Abstract
Amongst other immune cells, neutrophils play a key role in systemic inflammation leading to cardiovascular disease and can release inflammatory factors, including lipocalin-2 (LCN2). LCN2 drives cardiac hypertrophy and plays a role in maladaptive remodelling of the heart and has been associated with renal injury. While lifestyle factors such as diet and exercise are known to attenuate low-grade inflammation, their ability to modulate plasma LCN2 levels is unknown. Forty-eight endurance athletes and 52 controls (18–55 years) underwent measurement for various cardiovascular health indicators, along with plasma LCN2 concentration. No significant difference in LCN2 concentration was seen between the two groups. LCN2 was a very weak predictor or absent from models describing blood pressures or predicting athlete status. In another cohort, 57 non-diabetic overweight or obese men and post-menopausal women who fulfilled Adult Treatment Panel III metabolic syndrome criteria were randomly allocated into either a control, modified Dietary Approaches to Stop Hypertension (DASH) diet, or DASH and exercise group. Pre- and post-intervention demographic, cardiovascular health indicators, and plasma LCN2 expression were measured in each individual. While BMI fell in intervention groups, LCN2 levels remained unchanged within and between all groups, as illustrated by strong correlations between LCN2 concentrations pre- and 12 weeks post-intervention (r = 0.743, P < 0.0001). This suggests that circulating LCN2 expression are stable over a period of at least 12 weeks and is not modifiable by diet and exercise.
Similar content being viewed by others
Introduction
High body mass index (BMI) and obesity promote a low-grade chronic systemic inflammatory state, triggering a systemic acute phase response1. Pro-inflammatory acute phase response proteins are then produced and released, which can feedback into the chronic inflammatory phenotype2. Low-grade systemic inflammation also produces an ideal environment for the development of cardiovascular (CVD) and renal disease. Inflammatory biomarkers such as C-reactive protein (CRP) and the more recently implicated GlycA can predict the risk of CVD many years in advance3,4. Inflammatory pathways are critical for the pathological remodelling of the heart, for example, through stimulation of proteins involved in degradation and reconstruction of the interstitium5,6,7. This maladaptive remodelling is involved in multiple types of CVDs, and may lead to cardiac hypertrophy and heart failure8.
Neutrophils, the most abundant type of white blood cells in the body, are recognized for their role in the atherosclerotic process and their relevance for the development of heart failure9,10. Among a variety of inflammatory factors, neutrophils release lipocalin-2 (LCN2, otherwise known as neutrophil gelatinase-associated lipocalin or NGAL), a 25 kDa protein that is upregulated during the acute phase response, and primarily contributes to innate immunity through its function as an iron chelator11,12. It has been used clinically as a marker of renal failure and, more recently, as a marker of increased cardiovascular risk13,14,15. Other both immune and non-immune cell types such as cardiomyocytes, fibroendothelial cells, renal cells, and macrophages, have also been shown to release LCN216,17,18,19. In the context of cardiac remodelling, LCN2 has been shown to preserve the enzymatic activity of matrix metalloprotease-9 (MMP9), a protein heavily involved in the breakdown of the interstitium20,21. LCN2 has also been shown to drive cardiac hypertrophy in vitro and vivo, and was associated with left ventricular mass and BMI in humans19. Importantly, LCN2 was a stronger marker of innate immune response, in particular neutrophil function, than CRP or GlycA19.
The ability of a balanced diet and regular exercise to reduce BMI and the risk of CVD is well documented22,23,24. In particular, specific diets such as the Dietary Approach to Stop Hypertension (DASH) have been able to reduce low-grade inflammation observed as serum CRP levels25,26,27,28,29. However, whether LCN2 expression can be attenuated by exercise or diet interventions is currently unclear. Here we aimed to determine if LCN2 plasma levels are modulated by physical fitness and diet, and to investigate whether LCN2 levels are stable over time.
Results
Relative to controls, endurance athletes were taller, had superior cardiorespiratory fitness as indicated by peak oxygen uptake (\(\dot{V}\)O2peak) (controls vs athletes mean ± SD, units: 42.6 ± 7.1 v 58.0 ± 8.8, ml.kg−1 min−1), lower resting heart rate (68 ± 10.5 v 52 ± 7.8, beats min−1), and BMI (25.9 ± 2.8 v 22.6 ± 2.3) (all P < 0.001, Table 1). The difference in plasma LCN2 protein levels between controls and endurance athletes was of borderline statistical significance (68.3 ± 21.8 v 75.0 ± 14.8, ng/mL, P = 0.073, Fig. 1). Therefore, a sensitivity analysis was performed to determine which cardiovascular health indicators could predict whether an individual belonged to the athlete group or control group (athlete status) (Table 2). The analysis showed that athlete status could be predicted correctly in 94.8% of individuals using BMI, sex, age, and \(\dot{V}\)O2peak as predictors, with the model having an overall Cox & Snell β value of 0.683. LCN2 concentration, however, was not a significant predictor of athlete status in this model.
