be due to differences in the background diet, the initial bacterial populations and/or differences in host genetics. Prevotella and Bacteroides are the two main genera of the same Bacteroidetes phylum and are highly abundant in human stool samples36. It is generally accepted that vegetable-rich diets tend to give rise to a greater abundance of Prevotella whereas animal-based diets tend to be enriched in Bacteroides37,38. Similarly, vegetarians and vegans have greater Prevotella/Bacteroides ratio than Omnivore populations39,40, as well as rural populations having a greater Prevotella/Bacteroides ratio as compared with urban populations34,41. However, we did not find any differences in Prevotella/Bacteroides ratio in our study most likely due to dietary fibre in the above-mentioned dietary patterns driving such effects.
Indeed, previous studies found that increasing the intake of various polyphenols, such as dealcoholized red wine extracts rich in resveratrol42 and pomegranate rich extracts43 led to increases in Prevotella populations, although we did not observe this effect within our study population. In support of our study, a previous study reported that in vitro incubation of dietary polyphenolic components such as chlorogenic acid, caffeic acid, and rutin reduced the Bacteroides population under anaerobic conditions44. In vivo studies in rats also found a decrease in Bacteroides population upon feeding of polyphenol-rich cocoa45. Even within our lab, we have shown that tea phenolic metabolites can extensively suppress the growth of Bacteroides22. Thus, taking into account these previous findings, the modulation of Bacteroides by polyphenol-rich mixed spices observed in our present study is likely to be causally associated and further studies are needed to confirm this. It should also be noted that the median reduction in relative abundance of Bacteroides was driven by a subset of voulnteers (Fig. 5a) who mainly had higher level of this bacteria at Day 0 as discussed in detail below and presented in Fig. 6a. This is further supported by no differences in the median Bacteroides relative abundance between various spice dose at Day 2 (nor Day 0 as expected, as shown in Fig. 5a). The negative associations between both CNA and PAA with Bacteroides further reiterates the decreases in the relative abundance of this bacteria with increasing doses of mixed spice rich curries may well be causally linked, as we have previously shown that the concentrations of both CNA and PAA could be used as objective biomarkers of the intake of the mixed spices used in our study30.
Several previous studies have indicated that polyphenol-rich foods can increase the abundance of Bifidobacterium46. Among the randomized controlled trials, one study with dealcoholized red wine showed significant increases in Bifidobacterium47, which was in fact further associated with increases in secondary metabolites of anthocyanins including several phenolic acids48. Similarly, consumption of wild blueberries49 as well as tart cherries50 were also shown to increase Bifidobacteria. Furthermore, the study by Lu et al.27 with five mixed spices, mentioned earlier, also found increases in the abundance of several Bifidobacteria spp. This, together with the findings of our study of the increases in Bifidobacteria with the highest dose of spice (D2C) meal indicates that polyphenol-rich spices may have bifidogenic properties. Hence, our study supports the concept of polyphenols having prebiotic properties, as discussed previously51. However, it should also be noted that the median increase in relative abundance of Bifidobacterium at higher dose of spices was mainly driven by only a subset of voulnteers, with low relative abundance at baseline (as shown in Fig. 5b), as discussed in more detail below (and shown in Fig. 6b). This is further supported by no differences in the median Bifidobacterium relative abundance between various spice dose at Day 2 (nor Day 0 as expected, as shown in Fig. 5b). Nonetheless, the positive association between plasma CNA and Bifidobacteria could also be a causal effect, since various hydroxycinnamic acids such as chlorogenic acid and caffeic acid have been previously shown to stimulate the growth of Bifidobacteria44.
