Abstract
The new coronavirus disease (COVID-19) is a challenge for clinical decision-making and the effective allocation of healthcare resources. An accurate prognostic assessment is necessary to improve survival of patients, especially in developing countries. This study proposes to predict the risk of developing critical conditions in COVID-19 patients by training multipurpose algorithms. We followed a total of 1040 patients with a positive RT-PCR diagnosis for COVID-19 from a large hospital from São Paulo, Brazil, from March to June 2020, of which 288 (28%) presented a severe prognosis, i.e. Intensive Care Unit (ICU) admission, use of mechanical ventilation or death. We used routinely-collected laboratory, clinical and demographic data to train five machine learning algorithms (artificial neural networks, extra trees, random forests, catboost, and extreme gradient boosting). We used a random sample of 70% of patients to train the algorithms and 30% were left for performance assessment, simulating new unseen data. In order to assess if the algorithms could capture general severe prognostic patterns, each model was trained by combining two out of three outcomes to predict the other. All algorithms presented very high predictive performance (average AUROC of 0.92, sensitivity of 0.92, and specificity of 0.82). The three most important variables for the multipurpose algorithms were ratio of lymphocyte per C-reactive protein, C-reactive protein and Braden Scale. The results highlight the possibility that machine learning algorithms are able to predict unspecific negative COVID-19 outcomes from routinely-collected data.
Similar content being viewed by others
Introduction
The consequences of a long stay and demand for hospital resources due to COVID-19 have been disastrous for health systems in middle and low-income countries (LMICs)1,2, requiring immediate clinical decisions, especially when dealing with limited resources3,4. An accurate COVID-19 prognosis assessment is crucial for screening and treatment procedures and may increase patient survival5,6. In Brazil7, many cities are at their saturation capacity for the provision of clinical care, especially regarding ICU beds and mechanical ventilators8,9,10,11,12,13,14,15,16,17,18,19,20. Data-driven solutions are needed to support decision-making11.
COVID-19 has shown to rapidly worsen a few days after infection12,13. The median time from disease onset to ICU admission is 9–12 days14,15. About 26–32% of the hospitalized patients are eventually admitted to ICU, and mortality in this group ranges from 39 to 72%, depending on the local characteristics of patients14,15. The median length of ICU stay and use of mechanical ventilation is approximately 9 days (95% CI 6.5–11.2) and 8.4 days (95% CI 1.6–13.7), respectively16.
Previous studies have used blood tests17, CT images18,19, sociodemographic and comorbidities history20 to develop COVID-19 diagnostic and prognostic models, including machine learning techniques21,22,23. Biomarkers from blood tests have emerged as important variables for poor prognostic factors24, which are a promising tool in poorer regions, due to its low cost and inclusion in standard protocols for clinical care. However, the majority of studies25 rely on algorithms trained on a single prognostic outcome, which in theory require the training of specific algorithms for each distinct negative outcome.
This study proposes to develop multipurpose machine learning algorithms to analyze if it is possible to predict overall poor prognosis for COVID-19 patients. We aim to test if the algorithms can generalize risk patterns for severe conditions, so they can be used as tools to assist in the prognosis of distinct negative outcomes for COVID-19 patients.
Results
Descriptive statistics
Table 1 shows the descriptive statistics for the demographic characteristics of the patients. The sample of the study (1040 patients with COVID-19) was mostly comprised by men (53.3%), with an average age of 51.7 years, and the majority of patients (63.8%) were white. The full descriptive statistics for all variables are presented in Supplementary Table 1.
Algorithms performance
We analyzed the predictive performance of the algorithms for three negative prognostic outcomes: ICU admission (n = 263, 25.5%), mechanical ventilation (MV) intubation (n = 106, 10.2%) and death (n = 92, 9.4%).