To understand whether the borderline difference in plasma LCN2 expression could be explained through sex-specific differences, we analysed the cohort by splitting the controls and endurance athletes into male and female groups. Male athletes were more similar to sex-matched control group counterparts than when compared to female athletes compared to sex-matched controls in terms of cardiovascular health indicators (Tables 3, 4). Female athletes had a lower BMI (26.6 ± 3.4 v 20.9 ± 2.3), diastolic blood pressure (DBP) (76.6 ± 8.8 v 67.7 ± 7.4, mmHg), mean arterial pressure (MAP) (91.0 ± 9.3 v 82.7 ± 6.3, mmHg), central systolic blood pressure (SBP) (107.6 ± 11.3 v 96.9 ± 7.0, mmHg), central DBP (77.6 ± 8.8 v 67.8 ± 7.3, mmHg), central MAP (87.7 ± 9.3 v 77.5 ± 6.9, mmHg), resting heart rate (72.0 ± 9.6 v 52.5 ± 9.9, beats min−1), and higher \(\dot{V}\)O2peak (34.7 ± 4.4 v 54.6 ± 13.1, ml kg−1.min−1), while male athletes only differed from the controls when looking at BMI (25.6 ± 2.5 v 23.0 ± 2.2), resting heart rate (67.2 ± 10.7 vs 52.3 ± 7.3, beats min−1), and \(\dot{V}\)O2peak (45.8 ± 5.2 vs 59.0 ± 7.2, ml kg−1 min−1) (all P < 0.01). Importantly, the difference in plasma LCN2 expression was of borderline statistical significance in male controls compared to male athletes (67.1 ± 22.6 v 75.9 ± 15.3, ng/mL, P = 0.053), while there were no differences between controls and athletes in females (70.1 ± 20.6 v 71.5 ± 12.7, ng/mL, P = 0.847) (Fig. 2). Thus, a sensitivity analysis was again performed on the male data to test whether athlete status could be predicted from LCN2 expression, BMI, age, and \(\dot{V}\)O2peak (Table 5). Male athlete status was found to be able to be predicted in 90.5% of the data using age and \(\dot{V}\)O2peak as inputs, with the model showing an overall Cox & Snell β value of 0.676. LCN2 expression and BMI were not found to be significant predictors of athlete status. Therefore, males and females were grouped for all subsequent analyses.
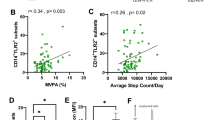
There was no significant difference in plasma lipocalin-2 (LCN2) protein levels between male controls (n = 34) and endurance athletes (n = 41) (P = 0.053), or between female controls (n = 14) and endurance athletes (n = 11) (P = 0.847). Data are expressed as mean ± S.E.M. from two-tailed Man-Whitney U-test.
We also performed sensitivity analyses to determine the influence of LCN2 on SBP, DBP, central SBP and central DBP (Tables 6, 7). While a generalised model predicting SBP did not show LCN2 concentration as a significant factor, models predicting DBP, central SBP, and central DBP found that LCN2 concentration was a significant predictor (P < 0.05, Tables 6, 7). All models explained 21.1% to 29.2% of the variability of the predicted variable around its mean as indicated by their r2 values. The β values associated with LCN2 concentration were small and ranged between 0.105 and 0.128, suggesting that the effect of LCN2 on these factors was limited in scale.
In the randomised control trial, we compared the difference between time points in all groups in the intervention study. We had previously reported that the diet + exercise group was the only group to have a significant increase in \(\dot{V}\)O2peak, which validates the exercise intensity methodology30. No groups differed significantly from one another in the context of age, sex, plasma LCN2 content, or cardiovascular factors, although the BMI between groups differed significantly post-intervention (P = 0.0008, Table 8). While LCN2 protein concentration did not differ substantially between any of the intervention groups (all P > 0.05, Fig. 3) or within each group when comparing initial and post-intervention expression (all P > 0.05), the diet and exercise intervention successfully reduced BMI (P = 0.04) compared to the control group (Table 8). This change was not reflected in the cardiovascular health indicators of the intervention groups, as both SBP and DBP remained similar between the control and integrated diet-exercise groups. We found that LCN2 levels remained stable over the 12-week interventions in all groups (Table 8). Furthermore, initial and post-intervention LCN2 levels were significantly correlated with each other between all groups (Table 9). Plasma CRP levels pre-intervention and post-intervention were also significantly correlated with each other, but did not correlate with plasma LCN2 levels (Table 10).
Plasma lipocalin-2 (LCN2) protein levels before and after a randomised control trial. There was no difference in LCN2 levels between control (n = 17), dietary intervention (n = 20), or dietary intervention and exercise arms (n = 20) (P = 0.37). Comparisons of initial and post-intervention LCN2 concentrations also showed no significant changes (P = 0.47, P = 0.60, P = 0.51 for comparisons in the control, diet intervention, and dual intervention group respectively). Data are expressed as mean ± S.E.M.
