Abstract
Pronounced temporal and spatial variation in the availability of food resources can produce energetic deficits in organisms. Fruit-dependent Bornean orangutans face extreme variation in fruit availability and experience negative energy and protein balance during episodes of fruit scarcity. We evaluate the possibility that orangutans of different sexes and ages catabolize muscle tissue when the availability of fruit is low. We assess variation in muscle mass by examining the relationship between urinary creatinine and specific gravity and use the residuals as a non-invasive measure of estimated lean body mass (ELBM). Despite orangutans having a suite of adaptations to buffer them from fruit scarcity and associated caloric deficits, ELBM was lower during low fruit periods in all age-sex classes. As predicted, adult male orangutans had higher ELBM than adult females and immatures. Contrary to expectation, flanged and unflanged males did not differ significantly in ELBM. These findings highlight the precarity of orangutan health in the face of rapid environmental change and add to a growing body of evidence that orangutans are characterized by unique metabolic traits shaped by their unpredictable forest environment.
Similar content being viewed by others
Introduction
The rainforests of Southeast Asia are characterized as challenging habitats for vertebrate frugivores1. In particular, these rainforests experience greater variation in annual fruit productivity than African and South American rainforests2. The island of Borneo is particularly limited, with lower overall fruit productivity compared to Sumatra3,4, and this low resource abundance has shaped the island’s fauna in various ways. For instance, Bornean mammal populations have smaller body sizes compared to their counterparts on Sumatra, Java, and the Malay/Thai Peninsula5. Larger than most other mammals on the island, orangutans cope with these conditions through unique behavioral, physiological, and morphological adaptations6,7,8. They experience extreme reductions in caloric intake when fruit is scarce, amounting to more than a 70% reduction in energy intake compared to high fruiting periods in some populations7,9,10. Behaviorally, orangutans are less gregarious than most haplorrhine primates, including all apes, and their semi-solitary nature has been attributed to their challenging foraging conditions11. During low fruit periods, orangutans spend less time traveling, have shorter active periods, and spend more time feeding7. Morphologically, Bornean orangutans have more robust mandibles for processing the tougher foods they consume compared to their Sumatran counterparts8 and other African apes12. Physiologically, orangutans have an extremely low basal metabolic rate and expend less energy than any other mammal that has been measured, except sloths13 and pandas14. In captivity, orangutans display a notable tendency to become obese15,16,17, and it has been argued that a propensity for storing fat during periods of fruit abundance which is then catabolized during lean periods sets orangutans apart from the other great apes18.
Despite this suite of adaptations to their challenging environment, low fruit periods are associated with declining ovarian hormone production, lower conception rates18,19, negative energy balance20, ketosis9, and negative protein balance21,22 in orangutans. Previous research has indicated that prolonged periods of low fruit availability on Borneo are associated with increased levels of δ15N in orangutan urine, indicating that somatic catabolism, or tissue wasting, was beginning to occur22. Thus, despite their enhanced tendency for fat storage15,16,17,18,23, orangutan fat reserves may not be sufficient and instead they may catabolize functional body tissues for energy. Here, we aim to evaluate this possibility by assessing whether variation in fruit availability predicts variation in estimated lean body mass among orangutans at the Tuanan Research Station.
Investigations of physiological variation via non-invasive means are integral to understanding the health, conservation status, and fitness consequences of behavioral and nutritional strategies in wild animals. While a wealth of research has evaluated the impact of ecological fluctuations on wild primate health through urinary and fecal indicators of energetic status, this research has been limited by an inability to assess body size and composition non-invasively24. Emery Thompson and colleagues24 proposed a solution to this challenge by measuring creatinine and specific gravity of urine collected from wild chimpanzees (Pan troglodytes schweinfurthii). Creatinine and specific gravity are both methods for assessing the water content of urine, but only creatinine is related to an individual’s muscle mass; individuals with greater muscle mass excrete more creatinine. Thus, the variation in creatinine that is unexplained by the variation in specific gravity represents an estimate of lean body mass25,26.
