Abstract
We present a cohort of patients with anterior communicating artery (ACoA) aneurysms to investigate morphological characteristics and clinical factors associated with rupture of the aneurysms. 505 patients with ACoA aneurysms were identified at the Brigham and Women’s Hospital and Massachusetts General Hospital between 1990 and 2016, with available CT angiography (CTA). Three-dimensional (3D) reconstructions were performed to evaluate aneurysmal morphologic features, including location, projection, irregularity, the presence of daughter dome, height, height/width ratio, and relationships between surrounding vessels. Patient risk factors assessed included patient age, sex, tobacco use, alcohol use, and family history of aneurysms and aneurysmal subarachnoid hemorrhage. Logistic regression was used to build a predictive ACoA score for rupture. Morphologic features associated with ruptured ACoA aneurysms were the presence of a daughter dome (OR 21.4, 95% CI 10.6–43.1), smaller neck diameter (OR 0.55, 95% CI 0.42–0.71), larger aspect ratio (OR 3.57, 95% CI 2.05–6.24), larger flow angle (OR 1.03, 95% CI 1.02–1.05), and smaller ipsilateral A2-ACoA angle (OR 0.98, 95% CI 0.97–1.00). Tobacco use was predominantly associated with morphological factors intrinsic to the aneurysm that were associated with rupture while younger age was also associated with morphologic features extrinsic to the aneurysm that were associated with rupture. The ACoA score had good predictive capacity for rupture with AUC = 0.92 using the 0.632 bootstrap cross-validation for correction of overfitting bias. Ruptured ACoA aneurysms were associated with morphological features that are simple to assess using a simple scoring system. Tobacco use and younger age were predominantly associated with intrinsic and extrinsic morphological features characteristic of rupture, respectively.
Similar content being viewed by others
Introduction
Rupture of an intracranial aneurysm is unpredictable and can lead to significant morbidity and mortality. There is ongoing effort into understanding the natural history of aneurysm growth and factors associated with rupture to improve our clinical assessment for treatment. Anterior communicating artery (ACoA) aneurysms are amongst the most common intracranial aneurysms to present after rupture and are estimated to account for up to 30% of all intracranial aneurysms1,2. The advancement of computed tomography angiography (CTA) and digital subtraction angiography (DSA) has provided a greater venue to assess morphologic features that may be associated with rupture of an aneurysm. Features previously proposed to be associated with ACoA aneurysm rupture included intrinsic aneurysm characteristics, such as aneurysm size, height and projection, and transitional characteristics, such as the relationship of the aneurysm with the parent vessels3,4. It has been postulated that the underlying mechanisms and determinants of aneurysm ruptures are multifactorial, but are associated with hemodynamic loads, wall biomechanics, and the local aneurysm environment5.
In addition to intrinsic aneurysm characteristics, patients’ clinical factors, such as tobacco use and hypertension, are well-established risk factors associated with intracranial aneurysm rupture6,7. Differences in aneurysm morphology and size have been proposed between smoking and non-smoking patients8,9. It is thought that nicotine promotes local inflammation and sustained angiogenesis, resulting in downstream changes and alteration of aneurysm morphology10. Here, we present a large cohort of 505 patients with anterior communicating artery (ACoA) aneurysms to assess morphological features associated with ruptured aneurysms, as well as, the association of patient characteristics, including age and tobacco use, with those morphological features.
Methods
Patient selection
Patients diagnosed with intracranial aneurysms at the Brigham and Women’s Hospital (BWH) and Massachusetts General Hospital (MGH) between 1990–2016 were identified using natural language processing (NLP), as well as prospectively, as previously described11. Manual medical record review was performed to confirm the diagnosis of a cerebral artery aneurysm. Briefly, both codified and NLP data were used to identify an initial set of patients with potential aneurysms from the Research Patient Data Registry (RPDR), the research version of our electronic medical record, 5,589 patients were initially identified11. 727 of these patients were also seen on clinical presentation from 2007–2013 with prospectively collected data. Additionally, 474 patients with prospectively collected data who were seen on clinical presentation from 2013–2016, were also included11. 4701 patients with definite saccular aneurysms were identified by manually reviewing (AC and RD) the electronic medical records of all 6063 patients11. Patients with non-saccular aneurysms, aneurysms associated with arteriovenous malformations, or aneurysms that lacked preoperative CT angiography (CTA) were excluded from this study.