Discussion
Emerging evidence proposes that LCN2 is not merely a biomarker of cardiac and renal diseases, but has a role in modulating chronic inflammation and other processes that can lead to these diseases19,31,32. Chronic inflammation is usually associated with poor diet, low physical activity and higher BMI, which are risk factors for all CVDs. Here we showed that LCN2 is not modulated by exercise in two independent cohorts. Moreover, a three-month intervention with the DASH diet, which is associated with lower inflammation, was not able to lower levels of LCN2. Finally, LCN2 levels were highly correlated three months apart, independently of the type of intervention and BMI changes. Together, our findings suggest that LCN2 is stable over time and is not modulated by exercise nor diet in healthy or overweight/obese people.
When comparisons were drawn between the control and athlete groups, we found no significant difference in LCN2 levels, even though conventional cardiovascular health markers were markedly different between the groups. Athletes had a significantly healthier cardiovascular profile, consistent with previous studies which have found athletes to have longer telomeres and lower general and CVD mortality33,34,35,36,37,38. Interestingly, while a recent study suggested that LCN2 expression is sex-specific in mice, we could not pinpoint any significant differences of plasma LCN2 expression in humans between athletes and controls in male or female groups39. The ability of repeated exercise to rapidly attenuate the initially-seen physiological stress response has been well reported, even when the exercise is scaled with the participant’s increasing fitness40,41. A strong acute phase response may have initially occurred in newer athletes which quickly diminished over their time spent training, causing plasma LCN2 expression to stabilise. In addition, not all components of the acute phase response may be activated by exercise, instead creating a diminished acute phase response. The literature surrounding this topic suggests that partial activation of the acute phase response after extreme endurance exercise is common42. It is possible that circulating LCN2 is unaffected by physiological stress associated with long-term, repeated endurance training, and, therefore, would be unchanged in athletes when compared to apparently healthy controls. However, multiple studies have found significant changes in post-aerobic exercise serum and plasma LCN2 concentrations, which suggests that the acute phase response exhibited in response to moderate to intense aerobic exercise is sufficient to cause expression of LCN243,44,45. Another possibility is that LCN2 is genetically determined, and is minimally influenced by lifestyle factors such as exercise. Indeed, in a previous study we found that the single nucleotide polymorphism rs13297295 located near the gene coding LCN2 was a cis-expression quantitative gene locus for LCN2 levels in a large cohort, suggesting that plasma LCN2 levels are primarily under genetic control19.
LCN2 has been proposed as an inflammatory marker for obesity, hyperglycaemia and insulin resistance, and has also been found to drive left ventricular pathological cardiac hypertrophy19,46. Due to LCN2 being under genetic control of the NF-kβ and MAPK pathway, both involved in chronic low-grade inflammation found in obese and hypertensive individuals, circulating LCN2 expression is possibly stable for as long as inflammation status remains relatively unchanged. Our data, however, suggested that plasma LCN2 levels are resistant to modification by short-term interventions known to decrease inflammation, such as diet or exercise, and levels were stable over a period of three months. Indeed, there was no change in LCN2 with a decrease in BMI. Whether lean tissue mass is associated with LCN2 remains to be determined. Although our sensitivity analyses also showed that LCN2 was associated with SBP and DBP, the ability of LCN2 to predict blood pressure values was low and other traditional risk factors, such as sex and BMI, were more important. Furthermore, the immutable nature of plasma LCN2 in response to the exercise or diet regimens and its lack of correlation to various cardiovascular health markers lends credence to the applicability of LCN2 as a potential biomarker for CVD, as it may be directly influenced by the development of CVD as opposed to being influenced by non-disease related cardiovascular health markers (e.g. diet and \(\dot{V}\)O2peak). This is further supported by previous research, where LCN2 has been found to be a predictor of cardiovascular mortality over an 11 year longitudinal study47.
Interestingly, we found that decreases in BMI nor changes in plasma CRP over the three month period were not associated with changes in LCN2 plasma concentration. The association of circulating LCN2 with BMI and other cardiovascular health markers has not been elucidated clearly in the literature, with multiple studies conflicting on whether LCN2 and BMI change in tandem48,49,50,51. LCN2 levels, however, are correlated with obesity and hyperglycaemia, both of which have been shown to induce and promote a chronic low-grade inflammatory state52,53,54. Our results support the hypothesis that BMI and LCN2 are not intrinsically linked together, and that independent modulation of either variable is possible. Plasma LCN2 levels, however, may be attenuated over longer time periods as participants recover from obesity and concomitant chronic low-grade inflammation.