First, we tested the predictive performance of the machine learning algorithms for a specific individual outcome (e.g. death) to get a baseline for comparison. Then, we used observations from patients who had the other two outcomes (in this specific example, mechanical ventilation and ICU admission) to train an aggregated model. In the aggregated model, we tested the performance when predicting the severe outcome not included in training (e.g. death). Finally, we compared the performance of the two strategies (e.g. individual against aggregated models) using the 95% confidence interval of the area under the receiver operating characteristic curve (AUROC).
Table 2 shows the results of the models trained with the aggregated outcomes and the models with a single outcome. Every model, even the ones trained with different outcomes, presented high predictive performance, always with an AUROC over 0.91 in the test set. The individual models presented better AUC compared to the aggregated models when predicting ICU, MV or death with AUROC over 0.959, 0.945 and 0.972 respectively.
Despite the individual models being overall better, the difference between the aggregated and individual models were all within the 95% confidence intervals. Supplementary Fig. 1 shows the AUROC for each model. The sensitivity and specificity of the machine learning algorithms were also very high, in most cases over 0.8, with an average sensitivity of 0.92 and specificity of 0.82.
The positive predictive values (PPV) for the aggregated models were higher than the individual models when predicting mechanical ventilation and ICU, reaching 0.398 and 0.729 respectively, while for death there was a decrease to 0.290. This means that two out of three of the aggregated models had higher PPV when predicting which patients would develop severe illness and require hospital resources than the individual models. In Supplementary Table 2 we present the final hyperparameters for each model.
Interpretability
Figure 1 presents the prediction density for each individual outcome according to the different training strategies. The results point to a low overlap between negative and positive cases, indicating a good discriminative ability of the algorithms irrespective of the training strategy.
Figure 2 presents the top five variables that most contributed to predict a severe outcome in the aggregated models, according to the Shapley values. The variables are ranked according to the contribution for each specific algorithm. The Braden score played an important role in the aggregated outcome algorithms, ranking as the most important predictor in two of the three models. Also, the C-reactive protein and ratio of lymphocytes per C-reactive protein were found to be good predictors, appearing in the top five in all three models. Urea, age, creatinine, and arterial lactate were important for only one of the aggregated models.
Top five feature contributions to predict severe outcome in the aggregated models, BP Hospital—A Beneficência Portuguesa de São Paulo, Brazil, 2020. (a) Combined outcomes (MV + ICU) to predict death (b) Combined outcomes (Death + ICU) to predict MV. (c) Combined outcomes (Death + MV) to predict ICU.
Discussion
Previous studies have used machine learning to develop early COVID-19 prognostic models for a specific severe outcome with overall good performance21,23, frequently reaching over 0.90 AUROC26. We used a different approach, by combining severe outcomes to train algorithms to predict another outcome, in order to test its potential for predicting multiple untrained outcomes.
We found that machine learning algorithms were able to predict negative prognostic outcomes with high overall performance for COVID-19, even when the specific outcome was not included in the training of the algorithms. All models presented an AUROC higher than 0.91 (average of 0.92) in the test set, with high sensitivity and specificity (average of 0.92 and 0.82, respectively). The results highlight the possibility that high-performance machine learning algorithms are able to predict unspecific negative COVID-19 outcomes using routinely-collected data.
The development of multipurpose prognostic algorithms, i.e. algorithms that identify nonspecific outcomes and overall future clinical deterioration, can be used in a large number of situations, especially in the case of complex and unknown diseases that lead to the development of several different negative outcomes. Instead of having to develop a different algorithm for each of the specific outcomes, multipurpose models can provide more comprehensive and clinically relevant information about the risks of future health problems of patients. The algorithms can be embedded in an app for smartphones or in electronic medical records to be used with routinely-collected data to perform simple predictions for each incoming patient, thus supporting screening procedures and decision-making. In the case of developing countries, while the issue of current availability of electronic medical records in poorer areas is still a challenge, in Brazil there have been promising recent advances regarding the use of electronic medical records27.
Brazil is currently the third country in the world in total number of cases and second in deaths from COVID-1928. There is a growing demand in Brazil, and in many other developing countries, for decision support in the allocation of scarce hospital resources, especially in relation to the availability of ICU beds and mechanical ventilators29,30. From a clinical standard, knowledge about immediate risks of negative prognosis can also contribute to the early start of preventive measures and new interventions, and thereby increase patient survival5,6.
