Abstract
In total, 90 gelatinous spheres, averaging one meter in diameter, have been recorded from ~ 1985 to 2019 from the NE Atlantic Ocean, including the Mediterranean Sea, using citizen science. More than 50% had a dark streak through center. They were recorded from the surface to ~ 60–70 m depth, mainly neutrally buoyant, in temperatures between 8 and 24°C. Lack of tissue samples has until now, prohibited confirmation of species. However, in 2019 scuba divers secured four tissue samples from the Norwegian coast. In the present study, DNA analysis using COI confirms species identity as the ommastrephid broadtail shortfin squid Illex coindetii (Vérany, 1839); these are the first confirmed records from the wild. Squid embryos at different stages were found in different egg masses: (1) recently fertilized eggs (stage ~ 3), (2) organogenesis (stages ~ 17–19 and ~ 23), and (3) developed embryo (stage ~ 30). Without tissue samples from each and every record for DNA corroboration we cannot be certain that all spherical egg masses are conspecific, or that the remaining 86 observed spheres belong to Illex coindetii. However, due to similar morphology and size of these spheres, relative to the four spheres with DNA analysis, we suspect that many of them were made by I. coindetii.
Similar content being viewed by others
Introduction
Rare, large (~ 1 m in diameter) gelatinous spheres from the NE Atlantic (Scandinavia and Mediterranean Sea), attributed to egg mass of ommastrephid squid (Oegopsida, Cephalopoda, Mollusca), have previously been reported1, but lack of tissue samples prevented molecular analysis and confirmation of species. Representatives of several squid families (e.g. Thysanoteuthidae Keferstein, 1866, Ommastrephidae Steenstrup, 1857 and possibly Lycoteuthidae Pfeffer, 1908) are known to produce large, neutrally buoyant structures, and egg masses of many squid families are unknown. Pelagic squid egg masses are rarely recorded, and studying in situ spheres is based on chance encounters. Such huge egg masses are thought to maintain their depth in the water column by floating on a pycnocline2,3,4,5,6,7,8,9.
Ommastrephids are the most abundant and widely distributed family of cephalopods, and are distributed throughout the world oceans from sub-Arctic seas to sub-Antarctic seas. They inhabit waters from the surface to depths of 2000 m (e.g. Todarodes sagittatus Steenstrup, 188010), but are mainly recorded above 1000 m11. Three main ecological groups (life forms); (1) Slope-shelf group, (2) Nerito-oceanic group and (3) Oceanic group, are distinguished among ommastrephids, by characteristics of habitat, especially spawning habitat, which includes the degree of relationship with the bottom. Many species within Illex, Todarodes, Todaropsis and Nototodarus genera belongs within the slope-shelf group where the main habitats are the waters of the continental slope and shelf12. Planktonic dispersal patterns of paralarvae in an Iberian-Canary Upwelling system show larvae of I. coindetii within the group using «coastal strategy»13.
The Ommastrephidae are also the most important cephalopods for commercial fisheries12. During 2010–2014, the annual world catch of ommastrephids varied between 1.6 and over 4 million metric tonnes, representing 70% of the total world cephalopod catch (Jereb et al.14). Of these, the genus Illex contributes a significant portion, corresponding to 9.6% of the total annual cephalopod catches in 201515. However, it is difficult to obtain specific statistics due to systematic uncertainties in the definition of each species and a common tendency of landing mixed squid species, and reporting even single species merely as “short-finned squid”.
Cephalopods have highly variable reproduction strategies16. Generally, cephalopods are short lived and semelparous17. Many observations indicate that I. coindetii females are “intermittent spawners” that spawn several times during a period of time ranging between a few days and a few weeks12, with a so-called intermittent terminal spawning pattern where oocyte maturation and egg-laying occur separately during the spawning period, which may last for several weeks18. The female does not grow in this period and dies shortly after. Statolith data indicate an average life span of one year or less19,20,21,22. Fertilization in ommastrephids takes place while spawning23,24,25. Knowledge on egg and juvenile development for I. coindetii is limited11, and egg masses have never been observed in situ except by Naef2 who reported «floating devices» from the Mediterranean Sea, together with his drawings of juvenile squid, later thought to be I. coindetii26. However, in vitro fertilization has been accomplished for I. coindetii, and hatchling morphologies have been described27.
The aim of this study is to determine which cephalopod species is responsible for the production of huge gelatinous spheres from NE Atlantic Ocean, using DNA analysis on sphere tissue samples. We also report on 90 huge spheres from the NE Atlantic extending ~ 35 years back in time (from ~ 1985 to 2019). Of these 90, 27 are reported in Ringvold and Taite1 and 63 are newly reported herein.
Material and methods
Collecting tissue samples
As a continuation of the study in Ringvold and Taite1, popular press articles and posters were published to obtain as many new sphere observations as possible, with associated ecological information, and to solicit collection of tissue samples for molecular identification. We focused these solicitations on Facebook Internet pages for diving clubs and diving centers in Norway, Sweden, England, Scotland, Ireland, France, Spain, Portugal, Italy, Malta, Cyprus and Croatia.
On 28 July 2019 at noon two experienced technical Scuba divers at Magerholm in Storfjorden near Ålesund, NW Norway, encountered a large gelatinous sphere floating at 43 m depth, 2 m above the sediment (Table 1, Fig. 1). The sphere measured approximately 1 m in diameter, and had a narrow streak, or structure, through the center. One diver had a 0.5 l plastic bottle with screw cap. Because one of the divers had heard about our citizen science project through media, and our desire for a tissue sample, they agreed to take a sample using the plastic bottle. After squeezing out the freshwater, the bottle opening was placed underneath the sphere, sucking out some tissue. Directing a flashlight towards the bottle confirmed that eggs from the sphere was successfully collected. The sample was secured at 12:45, and in situ water temperature was 8 °C, measured with a Shearwater Petrel computer. The sphere did not seem to be damaged by the sampling but kept the same spherical shape as it drifted off. No picture of this sphere was taken.