One limitation of this study is the singular type of exercise chosen as the intervention. Many aspects of exercise were controlled in our study, such as duration, frequency, intensity, and mode. However, other regimens such as high intensity interval training (HIIT) or resistance training have also shown to be effective exercises for cardiovascular health, with HIIT shown to improve both blood pressure and cardiovascular fitness55. Moreover, overweight/obese subjects may display chronotropic incompetence and as such exercise intensity based on percentage of maximal heart rate is not always ideal. However, we still observed a significant increase in \(\dot{V}\)O2peak in the diet + exercise group only30. Furthermore, as LCN2 levels are positively correlated with left ventricular hypertrophy, longitudinal measurements of LCN2 and cardiac size in long-term athletes may yield interesting results as exercise induces physiological hypertrophy of the heart19,56. Additionally, while our study measured plasma LCN2, LCN2 content in skeletal muscle or cardiac tissue was not measured. Due to LCN2 being linked to the MAPK pathway and because the pathway is heavily involved in muscle regeneration, it is possible that skeletal muscle LCN2 content is influenced by acute and long-term exercise57,58,59. Our study had several strengths including the use of two independent cohorts, one which was a cross-sectional cohort and a randomised control trial. Furthermore, it is possible that our intervention timeframe of 12 weeks was insufficient to significantly change the inflammatory phenotype that had been built over a long period in overweight or obese individuals. While we would expect to still see a change in plasma LCN2 expression within the intervention groups due to the positive correlations between BMI and LCN2 expression, along with correlations between BMI and inflammatory markers described in the current literature, the change may not have been large enough to elucidate significance46,60,61,62.
In conclusion, LCN2 levels were not different between athletes and controls, nor were they modified after three months of interventions with diet and exercise. While LCN2 was a significant variable included in models predicting blood pressure in athletes or controls, the low β values for each suggest that LCN2 has minimal effects. Importantly, our data shows that circulating LCN2 plasma concentration is not dependent on BMI, and is stable over a period of three months. Pre-intervention LCN2 levels were found to be strongly correlated with post-intervention expression. Further research may elucidate a molecular pathway between LCN2 expression and multiple renal and CVDs.
Methods
All participants gave written informed consent. The studies were approved by the Alfred Hospital and the Federation University Australia Human Research Ethics Committees, and followed the Declaration of Helsinki.
Athlete and control participants
Endurance athletes and control subjects involved in a cross-sectional analyses were described elsewhere63,64. Briefly, 52 endurance athletes (cyclists, runners and triathletes) and 48 controls (aged 18–55) were recruited at Federation University Australia from local advertisements, social media and word of mouth (Table 1). Athletes engaged in vigorous endurance exercise at least three times per week, for a minimum of one year. Endurance athletes were cyclists, triathletes, middle- or long-distance runners, and ultra-marathon runners at state through to international level. The apparently healthy controls were recreationally active but were not engaged in any structured aerobic or resistance exercise training. All subjects were non-smoking, free from any age-related chronic diseases and not taking any medications. The controls were recreationally active but were not engaged in any structured aerobic or resistance exercise training. All BP assessments were conducted in the AM following an overnight fast. Brachial and central BP was measured with participants in a seated position after a 10-min rest period using the SphygmoCor Device (AtCor Medical, Australia). Blood pressures were calculated from the average of three measurements, recorded one minute apart63.
Diet and exercise intervention participants
Overweight and obese middle-aged (45–65 year old) men and post-menopausal women who fulfilled Adult Treatment Panel III criteria for metabolic syndrome (MetS) were recruited at the Alfred Hospital through newspaper advertisements (clinicaltrials.gov registration number NCT00163943, first registration 14/09/2005), as previously reported30. All subjects were sedentary and not taking any medication while being free from non-communicable diseases, but were overweight or obese (waist circumference: ≥ 102 cm for men and ≥ 88 cm for women). None of the participants had type 2 diabetes or a history of secondary hypertension, cardiovascular, cerebrovascular, renal, liver, thyroid disease, or drug use known to affect these parameters. Supine blood pressure was measured in the clinic as the average of five readings after a 5-min rest (Dinamap, Model 1846SX; Critikon, Tampa, FL). Body weight was measured in light indoor clothes without shoes, using a digital scale. Waist circumference was measured at the midpoint between the lowest rib and iliac crest and hip circumference at the level of the greater trochanters. Stratified randomization by sex and hypertension status in blocks of six was used to allocate subjects to one of three groups. Participants were randomly allocated into three arms of a 12-week intervention: control arm (no intervention), diet arm, or exercise and diet arm. The diet and exercise intervention protocol has also been reported, which consisted of 40 min of bicycle riding on alternating days over 12 weeks30. Exercise was moderate, with participants reaching 65% of a predetermined maximum heart rate throughout. Maximum work capacity and maximum oxygen consumption were measured during a "sprint" exercise test. The exercise test was supervised by a cardiologist and was performed on a bicycle ergometer, commencing at zero work rate. Each minute the work rate was increased 20 W until any further increase was prevented by fatigue. The 14-day menu plans, recipes, and prepared meals followed a modified Dietary Approaches to Stop Hypertension (DASH) diet, and was composed of 30% fat (6% polyunsaturated, 15% monounsaturated, and 9% saturated), 22% protein, and 48% carbohydrate as previously described65,66. They were provided with a 14-day menu plan and recipes, and prepared meals in their homes. High sensitivity CRP was measured in fasting samples by immunoturbidimetric assay.