For every outcome, variable importance analysis identified that age, C-reactive protein (CRP), creatinine, urea and the Braden Scale were usually among the most important. While the age of the patient is widely found to be an important predictor for most negative health outcomes, CRP has been increasingly included among the main inflammatory biomarkers for the prognosis of cardiovascular31 and respiratory diseases32. High levels of CRP have been also previously associated with individual severity of SARS-CoV-233,34. Interestingly, previous studies have also identified that chronic kidney disease is associated with developing severe conditions in COVID-19 patients35,36,37, where it has been observed that patients with higher levels of creatinine and urea are more at risk38. The Braden Scale is often used as a predictor for pressure ulcers, a common clinical classification scale for predicting pneumonia39 during clinical reception, and in this study, it was an important predictor for negative prognosis in COVID-19 patients. The scale has a score between 1 (worst score) and 4 (best score) where the factors included are sensory perception, skin moisture, activity, mobility, nutritional status and friction40. The percentage of lymphocytes in the blood has been described as a strong predictor of prognosis for the severity of the new coronavirus. A randomized study by Tan et al.41 suggested that, in most confirmed cases, the percentage of lymphocytes was reduced to 5% in 2 weeks after the onset of COVID-19, in line with other studies findings42.
The study has a few limitations that need to be mentioned. First, some of the outcomes overlap, which may have helped the performance of the aggregated models, even though in the majority of cases the outcomes were independent. In the case of ICU admission, 55% of the patients did not die or used MV, while in the case of MV and death, 63% and 70% of their respective aggregated model was trained on other outcomes. Ideally, the outcomes would never overlap, but this is clinically unfeasible given the interlaced nature of negative prognostic outcomes. Another limitation is that we analyzed data from an urban COVID-19 hotspot in Brazil, in a period where clinical protocols for the disease were still being established, so this could affect the incidence of prognostic outcomes and may not directly generalize to other periods.
In conclusion, we found that machine learning algorithms can predict severe outcomes in COVID-19 patients with high performance, including previously unobserved outcomes, using only routinely-collected laboratory, clinical and demographic data. The use of multipurpose algorithms for the prediction of overall negative prognosis is a promising new area that can support doctors with clinical and administrative decisions, especially regarding priorities for hospital admission and monitoring.
Methods
Data source
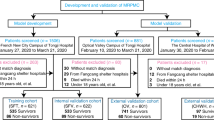
We followed a cohort of 3280 patients with a RT-PCR diagnostic exam for COVID-19 from a large hospital chain in the city of São Paulo (BP-A Beneficência Portuguesa de São Paulo) between March 1, 2020, and 28 June, 2020. Of these, 1040 (31.7%) patients were positive for COVID-19 and were included in the analysis. The study was approved by the Institutional Review Board (IRB) of BP—A Beneficência Portuguesa de São Paulo (CAAE:31177220.4.3001.5421), including a waiver of informed consent. The study followed the guidelines of the transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD)43.
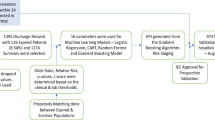
Individual patient data was collected from electronic medical records. We included as predictors only variables collected in early hospital admission, i.e. within 24 h before and 24 h after the RT-PCR exam. From a total of 82 routinely-collected variables from the hospital, 57 variables were selected for the development of the predictive models, after removing variables with 90% or higher missing values, highly-correlated variables (above 0.9) and identifying variables such as patient number and hospital identification variables. The flowchart for feature selection is described in Supplementary Fig. 2 and the complete variable list, including demographic data, laboratory tests and vital signs is described in Supplementary Table 1. Figure 3 illustrates the overall process.