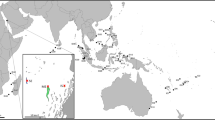
Locations where huge gelatinous spheres have been observed in the NE Atlantic Ocean, including the Mediterranean Sea (red dots), combining data from this study and Ringvold and Taite (1, Fig. 2). Locations of DNA tested spheres (green stars). Figure generated with Qgis 3.2. (www.qgis.org) (Credit: Halldis Ringvold/Sea Snack Norway).
When ashore the bottle was transported to the home of one of the divers, in an air conditioned car, and placed in the freezer. The next day transport was arranged for the sample to the nearest research station (Møreforsking in Ålesund) 10 min away, and the bottle contents were thawed after arrival at the station.
At the laboratory the bottle contents were poured into a 1 l sterile beaker. The first attempt to separate eggs from their mucous matrix led to the pipette being clogged with slimy mucus. The bottle contents were therefore poured into several petri dishes, and the eggs were successfully pipetted into seven 15 ml tubes. A ratio of about 2 ml of eggs and some gelatinous matrix to 13 ml 96% ethanol was used, and eggs were visible in the ethanol solution. Two eggs were preserved in a 4% formalin solution. Photos of a few thawed elipsoid eggs were taken with a handheld mobile camera through a microscope. Thawed egg length was estimated by eye. After making sure that all eggs had been extracted from the sample, the excess seawater and gelatinous matrix was transferred to five 50 ml tubes with 96% ethanol, which were stored cool (4 °C). All samples were shipped immediately to Sea Snack Norway (< 24 h transport).
On 7 and 8 August 2019 two additional sphere tissue samples were obtained near Kilsund in Arendal, SE Norway, at 17 and 15 m depth, respectively. The first sphere, collected 7 August, was disintegrating with the sphere wall rupturing. Tissue was secured in a small plastic jar with screw cap. After returning to the surface, the divers transported the jar to a home freezer (approx. − 20 °C). A few days later, it was delivered to Flødevigen. When thawed, only a small part of the sample was left in the jar. The sample seemed to be a part of the outermost section of the sphere, containing a mix of gelatinous matrix and algae. The sample was investigated in a petri dish under the microscope and a single developed embryo was found and measured.
A sample of the second sphere, collected 8 August, was secured in a 0.5 l plastic bottle with screw cap, using the same method as for the Ålesund sample, and delivered to the nearest research station, Institute of Marine Research at Flødevigen the same day. The eggs could be seen floating in the mass of clear gelatinous matrix mixed with seawater. The volume of gelatinous matrix was estimated as 2–3 deciliters. The sample was poured into a big petri dish, put under a dissecting microscope and live eggs were isolated from the sticky egg mass using a dissection needle and a disposable pipette cut to a suitable size. The eggs, about 65 in total, were transferred to a small staining jar and investigated further under the microscope. Pictures of live embryos were taken through the eyepiece of the microscope. The size of the embryos was also measured and checked with a calibrated metal sphere of 1.00 mm.
Tissue from both spheres was preserved directly in 96% ethanol. A few eggs collected on 8 August were preserved in 4% formalin, and a few were conserved in separate tubes with 96% ethanol, for possible additional analysis (not herein).
On 16 August 2019, tissue of a fourth sphere was collected at Søgne, at 10 m depth. The diver secured a sample of a dissolving sphere using a 0.5 l plastic bottle with screw cap, and when at home the sample was placed in the freezer. A few days later sample transport to University of Agder in Kristiansand was arranged. After defrosting, the content of the bottle was placed in petri dishes and analysed through a Zeiss Stemi DRC stereo microscope with ocular micrometer scaling slide using 16 * ocular and 1.6 * object zoom, where 10 sublines corresponded to 0.55 mm. Pictures were taken using an iphone 7 mobile camera through the eyepiece of the stereo microscope. Whole embryos and most pieces of embryo in the sample were pipetted out and conserved in 96% ethanol for DNA analysis. A few embryos were also preserved in 4% formalin. All samples were shipped to Sea Snack Norway.
Ethanol-preserved tissue samples of adult Todarodes sagittatus (Lamarck, 1798) were provided from cruises by Faroe Marine Research Institute (Faroe Islands) and Marine & Freshwater Research Institute (Iceland). Ethanol-preserved tissue samples of adult Illex coindetii were provided by Stavanger Museum.
All ethanol samples were shipped from Sea Snack Norway to University of Ireland, Galway, for DNA analysis. Voucher samples are deposited in the collections of Stavanger Museum, Norway.
Other observations
DORIS (Données d'Observations pour la Reconnaissance et l'Identification de la faune et la flore Subaquatiques) is a citizen science project initiated in 2006 by the “environment and biology commission” of the French Underwater Federation (FFESSM). The aim was to construct a web site presenting French aquatic species (including French overseas territories). More than 2.600 species are described and illustrated by about 26.000 pictures. The DORIS network was contacted by Professor Helmut Zibrowius (Marseille Oceanology Centre) in 2007, who asked about observations, photos, and samples of an unknown huge sphere observed in Croatia in 2006. The huge sphere was named GST (“Grande Sphère Transparente” i.e. huge transparent sphere). The DORIS network enabled recording of sphere observations from France more than 10 years back in time—to the present; these observations are included in this study. Unfortunately, no sphere tissue samples have yet been taken from French, or Mediterranean, waters. One observation was also mentioned from Croatia, by Miro Andric28.
Molecular analysis
The four gelatinous sphere tissue samples collected from Norwegian waters were well preserved in ethanol and eggs were clearly present, some with visible embryos. DNA was extracted from the eggs from the spheres, as well as from separate tissue samples of adult T. sagittatus and I. coindetii, using a Purelink genomic DNA mini kit following the manufacturer’s instructions. Universal Folmer primers LCO1490 (5′-GGT CAA CAA ATC ATA AAG ATA TTG G-3′) and HCO2198 (5′-TAA ACT TCA GGG TGA CCA AAA AAT CA-3′)29 were used to amplify the Folmer region of the cytochrome oxidase subunit I (COI) gene. Each PCR contained 12.5 μl DreamTaq Green PCR Master Mix (Thermo Scientific), 0.5 μl of each primer (10 μM), 9 μl nuclease-free water (Thermo Scientific) and 2.5 μl DNA template (20 ng). The PCR thermocycling program included an initial step at 94° C for 2 min, followed by 35 cycles at 94 °C for 40 s, 50 °C for 40 s and 72 °C for 90 s which was followed by a final step at 72 °C for 10 min. The sample was assessed by electrophoresis on a 1% agarose gel stained with SYBR Safe DNA Gel Stain (Invitrogen, LifeTech). PCR products were purified using the Purelink PCR purification kit following the manufacturer’s instructions and sequenced by GATC Biotech (Constance, Germany) on a Sanger ABI 3730xl.