505 patients with anterior communicating (ACoA) aneurysms were included in this study. CT angiography (CTA) images were obtained using mi2b2 open-source software to comply with research privacy requirements12. Relevant demographic and clinical data, including age, sex, hypertension, current tobacco use, heavy tobacco use > 1 pack per day, years since quitting tobacco, alcohol use, hypertension, atrial fibrillation, history of ischemic stroke, coronary artery disease, history of myocardial infarction, family history of intracranial aneurysms and aneurysmal subarachnoid hemorrhage, and aspirin anticoagulant, and antihyperlipidemic agent use at diagnosis, were retrieved from the medical records. This study was approved by the Partners Human Research Committee which also waived the requirement for informed consent. All procedures performed were in accordance with the ethical standards of the institutional review board and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Reconstruction of 3-dimensional models
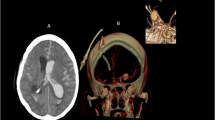
Three-dimensional (3D) models of aneurysms and their surrounding vasculature were generated from DICOM (Digital Images and Communication in Medicine) format using preoperative CTA by the Vitrea Advanced Visualization software (version 6.9.68.1, Vital Images, Minnetonka, MN)13. Images were evaluated by an attending neurosurgeon (JZ) with verification by a second neurosurgeon (RD) when needed. Detailed protocol on the construction of the 3-dimensional images and aneurysm measurements were previously described14. Intrinsic, transitional and extrinsic morphological parameters of the aneurysms were studied to assess their risk for rupture (Fig. 1). Intrinsic characteristics are those that involve the aneurysm only, extrinsic characteristics involve only the surrounding vessels, and transitional ones involve both the aneurysm and the surrounding vessels15.
(A) Illustration of morphological parameters for anterior communicating artery (ACoA) aneurysm. (B) Projection of ACoA aneurysm based on sagittal view. Horizontal line is parallel to the anterior skull base. (C) Location of ACoA aneurysm based on coronal projections. Medial location is defined as projections within 15° of midline, between lines RC and LC. Image was partially created by Vitrea Advanced Visualization software (version 6.9.68.1).
Aneurysms were assessed for their location (middle, left or right), projection (anterior, posterior, superior or inferior), the smoothness of the wall (smooth or irregular), and the presence of a daughter dome. The maximum height of the aneurysm was measured from the center of the aneurysm neck to the greatest distance on the aneurysm dome. Maximum perpendicular height was measured as the distance from the center of the aneurysm neck to the perpendicular height of the aneurysm dome. Neck diameter was defined as the width of the aneurysm neck. Aspect ratio was calculated as the ratio of maximum perpendicular height to the neck diameter. In addition, the maximum width of the aneurysm was measured, and a height/width ratio was calculated by dividing the maximum perpendicular height by the width.
Extrinsic parameters of the aneurysms involving the surrounding vasculature were also assessed. The average A1 length and the diameters of the ACoA, ipsilateral A1, ipsilateral A2, contralateral A1, and contralateral A2 were measured. Mean diameter of the vessels was measured by averaging the diameter of the cross-section of a vessel just proximal to the neck of the aneurysm, and the diameter of the cross-section at 1.5 times from the neck of the aneurysm. Anatomical variations were also noted, including ipsilateral hypoplastic A1, and ipsilateral or contralateral aplastic A1. The angles between ipsilateral A1-A2, ipsilateral A1-ACoA, ipsilateral A2-ACoA, contralateral A1–A2, contralateral A1-ACoA, and contralateral A2-ACoA were also measured.
Transitional parameters evaluated included size ratio and flow angle. Size ratio was defined as the ratio between the maximum aneurysm height and the mean vessel diameter of all branches associated with the aneurysm. Flow angle is the angle between the maximum height of the aneurysm and the parent vessel.
Statistical analysis
Demographic baseline characteristics between ruptured and unruptured ACoA aneurysms were evaluated using the student t-test for continuous variables and the Pearson’s chi-square test for categorical variables. Univariate and multivariate logistic regression models were used to study different morphological parameters on the rupture status of ACoA aneurysms, with a backward elimination procedure to identify significant confounders with a cut-off value of p = 0.1 for the initial selection of variables. Additional univariate and multivariate analyses were performed to study patient characteristics and risk factors and their associations with morphologic parameters that met statistical significance.