Machine learning techniques
Five of the most popular machine learning models for structured data (artificial neural networks44, extra trees45, random forests46, catboost47, and extreme gradient boosting48) were trained with 70% of the data, and tested in the other 30%, simulating new unknown data. All the results reported in this study are from the test set. K-fold cross-validation with 10 folds was used to adjust the hyperparameters with Bayesian optimization (HyperOpt). Due to the unbalanced nature of the outcomes, random undersampling was performed in the training set, by randomly selecting examples from the majority class for exclusion. This technique was implemented using the RandomUnderSampler imbalanced-learn class49.
Variables with more than two categories were represented by a set of dummy variables, with one variable for each category. Continuous variables were standardized using the z-score. Variables with a correlation greater than 0.90 (mean arterial pressure, total bilirubin, and creatine kinase) were discarded, and missing values were imputed by the median. To assess the performance of the models, measures such as accuracy, sensitivity (also known as recall), specificity, positive predictive value (PPV) (also known as precision), negative predictive value (NPV), and F1 score were analyzed. The value of the AUROC was used to select the best model. To understand the individual contribution of each variable to the predictive models, we calculated their respective Shapley values. All the analyzes were performed using the Python programming language with the scikit-learn library.
Data availability
The data comes from electronic medical records from BP—A Beneficência Portuguesa de São Paulo Hospital in Brazil and it is not publicly available as it contains sensitive information of patients.
Code availability
All the code written to process and analyze the data can be made available upon request to the corresponding author.
References
Bong, C.-L. et al. The COVID-19 pandemic: Effects on low- and middle-income countries. Anesth. Analg. 131, 86–92 (2020).
Stewart, R., El-Harakeh, A. & Cherian, S. A. Evidence synthesis communities in low-income and middle-income countries and the COVID-19 response. Lancet 396, 1539–1541 (2020).
Walker, P. G. T. et al. The impact of COVID-19 and strategies for mitigation and suppression in low- and middle-income countries. Science 369(6502), 413–422. https://doi.org/10.1126/science.abc0035 (2020).
Da Silveira, M. R. COVID-19: Intensive care units, mechanical ventilators, and latent mortality profiles associated with case-fatality in Brazil. Cad. Saude Publica. 36(5), 1–12 (2020).
Cheng, F.-Y. et al. Using machine learning to predict ICU transfer in hospitalized COVID-19 patients. J. Clin. Med. 9(6). https://doi.org/10.3390/jcm9061668 (2020).
Cao, X. COVID-19: Immunopathology and its implications for therapy. Nat. Rev. Immunol. Internet. 20, 269–270. https://doi.org/10.1038/s41577-020-0308-3 (2020).
Candido, D. et al. Evolution and epidemic spread of SARS-CoV-2 in Brazil. Science 369(6508), 1255–1260. https://doi.org/10.1126/science.abd2161 (2020).
Noronha, K. V. M. S. et al. The COVID-19 pandemic in Brazil: Analysis of supply and demand of hospital and ICU beds and mechanical ventilators under different scenarios. Cad. Saude Publica 36, 1–17 (2020).
Palamim, C. V. C. & Marson, F. A. L. COVID-19—The availability of ICU beds in Brazil during the onset of pandemic. Ann. Glob. Heal. 86, 100 (2020).
Castro, M. C., Carvalho, L. R. De, Chin, T. & Kahn, R. Demand for hospitalization services for COVID-19 patients in Brazil. medRxiv. https://doi.org/10.1101/2020.03.30.20047662 (2020).
Souza, W. M. et al. Epidemiological and clinical characteristics of the COVID-19 epidemic in Brazil. Nat. Hum. Behav. 4, 856–865 (2020).
Zhou, F. et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet [Internet]. 395(10229), 1054–1062. https://doi.org/10.1016/S0140-6736(20)30566-3 (2020).
Hirayama, A. et al. The characteristics and clinical course of patients with COVID-19 who received invasive mechanical ventilation in Osaka, Japan. Int. J. Infect. Dis. 102, 282–284 (2020).
CDC. Interim clinical guidance for management of patients with confirmed coronavirus disease (COVID-19). (2020). https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html. (Accessed 7 December 2020)
Yang, X. et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study. Lancet Respir. Med. 8, 475–481 (2020).