BLAST30 indicated that the DNA extracted from the spheres probably originated from Illex sp.. Thus all available Illex COI sequences were downloaded from GenBank31, excepting those where the squid had not been identified to species using morphological methods (i.e., excluding studies which were themselves using DNA to identify species), those that were unusually short, and known contaminants (e.g., «Illex argentinus» from32 from a Korean fishmarket which are now known to have originated from Nototodarus sloanii). Downloaded sequences were combined in a fasta file with those sequences generated herein, and four additional sequences of T. sagittatus (also from Genbank). Sequences were imported to Unipro UGENE33, aligned with Muscle. The resulting alignment contained 73 sequences (13 new) and was 624 base pairs long. A maximum likelihood tree, rooted on the Todarodes sequences, was built in RAxML version 834 using raxmlGUI 2.0 beta35. 100 fast boostraps (BS) were generated using the GTRGAMMA model and default settings.
Ethical approval
In accordance to Norwegian and European legislation related to animal research, formal approval of the experimental protocol by the Norwegian Animal Research Authority (NARA) is not required because the experimental conditions are practices undertaken for the purpose of recognized animal husbandry. Such practices are excepted from the European convention on the protection of animals used for scientific purposes (2010/63/EU), cf. article 5d. Also, these practices do not require approval by the Norwegian ethics board according to the Norwegian regulation on animal experimentation, § 2, 5a, d “non-experimental husbandry (agriculture or aquaculture)” and “procedures in normal/common breeding and husbandry”. Norway has implemented the European Directive according to the EEA agreement. This explanation may be viewed as a waiver. The Norwegian Animal Research Authority does normally not give formal waivers. I.e. in clear cases, such as this one. Experiments involving in situ captured, fertilized squid eggs prior to exogenous feeding are exempted from the Norwegian Regulation on Animal Experimentation.
Results
Molecular analysis
The newly generated sequences are available through Genbank with Accession Numbers MW444369-MW444381. They include four COI sequences of egg masses, and additional COI sequences from adult squids as indicated in methods. The corresponding specimens are deposited in Stavanger Museum, Norway. All sequences from egg masses resolved in a highly supported clade (BS) together with new sequences obtained from specimens identified as I. coindetii from Norwegian waters (Fig. 2).
Morphology
Embryos from four spheres where recorded, at four different embryonic stages. When describing stages, we follow Sakai et al.36.
Ålesund sample
The sphere was whole, and when the diver poked it with his hand the consistency seemed firm. The gelatinous matrix and eggs from the Ålesund sample were all transparent and sticky. About 15–20 eggs were isolated, all approximately developmental stage ~ 3. Estimated egg length was between 1–2 mm (estimation by eye). All eggs were ellipsoid, and recently fertilized, with micropyle visible. The yolk sac, the embryo, and the chorion are visible at this stage (Fig. 3). The embryo has also covered the animal pole of the yolk with a layer a few cells thick and is spreading to cover the entire yolk. On the formalin-fixed egg (Fig. 3), the darkened chorion is visible in the jelly envelope. Chorion length and width for one egg were measured with a Leica stereomicroscope to 1.13 and 0.91 mm, respectively, and the jelly envelope length and width were measured to 1.65 and 1.59 mm, respectively.
(1) Ålesund sphere sample (thawed): (A) Recently fertilized, elipsoid egg of Illex coindetii measuring between 1–2 mm in length (by eye). (B) Formalin fixated egg. Chorion length and width for one egg measured 1.13 and 0.91 mm, respectively, and the jelly envelope length and width were measured to 1.65 and 1.59 mm, respectively. (2) Arendal sphere sample: (A) Live embryos of Illex coindetii; 16 embryos measured width of 0.77 ± 0.047 mm and length of 1.15 ± 0.119 mm (from microscope pictures (collected 8 August). Black measurement sphere is 1 mm in diameter. (B) Arrangement of live embryos within the egg mass in the internal mucous matrix. (3) Søgne sphere sample (thawed): Seven developed embryos of Illex coindetii, of which five were measured, showing total length of 1.83 ± 0.139 mm (from microscope pictures). (4) Arendal sphere sample (thawed): Developed embryo measuring approximately 2 mm in length (by microscope) (collected 7 August). (Photo credits: Martin Hansen/Oceanfjord as, Snorre Bakke/Møreforsking, Arne Hassel and Hanne Sannæs/IMR, Ann-Elin Wårøy Synnes/University of Agder. Collage Halldis Ringvold/Sea Snack Norway.).
Two Arendal samples
The sphere sampled 7 August was disintegrating, and of loose consistency. The internal mucous matrix was sticky. A single developed embryo (around stage 30) was found, not in the matrix but in sea water associated with the sample. It measured about 2 mm in total length (Fig. 3, video 1).
The sphere sampled 8 August was whole and of firm consistency (video 2). Eggs were elipsoid, and both eggs and the internal mucous matrix were sticky. Embryos had reached organogenesis stages 17–19, and 16 of the embryos measured width of 0.77 ± 0.047 and length of 1.15 ± 0.119 (Fig. 3).
Søgne sample
The sphere was dissolving (video 3) so much that when a hand was waved close to the sphere wall, it ruptured. The internal mucous matrix was sticky. Seven whole embryos were found, as well as a few more head fragments, from developmental stage ~ 23, and total length measured 1.83 ± 0.139 mm (Fig. 3).