Basal energy requirements were calculated for each individual by indirect calorimetry and energy intake was reduced by 600 cal per day as previously described. Subjects were provided with 14-day menu plans and recipes and prepared meals in their homes. Participants attended at two weekly intervals for dietary counselling. Compliance was assessed by prospective 4-day diet records, which were analysed using Australian Food Composition Tables (FoodWorks Professional Version 3.02; Xyris Software, Highgate Hill, Australia https://xyris.com.au/products/foodworks-10-professional/). Sodium, potassium, and protein intake were quantified by 24-h urine collections.
Exercise training comprised 40 min bicycle riding on alternate days at a moderate intensity of 65% of predetermined maximum heart rate67. This corresponded to target heart rates within the range 120 –145 bpm during exercise. Workload was increased as necessary to maintain target heart rate. Once a week, exercise was performed under supervision in our clinical laboratory at the Alfred Hospital Heart Centre. Other sessions were performed at the subjects’ homes using provided exercise bicycles and heart rate monitors. Subjects kept records of average heart rate during each exercise session. Compliance was assessed by review of heart rate records and at the end of the intervention from measures of maximal oxygen consumption during a continuous incremental cycle ergometry protocol, during which workload was increased by 20 W/min.
Participants in the control group were instructed to maintain their usual dietary and exercise habits. They attended our laboratory at the Heart Centre every 3 weeks for body weight and blood pressure assessment.
Plasma measurement of LCN2
All participants gave a fasted (12 h) blood sample collected from the antecubital vein using standard phlebotomy procedures. A resting blood sample was collected with participants seated. Subjects were asked to refrain from alcohol, caffeine and exercise training at least 24 h before blood donation. Plasma was isolated from peripheral blood that was collected into EDTA tubes and stored at − 20 °C. Plasma LCN2 abundance was measured in duplicates by enzyme-linked immunosorbent assay (ELISA) run in duplicate using the Human NGAL ELISA Kit (ElabScience, E-EL-H0096) according to the supplier’s guidelines. Intra-assay coefficients of variability were calculated and only samples with less than 15% variability were accepted. Overall inter-assay coefficient of variability was 5.4–9.7%.
Statistical analyses
Outliers were removed using the robust regression and outlier removal algorithm in GraphPad Prism (version 8.1.2, GraphPad Software, CA, USA https://www.graphpad.com/scientific-software/prism/), using a Q value of 2%. The ROUT algorithm is based on a nonlinear regression to fit an outlier-independent curve, which is then used to identify and remove outliers. Two controls and one athlete were excluded from the athlete and control cohorts based on the ROUT algorithm, with LCN2 concentrations of 1.638 ng/mL, 147.714 ng/mL, and 1.514 ng/mL. No participants were found to be outliers for the intervention cohorts. Data was first tested for normality using the D’Agostino-Pearson test for normality, and comparisons between controls and athletes were performed using two-tailed independent samples t-tests or Mann–Whitney U-tests for continuous variables, and chi-squared tests for categorical variables (i.e. biological sex) in SPSS (version 24, IBM, NY, USA https://www.ibm.com/au-en/analytics/spss-statistics-software), and the equalities of variances of variables between groups was computed using Levene’s test. Correlation between LCN2 levels and cardiovascular heath indicators (including systolic and diastolic blood pressure, mean arterial pressure, and central blood pressures) were calculated using Pearson’s correlation, with P-values further corrected for multiple comparisons using the ‘p.adjust’ function with the false discovery rate (FDR) correction argument in R (version 3.6.1, R Foundation, Vienna, Austria https://www.r-project.org/). Sensitivity analysis of LCN2 levels and other significant cardiovascular factors was performed through usage of a forward stepwise linear regression analysis in SPSS, using the probability of F as an entry and exit factor at 0.15 and 0.20 respectively. An additional sensitivity analysis in the form of a forward stepwise binary logistic regression model was performed for the entire cohort as well as sex-specific subgroups, testing the predictability of athlete status from LCN2 concentration, cohort characteristics, and cardiovascular health indicators. The R2 (adjusted R2 values for linear regression models, Cox-Snell R2 values for binary logistic regression models), β, and P-values were reported alongside the general model equations.
Comparisons between controls, diet, or diet and exercise arms were performed using a mixed analysis of variance (MANOVA), with the comparison between the baseline and post-intervention observations as the within-group effect and the intervention type as the between-group effects. Multiple comparisons were subsequently performed using Tukey’s honest significance test. Correlations between initial and post-intervention plasma LCN2 concentration were performed using a Pearson’s correlation, with r and FDR-adjusted q-values reported. Data are reported as means, with error bars representing standard error of the mean (S.E.M.) or standard deviation (SD). The mean and standard deviation (S.D.) are reported for cohort characteristics. P or FDR q-values below 0.05 were considered significant.