In our study, the consumption of curry meals did not alter the alpha diversity of the gut microbiome of the tested subjects (Fig. 1). This may be due to the acute nature of the intervention as well as human gut microbiome, particularly the core members which account for most of the abundance in the gut microbiome that has been found to possess a high functional response diversity through functionally genetic redundancy52. This allows them to adapt well to short-term changes in the diet, which could explain why there were rarely large-scale changes in gut microbiota profile despite daily and seasonal variation in dietary ingredients and preparation53,54. Despite this adaptive capability, we have shown that a single bout of mixed spices can lead to, or restore compared to control dose (D0C session) significant modulation of certain gut microbes, particularly at the higher dose of curry (i.e., D2C meal). However, it remains to be established whether similar changes occur following longer-term consumption of polyphenol-rich spices and whether such changes are stable over time. Indeed this is one of the main limitations of this study in that finding reported here only describes acute modifications in gut microbiome. Another limitation of this study is that we only looked at associations of gut microbial changes with specific targeted metabolites in plasma/urine. Therefore future studies with individual and/or mixed spices should not only be undertaken with a longer duration of dietary intervention but should also explore how the changes in gut microbiome may be resulting from global changes in the metabolome in plasma and/or stool samples, arising from the intake of specific foods and/or food groups, in order to determine known/unknown metabolites that may directly influence certain bacterial populations. Nonetheless, with the globalization of dietary habits and the increasing amount of spices being consumed globally, the data from our study indicate an opportunity for bringing about beneficial changes in the gut microbiome through the intake of polyphenol-rich spices. Moreover, characterization of the gut microbiome across various parts of the world and various ethnic groups and/or cultures is an expanding area of research43,55 and further work needs to be done on the extent to which specific diet and lifestyle practices can modulate microbiome within these populations. Importantly, the functional consequences of the modulation of the microbiota, including what constitutes a ‘healthy microbiome’ are yet to be fully established56,57. Furthermore, recent reviews highlighted the utility of these polyphenol-associated gut microbiota changes on several anthropometric and clinical parameters related to cardiometabolic health18,58,59 and therefore changes in gut microbiome may be one of the major mechanisms through which dietary polyphenols may exert their widely reported benefits. The findings in our study also support these previous reports that some downstream metabolites of dietary polyphenols present in the mixed spices such as CNA and PAA may be directly influencing the bacterial changes observed with the intake of mixed spices used in our study.
Finally, the relative abundance at baseline of both Bacteroides and Bifidobacterium influenced the extent to which the changes in relative abundances of these two bacteria occurred at the various doses of curry. The regression plot in Fig. 6a shows a greater decrease in the relative abundance in Bacteroides in those individuals with a greater baseline relative abundance in this bacteria, particularly for Dose 2 curry, although this association was not present during the Dose 0 control session. Similarly, the regression plot in Fig. 6b shows that those with a lower relative abundance of Bifidobacterium had the greatest increase in the relative abundance (positive change) of the bacteria between Day 0 and Day 2 for both Dose 1 curry and Dose 2 curry. In comparison, majority of the individuals during Dose 0 control either had a reduction (negative change) in the Bifidobacterium relative abundance or did not change. This further highlights that the effects of dietary components on gut bacteria are likely to be dependent on the individual microbiome profile and hence may explain the inter-individual differences in responses to various dietary interventions.
Conclusions
This dose–response study has shown that even a single dose of mixed spices curry can modify/restore gut microbiome as compared with a control diet which was low in polyphyenols, within a 24–48-h timeframe. While the overall alpha-diversity of gut microbiome did not change as a result of this dietary intervention, there were in fact decrease/restoration in Bacteroides populations with increasing mixed spices doses relative to the dose 0 control (D0C) diet, when there was an increase in this bacteria. Similarly, our study also showed an increase/restoration in Bifidobacteria populations in the highest curry dose (D2C) compared with control (D0C), which further supports the bifidogenic potential of polyphenols. It should also be noted that the relative abundance changes in both Bacteroides and Bifidobacterium observed within each individual dose of spices (D0C, D1C and D2C) were mainly driven by a subset of individuals (Figs. 4 and 5), indicating large intra and inter-individual variabilities in responses to the dietary intervention. The reasons for this variability remains to be additionally investigated and was beyond the remit of this study. Furthermore, longer term studies are required to confirm our acute findings and a more detailed array of metabolites should be measured in order to establish mechanisms responsible for such changes.