In summary, the consistency of the four spheres varied, and changed from firm to disintegrating stage—as the embryos became more developed (Table 2).
Observations of gelatinous spheres
Spheres have mostly been recorded from Norway and the Mediterranean Sea. In Norway, they have been recorded from Nordland County in the north to Østfold County in the south. Also a few spheres have been recorded from the Swedish westcoast (outlet of Gulmarsfjorden) (Fig. 4). In the Mediterranean Sea, they have mainly been recorded from the western basin (Spain, including Mallorca, France, Italy and Malta), but two observations are from Croatia (Fig. 5, video 4.). Since the study published by Ringvold and Taite1, observations have also been made in two new areas—NW coast of Spain (Bay of Biscay) (video 5) and NE of England, close to the Scottish border (Fig. 4).
Huge gelatinous spheres from the NE Atlantic Ocean (Norway, Sweden and England) attributed to squid egg mass. (Photo credits: (A) Robert Fiksdal, (B) Harald Woldsund, (C) Birgitta Lantto, (D) Tapio Salakari, (E) Geir Eliassen, (F) Steve Saunders, (G) Tor Erik Meland, (H) Nils Baadnes, (I) Grzegorz Marsicki. Collage by Halldis Ringvold/Sea Snack Norway).
Huge gelatinous spheres from the Mediterranean Sea and the Spanish Atlantic coast, attributed to squid egg mass. (Photo credits: (A) Franc Jourdan, (B) Thomas Brelet, (C) Edouard Bard, (D) Eduardo Losada Lage, (E) José Coronel, (F), Philippe Le Roy, (G) Christine Chesnay, (H) Pietro Crovetto, (I) Simone Ulzega, (J) Alain Beauté, (K) Patrick Carreno, (L) Emmanuel Roguet. Collage by Halldis Ringvold/Sea Snack Norway).
Spheres were observed from March to October (all areas combined) with a peak in July and August in Norwegian waters, and August and September in the Mediterranean Sea (Fig. 6). Based on all data we have compiled (Table 1), the egg spheres are divided into three sphere size ranges, < 0.5 m, 0.5–1 m and > 1 m in diameter. More than half (58%) of the spheres measured between 0.5 and 1 m in diameter. The depths of these spheres were from the surface to ~ 60 m (Fig. 7), but most spheres near Norway were observed from surface to 20 m depth whereas from the Mediterranean region most were from the surface to 30–50 m depth. Depths down to approximately 30 m are those visited by recreational sports divers.
Most spheres were observed during daytime (all areas combined), but one is also observed during night diving. Night diving is a more rare activity. Temperatures ranged (for all areas combined) 8–24 °C. For Norway the temperature range was 8–18 °C, and for the Mediterranean region 14–24 °C (Fig. 8). Around 55% of spheres from all areas combined had a dark streak, or structure, through the center.
In 2019 the same divers observed four spheres in the Arendal-Kristiansand area (S/SE Norway) from 28 July to 24 August (Fig. 9). There were also several reports from Lillesand to Mandal area the same year, as well as from Møre & Romsdal county (video 6). At an aquaculture farm in Stavanger area, Ombo, several spheres were observed by professional divers through a 13 year period, as spheres been bumping into, and getting entangled in, aquaculture nets.
Whole sphere (deformed) with live embryos, photographed in situ from Kristiansand area (sphere observation 24.08.2019). Note the different orientation patterns of the embryos within the jelly envelope, with more or less all heads facing downwards. This sphere has not been genetically tested, but is high likely made by Illex coindetii. (Photo credits: Anita Eliassen (sphere) and Geir Eliassen (embryos)/Arendal undervannsklubb. Collage Halldis Ringvold/Sea Snack Norway).
One French diver reports, «despite diving every week at Le Mejean, Frapao, throughout the year in the same area of the Mediterranean, only one sphere has been seen (15 August 2014). There is a strong seasonal thermocline there and the temperature is usually higher than 22 °C at depths of 5–10 m». The one sphere seen there had a brown streak, or structure, in the middle (Pers. com. Edouard Bard).
Discussion
Confirmation of species, using DNA analysis
Because the DNA of our sphere samples matches that of adult squid identified as I. coindetii from Norwegian waters we infer that the spheres are from I. coindetii. Much has been written about taxonomic difficulties in Illex. The COI tree comprises four clades of Illex, one of which clearly pertains to Illex argentinus (Castellanos, 1960). There are three other described species: Illex coindetii, Illex illecebrosus (Lesueur, 1821), and I. oxygonius Roper, Lu & Mangold, 1969. We labelled our clades A, B, and C, to indicate their correspondence with the findings of Carlini et al.32, and assume that each pertains to one of the described species of Illex. Carlini was unable to match species to clades, but Clade A not only contains the adults identified in this project as I. coindetii, but also contains specimens from the Mediterranean (DQ373941). Since I. coindetii is the only species of Illex known from the Mediterranean, this is further confirmation of the identity of Clade A, and thus our spheres, as Illex coindetii.
Using citizen science from roughly 200 divers secured observations of 90 spheres, including rare tissue samples of four of them, thus enabling a molecular approach towards the first confirmation of egg masses in situ as those of the broadtail shortfin squid, Illex coindetii. Illex coindetii was named in honour of Dr. Coindet from Geneva in 185137. It took 180 years from the description of the adult to identification of its egg mass in the wild. To our knowledge no whole egg mass of Illex spp. has previously been reported from the wild, except by Adolf Naef, who reported on live ommastrephid embryos and paralarvae from Naples, Italy2. The embryos were pulled out of a floating spawn or floating egg mass, or as he describes «Fig. 1 und 2 sind aus einem flottierenden Laich gezogene Larven von Ommatostrephiden». These illustrations were later identified as Illex coindetii by Boletzky et al.26, studying egg development of I. coindetii in the laboratory, claiming «The general characteristics of the embryonic developement observed by us match the figures given by Naef (1923 : plts 9–12) of an unidentified egg mass of a member of the Ommastrephidae (Naef 1921)». However, no drawing of the «laich» was provided.