Serafim, R. B., Póvoa, P., Souza-Dantas, V., Kalil, A. C. & Salluh, J. I. F. Clinical course and outcomes of critically ill patients with COVID-19 infection: A systematic review. Clin. Microbiol. Infect. https://doi.org/10.1016/j.cmi.2020.10.017 (2020).
Zhang, L. et al. D-dimer levels on admission to predict in-hospital mortality in patients with COVID-19. J. Thromb. Haemost. 18, 1324–1329 (2020).
Qin, L. et al. A predictive model and scoring system combining clinical and CT characteristics for the diagnosis of COVID-19. Eur. Radiol. https://doi.org/10.1007/s00330-020-07022-1 (2020).
Wang, S. et al. A fully automatic deep learning system for COVID-19 diagnostic and prognostic analysis. Eur. Respir. J. https://doi.org/10.1183/13993003.00775-2020 (2020).
DeCaprio, D. et al. Building a COVID-19 vulnerability index. medRxiv https://doi.org/10.1101/2020.03.16.20036723 (2020).
Yan, L. et al. An interpretable mortality prediction model for COVID-19 patients. Nat. Mach. Intell. 2, 283–288 (2020).
Batista, A. F. M., Miraglia, J. L., Donato, H. R. & Chiavegatto Filho, A. D. P. COVID-19 diagnosis prediction in emergency care patients: A machine learning approach. medRxiv. https://doi.org/10.1101/2020.04.04.20052092 (2020).
Heldt, F. S. et al. Early risk assessment for COVID-19 patients from emergency department data using machine learning. medRxiv. https://doi.org/10.1101/2020.05.19.20086488 (2020).
Terpos, E. et al. Hematological findings and complications of COVID-19. Am. J. Hematol. 95(7), 834–847 (2020).
Wynants, L. et al. Prediction models for diagnosis and prognosis of COVID-19 infection: Systematic review and critical appraisal. BMJ 369. https://doi.org/10.1136/bmj.m1328 (2020).
Gao, Y. et al. Machine learning based early warning system enables accurate mortality risk prediction for COVID-19. Nat. Commun. 11, 5033 (2020).
Junior, J. C., Andrade, A. B. & Carvalho, W. B. Evaluation of the use of electronic medical record systems in Brazilian intensive care units. Rev. Bras. Ter. Intensiva 30, 338–346 (2018).
WHO. Coronavirus disease (COVID-19) weekly epidemiological update and weekly operational update. (2020). https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports. (Accessed 9 December 2020).
Satomi, E. et al. Fair allocation of scarce medical resources during COVID-19 pandemic: ethical considerations. Einstein. 18. https://doi.org/10.31744/einstein_journal/2020ae5775 (2020).
Dondorp, A. M., Hayat, M., Aryal, D., Beane, A. & Schultz, M. J. Respiratory support in COVID-19 patients, with a focus on resource-limited settings. Am. J. Trop. Med. Hyg. 102, 1191–1197 (2020).
Rath, D. et al. Impaired cardiac function is associated with mortality in patients with acute COVID-19 infection. Clin. Res. Cardiol. https://doi.org/10.1007/s00392-020-01683-0 (2020).
Bajwa, E. K. et al. Plasma C-reactive protein levels are associated with improved outcome in ARDS. Chest 136(2), 471–480 (2009).
Chen, W. et al. Plasma CRP level is positively associated with the severity of COVID-19. Ann. Clin. Microbiol. Antimicrob. 19, 18 (2020).
Wang, G. et al. C-Reactive protein level may predict the risk of COVID-19 aggravation. Open Forum Infect. Dis. 7. https://doi.org/10.1093/ofid/ofaa153 (2020)
Kermali, M., Khalsa, R. K., Pillai, K., Ismail, Z. & Harky, A. The role of biomarkers in diagnosis of COVID-19—A systematic review. Life Sci. 254, 117788 (2020).