References
Rodríguez-Hernández, H., Simental-Mendía, L. E., Rodríguez-Ramírez, G. & Reyes-Romero, M. A. Obesity and inflammation: epidemiology, risk factors, and markers of inflammation. Int. J. Endocrinol. 2013, 678159–678159 (2013).
Gregor, M. F. & Hotamisligil, G. S. Inflammatory mechanisms in obesity. Annu. Rev. Immunol. 29, 415–445 (2011).
Eltoft, A. et al. C-reactive protein in atherosclerosis—a risk marker but not a causal factor? A 13-year population-based longitudinal study: the Tromso study. Atherosclerosis 263, 293–300 (2017).
Joshi-Aditya, A. et al. GlycA is a novel biomarker of inflammation and subclinical cardiovascular disease in psoriasis. Circ. Res. 119, 1242–1253 (2016).
Frieler, R. A. & Mortensen, R. M. Immune cell and other noncardiomyocyte regulation of cardiac hypertrophy and remodeling. Circulation 131, 1019–1030 (2015).
Palmieri, V. et al. Relation of left ventricular hypertrophy to inflammation and albuminuria in adults with type 2 diabetes: the strong heart study. Diabetes Care 26, 2764–2769 (2003).
Salles, G. F., Fiszman, R., Cardoso, C. R. & Muxfeldt, E. S. Relation of left ventricular hypertrophy with systemic inflammation and endothelial damage in resistant hypertension. Hypertension 50, 723–728 (2007).
Shimizu, I. & Minamino, T. Physiological and pathological cardiac hypertrophy. J. Mol. Cell. Cardiol. 97, 245–262 (2016).
Gaul, D. S., Stein, S. & Matter, C. M. Neutrophils in cardiovascular disease. Eur. Heart J. 38, 1702–1704 (2017).
Swirski, F. K. & Nahrendorf, M. Leukocyte behavior in atherosclerosis, myocardial infarction, and heart failure. Science 339, 161–166 (2013).
Srinivasan, G. et al. Lipocalin 2 deficiency dysregulates iron homeostasis and exacerbates endotoxin-induced sepsis. J. Immunol. 189, 1911–1919 (2012).
Xiao, X., Yeoh, B. S. & Vijay-Kumar, M. Lipocalin 2: an emerging player in iron homeostasis and inflammation. Annu. Rev. Nutr. 37, 103–130 (2017).
Leoncini, G. et al. Combined use of urinary neutrophil gelatinase-associated lipocalin (uNGAL) and albumin as markers of early cardiac damage in primary hypertension. Clin. Chim. Acta 412, 1951–1956 (2011).
Maisel, A. S. et al. Prognostic utility of plasma neutrophil gelatinase-associated lipocalin in patients with acute heart failure: the NGAL EvaLuation Along with B-type NaTriuretic Peptide in acutely decompensated heart failure (GALLANT) trial. Eur. J. Heart Fail. 13, 846–851 (2011).
Viau, A. et al. Lipocalin 2 is essential for chronic kidney disease progression in mice and humans. J. Clin. Investig. 120, 4065–4076 (2010).
Eilenberg, W. et al. Neutrophil gelatinase-associated lipocalin (NGAL) is associated with symptomatic carotid atherosclerosis and drives pro-inflammatory state in vitro. Eur. J. Vasc. Endovasc. Surg. 51, 623–631 (2016).
Flo, T. H. et al. Lipocalin 2 mediates an innate immune response to bacterial infection by sequestrating iron. Nature 432, 917–921 (2004).
Liu, F., Yang, H., Chen, H., Zhang, M. & Ma, Q. High expression of neutrophil gelatinase-associated lipocalin (NGAL) in the kidney proximal tubules of diabetic rats. Adv. Med. Sci. 60, 133–138 (2015).
Marques, F. Z. et al. Experimental and human evidence for lipocalin-2 (Neutrophil Gelatinase-Associated Lipocalin [NGAL]) in the development of cardiac hypertrophy and heart failure. J. Am. Heart Assoc. 6, e005971. https://doi.org/10.1161/JAHA.117.005971 (2017).
Halade, G. V., Jin, Y. F. & Lindsey, M. L. Matrix metalloproteinase (MMP)-9: a proximal biomarker for cardiac remodeling and a distal biomarker for inflammation. Pharmacol. Ther. 139, 32–40 (2013).
Yan, L., Borregaard, N., Kjeldsen, L. & Moses, M. A. The high molecular weight urinary matrix metalloproteinase (MMP) activity is a complex of gelatinase B/MMP-9 and neutrophil gelatinase-associated lipocalin (NGAL). Modulation of MMP-9 activity by NGAL. J. Biol. Chem. 276, 37258–37265 (2001).
Eilat-Adar, S., Sinai, T., Yosefy, C. & Henkin, Y. Nutritional recommendations for cardiovascular disease prevention. Nutrients 5, 3646–3683 (2013).