Methods
Dietary intervention design
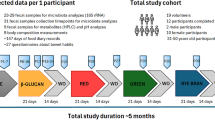
The study protocol, including the inclusion and exclusion criteria, has been detailed elsewhere15. In brief, the study was a randomized, crossover, acute, food based intervention trial specifically undertaken in 21–40-year-old, healthy, Chinese men, with a BMI between 18.5 and 27.5 kg/m2, who would otherwise typically not consume large amounts of Indian spices as part of their habitual diet. This secondary analysis was only undertaken in a subset of 15 volunteers who provided stool samples for the study and have completed all three doses of the dietary intervention. A per-protocol approach was utilized and data from three volunteers were excluded. These volunteers did not complete all 3 doses of intervention since we used matching analyses across all 3 doses. The study was approved by the Domain Specific Research Board (DSRB) ethics committee, Singapore (Reference: C/2015/00729) and was registered at clinicaltrials.gov (ID: NCT02599272) and was undertaken in accordance with the Declaration of Helsinki, revised in 2013 and as per Singapore Good Clinical Practice Guidelines. Informed consents from all volunteers were obtained before the intervention study.
Each volunteer in the study undertook 3 intervention sessions which were completed in random order which was obtained using an online randomization generator (http://randomizer.org). These sessions included a Dose 0 Control (D0C, no spices) session, Dose 1 Curry (D1C, low spices) session, and the Dose 2 Curry (D2C, high spices) session. The test meals for the D1C and D2C sessions included a mixture of 7 dried spice powders at doses 6 g and 12 g respectively. The spice mix was made up of turmeric, cumin, coriander, amla (Indian gooseberry), cinnamon, clove, and cayenne pepper mixed in the ratios of 8:4:4:4:2:1:1 respectively. The individual polyphenols present in the ingredients used to prepare the various curries (D1C and D2C) or the control (D0C) are listed in Supplementary Table S3.
The schematics of the study design is shown in Supplementary Fig. S1. In the 3 days prior to the main study day (Day 1) when the mixed spice containing meals (D1C or D2C) or the control meal (D0C) were consumed, all participants were asked to avoid consumption of any spice or any other polyphenol-rich foods (i.e., the ‘run-in period’). To aid their compliance, a list of common foods rich in polyphenols and common spices were provided to them. Furthermore, a 3-day food diary was completed during the ‘run-in period’ for each study session to record all foods that they consumed to further facilitate the detection of erroneous spices or polyphenol-rich food consumption during these periods and to improve compliance to the dietary intervention. One day before the main study day of each session, the volunteers were required to provide their baseline (Day 0) stool sample prior to the food based intervention on Day 1. On the main study day (Day 1), in the morning after an overnight fast, the volunteer consumed one of 3 intervention meals (D0C, D1C or D2C test meals). The total energy, macronutrients as well as total vegetable contents of the 3 intervention meals were comparable as reported previously15. The rest of the meals and snacks on Day 1 were standardized and contained low amounts of polyphenol containing ingredients and no spices within them. On the following day (Day 2), another stool sample was provided. Therefore, Day 0 stool represented the gut microbiome of the participants prior to each intervention session, after abstaining from polyphenol-rich foods and spices for the previous three days, whereas, Day 2 stool sample represented the gut microbiome immediately after each dietary intervention. Between each study session, each participant had at least a 14-day ‘washout period’ during which the participants resumed their habitual diet. This was done to avoid any ‘carry-over’ effect from one intervention session to the next.
Polyphenol analyses in urine and plasma
Urine samples were also collected over 2 × 24 h periods between Day 0 to Day 2, in 2 × 3 L plastic containers (Simport, Canada), to measure total polyphenol excretion (TPE) using the Folin-Ciocalteu assay as described in detail previously29. ‘Day 0’ urine was collected in the first urine container from the morning of Day 0 morning until Day 1 morning immediately prior to the consumption of test meals. ‘Day 1’ urine was collected during the subsequent 24 h period immediately after the test meal consumption between Day 1 morning until Day 2 morning. Blood samples were also collected in K2 EDTA vacutainer tubes (BD, Franklin Lakes, NJ, USA) immediately after the consumption of test meals on Day 1, at regular intervals (12 time points) to measure concentrations of various phenolic acids in plasma using a UHPLC-MS/MS method, as described in detail previously30. The areas under the curve (AUC) of postprandial concentrations of plasma phenolic acids were calculated using the trapezoid method.