Following Emery Thompson et al.24,25, we examined creatinine and specific gravity of wild orangutan urine as a non-invasive estimate of lean body mass (ELBM). Due to significant reductions in caloric intake when fruit is scarce, we predicted that episodes of fruit scarcity would be associated with skeletal muscle wasting (e.g., lower ELBM). Based on body weight and size27,28, muscle mass is also expected to vary among the age-sex classes, with adult flanged male orangutans having the highest muscle mass, followed by adult unflanged males, then adult females, and then subadult individuals.
Results
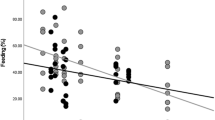
To detect changes in muscle mass, we measured the concentration of creatinine (range: 0.022–3.10 mg/ml) and the specific gravity of each urine sample (range: 1.003–1.055). ELBM was predicted by both age-sex class and FAI (Table 1). Episodes of fruit scarcity were associated with lower ELBMs (F(5.094) = 13.6, p < 0.001); this lower estimated muscle mass measured during periods of fruit scarcity was consistent across all age-sex classes (Figs. 1, 2). This result held using FAI as a continuous variable or split into categorical high/low periods based on the 50% median (Fig. 2, see also S1).
Predicted ELBM of each age-sex class using FAI as a binary predictor (high and low fruit, see S1 for GLMM results).
ELBM varied among the age-sex classes in the general predicted pattern, with significantly greater ELBM for flanged males and adult unflanged males compared to all other age-sex classes (Table 1). ELBM for adult females, was not significantly different from adolescents and dependents although trended in the predicted direction (Fig. 1; Table 1).
Discussion
Our results highlight that despite several well-documented adaptations to cope with unpredictable episodes of fruit scarcity in orangutans, lower ELBMs during low fruit periods suggest they still enter a critical state of muscle catabolism when fruit is scarce. Orangutans rely on less energy dense fallback foods when fruit availability is low7, and at several sites it has been demonstrated that they catabolize fat stores for energy during these periods9,10,29. Our findings extend previous evidence from another study site that orangutans enter into the early stages of a protein deficit after prolonged periods of fruit scarcity at Gunung Palung (West Kalimantan, Borneo). Gunung Palung is dominated by dipterocarp trees and, thus, experiences the masting phenomenon that is characteristic of these forest types30,31. Tuanan is a non-masting forest, with lower but more consistent fruit availability patterns than Gunung Palung32. Future research should compare patterns of muscle loss between these study sites to better understand variation in orangutan physiology in relationship to local ecologies.
The consistency of the suggested pattern of muscle wasting during low fruit periods across all age-sex classes raises questions about the impacts of muscle catabolism. Nutritional and energetic constraints have clearly shaped orangutan life history; they have the longest developmental period among apes33 and the slowest pace of reproduction among mammals34. This life history strategy may help buffer individuals against energy deficits by reducing the energetic requirements of growth or reproduction at a given time, which may reduce the need for muscle catabolism for energy. Future research should examine the long-term health and fitness impacts of more severe or frequent muscle wasting on individuals, particularly during sensitive windows such as juvenility or lactation.
We also predicted that there would be general differences in muscle mass among the age-sex classes. In chimpanzees, ELBM is highest in adult males, followed by sub-adult males, then adult females and sub-adult females24. Thus, we predicted a similar directional difference in ELBM for orangutans. In our study, ELBM differed among the age-sex classes in the expected pattern with some exceptions. Both adult male classes had higher ELBM than adult females and immature classes, but adult females did not differ from immature orangutans. There was no significant difference between independent immatures and dependent immatures in our sample. Figures 1 and 2 suggest that adult females trend towards greater ELBM than both classes of immatures, and we cannot rule out that the smaller sample size of the two immature groups (22 samples for dependents and 113 samples for adolescents, compared to 511 for adult females) obscured the differences. While it is less surprising that adult females and adolescent females did not show significant differences in ELBM, it is reasonable to expect a profound difference between adults and dependent individuals. Newborn human babies have high creatinine levels35, as do chimpanzees under the age of 324. While no individuals in our sample were under the age of 3.5, it is possible that immature kidney functioning leading to reduced glomerular filtration36 caused higher creatinine levels in these young orangutans.