Foster-Schubert, K. E. et al. Effect of diet and exercise, alone or combined, on weight and body composition in overweight-to-obese postmenopausal women. Obesity (Silver Spring, Md.) 20, 1628–1638 (2012).
Wannamethee, S. G., Shaper, A. G. & Walker, M. Physical activity and mortality in older men with diagnosed coronary heart disease. Circulation 102, 1358–1363 (2000).
Asemi, Z., Samimi, M., Tabassi, Z., Sabihi, S. S. & Esmaillzadeh, A. A randomized controlled clinical trial investigating the effect of DASH diet on insulin resistance, inflammation, and oxidative stress in gestational diabetes. Nutrition (Burbank, Los Angeles County, Calif.) 29, 619–624 (2013).
Calder, P. C. et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br. J. Nutr. 106, S1–S78 (2011).
Mozaffarian, D. Dietary and policy priorities for cardiovascular disease, diabetes, and obesity: a comprehensive review. Circulation 133, 187–225 (2016).
Silveira, B. K. S. et al. Dietary pattern and macronutrients profile on the variation of inflammatory biomarkers: scientific update. Cardiol. Res. Pract. 2018, 4762575 (2018).
Smidowicz, A. & Regula, J. Effect of nutritional status and dietary patterns on human serum C-reactive protein and interleukin-6 concentrations. Adv. Nutr. (Bethesda, Md.) 6, 738–747 (2015).
Straznicky, N. E. et al. Sympathetic neural adaptation to hypocaloric diet with or without exercise training in obese metabolic syndrome subjects. Diabetes 59, 71–79 (2010).
Aghel, A., Shrestha, K., Mullens, W., Borowski, A. & Tang, W. H. W. Serum neutrophil gelatinase-associated lipocalin (NGAL) in predicting worsening renal function in acute decompensated heart failure. J. Cardiac Fail. 16, 49–54 (2010).
Bolignano, D. et al. Neutrophil gelatinase-associated lipocalin (NGAL) as a marker of kidney damage. Am. J. Kidney Dis. 52, 595–605 (2008).
Fagard, R. Athlete’s heart. Heart (Br. Cardiac Soc.) 89, 1455–1461 (2003).
Garatachea, N. et al. Elite athletes live longer than the general population: a meta-analysis. Mayo Clin. Proc. 89, 1195–1200 (2014).
Lemez, S. & Baker, J. Do elite athletes live longer? A systematic review of mortality and longevity in elite athletes. Sports Med. Open 1, 16 (2015).
Muniesa, C. A. et al. Telomere length in elite athletes. Int. J. Sports Physiol. Perform. 12, 994–996 (2017).
Simoes, H. G. et al. Longer telomere length in elite master sprinters: relationship to performance and body composition. Int. J. Sports Med. 38, 1111–1116 (2017).
Teramoto, M. & Bungum, T. J. Mortality and longevity of elite athletes. J. Sci. Med. Sport 13, 410–416 (2010).
Chella-Krishnan, K. et al. Sex-specific metabolic functions of adipose Lipocalin-2. Mol. Metab. 30, 30–47 (2019).
Hackney, A. C. Stress and the neuroendocrine system: the role of exercise as a stressor and modifier of stress. Expert Rev. Endocrinol. Metab. 1, 783–792 (2006).
JanssenDuijghuijsen, L. M. et al. Adaptation of exercise-induced stress in well-trained healthy young men. Exp. Physiol. 102, 86–99 (2017).
Fallon, K. E. The acute phase response and exercise: the ultramarathon as prototype exercise. Clin. J. Sport Med. 11, 38–43 (2001).
Andreazzoli, A. et al. Assessment of pN-GAL as a marker of renal function in elite cyclists during professional competitions. J. Biol. Regul. Homeost. Agents 31, 829–835 (2017).
Lippi, G. et al. Variation of serum and urinary neutrophil gelatinase associated lipocalin (NGAL) after strenuous physical exercise. Clin. Chem. Lab. Med. (CCLM) 50, 1585 (2012).
Machado, J. C. Q., Volpe, C. M. O., Vasconcellos, L. S. & Nogueira-Machado, J. A. Quantification of NGAL in urine of endurance cycling athletes. J. Phys. Activ. Health 15, 679 (2018).
Wang, Y. et al. Lipocalin-2 is an inflammatory marker closely associated with obesity, insulin resistance, and hyperglycemia in humans. Clin. Chem. 53, 34–41 (2007).
Daniels, L. B. et al. Plasma neutrophil gelatinase-associated lipocalin is independently associated with cardiovascular disease and mortality in community-dwelling older adults. Rancho Bernardo Study 59, 1101–1109 (2012).
Auguet, T. et al. Upregulation of lipocalin 2 in adipose tissues of severely obese women: positive relationship with proinflammatory cytokines. Obesity (Silver Spring, Md.) 19, 2295–2300 (2011).