A predictive score for rupture was created using the results of the multivariate model including covariates with p < 0.05. Points were allocated according to OR, with -3 points for OR < 1/2, -2 points for OR [1/2,2/3), − 1 point for OR [2/3,1.0), 1 point for OR [1.0,1.5), 2 points for OR [1.5,2.0), 3 points for OR [2.0,3.0), and 4 points for OR ≥ 2.0. The thresholds used for continuous variables were the mean value between the ruptured and unruptured groups. The predictive capacity of the score was evaluated by using a logistic regression with lasso penalty. The area under the receiver operating curve (AUC) was obtained using the 0.632 bootstrap cross-validation for correction of overfitting bias via the ROC63216 package in R17 (version 3.6.2). The goodness-of-fit of the model was evaluated with the Hosmer–Lemeshow test using quartiles for risk intervals. Statistical analyses were performed using the Stata statistical software (version 14, StatCorp. College Station, TX) and R17 (version 3.6.2). A sensitivity analysis was performed for the predictive score by removing variables that may be affected by the rupture process such as the presence of a daughter dome. A p value of less than 0.5 was considered statistically significant.
Results
Baseline characteristics of ruptured and unruptured ACoA aneurysms
505 patients with ACoA aneurysms were identified in this study. Table 1 shows the demographics of the study population. The mean age at presentation was 57 ± 13 years, with females representing 62% of the cohort. Patients with ruptured aneurysms presented earlier than those with unruptured aneurysms (54 ± 13 years vs 61 ± 12 years, p < 0.01), with a higher rate of tobacco use (38% vs 28%, p < 0.01) and alcohol use (57% vs 45%, p = 0.01). Female sex, hypertension, family history of SAH and family history of intracranial aneurysms were not different between the ruptured and unruptured cohorts.
Further analyses for association between patient characteristics demonstrated an inverse relationship between tobacco use and age (OR0.96 [95% CI 0.94–0.97], p < 0.01), and an association with tobacco use and alcohol use (OR1.33 [95% CI 1.1–1.6], p < 0.01). The associations between tobacco use and aspirin use (OR1.32 [95% CI 0.58–3.0], p = 0.51), anticoagulant use (OR1.94 [95% CI 0.32–11.9] p = 0.47), and antihyperlipidemic agent use (OR0.97 [95% CI 0.47–1.97], p = 0.93) were not significant.
Table 1 illustrates baseline aneurysm characteristics of patients with unruptured and ruptured ACoA aneurysms. For intrinsic morphological parameters, ruptured aneurysms tend to be anteriorly projecting, had a larger maximal height, perpendicular height, height/width ratio, aspect ratio, but a smaller neck diameter. Both size ratio and flow angle were larger in ruptured aneurysms. For extrinsic parameters, ruptured aneurysms were associated with larger ipsilateral A1-A2 angles, smaller ipsilateral A1 and A2 diameters, and smaller ipsilateral and contralateral A2-ACoA angles.
Morphological features associated with ruptured ACoA aneurysms
Table 2 illustrates intrinsic, transitional, and extrinsic aneurysm characteristics associated with rupture. Univariate analyses showed an association of rupture with the presence of irregular domes and daughter domes, larger maximal height, perpendicular height, height/width ratio, aspect ratio, ipsilateral A1–A2 angle, flow angle, and size ratio. Aneurysm rupture was inversely associated with neck diameter, ipsilateral A1 diameter, ipsilateral A2 diameter, and ipsilateral A2-ACoA angle.
After multivariate adjustment, the presence of a daughter dome (OR21.4 [95% CI 10.6–43.1]), larger aspect ratio (OR3.57 [95% CI 2.05–6.24]), smaller neck diameter (OR0.55 [95% CI 0.42–0.71]), larger flow angle (OR1.03 [95% CI 1.02–1.05]), and smaller ipsilateral A2-ACoA angle (OR0.98 [95% CI 0.97–1.00]) remained associated with rupture status.
Patient risk factors associated with morphological features
Morphological features associated with ruptured ACoA on univariate analyses were subsequently assessed to determine their association with clinical risk factors (Table 3 and Supplemental Tables 1–3). In the univariate analyses, age at diagnosis was associated with larger neck diameter, ipsilateral A1 diameter, ipsilateral A2 diameter, ipsilateral A2-ACoA angle, and inversely associated with aspect ratio, height/width ratio, ipsilateral A1–A2 angle, contralateral A1–A2 angle, flow angle, and the presence of daughter domes and irregularity. Active tobacco use was associated with larger maximal height, perpendicular height, aspect ratio, height/width ratio and size ratio on univariate analyses.