We also did not find a difference in ELBM between flanged and unflanged males. Flanged and unflanged male orangutans, while both fully sexually mature, have different appearances and body masses, with adult flanged males ranging from 74 to 90 kg27,28 and unflanged males 40.5 kg on average at one Bornean site (this average for unflanged males may be low as it comes from a disturbed forest site where orangutans tend to be thinner)28. These two male morphs differ in androgen levels37,38,39, with flanged males having higher levels of testosterone and DHT, which should promote muscle growth40. Thus, we expected substantial differences in ELBM between these two classes. It has been shown that male chimpanzees lose muscle mass as part of the ageing process. While it is possible that at least some of the flanged males in our sample were of advanced age, it is exceedingly difficult to track adult male orangutans over the long time periods to obtain an accurate estimate of age41, so we were unable to account for individual age in our study. Additionally, our sample size for unflanged males (53 samples among 15 individuals) was small compared to that of flanged males (431 samples among 31 individuals). Tuanan is characterized by a greater ratio of flanged relative to unflanged males, which is also true at nearby Sabangau39, but differs from a similar habitat site in Sumatra42. It is possible that with a more robust sampling of unflanged males this result could change, however obtaining a more robust sample at Tuanan would be challenging given the infrequent encounter rate for unflanged males. However, if indeed these two morphs do not differ in lean muscle mass, the difference in overall body mass between them would be the result, perhaps, of profoundly higher fat deposition in flanged males and/or a lower basal metabolic rate in the larger flanged males43. Male bimaturism in orangutans remains poorly understood, and these findings raise new questions about the eco-physiological differences between them.
These findings emphasize the ecological challenges that the critically endangered Bornean orangutan faces. Borneo is heavily impacted by El Niño events, and local climate shifts due to land conversion exacerbate the susceptibility of forests to fires during these periods44. As global climate change threatens to amplify El Niño events45, droughts and wildfires may worsen, having detrimental impacts on orangutan habitat and their food supply. Simple methods like the one used here will allow for better tracking of the health of Bornean orangutan populations. Long-term non-invasive monitoring of orangutan health status is critical, as it will provide insight into the effectiveness of current conservation practices and the types of new protections that must be implemented to protect vulnerable individuals and populations. These findings suggest that access to fruit is critical for orangutans, and emphasize the need for careful conservation planning, including thorough plant species surveys in forests being considered for the release of rehabilitants or translocated orangutans. The establishment and maintenance of forest corridors that connect fruit-poor fragments with more productive forest areas is imperative for orangutan survival. As demonstrated by variation among the age-sex classes in the relationship between muscle wasting and FAI, individual variation in vulnerability to different ecological challenges must be considered as part of effective conservation plans.
Methods
Data were collected at Tuanan Research Station in Central Kalimantan, Indonesia from 2009 to 2017. The study site contains only wild orangutans, with no ex-captives or translocated individuals released at the study site. A fruit abundance index (FAI) was determined each month as the percent of stems with a diameter at breast height (dbh) greater than 10 cm that were fruiting (n = 2400 stems) within phenology plots6. The phenology plots cover 2.3 ha within the 900 ha research site and are spread across the home ranges of the most heavily sampled orangutans in the dataset. Age-sex classes were determined by developmental status and sex7, and included adult females N = 32; adult flanged males N = 31; adult unflanged males N = 15; adolescent males and females, which includes both independent and semi-independent immatures N = 11 (weaned and/or older sibling animals of smaller body size than adults—independents rarely travel with mother, semi-independent travel with mother most of the time); and dependent male and female offspring N = 11 (clinging or non-clinging unweaned animals sleeping in mother’s nest and in daily association with mother).