Challenges collecting in situ material
Huge gelatinous spheres from squid are difficult to study in situ. They are rarely reported, and hard to sample. We have collected 90 sphere observations from ~ 35 years back (~ 1985 to 2019), from an area stretching from the Mediterranean Sea north to the Norwegian Sea, which gives a good illustration on sphere findings of ~ 2.6 sphere observations per year. In addition, the spheres most likely have a short-life span. Life span of spheres spawned and reared in aquaria (between 40 and 120 cm in diameter) of Todarodes pacificus (Steenstrup, 1880) is 5–7 days, with the smallest disintegrating first38.
Sphere shape and size
Gelatinous egg masses of cephalopods vary in size and form among species. Some egg masses are spherical, but there are also examples of oblong structures39,40,41. Sphere size may be up to 4 m in diameter1,5,42. Ringvold and Taite (op. cit.) collected information on a total of 27 spheres recorded in European waters varying from 0.3 to 2 m in diameter, as also for the additional spheres from this study. The four spheres in our study, confirmed to belong to I. coindetii, measured between 0.5 and 1 m in diameter.
Egg mass of another ommastrephid squid, Todarodes sagittatus, has yet to be found in situ. The species is known to be larger than Illex species, and egg mass is also most probably larger. The largest spheres recorded in our study measured up to 2 m in diameter, but none of these were sampled for molecular analysis, nor were pictures taken. It is uncertain whether they could belong to other species e.g., Todaropsis eblanae (Ball, 1841), Todarodes sagittatus or Ommastrephes sp..
Dark streak through core
Almost 60% of the spheres had a dark streak through the center. This feature might be ink, one important characteristic of cephalopods, produced by most cephalopod orders. The ink sac with its ink glands produces black ink containing melanin43. During fertilization, sperm are released—as well as possibly some ink. Spheres with or without ink may be a result of spheres beeing at different maturity stages1, where spheres with ink are freshly spawned. After a while, when embryos starts developing, the whole sphere, including the streak, will start to disintegrate.
Some of us speculate that one function of the streak through the center might serve as visual mimics e.g. of a large fish in order to scare off predators. Other possible functions discussed are also if the streak/structure can be caused by a sphere strengthening structure which is denser or having a higher optical density than the sourrounding structure. A disadvantage with the streak is that it might reveal the whole transparent sphere in the water, visible to e.g. scuba divers.
Function of the gelatinous matrix
Observations in captivity3,44 showed that species within the genus Illex produce gelatinous egg masses while swimming in open water. Gel functions as a buoyancy mechanism that prevents eggs from sinking, and complete density equilibration requires many days under most conditions44. Such a buoyancy mechanism keeps pelagically spawned eggs of Illex in areas where temperatures are most optimal for embryonic development. Optimal environmental conditions will likely have a positive effect on survival of both hatchlings and paralarvae. Despite consistency in where spawning areas are found, interannual variability has been recorded in the main recruitment areas, which could be related to e.g. mesoscale eddies and/or affecting post-hatching dispersal45.
Huge spheres are formed of mucus produced by the nidamental glands, situated inside the mantle cavity of the female46,47. When fully developed, hatchlings emit an enzyme which starts to dissolve the mucus. Eggs and embryos from our four spheres were covered in sticky gelatinous mass, except for a few specimens (from Arendal, collected 7 August, and Søgne) laying in the petri dish outside the sticky gel, in the surrounding sea water following the tissue sample, and might have been old enough to start producing such enzymes.
When at hatching, Illex coindetii eggs are about 2 mm long26,48, in line with other ommastrephids12. The longest of our embryos (from Arendal, collected 7 August) measured ~ 2 mm, a developed embryo with long proboscis, mantle about ½ of total body length, as well as chromatophores, large eyes and funnel visible (Fig. 3). It could possibly be a hatchling.
Abiotic factors and locations
The success and duration of embryonic development is related to water temperature. All observations available to date indicate that successful embryonic development for I. coindetii takes ca. 10–14 days at 15 °C; this temperature corresponds to the median temperature value reported for Mediterranean Sea midwater48. Boletzky et al.26 reports on a temperature minimum above 10 °C. Spheres in the Mediterranean were observed in temperatures ranging between 14 and 24 °C. Watermass temperature for one sphere with recently fertilized eggs (Ålesund sample, embryos stage ~ 3) from Norway was 8 °C. It was also observed north of the existing known distribution range for I. coindetii, in the Norwegian Sea, at 43 m depth. Most spheres from Norway were observed from July and August, in water mass 10–14 °C, with maximum temperature at 18 °C.
It is unknown whether some of the observed spheres had drifted to water layers unsuitable for the development of the eggs, and, eventually, would have died due to unfavourable abiotic conditions (e.g. transport outside the optimal temperature- or depth range for that particular species), but most likely they were in an area where they would survive. Higher occurrence of sphere sightings from 2017 to 2019, could be a combination of higher abundance of these squid in the area as well as increased knowledge regarding our Citizen Science Sphere Project, and thereby increased reports of observations.
Illex coindetii may be considered as an intermittent spawner with a spawning season extending throughout the year, reaching a peak in July–August18.
Our sphere observations from all areas were made from March to October: The earliest sphere which can be documented (to month) in the North Sea to date was observed 27 May (2001), and the last sphere was reported on 20 October (2019), coinciding with a study on adult Illex condetii from the North Sea where the spawning season has been suggested to be between spring and autumn49. However, our data show a peak of sphere observations from July to September (all areas combined), from July to August in Norway and from August to September in the Mediterranean Sea. The two recordings from Galicia in Spain, and Seiano in Italy, were the earliest recordings of the year, observed 24 March (in 2017 and 2019, respectively). For all areas combined, no observations during wintertime (November to February) have been recorded.