After multivariate adjustment, age at diagnosis remains associated with larger neck diameter (β0.02 [95% CI 0.02–0.03]), and inversely associated with ipsilateral A1-A2 angle (β− 0.21 [95% CI − 0.41 to − 0.01]), contralateral A1–A2 angle (β− 0.34 [95% CI − 0.57 to − 0.12]), flow angle (β− 0.24 [95% CI − 0.39 to 0.078]). and the presence of irregular domes (OR0.98 [95% CI 0.96–0.99]) and daughter domes (OR 0.98 [0.96–0.99]). Current tobacco use was associated with larger maximal height (β1.1 [95% CI 0.44–1.8]), perpendicular height (β1.1 [95% CI 0.38–1.8]), aspect ratio (β0.22 [95% CI 0.06–0.37]), and size ratio (β0.19 [95% CI 0.06–0.31]). Other associations with female sex, years since quitting smoking, alcohol use, hypertension, coronary artery disease, history of ischemic stroke, and family history of aneurysms and SAH were also found.
Predictive ACoA score
The predictive ACoA score obtained from the multivariable regression model is shown in Table 4. The AUC of the ACoA score from the 0.632 bootstrap cross-validation was 0.92. The p-value for the Hosmer–Lemeshow test was 0.15, signifying good fit. The proportion of ruptured aneurysms stratified by ACoA score is shown in Fig. 2.
Percent ruptured aneurysms stratified by predictive anterior communicating artery aneurysm score defined in Table 4.
A sensitivity analysis was performed by removing the presence of a daughter dome as that may occur as a result of the rupture process. The predictive score using this reduced model is shown in Supplemental Table 4. The AUC from the 0.632 bootstrap cross-validation was 0.89. The p-value for the Hosmer–Lemeshow test was 0.55. The proportion of ruptured aneurysms stratified by the reduced score is shown in Supplemental Fig. 1.
Discussion
In this study, we present a cohort of 505 patients with ACoA aneurysms, and studied aneurysm morphological factors and patient characteristics associated with aneurysm rupture. We demonstrated that the presence of a daughter dome, smaller neck diameter, larger aspect ratio, larger flow angle, and smaller ipsilateral A2-ACoA angle were associated with ACoA aneurysm rupture. We also demonstrated that tobacco use was predominantly associated with intrinsic aneurysm characteristics associated with rupture including larger aspect ratio. In contrast, younger age was also associated with extrinsic aneurysm characteristics associated with rupture, including smaller neck diameter, the presence of irregularity and a daughter dome, larger flow angle, and larger ipsilateral A1–A2 angle and contralateral A1–A2 angle.
Morphological risk factors
We identified several intrinsic morphological features that were associated with rupture of ACoA aneurysms. We found the presence of a daughter dome to have the highest association with rupture risk. This is in support of prior studies which demonstrated daughter sacs to be highly associated with rupture status18,19,20. We also found aneurysm rupture to be associated with a larger aspect ratio and a smaller neck diameter. While aspect ratio and similar parameters such as height/width ratio have been found to be significant in prior studies21, neck diameter has not previously been found to be significant and may be due to our large sample size. A smaller neck diameter may affect the location and size of the impingement zone, size and location of the inflow jet, and the area of low wall shear stress22. These changes may contribute to the increased rupture risk. We did not find an association of aneurysm rupture with projection or the location of the ACoA aneurysm.
Our analysis of the extrinsic and transitional morphological features demonstrated larger flow angle and smaller ipsilateral A2-ACoA angle to be associated with rupture. Flow angle reflects the relationship between the aneurysm and the parent vessel and has previously been described to be associated with rupture23, but not all studies found this association24,25. Computational fluid dynamics modeling has demonstrated that larger flow angles resulted in higher flow velocity and wall shear stress, which may explain the association of flow angle with aneurysm rupture27. Our study also found a smaller ipsilateral A2-ACoA angle to be associated with rupture which has not previously been shown. The A2-ACoA angle has been shown to be inversely associated with ipsilateral A1 and A2 diameter, which was thought to explain its contribution to hemodynamic stress, but we did not find this association26.
Patient risk factors
Analysis of patient characteristics demonstrated tobacco use to be predominantly associated with intrinsic morphological features of ruptured aneurysms. Specifically, tobacco use was associated with larger maximal and perpendicular heights, aspect ratio, and size ratio. Smoking is a known independent risk factor for aneurysm formation and rupture, with a dose–response curve with the duration of smoking and increased rupture risk6,10,27,28,29. Our study further demonstrated that ACoA aneurysms in patients with active tobacco use have morphological features associated with rupture. It can be postulated that tobacco use may alter local aneurysm environment and architecture through its inflammatory and downstream pathways, resulting in morphological changes associated with higher rupture risk10.