Stool sample collection and 16 s rRNA gene sequencing analysis
For each stool sample collection (Day 0 and Day 2), the volunteers were asked to provide approximately 5 g of stool sample which was dissolved in a universal tube containing 2 ml of RNAlater solution (Ambion, Inc., Texas, USA) and were analysed for gut microbiota profiling with 16 s rRNA gene sequencing. To do this, 0.2 ml of faecal homogenate from approximately 1 g of collected stool was extracted using the phenol–chloroform method after washing with Phosphate-buffered saline (PBS). Along with the glass-beads mechanical extraction, Tris-SDS, TE-saturated phenol (Sigma-Aldrich, Cor., St.Louis, Missouri, USA) and phenol/chloroform/isoamyl alcohol (25:24:1) solutions were used. DNA was precipitated with sodium acetate and isopropanol followed by washing with ethanol and eluted in TE. The quantified DNA was normalised to 12.5 ng and amplified with a primer set that targeted at the regions of v3 and v4 of the 16 s rRNA gene. Once the amplicons were purified with Agencourt AMPure XP beads (Beckman Coulter, Inc., Fullerton, CA, USA), they were amplified with the Nextera XT index primers (Illumina, Inc., San Diego, USA). The quantified and normalised library was denatured and spiked with the PhiX control library followed by sequencing in the Miseq system (Illumina, Inc., San Diego, USA).
Bioinformatics and statistical analyses
The 16 s rRNA gene sequence outputs were analysed with Quantitative Insights Into Microbial Ecology (QIIME) version 1.9.160. After joining the reads and quality filtering, the chimeric sequences were removed with USEARCH v6.161. The non-chimeric sequences were picked to get the operational taxonomic unit (OTU) using 97% similarity sequences of Greengenes v13_8 database. The assigned OTUs were summarised into the taxonomical category for the bacterial profiling of samples in three intervention sessions. Chao 1 and Shannon diversity indexes of alpha diversity were computed using the OTU table. The 3-day food diary data were converted into the average energy and nutrients values per day by an online tool of energy and nutrient composition of food Health Promotion Board, Singapore62.
The bacterial genus taxonomy data, alpha diversity data and the dietary nutrients data were further, analysed and normality tests were performed. Bacterial genus with more than 1% of the relative abundance of the total bacterial population were included in this study and 24 major bacterial genera were found. Mean and Standard Deviation (SD) were described for the average energy and nutrients such as energy (kcal), protein (g), total fats (g), saturated fat (g), fibre (g), carbohydrate (g), cholesterol (mg) and sodium (mg) per day. Friedman’s test with Dunn’s multiple comparison tests were compared the different intervention groups at baseline (Day 0). The consumption of the dietary energy and nutrients were analysed with one-way ANOVA mixed-effects analysis. For comparison of changes of major bacteria, abundances of Bacteroides and Bifidobacterium and Alpha diversity from Day 2 to Day 0 for the three spices intervention groups, Friedman with Dunn’s multiple comparison tests were performed. Paired individual changes of above two bacteria were analysed by Wilcoxon matched-pairs signed rank test. A non-parametric Spearman rho and two-tailed p values were calculated for the correlation between changes of three metabolites and relative abundance of bacterial genera. The changes in the relative abundances of two bacteria (Bacteroides and Bifidobacterium) in relation to their absolute abundance at baseline (Day 0) for the various curry doses (D0C, D1C and D2C) were analysed using simple linear regression. The comparison of relative abundance of major bacteria at baseline were provided and described as Mean ± SD and the intra-individual variation at baseline between the three doses is presented as mean % Coefficient of Variation (%CV). All the statistical analyses were performed by GraphPad Prism 8 (GraphPad Software Inc., San Diego, USA).
Data availability
The relevant data are provided in the paper and the raw sequencing data can be found at the EBI repository (accession no: PRJEB35853).
References
Khine Wei, W. T. et al. Gut microbiome of pre-adolescent children of two ethnicities residing in three distant cities. Sci. Rep. 9, 7831 (2019).
Rothschild, D. et al. Environment dominates over host genetics in shaping human gut microbiota. Nature 555(7695), 210–215 (2018).
Wen, L. & Duffy, A. Factors influencing the gut microbiota, inflammation, and type 2 diabetes. J. Nutr. 147(7), 1468S-1475S (2017).