Urine was collected from first-morning voids during nest-nest focal follows and kept on ice in a thermos. Upon return to the research camp, SG was measured using a digital refractometer (Atago PAL-10S), and samples were then frozen at − 20 °C. Samples were shipped to Rutgers University on dry ice and stored at − 80 °C until analyzed for creatinine following Emery Thompson et al.24,25. We followed previously validated methods to estimate lean body mass from creatinine adjusted by SG in spot urine samples24,25. We first removed all overly dilute samples with SG less than 1.003 (n = 14), resulting in 1,130 samples from 70 individuals that were used in our analyses (Table 2). Water has a SG of 1.0 and a creatinine level of 0.0 mg/ml, so we subtracted 1 from each SG reading and used the resulting values to calculate a global fit for creatinine levels against [(SG − 1) + (SG − 1)2] via linear regression forced through the origin (r2adj = 0.812, p < 0.0001). We used both the linear and quadratic terms for specific gravity to account for the curvilinear relationship between creatinine and SG. The residuals from this regression represented the variation in creatinine unexplained by SG, indicating an estimate of lean body mass25. The residuals were used as the target in a generalized additive mixed model (GAMM) with FAI and age-sex class as fixed effects and individual ID as a random factor. Pairwise comparisons were achieved via the relevel function for age-sex class in the GAMM. A generalized linear mixed model (GLMM) with FAI (binned as High and Low) and age-sex class was used for visualization (Fig. 2, see S1 for results). All statistical analyses were run in R version 3.6.146. R code is available at https://github.com/adigiorgio17/sg-cre-ou.
Ethical approval
This research followed all international, national, and institutional guidelines for the care and use of animals. This protocol was approved by the Institutional Animal Care and Use Committee of Rutgers, the State University of New Jersey, PROTO999900338. A CITES export permit was obtained to transport urine samples (permit #s:15497/IV/SATS-LN/2019, 22655/IV/SATS-LN/2017, 17/BKSDA.KALTENG-1/2015) from Indonesia.
Data availability
The dataset analyzed for the current study are available from the authors upon reasonable request.
References
Fleming, T. H., Breitwisch, R. & Whitesides, G. Patterns of tropical vertebrate frugivore diversity. Annu. Rev. Ecol. Syst. 18, 91–109 (1987).
van Schaik, C. P. & Pfannes, K. R. Tropical climates and phenology: a primate perspective. In Seasonality in Primates: Studies of Living and Extinct Human and Non-Human Primates (eds Brockman, D. K. & van Schaik, C. P.) (Cambridge University Press, 2005).
Marshall, A. J., Boyko, C. M., Feilen, K. L., Boyko, R. H. & Leighton, M. Defining fallback foods and assessing their importance in primate ecology and evolution. Am. J. Phys. Anthropol. 140, 603–614 (2009).
Wich, S. A. et al. Forest fruit production is higher on Sumatra Than on Borneo. PLoS ONE 6, e21278 (2011).
Meiri, S., Meijaard, E., Wich, S. A., Groves, C. P. & Helgen, K. M. Mammals of Borneo: small size on a large island. J. Biogeogr. 35, 1087–1094 (2008).
Vogel, E. R. et al. Functional ecology and evolution of hominoid molar enamel thickness: Pan troglodytes schweinfurthii and Pongo pygmaeus wurmbii. J. Hum. Evol. 55, 60–74 (2008).
Vogel, E. R. et al. Nutritional ecology of wild Bornean orangutans (Pongo pygmaeus wurmbii) in a peat swamp habitat: effects of age, sex, and season. Am. J. Primatol. 79, e22618 (2017).
Vogel, E. R. et al. Food mechanical properties, feeding ecology, and the mandibular morphology of wild orangutans. J. Hum. Evol. 75, 110–124 (2014).
Knott, C. D. Changes in orangutan caloric intake, energy balance, and ketones in response to fluctuating fruit availability. Int. J. Primatol. 19, 1061–1079 (1998).
Harrison, M. E., Morrogh-Bernard, H. C. & Chivers, D. J. Orangutan energetics and the influence of fruit availability in the nonmasting peat-swamp forest of Sabangau, Indonesian Borneo. Int. J. Primatol. 31, 585–607 (2010).
van Schaik, C. P. The socioecology of fission-fusion sociality in Orangutans. Primates 40, 69–86 (1999).
Taylor, A. B., Vogel, E. R. & Dominy, N. J. Food material properties and mandibular load resistance abilities in large-bodied hominoids. J. Hum. Evol. 55, 604–616 (2008).
Pontzer, H., Raichlen, D. A., Shumaker, R. W., Ocobock, C. & Wich, S. A. Metabolic adaptation for low energy throughput in orangutans. Proc. Natl. Acad. Sci. 107, 14048–14052 (2010).