Elkhidir, A. E., Eltaher, H. B. & Mohamed, A. O. Association of lipocalin-2 level, glycemic status and obesity in type 2 diabetes mellitus. BMC Res. Notes 10, 285 (2017).
Luo, Y. et al. Serum lipocalin-2 levels are positively associated with not only total body fat but also visceral fat area in Chinese men. Medicine 95, e4039 (2016).
Wu, G. et al. Elevated circulating lipocalin-2 levels independently predict incident cardiovascular events in men in a population-based cohort. Arterioscler. Thromb. Vasc. Biol. 34, 2457–2464 (2014).
Castro, A. M., Macedo-de la Concha, L. E. & Pantoja-Meléndez, C. A. Low-grade inflammation and its relation to obesity and chronic degenerative diseases. Revista Médica del Hospital General de México 80, 101–105 (2017).
de Rekeneire, N. et al. Diabetes, hyperglycemia, and inflammation in older individuals: the health, aging and body composition study. Diabetes Care 29, 1902–1908 (2006).
Mraz, M. & Haluzik, M. The role of adipose tissue immune cells in obesity and low-grade inflammation. J. Endocrinol. 222, R113 (2014).
Angadi, S. S. et al. High-intensity interval training vs. moderate-intensity continuous exercise training in heart failure with preserved ejection fraction: a pilot study. J. Appl. Physiol. 119, 753–758 (2015).
McMullen, J. R. & Jennings, G. L. Differences between pathological and physiological cardiac hypertrophy: novel therapeutic strategies to treat heart failure. Clin. Exp. Pharmacol. Physiol. 34, 255–262 (2007).
Keren, A., Tamir, Y. & Bengal, E. The p38 MAPK signaling pathway: a major regulator of skeletal muscle development. Mol. Cell. Endocrinol. 252, 224–230 (2006).
Kramer, H. F. & Goodyear, L. J. Exercise, MAPK, and NF-κB signaling in skeletal muscle. J. Appl. Physiol. 103, 388–395 (2007).
Segales, J., Perdiguero, E. & Munoz-Canoves, P. Regulation of muscle stem cell functions: a focus on the p38 MAPK signaling pathway. Front. Cell Dev. Biol. 4, 91 (2016).
Auguet, T. et al. Upregulation of lipocalin 2 in adipose tissues of severely obese women: positive relationship with proinflammatory cytokines. Obesity 19, 2295–2300 (2011).
Lee, H., Lee, I. S. & Choue, R. Obesity, inflammation and diet. Pediatr. Gastroenterol. Hepatol. Nutr. 16, 143–152 (2013).
Straub, R. H. et al. Hormone replacement therapy and interrelation between serum interleukin-6 and body mass index in postmenopausal women: a population-based study. J. Clin. Endocrinol. Metab. 85, 1340–1344 (2000).
Denham, J. et al. Aortic augmentation index in endurance athletes: a role for cardiorespiratory fitness. Eur. J. Appl. Physiol. 116, 1537–1544 (2016).
Denham, J., O’Brien, B. J., Prestes, P. R., Brown, N. J. & Charchar, F. J. Increased expression of telomere-regulating genes in endurance athletes with long leukocyte telomeres. J. Appl. Physiol. (Bethesda, Md.: 1985) 120, 148–158 (2016).
Appel, L. J. et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med. 336, 1117–1124 (1997).
Straznicky, N. E. et al. Effects of dietary weight loss on sympathetic activity and cardiac risk factors associated with the metabolic syndrome. J. Clin. Endocrinol. Metab. 90, 5998–6005 (2005).
Meredith, I. T. et al. Exercise training lowers resting renal but not cardiac sympathetic activity in humans. Hypertension 18, 575–582 (1991).
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: F.Z.M. Performed experiments: J.D., P.R.P., N.E., E.A.L., N.E.S. Contributed data or analysis tools: J.D., P.R.P., N.E., E.A.L., N.E.S., M.P.S., M.D.E., B.J.O.B., F.C., G.W.L. Performed analyses: M.E.N. Wrote the paper: M.E.N., F.Z.M. Reviewed the paper: all authors. Secured funding: F.Z.M., F.C.
Corresponding author
Ethics declarations
Competing interests
MPS has received research support from Medtronic, Abbott, Novartis, Servier, Pfizer, and Boehringer-Ingelheim. GWL has acted as a consultant for Medtronic. FZM is supported by a National Heart Foundation Future Leader Fellowship, a National Health and Medical Research Council Project Grant and a National Heart Foundation Vanguard Grant. GWL, NE and EAL are supported by Swinburne Research, Swinburne University of technology. MPS is supported by an NHMRC Research Fellowship. All other authors declare no competing interests. Data used in this manuscript is available upon request.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nakai, M.E., Denham, J., Prestes, P.R. et al. Plasma lipocalin-2/NGAL is stable over 12 weeks and is not modulated by exercise or dieting. Sci Rep 11, 4056 (2021). https://doi.org/10.1038/s41598-021-83472-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-83472-x
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.