Oluwagbemigun, K. et al. Dietary patterns are associated with serum metabolite patterns and their association is influenced by gut bacteria among older German adults. J. Nutr. 150(1), 149–158 (2020).
Garcia-Mantrana, I., Selma-Royo, M., Alcantara, C. & Collado, M. C. Shifts on gut microbiota associated to Mediterranean diet adherence and specific dietary intakes on general adult population. Front. Microbiol. 9, 890 (2018).
Barone, M. et al. Gut microbiome response to a modern paleolithic diet in a Western lifestyle context. PLoS ONE 14(8), e0220619 (2019).
Berding, K., Holscher, H. D., Arthur, A. E. & Donovan, S. M. Fecal microbiome composition and stability in 4- to 8-year old children is associated with dietary patterns and nutrient intake. J. Nutr. Biochem. 56, 165–174 (2018).
Trošt, K. et al. Host: Microbiome co-metabolic processing of dietary polyphenols- an acute, single blinded, cross-over study with different doses of apple polyphenols in healthy subjects. Food Res. Int. 112, 108–128 (2018).
Mosele, J. I. et al. Effect of daily intake of pomegranate juice on fecal microbiota and feces metabolites from healthy volunteers. Mol. Nutr. Food Res. 59(10), 1942–1953 (2015).
Chiu, H.-F. et al. Regulatory/modulatory effect of prune essence concentrate on intestinal function and blood lipids. Pharm. Biol. 55(1), 974–979 (2017).
Hiel, S. et al. Effects of a diet based on inulin-rich vegetables on gut health and nutritional behavior in healthy humans. Am. J. Clin. Nutr. 109(6), 1683–1695 (2019).
Kopf, J. C. et al. Role of whole grains versus fruits and vegetables in reducing subclinical inflammation and promoting gastrointestinal health in individuals affected by overweight and obesity: A randomized controlled trial. Nutr. J. 17(1), 72 (2018).
Klinder, A. et al. Impact of increasing fruit and vegetables and flavonoid intake on the human gut microbiota. Food Funct. 7(4), 1788–1796 (2016).
Clark, R. L. et al. Educational intervention improves fruit and vegetable intake in young adults with metabolic syndrome components. Nutr. Res. 62, 89–100 (2019).
Martínez, I. et al. Gut microbiome composition is linked to whole grain-induced immunological improvements. ISME J. 7(2), 269–280 (2013).
Duda-Chodak, A., Tarko, T., Satora, P. & Sroka, P. Interaction of dietary compounds, especially polyphenols, with the intestinal microbiota: A review. Eur. J. Nutr. 54(3), 325–341 (2015).
Selma, M. V., Espín, J. C. & Tomás-Barberán, F. A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 57(15), 6485–6501 (2009).
Loo, Y. T., Howell, K., Chan, M., Zhang, P. & Ng, K. Modulation of the human gut microbiota by phenolics and phenolic fiber-rich foods. Compr. Rev. Food Sci. Food Saf. 19, 1268–1298 (2020).
Viuda-Martos, M., Ruiz-Navajas, Y., Fernández-López, J. & Pérez-Álvarez, J. A. Spices as functional foods. Crit. Rev. Food Sci. Nutr. 51(1), 13–28 (2010).
Opara, E. I. & Chohan, M. Culinary herbs and spices: Their bioactive properties, the contribution of polyphenols and the challenges in deducing their true health benefits. Int. J. Mol. Sci. 15(10), 19183–19202 (2014).
Etxeberria, U. et al. Impact of polyphenols and polyphenol-rich dietary sources on gut microbiota composition. J. Agric. Food Chem. 61(40), 9517–9533 (2013).
Lee, H. C., Jenner, A. M., Low, C. S. & Lee, Y. K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 157, 876–884 (2006).
Cardona, F., Andrés-Lacueva, C., Tulipani, S., Tinahones, F. J. & Queipo-Ortuño, M. I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 24(8), 1415–1422 (2013).
Gorvitovskaia, A., Holmes, S. P. & Huse, S. M. Interpreting Prevotella and Bacteroides as biomarkers of diet and lifestyle. Microbiome. 4, 15 (2016).