Nie, Y. et al. Exceptionally low daily energy expenditure in the bamboo-eating giant panda. Science 349, 171–174 (2015).
Zihlman, A. L., Mcfarland, R. K. & Underwood, C. E. Functional anatomy and adaptation of male gorillas (Gorilla gorilla gorilla) with comparison to male orangutans (Pongo pygmaeus). Anat. Rec. 294, 1842–1855 (2011).
Gresl, T. A., Baum, S. T. & Kemnitz, J. W. Glucose regulation in captive Pongo pygmaeus abeli, P. p. pygmaeus, and P. p. abeli x P. p. pygmaeus orangutans. Zoo Biol. 19, 193–208 (2000).
Jones, M. L. The orang utan in captivity. In The Orang Utan: Its Biology and Conservation (ed. de Boer, L. E. M.) 17–37 (Dr. W. Junk Publishers, 1982).
Knott, C. D. Energetic responses to food availability in the great apes: implications for hominin evolution. In Seasonality in Primates: Studies of Living and Extinct Human and Non-Human Primates (eds Brockman, D. K. & van Schaik, C. P.) 351–378 (Cambridge University Press, 2005).
Knott, C. D. Reproductive, physiological and behavioral responses of orangutans in Borneo to fluctuations in food availability. Doctoral Dissertation (Harvard University, 1999).
Emery Thompson, M. & Knott, C. D. Urinary C-peptide of insulin as a non-invasive marker of energy balance in wild orangutans. Horm. Behav. 53, 526–535 (2008).
Vogel, E. R. et al. A noninvasive method for estimating nitrogen balance in free-ranging primates. Int. J. Primatol. 33, 567–587 (2012).
Vogel, E. R. et al. Bornean orangutans on the brink of protein bankruptcy. Biol. Lett. 8, 333–336 (2012).
Wheatley, B. P. The evolution of large body size in orangutans: a model for hominoid divergence. Am. J. Primatol. 13, 313–324 (1987).
Emery Thompson, M., Muller, M. N. & Wrangham, R. W. Technical note: variation in muscle mass in wild chimpanzees: application of a modified urinary creatinine method. Am. J. Phys. Anthropol. 149, 622–627 (2012).
Emery Thompson, M. et al. Evaluating the impact of physical frailty during ageing in wild chimpanzees (Pan troglodytes schweinfurthii). Philos. Trans. R. Soc. B Biol. Sci. 375, 20190607 (2020).
Bergstrom, M. L., Thompson, M. E., Melin, A. D. & Fedigan, L. M. Using urinary parameters to estimate seasonal variation in the physical condition of female white-faced capuchin monkeys (Cebus capucinus imitator). Am. J. Phys. Anthropol. 163, 707–715 (2017).
Markham, R. & Groves, C. P. Brief communication: weights of wild orang utans. Am. J. Phys. Anthropol. 81, 1–3 (1990).
Rayadin, Y. & Spehar, S. N. Body mass of wild bornean orangutans living in human-dominated landscapes: implications for understanding their ecology and conservation. Am. J. Phys. Anthropol. 157, 339–346 (2015).
Vogel, E. R. et al. The power of protein: protein regulation, energetics, and health in wild Bornean orangutan (Pongo pygmaeus wurmbii). Am. J. Phys. Anthropol. 162, 397 (2017).
Ashton, P. S., Givnish, T. J. & Appanah, S. Staggered flowering in the dipterocarpaceae: new insights into floral induction and the evolution of mast fruiting in the aseasonal tropics. Am. Nat. 132, 44–66 (1988).
Cannon, C. H., Curran, L. M., Marshall, A. J. & Leighton, M. Long-term reproductive behaviour of woody plants across seven Bornean forest types in the Gunung Palung National Park (Indonesia): suprannual synchrony, temporal productivity and fruiting diversity. Ecol. Lett. 10, 956–969 (2007).
Vogel, E. R. et al. Nutritional differences between two orangutan habitats: implications for population density. PLoS ONE 10, e0138612 (2015).
Wich, S. A. et al. Life history of wild Sumatran orangutans (Pongo abelii). J. Hum. Evol. 47, 385–398 (2004).
van Noordwijk, M. A. et al. The slow ape: high infant survival and long interbirth intervals in wild orangutans. J. Hum. Evol. 125, 38–49 (2018).