Embryonic development and consistencies of spheres
We collected tissue mass of four different spheres of I. coindetii, and embryos in each sphere were at different developmental stages, ~ 3 to 30, according to Sakai et al.36 based on I. argentinus. The sphere walls of the four spheres were also of different consistencies (Table 2); from Ålesund sphere with recently fertilized eggs and firm, transparent sphere wall to Søgne and Arendal spheres (the latter collected 7 August) with developed embryos and disintegrating sphere walls. The remains of the Arendal sphere was hanging as a long «scarf» in the water. Experienced divers, who previously had seen a few spherical spheres, recognized this disintegrating sphere.
Function of spheres
Ommastrephidae fecundity is extremely high, and a single sphere may contain thousands to several hundred thousands of eggs41,50,51,52. The function of the spheres is protection and transport of the offspring by sea currents for paralarval dispersal. Inside these gelatinous structures, the eggs and newly hatched paralarvae are protected from predation by e.g. fish, parasite infection and infestation by crustaceans and protozoans during a first relative short period of their lives5,51. Bottom trawlers operate in spawning areas of squids, exposing them to a risk of egg loss, as also for our fisherman at Askøy, Norway, who caught a sphere in his trawl1,5.
Scientific cruises and fishery
The Institute for Marine Research in Norway started identification of cephalopods on their regular scientific cruises in 2013, but no Illex coindetii was recorded that year. However, data show increasing catches from 2014 to 2019 (unpublished). No spheres are reported from Norway in 2013, but between 1995 to 2010, and from 2015 to 2019, observations were made. Most observations are between 2017 and 2019, indicating more frequent squid visits/spawnings. This coincides with more frequent sphere observations from 2017 to 2019.
The broadtail shortfin squid, Illex coindetii, is probably the most widespread species found on both sides of the Atlantic and throughout the Mediterranean Sea12. In the NE Atlantic, it has been reported from Oslofjorden, Norway (59°N);53 and the Firth of Forth, east Scotland54, southwards along the European and African coasts to Namibia, including Hollam’s Bird Island (24°S) and Cape Frio (18°S)55. For example, I. coindetii is periodically very abundant in coastal waters of the eastern North Atlantic off Scotland, Ireland and Spain, where it supports opportunistic fisheries. However, the oceanographic and biological factors that drive this phenomenon, are still unknown12.
Illex coindetii is widely distributed throughout the Mediterranean Sea11, where it is caught commercially mostly by Italian trawlers, usually as a by-catch, but also by recreational fishing, by means of squid jigging. Annual Italian landings during the last five years have varied between two and three thousand tonnes, but with historical landings reaching numbers of more than eight thousand tonnes during the 1980s and 1990s (FAO 2019)15.
In the North Sea, studies show that inshore squids (Alloteuthis subulata (Lamarck, 1798) and Loligo forbesii Steenstrup, 1856) are more abundant than short-finned squid (Illex coindetii, Todaropsis eblanae and Todarodes sagittatus), and I. coindetii is among the rarest ommastrephid species caught49,56. However, two recent studies (1) on summer spawning stock of Illex coindetii in the North Sea57 and (2) I. coindetii recorded from the brackish Baltic Sea58 suggest more frequent visits to this area. Reports on Illex coindetii from Norwegian waters are scarce, but it has been reported from Oslofjorden53, and recently as by-catch from Stavanger area, and by divers from Oslofjorden and Bergen.
Conclusion
Without tissue samples from each and every record for DNA corroboration we cannot prove that all spherical egg masses are conspecific, or that the rest of them (86 spheres) also belong to I. condetii. However, given similarities between all these spheres and known egg masses spawned by ommastrephid squid, we are confident in attributing these to ommastrephids.
We would like to continue recording sphere observations, hopefully revealing egg masses of other ommastrephids, such as e.g. Todarodes sagittatus and Todaropsis eblanae—of which egg mass has yet to be described.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ringvold, H. & Taite, M. Using citizen science to obtain data on large, floating gelatinous spheres from NE Atlantic, attributed to egg mass of ommastrephid squid (Oegopsida, Cephalopoda, Mollusca). Mar. Biol. Res. 14, 672–681 (2018).
Naef, A. Die Cephalopoden I. Teil (R. Friedländer & Sohn, Berlin, 1921).
Durward, R., Vessey, E., O’Dor, R. & Amaratunga, T. Reproduction in the Squid,Illex illecebrosus: first observations in captivity and implications for the life cycle. (ICNAF Selected Papers No. 6, 1980).
Sakurai, Y., Kiyofuji, H., Saitoh, S., Goto, T. & Hiyama, Y. Changes in inferred spawning areas of Todarodes pacificus (Cephalopoda: Ommastrephidae) due to changing environmental conditions. ICES J. Mar. Sci. 57, 24–30 (2000).
O’Shea, S., Bolstad, K. & Ritchie, P. First records of egg masses of Nototodarus gouldi McCoy, 1888 (Mollusca: Cephalopoda: Ommastrephidae), with comments on egg-mass susceptibility to damage by fisheries trawl. N. Z. J. Zool. 31, 161–166 (2004).
Boyle, P. & Rodhouse, P. Cephalopods: Ecology and Fisheries (Wiley, London, 2005).
Roberts, M., Zemlak, T. & Connell, A. Cyclonic eddies reveal Oegopsida squid egg balloon masses in the Agulhas Current, South Africa. Afr. J. Mar. Sci. 33, 239–246 (2011).
Hoving, H. et al. The study of deep-sea cephalopods. Adv. Mar. Biol. 67, 235–359 (2014).
Puneeta, P., Vijai, D., Yoo, H., Matsui, H. & Sakurai, Y. Observations on the spawning behavior, egg masses and paralarval development of the ommastrephid squid Todarodes pacificus in a laboratory mesocosm. J. Exp. Biol. 218, 3825–3835 (2015).
Moiseev, S. Underwater observations of North Atlantic squids from the manned submersible “Sever 2.” In Underwater Fishery Investigations. Collection of Scientific Papers (ed. Nizovtsev, G.) 71–78 (PINRO Publishing, Murmansk, 1987).
Jereb, P. et al. Cephalopod Biology and Fisheries in Europe: II. Species Accounts (International Council for the Exploration of the Sea, Copenhagen, 2015).