Dueñas, M. et al. A survey of modulation of gut microbiota by dietary polyphenols. BioMed Res. Int. https://doi.org/10.1155/2015/850902 (2015).
Rastmanesh, R. High polyphenol, low probiotic diet for weight loss because of intestinal microbiota interaction. Chem. Biol. Interact. 189(1–2), 1–8 (2011).
Lu, Q.-Y. et al. Mixed spices at culinary doses have prebiotic effects in healthy adults: A pilot study. Nutrients 11(6), 1425 (2019).
Halder, S., Chia, S. C. & Henry, C. J. Polyphenol-rich curry made with mixed spices and vegetables increase postprandial plasma GLP-1 concentration in a dose-dependent manner. Eur. J. Nutr. 72, 297–300 (2018).
Haldar, S. et al. Polyphenol-rich curry made with mixed spices and vegetables benefits glucose homeostasis in Chinese males (Polyspice Study): A dose-response randomized controlled crossover trial. Eur. J. Nutr. 58(1), 301–313 (2019).
Haldar, S. et al. Dose-dependent increase in unconjugated cinnamic acid concentration in plasma following acute consumption of polyphenol rich curry in the polyspice study. Nutrients 10(7), 934 (2018).
Espín, J. C., González-Sarrías, A. & Tomás-Barberán, F. A. The gut microbiota: A key factor in the therapeutic effects of (poly) phenols. Biochem. Pharmacol. 139, 82–93 (2017).
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505(7484), 559–563 (2014).
David, A. L. et al. Host lifestyle affects human microbiota on daily timescales. Genome Biol. 15, R89 (2014).
Wu, G. D. et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 334, 105–108 (2011).
Lu, Q.-Y. et al. Prebiotic potential and chemical composition of seven culinary spice extracts. J. Food Sci. 82(8), 1807–1813 (2017).
Zitomersky, N. L., Coyne, M. J. & Comstock, L. E. Longitudinal analysis of the prevalence, maintenance, and IgA response to species of the order Bacteroidales in the human gut. Infect Immun. 79, 2012–2020 (2011).
De Filippo, C. et al. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. PNAS 107(33), 14691–14696 (2010).
Yatsunenko, T. et al. Human gut microbiome viewed across age and geography. Nature 486(7402), 222–227 (2012).
Ferrocino, I. et al. Faecal microbiota in healthy subjects following omnivore, vegetarian and vegan diets: Culturable Populations and rRNA DGGE Profiling. PLoS ONE 10(6), e0128669 (2015).
Wu, G. D. et al. Comparative metabolomics in vegans and omnivores reveal constraints on diet-dependent gut microbiota metabolite production. Gut 65, 308209 (2016).
Shankar, V. et al. Differences in gut metabolites and microbial composition and functions between Egyptian and U.S. children are consistent with their diets. mSystems 2(1), e00169-16 (2017).
Queipo-Ortuño, M. I. et al. Influence of red wine polyphenols and ethanol on the gut microbiota ecology and biochemical biomarkers. Am. J. Clin. Nutr. 95(6), 1323–1334 (2012).
Neyrinck, A. M. et al. Polyphenol-rich extract of pomegranate peel alleviates tissue inflammation and hypercholesterolaemia in high-fat diet-induced obese mice: Potential implication of the gut microbiota. Br. J. Nutr. 109, 802–809 (2013).
Parker, S. G., Trower, T. M. & Stevenson, D. E. Faecal microbial metabolism of polyphenols and its effects on human gut microbiota. Anaerobe 23, 12–19 (2013).
Massort-Cladera, M., Pérez-Berezo, T., Franch, A., Castell, M. & Pérez-Cano, F. J. Cocoa modulatory effect on rat faecal microbiota and colonic crosstalk. Arch. Biochem. Biophys. 527(2), 105–112 (2012).
Gwiazdowska, D., Juś, K., Jasnowska-Malecka, J. & Kluczyńska, K. The impact of polyphenols on Bifidobacterium growth. Acta Biochim. Pol. 62(4), 895–901 (2015).
Clemente-Postigo, M. et al. Effect of acute and chronic red wine consumption on lipopolysaccharide concentrations. Am. J. Clin. Nutr. 97(5), 1053–1061 (2013).