Finney, H., Newman, D., Thakkar, H., Fell, J. & Price, C. Reference ranges for plasma cystatin C and creatinine measurements in premature infants, neonates, and older children. Arch. Dis. Child. 82, 71–75 (2000).
Stonestreet, B. S., Bell, E. F. & Oh, W. Validity of endogenous creatinine clearance in low birthweight infants. Pediatr. Res. 13, 1012–1014 (1979).
Kingsley, S. Causes of non-breeding and the development of the secondary sexual characteristics in the male orangutan: a hormonal study. In The Orang-Utan, Its Biology and Conservation (ed. de Boer, L. E. M.) 215–229 (Dr. W. Junk Publishers, 1982).
Maggioncalda, A. N., Sapolsky, R. M. & Czekala, N. M. Reproductive hormone profiles in captive male orangutans: implications for understanding developmental arrest. Am. J. Phys. Anthropol. 109, 19–32 (1999).
Marty, P. R. et al. Endocrinological correlates of male bimaturism in wild Bornean orangutans. Am. J. Primatol. 77, 1170–1178 (2015).
Bhasin, S. et al. The effects of supraphysiologic doses of testosterone on muscle size and strength in normal men. N. Engl. J. Med. 335, 1–7 (1996).
Buckley, B. J. W. Ranging behaviour of male Orang-Utans in an unfragmented Bornean habitat and implications for mating-system mechanics. Doctoral Dissertation (Cambridge University, 2015).
Dunkel, L. P. et al. Variation in developmental arrest among male orangutans: a comparison between a Sumatran and a Bornean population. Front. Zool. 10, 1–11 (2013).
Kleiber, M. Body size and metabolic rate. Physiol. Rev. 27, 511–541 (1947).
Chapman, S. et al. Compounding impact of deforestation on Borneo’s climate during El Niño events. Environ. Res. Lett. 15, 084006 (2020).
Cai, W. et al. Increased variability of eastern Pacific El Niño under greenhouse warming. Nature 564, 201–206 (2018).
R Core Team. R: A Language and Environment for Statistical Computing (R Core Team, 2019).
Acknowledgements
We thank the Tuanan assistants, project managers, staff, and students for data collection, Tatang Mitra Setia, Carel van Schaik, Maria van Noordwijk, Jito Sugardjito, and the Biology Department at Universitas Nasional (UNAS) for their partnership and support. We thank Melissa Emery Thompson for helpful advice on methodology. We thank two anonymous reviewers, Charles Janson, and the Vogel lab for their insightful feedback on the manuscript. We are grateful to the Indonesian State Ministry for Research and Technology (RISTEK), the Indonesian Institute of Science (LIPI), the Directorate General of Forest Protection and Nature Conservation (PHKA), Ministry of Internal Affairs, the Nature Conservation Agency Palangkaraya (BKSDA), the local government in Central Kalimantan, the Borneo Orangutan Survival Foundation (BOSF Indonesia) and BOSF Mawas in Palangkaraya for their permission and support of the project. We thank Jesse McCann for code assistance. This research was funded by the National Science Foundation (BCS-0643122; BCS-1719825), The L.S.B. Leakey Foundation, United States Agency for International Development, Orangutan Outreach, Rutgers University, and The Center for Human Evolutionary Studies.
Author information
Authors and Affiliations
Contributions
C.O. conceptualized the study, prepared and helped analyze the data, drafted the manuscript, A.D. conceptualized the study, conducted statistical analysis, created graphics, helped draft the manuscript, A.U. helped conceive of the study and analyzed samples, R.B. directed and carried out sample collection, edited the manuscript, D.N. conducted sample analysis and assisted with graphics and draft revisions, S.U. served as research counterpart, and critically revised the manuscript, E.V. conceived of the study, coordinated the study as PI, assisted with statistical analysis and helped draft the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
O’Connell, C.A., DiGiorgio, A.L., Ugarte, A.D. et al. Wild Bornean orangutans experience muscle catabolism during episodes of fruit scarcity. Sci Rep 11, 10185 (2021). https://doi.org/10.1038/s41598-021-89186-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-89186-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.