In our series, patients with ruptured aneurysms presented at a younger age and moreover, younger age was associated with intrinsic, transitional, and extrinsic morphological features associated with aneurysm rupture, namely, the presence of irregular domes and daughter domes, smaller neck diameter, larger flow angle, and larger ipsilateral and contralateral A1–A2 angles. In contrast with tobacco use, these morphological features also include those that are extrinsic to the aneurysm. While older age is considered a risk factor for aneurysm rupture30, we found an opposite effect where ruptured ACoA aneurysms occurred at a younger age than unruptured ACoA aneurysms, consistent with prior observations31,32,33. This increase in rupture among younger patients may be explained by the changes to the surrounding vasculature, such as the A1–A2 angles, that occur with age.
Limitations
While an association with morphological patterns and rupture status can be investigated, these factors do not necessarily predict rupture risk as these patterns may reflect post-rupture changes. However, the extrinsic morphological factors and other intrinsic parameters such as neck diameters are unlikely to alter with aneurysm rupture and can serve as more robust parameters for risk prediction. To address the potential post-rupture changes, we have performed a sensitivity analysis with the presence of a daughter dome removed from our predictive score and achieved comparable results. A final limitation is that our prediction model is not independently validated. Despite these limitations, this is the largest series of ACoA aneurysms to our knowledge that assesses morphological features and patient risk factors associated with rupture.
Conclusion
We found that the presence of a daughter dome, smaller neck diameter, larger aspect ratio, larger flow angle and smaller ipsilateral A2-ACoA angle were associated with rupture. In addition, tobacco use and younger age were associated with predominantly intrinsic and extrinsic morphological parameters associated with rupture, respectively. The assessment of these morphologic features using a simple scoring system in addition to clinical risk factors may help in the risk assessment and management of patients with ACoA aneurysms.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Wiebers, D. O. et al. Unruptured intracranial aneurysms: Natural history, clinical outcome, and risks of surgical and endovascular treatment. Lancet 362, 103–110 (2003).
Zhao, L. et al. An analysis of 1256 cases of sporadic ruptured cerebral aneurysm in a single chinese institution. PLoS ONE 9, e85668 (2014).
Cai, W., Hu, C., Gong, J. & Lan, Q. Anterior communicating artery aneurysm morphology and the risk of rupture. World Neurosurg. 109, 119–126 (2018).
Ho, A. L., Lin, N., Frerichs, K. U. & Du, R. Intrinsic, transitional, and extrinsic morphological factors associated with rupture of intracranial aneurysm. Neurosurgery. 77, 433–441 (2015).
Sforza, D. M., Putman, C. M. & Cebral, J. R. Hemodynamics of cerebral aneurysms. Annu. Rev. Fluid Mech. 41, 91–107 (2009).
Can, A. et al. Association of intracranial aneurysm rupture with smoking duration, intensity, and cessation. Neurology. 89, 1408–1415 (2017).
Vlak, M. H., Rinkel, G. J., Greebe, P. & Algra, A. Risk of rupture of an intracranial aneurysm based on patient characteristics: A case-control study. Stroke 44, 1256–1259 (2013).
Ho, A. L., Lin, N., Frerichs, K. U. & Du, R. Smoking and intracranial aneurysm morphology. Neurosurgery. 77, 59–66 (2015).
Xia, N. et al. Smoking associated with increased aneurysm size in patients with anterior communicating artery aneurysms. World Neurosurg. 87, 155–161 (2016).
Kamio, Y. et al. Roles of nicotine in the development of intracranial aneurysm rupture. Stroke 49, 2445–2452 (2018).
Castro, V. M. et al. Large-scale identification of patients with cerebral aneurysms using natural language processing. Neurology. 88, 164–168 (2017).
Murphy, S. N. et al. High throughput tools to access images from clinical archives for research. J. Digit. Imaging. 28, 194–204 (2015).
Zhang J, Can A, Lai PMR, Mukundan S, Jr., Castro VM, Dligach D, et al. Vascular geometry associated with anterior communicating artery aneurysm formation. World Neurosurg. 2020
Zhang, J. et al. Morphological variables associated with ruptured middle cerebral artery aneurysms. Neurosurgery. 85, 75–83 (2019).
Ho, A. L., Lin, N., Frerichs, K. U. & Du, R. Intrinsic, transitional, and extrinsic morphological factors associated with rupture of intracranial aneurysms. Neurosurgery. 77, 433–441 (2015).