Roper, C., Nigmatullin, C. & Jereb, P. Family ommastrephidae in cephalopods of the world. An annotated and illustrated catalogue of species known to date. 2. Myopsid and oegopsid squids. In FAO Species Catalogue for Fishery Purposes, 4/2 (eds. Jereb, P. & Roper, C.) 69–347 (FAO, 2010).
Roura, A. et al. Oceanographic processes shape genetic signatures of planktonic cephalopod paralarvae in two upwelling regions. Prog. Oceanogr. 170, 11–27 (2019).
Jereb, P., Ceriola, L. & Ragonese, S. Illex coindetii. Life history, ecological importance and potential for a ‘key’ role in the Mediterranean context. 69. https://www.researchgate.net/publication/322144435_Illex_coindetii_Life_history_ecological_importance_and_potential_for_a_key_role_in_the_Mediterranean_context (2017).
FAO. FAO, 2006–2017. Fisheries and aquaculture software. FishStat Plus—Universal software for fishery statistical time series. Rome. http://www.fao.org/fishery/ (2017).
Rocha, F., Guerra, A. & González, A. A review of reproductive strategies in cephalopods. Biol. Rev. Camb. Philos. Soc. 76, 291–304 (2001).
Cole, L. The population consequences of life history phenomena. Q. Rev. Biol. 29, 103–137 (1954).
Gonzalez, A. & Guerra, A. Reproductive biology of the short-finned squid Illex coindetii (Cephalopoda, Ommastrephidae) of the Northeastern Atlantic. Sarsia 81, 107–118 (1996).
Rodhouse, P. & Hatfield, E. Dynamics of growth and maturation in the cephalopod Illex argentinus de Castellanos, I960 (Teuthoidea: Ommastrephidae). Philos. Trans. R. Soc. B 329, 229–241 (1990).
Sanchez, P. et al. Illex coindetii. In Squid recruitment dynamics. The genus Illex as a model. The commercial Illex species. Influences on variability (eds. Rodhouse, P., Dawe, E. & O’Dor, R.) 273 (FAO, 1998).
Ceriola, L. & Milone, N. Growth and reproduction of the squid Illex coindetii Verany, 1839 in the central Mediterranean Sea. Mediterr. Mar. Sci. 18, 107–120 (2017).
Salman, A. Fecundity and spawning strategy of shortfin squid Illex coindetii (Oegopsida: Ommastrephidae), in the Eastern Mediterranean. Turk. J. Fish. Aquat. Sci. 17, 841–849 (2017).
Hamabe, M. Embryological studies on the common squid, Ommastrephes sloani pacificus Steenstrup, in the southwestern waters of the Sea of Japan. Bull. Jpn. Sea Reg. Fish. Res. Lab. 10, 1–45 (1962).
O’Dor, R. & Dawe, E. Illex illecebrosus, Northern short-finned squid. In Advances in Squid Biology, Ecology and Fisheries Vol. II 36 (eds Rosa, R. et al.) (Nova Science Publishers Inc, Hauppauge, 2013).
Vijai, D. Egg masses of flying squids (Cephalopoda: Ommastrephidae). J. Shellfish Res. 35, 1007–1012 (2016).
Boletzky, S., Rowe, L. & Aroles, L. Spawning and development of the eggs in the laboratory of Illex coindetii (Mollusca: Cephalopoda). Veliger 15, 257–258 (1973).
Fernández-Álvarez, F., Martins, C., Vidal, E. & Villanueva, R. Towards the identification of the ommastrephid squid paralarvae (Mollusca: Cephalopoda): morphological description of three species and a key to the north-east Atlantic species. Zool. J. Linn. Soc. 180, 268–287 (2017).
Andric, M. Croatia’s Undersea World (Car Herc, Zagreb, 1999).
Folmer, O., Black, M., Hoeh, W., Lutz, R. & Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 3, 294–299 (1994).
Altschul, S., Gish, W., Miller, W., Myers, E. & Lipman, D. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Benson, D. et al. GenBank. Nucleic Acids Res. 41, D36-42 (2013).
Carlini, D., Kunkle, L. & Vecchione, M. A molecular systematic evaluation of the squid genus Illex (Cephalopoda: Ommastrephidae) in the North Atlantic Ocean and Mediterranean Sea. Mol. Phylogenet. Evol. 41, 496–502 (2006).
Okonechnikov, K., Golosova, O. & Fursov, M. Unipro UGENE: a unified bioinformatics toolkit. Bioinformatics 28, 1166–1167 (2012).
Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30, 1312–1313 (2014).
Edler, D., Klein, J., Antonelli, A. & Silvestro, D. raxmlGUI 2.0 beta: a graphical interface and toolkit for phylogenetic analyses using RAxML. bioRxiv 800912. https://doi.org/10.1101/800912 (2019).
Sakai, M., Bruneiti, N., Elena, B. & Sakurai, Y. Embryonic development and hatchlings of Illex argentinus derived from artificial fertilization. South Afr. J. Mar. Sci. 20, 255–265 (1998).
Verany, G. Mollusques méditeranéens observés, décrits, figurés et chromolitographiés d’après le vivant. 1. Céphalopodes de la Méditerranée, Vols. 1–16 (Genes, 1851).
Puneeta, P. et al. Structure and properties of the egg mass of the ommastrephid squid Todarodes pacificus. PLoS ONE 12, e0182261 (2017).
Quoy, I. & Gaimard, J. Observations sur les oeufs des Mollusques. Ann. Sci. Nat. 20, 472 (1830).
Sanzo, L. Nidamento pelagico, uova e larve di Thysanoteuthis rhombus Troschel. Mem. R. Com. Talassogr. Ital. 161, 1–10 (1929).
Guerra, A. Planktonic egg masses of the diamond-shaped squid Thysanoteuthis rhombus in the eastern Atlantic and the Mediterranean Sea. J. Plankton Res. 24, 333–338 (2002).
Lee, J. What is this car-size ball of jelly that mystified divers? Natl. Geogr. Weird & Wild (2015).
Derby, C. Cephalopod ink: production, chemistry, functions and applications. Mar. Drugs 12, 2700–2730 (2014).