Boto-Ordonez, M. et al. High levels of Bifidobacteria are associated with increased levels of anthocyanin microbial metabolites: A randomized clinical trial. Food Funct. 5(8), 1932–1938 (2014).
Guglielmetti, S. et al. Differential modulation of human intestinal Bifidobacterium populations after consumption of a wild blueberry (Vaccinium angustifolium) drink. J. Agric. Food Chem. 61(34), 8134–1840 (2016).
Mayta-Apaza, A. C. et al. Impact of tart cherries polyphenols on the human gut microbiota and phenolic metabolites in virto and in vivo. J. Nutr. Biochem. 59, 160–172 (2018).
Marchesi, J. R. et al. The gut microbiota and host health: A new clinical frontier. Gut 65(2), 330–339 (2016).
Lloyd-Price, J., Abu-Ali, G. & Huttenhower, C. The healthy human microbiome. Genome Med. 8(1), 51 (2016).
Lozupone, C. A., Stombaugh, J. I., Gordon, J. I., Jansson, J. K. & Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 489(7415), 220–230 (2012).
Zhang, C. et al. Structural resilience of the gut microbiota in adult mice under high-fat dietary perturbations. ISME J. 6(10), 1848–1857 (2012).
Gupta, V. K., Paul, S. & Dutta, C. Geography, ethnicity or subsistence-specific variations in human microbiome composition and diversity. Front. Microbiol. 8, 1162 (2017).
Rinninella, E. et al. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet and diseases. Microorganisms 7(1), 14 (2019).
McBurney, M. I. et al. Establishing what constitutes a healthy human gut microbiome: State of the science, regulatory considerations, and future directions. J. Nutr. 149(11), 1882–1895 (2019).
Moorty, M., Chaiyakunapruk, N., Jacob, S. A. & Palanisamy, U. D. Prebiotic potential of polyphenols, its effect on gut microbiota and anthropometric/clinical markers: A systemic review of randomized controlled trials. Trends Food Sci. Technol. 99, 634–649 (2020).
Gowd, V., Karim, N., Shishir, M. R. I., Xie, L. & Chen, W. Dietary polyphenols to combat the metabolic diseases via altering gut microbiota. Trends Food Sci. Technol. 93, 81–93 (2019).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 7, 335–336 (2010).
Edgar, R. C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–3246 (2010).
Energy & Nutrient Composition of Food, Health Promotion Board, Singapore. https://focos.hpb.gov.sg/eservices/ENCF/. 14 March 2011. Accessed on 14th June 2018.
Acknowledgements
We thank Prof. Christiani Jeyakumar Henry for part-funding this project from Singapore Institute for Clinical Sciences (SICS) core budget, for Ms. Siok Ching Chia for her assistance with sample collection and the volunteers for taking part in the study. We also thank Prof Eric Chan and Dr Sze Han Lee from the Department of Pharmacy, National University of Singapore, for their help with urinary total polyphenol excretion and plasma phenolic acid analyses. Financial support from Singapore Institute for Clinical Science, core budget and the research funding under Principle Investigator, National University of Singapore are acknowledged.
Author information
Authors and Affiliations
Contributions
The author contributions are as follows: conceptualisation, S.H. and Y.K.L; methodology, S.H., W.W.T.K. and S.D.L; software, W.W.T.K.; validation, W.W.T.K., S.H. and Y.K.L.; formal analysis, W.W.T.K.; investigation, W.W,T.K and S.H.; resources, S.H., Y.K.L.; data curation, W.W.T.K and S.D.L.; writing—original draft preparation, W.W.T.K, S.H. and S.D.L.; writing—review and editing, S.H., W.W.T.K and Y.K.L; visualization, W.W.T.K and S.H.; supervision, Y.K.L. and S.H.; project administration, S.H.; funding acquisition, Y.K.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Khine, W.W.T., Haldar, S., De Loi, S. et al. A single serving of mixed spices alters gut microflora composition: a dose–response randomised trial. Sci Rep 11, 11264 (2021). https://doi.org/10.1038/s41598-021-90453-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-90453-7
This article is cited by
-
Probiotic effect of Lactococcus lactis subsp. cremoris RPG-HL-0136 on intestinal mucosal immunity in mice
Applied Biological Chemistry (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.