Foucher R. Roc632: Construction of diagnostic or prognostic scoring system and internal validation of its discriminative capacities based on roc curve and 0.633+ boostrap resampling. 2013
Team RC. R: A language and environment for statistical computing. 2020
Bjorkman, J. et al. Irregular shape identifies ruptured intracranial aneurysm in subarachnoid hemorrhage patients with multiple aneurysms. Stroke 48, 1986–1989 (2017).
Investigators, U. J. et al. The natural course of unruptured cerebral aneurysms in a japanese cohort. N. Engl. J. Med. 366, 2474–2482 (2012).
Kang, H. et al. Aneurysm characteristics associated with the rupture risk of intracranial aneurysms: A self-controlled study. PLoS ONE 10, e0142330 (2015).
Kim, M. C. & Hwang, S. K. The rupture risk of aneurysm in the anterior communicating artery: A single center study. J Cerebrovasc Endovasc Neurosurg. 19, 36–43 (2017).
Schneiders, J. J. et al. Intracranial aneurysm neck size overestimation with 3d rotational angiography: The impact on intra-aneurysmal hemodynamics simulated with computational fluid dynamics. AJNR Am. J. Neuroradiol. 34, 121–128 (2013).
Duan, Z. et al. Morphological parameters and anatomical locations associated with rupture status of small intracranial aneurysms. Sci. Rep. 8, 6440 (2018).
Maiti, T., Bir, S. C., Patra, D. P., Cuellar, H. & Nanda, A. 158 morphological parameters for anterior communicating artery aneurysm rupture risk assessment. Neurosurgery. 63, 163–164 (2016).
Shao, X. et al. The effect of anterior projection of aneurysm dome on the rupture of anterior communicating artery aneurysms compared with posterior projection. Clin. Neurol. Neurosurg. 143, 99–103 (2016).
Zhang, X. J., Gao, B. L., Hao, W. L., Wu, S. S. & Zhang, D. H. Presence of anterior communicating artery aneurysm is associated with age, bifurcation angle, and vessel diameter. Stroke 49, 341–347 (2018).
Feng, X. et al. Number of cigarettes smoked per day, smoking index, and intracranial aneurysm rupture: A case-control study. Front. Neurol. 9, 380 (2018).
Bonita, R. Cigarette smoking, hypertension and the risk of subarachnoid hemorrhage: A population-based case-control study. Stroke 17, 831–835 (1986).
Teunissen, L. L., Rinkel, G. J., Algra, A. & van Gijn, J. Risk factors for subarachnoid hemorrhage: A systematic review. Stroke 27, 544–549 (1996).
Brinjikji, W. et al. Risk factors for growth of intracranial aneurysms: A systematic review and meta-analysis. AJNR Am. J. Neuroradiol. 37, 615–620 (2016).
Matsukawa, H. et al. Morphological and clinical risk factors for the rupture of anterior communicating artery aneurysms. J. Neurosurg. 118, 978–983 (2013).
Kamitani, H., Masuzawa, H., Kanazawa, I. & Kubo, T. Saccular cerebral aneurysms in young adults. Surg. Neurol. 54, 59–66 (2000).
Detmer, F. J. et al. Associations of hemodynamics, morphology, and patient characteristics with aneurysm rupture stratified by aneurysm location. Neuroradiology 61, 275–284 (2019).
Funding
This study was supported by Partners Personalized Medicine (RD), the National Institutes of Health (U54 HG007963: TC and SM, U01 HG008685: SM, and R01 HG009174: SM), and National Natural Science Foundation of China (81571121).
Author information
Authors and Affiliations
Contributions
All authors critically reviewed the manuscript. Study concept and design: J.Z., A.C., R.D. Acquisition of data: J.Z., A.C., P.L., V.C., D.D., S.F., V.G., R.D. Analysis and interpretation of data: J.Z., A.C., P.L., S.M., N.S., G.S., S.M., T.C., S.W., R.D. Drafted the manuscript: A.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, J., Lai, P.M.R., Can, A. et al. Tobacco use and age are associated with different morphologic features of anterior communicating artery aneurysms. Sci Rep 11, 4791 (2021). https://doi.org/10.1038/s41598-021-84315-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-84315-5
This article is cited by
-
The frequency and impact of tramadol addiction on acute aneurysmal subarachnoid hemorrhage: cross-sectional multicenter study
The Egyptian Journal of Neurology, Psychiatry and Neurosurgery (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.