O’Dor, R. & Balch, N. Properties of Illex illecebrosus egg masses potentially influencing larval oceanographic distribution Vol. N899 (North West Atlantic Fisheries Organization, Dartmouth, 1985).
Lefkaditou, E. et al. Influences of environmental variability on the population structure and distribution patterns of the short-fin squid Illex coindetii (Cephalopoda: Ommastrephidae) in the Eastern Ionian Sea. In Essential fish habitat mapping in the Mediterranean (ed. Valavanis, V.) 71–90 (Springer, Dordrecht, 2008). https://doi.org/10.1007/978-1-4020-9141-4_6.
Kimura, S., Higuchi, Y., Aminaka, M., Bower, J. & Sakurai, Y. Chemical properties of egg-mass mucin complexes of the ommastrephid squid Todarodes pacificus. J. Molluscan Stud. 70, 117–121 (2004).
Nair, J. et al. Cephalopod research and bioactive substances. Int. J. Mol. Sci. 40, 13–27 (2011).
Villanueva, R., Quintana, D., Petroni, G. & Bozzano, A. Factors influencing the embryonic development and hatchling size of the oceanic squid Illex coindetii following in vitro fertilization. J. Exp. Mar. Biol. Ecol. 407, 54–62 (2011).
Oesterwind, D., Piatkowski, U. & Brendelberger, H. On distribution, size and maturity of shortfin squids (Cephalopoda, Ommastrephidae) in the North Sea. Mar. Biol. Res. 11, 188–196 (2015).
Laptikhovsky, V. & Nigmatullin, C. Egg size, fecundity, and spawning in females of the genus Illex (Cephalopoda: Ommastrephidae). ICES J. Mar. Sci. 50, 393–403 (1993).
Bower, J. & Sakurai, Y. Laboratory observations on Todarodes pacificus (Cephalopoda: Ommastrephidae) egg masses. Am. Malacol. Bull. 13, 65–71 (1996).
Snyder, R. Aspects of the biology of the giant form of Sthenoteuthis oualaniensis (Cephalopoda: Ommastrephidae) from the Arabian Sea. J. Molluscan Stud. 64, 21–34 (1998).
Lu, C. Systematics and Zoogeography of the Squid Genus Illex (Oegopsida; Cephalopoda) (Memorial University of Newfoundland, St. John’s, 1973).
Norman, A. Revision of the British Mollusca. Ann. Mag. Nat. Hist. 6, 452–485 (1890).
Roeleveld, M. The status and importance of cephalopod systematics in southern Africa. Afr. J. Mar. Sci. 20, 1–16 (1998).
Oesterwind, D., ter Hofstede, R., Harley, B., Brendelberger, H. & Piatkowski, U. Biology and meso-scale distribution patterns of North Sea cephalopods. Fish. Res. Int. J. Fish. Sci. Fish. Technol. Fish. Manag. 106, 141–150 (2010).
Oesterwind, D. & Schaber, M. First Evidence of Illex coindetii (Vérany, 1839) in the Baltic Sea and the Kattegat. Fish Res. 221, 105384. https://doi.org/10.1007/s41208-019-00178-8 (2019).
Oesterwind, D. et al. First evidence of a new spawning stock of Illex coindetii in the North Sea (NE-Atlantic). Fish. Res. 221, 105384 (2020).
Acknowledgements
Sea Snack Norway (Project J. nr. Gel-Sph 17-20) and University of Ireland/Galway mainly funded this research. This work was supported by the Research Council of Norway through the Centres of Excellence funding scheme (Grant No. 223254 – NTNU AMOS) given to GJ. MT was funded by a Ph.D. fellowship from the Irish Research Council and was supported by the Dr. Tony Ryan Research Fund. We would like to thank everybody who have reported observations to our citizen science project, and Gro van der Meeren, Elaina Jorgensen and Manuel AE Malaquias who participated in discussions and media coverage in an early phase, and John Turriff, Mark Shea and Roddy Hay with field- and laboratory work in a later phase. Zoea Mallorca dive center, with Director and Dive Instructor Luis Comenge, is thanked for producing posters and distributing them to diving centers in Mallorca, and Raúl Frisuelos at Aolde radio for broadcasting radioshows for divers across Spain. Arne Hassel is thanked for measurements and picture of formalin fixated egg. We also wish to thank Faroe Marine Research Institute (Faroe Islands), Marine & Freshwater Research Institute (Iceland) and Alf Tore Mjøs at Stavanger Museum (Norway) for tissue samples of adult squid.
Author information
Authors and Affiliations
Contributions
All authors have contributed to this manuscript in form of writing or providing essential tissue samples and observations of spheres. H.R. was the project leader and contributed with writing, conseptualization, planning, posting and updating posters in media to reach divers, collecting observations as well as pictures and videos, organizing tissue samples, analyzing and interpreting data, making map and picture collages. M.T. and L.A. designed, carried out, and wrote up the molecular genetic studies. M.V., G.J. and A.F. participated in discussions and writing. M.P. collected many of the sphere observations from France, through DORIS, and writing. R.S. participated in media stunts in Italy, and writing. J.C. contributed with designing poster for media, media stunts in Spain, as well as own observation of sphere. S.B., H.S. and A.E.W.S. contributed in handling tissue samples at the laboratory, and writing. Divers M.F., P.G.O., A.E., G.E. and K.K. contributed with sphere tissue samples, pictures and videos. All authors read, commented on and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Video 1.
Supplementary Video 2.
Supplementary Video 3.
Supplementary Video 4.
Supplementary Video 5.
Supplementary Video 6.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ringvold, H., Taite, M., Allcock, A.L. et al. In situ recordings of large gelatinous spheres from NE Atlantic, and the first genetic confirmation of egg mass of Illex coindetii (Vérany, 1839) (Cephalopoda, Mollusca). Sci Rep 11, 7168 (2021). https://doi.org/10.1038/s41598-021-86164-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-86164-8
This article is cited by
-
Climate change-related changes in cephalopod biodiversity on the North East Atlantic Shelf
Biodiversity and Conservation (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.