Henry, B. M. & Lippi, G. Chronic kidney disease is associated with severe coronavirus disease 2019 (COVID-19) infection. Int. Urol. Nephrol. 52(6), 1193–1194 (2020).
Cheng, Y. et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 97, 829–838 (2020).
Xiang, J. et al. Potential biochemical markers to identify severe cases among COVID-19 patients. medRxiv. https://doi.org/10.1101/2020.03.19.20034447 (2020).
Ding, Y. et al. Braden scale for assessing pneumonia after acute ischaemic stroke. BMC Geriatr. 19, 259 (2019).
Suttipong, C. & Sindhu, S. Predicting factors of pressure ulcers in older Thai stroke patients living in urban communities. J. Clin. Nurs. 21(3–4), 372–379 (2011).
Tan, L. et al. Lymphopenia predicts disease severity of COVID-19: A descriptive and predictive study. Signal Transduct. Target Ther. [Internet]. 5(1), 33. https://doi.org/10.1038/s41392-020-0148-4 (2020).
Huang, I. & Pranata, R. Lymphopenia in severe coronavirus disease-2019 (COVID-19): Systematic review and meta-analysis. J. Intensive Care 8, 36 (2020).
Moons, K. G. M. et al. Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): Explanation and elaboration. Ann. Intern. Med. 162(1), W1-73 (2015).
Bishop, C. Neural Networks for Pattern Recognition (Oxford University Press, Oxford, 1995).
Geurts, P., Ernst, D. & Wehenkel, L. Extremely randomized trees. Mach. Learn. https://doi.org/10.1007/s10994-006-6226-1 (2006).
Breiman, L. Random forests. Mach. Learn. 45, 5–32 (2001).
Dorogush, A. V., Ershov, V. & Gulin, A. CatBoost: Gradient boosting with categorical features support. arXiv. https://arxiv.org/abs/1810.11363 (2018)
Chen, T. & Guestrin, C. XGBoost: A scalable tree boosting system. Proc. ACM SIGKDD Int. Conf. Knowl. Discov. Data Mining https://doi.org/10.1145/2939672.2939785 (2016).
He, H. & Ma, Y. Imbalanced Learning: Foundations, Algorithms, and Applications (Wiley, New York, 2013).
Acknowledgements
We would like to thank the BP—A Beneficência Portuguesa de São Paulo Hospital for its willingness to contribute to the research. This work was supported by National Council for Scientific and Technological Development (CNPq) under Grant Number 402626/2020-6 and Paraíba Research Foundation FAPESQPB with Grant Number 206/2020.
Author information
Authors and Affiliations
Contributions
Initial study concept and design: A.D.P.C.F. Acquisition of data: G.D.C. Model training: F.T.F, T.A.O, C.E.T, A.F.M.B. Analysis and interpretation of data: F.T.F, T.A.O, C.E.T, G.D.C., A.D.P.C.F. Drafting of the paper: All authors contributed for drafting the manuscript. Critical revision of the manuscript: all authors provided critical review of the manuscript and approved the final draft for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fernandes, F.T., de Oliveira, T.A., Teixeira, C.E. et al. A multipurpose machine learning approach to predict COVID-19 negative prognosis in São Paulo, Brazil. Sci Rep 11, 3343 (2021). https://doi.org/10.1038/s41598-021-82885-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-82885-y
This article is cited by
-
Immune landscape and redox imbalance during neurological disorders in COVID-19
Cell Death & Disease (2023)
-
Generalizable machine learning approach for COVID-19 mortality risk prediction using on-admission clinical and laboratory features
Scientific Reports (2023)
-
An overview of deep learning techniques for COVID-19 detection: methods, challenges, and future works
Multimedia Systems (2023)
-
SARS-CoV-2 Diagnosis Using Transcriptome Data: A Machine Learning Approach
SN Computer Science (2023)
-
Proof of concept of the potential of a machine learning algorithm to extract new information from conventional SARS-CoV-2 rRT-PCR results
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.