Abstract
Logging causes changes in habitat structure, which can potentially lead to variations in taxonomic and functional richness of biodiversity. Studies on how functional traits in birds are affected by logging operations can provide an important element for the understanding of ecosystem processes. In this paper, we examined how logging in subtropical Andean forests influenced taxonomic and functional diversity of cavity-nesting birds. We used these results to compare how logging affected ecosystem functions in temperate and subtropical forests of the Americas. We used point-counts to examine the effects of logging on taxonomic and functional traits in avian communities (Functional Richness, Functional evenness, Functional Divergence, and Community-weighted mean). We found that logging changed bird richness and abundance, although it had no effect on the functional response to the measured traits. The comparison of our results with those of temperate forests of Canada and Chile reveals differences in the functional richness of birds in these habitats, with a lower impact of logging on functional traits. We highlight the importance of including functional traits in the analyses, since the reduction in the species richness and abundance may not be translated into functional changes within the ecosystem.
Similar content being viewed by others
Introduction
Severe anthropogenic disturbances, such as fragmentation, habitat loss, and selective logging are major threats to worldwide biodiversity1,2. In particular, the loss of tropical and subtropical native forests due to selective logging can result in negative changes to vertebrates’ community composition3,4 and, particularly that of birds. When logging is conducted, the extraction of different tree species changes the availability of food and nesting resources5,6. In general, logging leads to an increase in the abundance of widely distributed bird species with low resource use specificity (e.g. feeding or nesting)7,8,9. Other bird species, however, may respond negatively to these changes, with a significant decrease in the composition, number and abundance of birds9,10,11,12.
Approximately 18% of all the bird species in the world use tree cavities as nesting sites13, resulting in trees being a key resource in forest ecosystems for the reproduction of birds14,15,16. Some species excavate their own cavities (e.g. woodpeckers and trogons), whereas non-excavator birds (e.g. owls, parrots) use cavities excavated by woodpeckers or cavities formed through decaying wood13,15. Current evidence in tropical and subtropical forests indicates that cavity-nesting birds are one of the most sensitive groups to forest exploitation9,17,18,19,20. Logging may remove essential trees for reproduction of cavity-nesting birds, since certain species use trees with specific features (e.g. size, cavity depth, tree species)14,21. For example, large secondary cavity-nesting bird species (such as ducks, parrots and toucans) depend on trees with an appropriate entrance size and depth for nesting, which can be scarce or absent in logged forests14,22,23. For woodpeckers, habitat alteration can lead to difficulties in the use of substrates suitable for excavation24,25,26 and it can reduce the abundance of these birds26,27. Many of the species affected by logging play an important role as pest controllers (e.g. owls and raptors) or seed dispersers (e.g. toucans); therefore, the diversity and abundance of these species are crucial to the persistence of the ecosystem services in the forest8,17,28,29.
Traditionally, taxonomic indices analyzed changes in community diversity30,31. Nowadays these indices are complemented with functional diversity indices which describe how different species traits respond to ecosystem changes32,33. For birds, these indices have often been based on morphological (e.g. body mass) and ecological traits (e.g. trophic guilds, seed dispersal, pest control)29,32. For instance, species richness may increase or remain unchanged in response to environmental disturbance, whereas functional diversity can be sensitive to these changes31,33,34. This may occur because, in disturbed environments, there may be a replacement or decline of habitat-specialist species, with a consequent increase of generalist species35,36,37. Thus, measuring functional traits is important and necessary to understand the effect of environmental perturbation38,39.
Regarding cavity-nesting birds, preliminary work focusing on the effects of logging on species richness and functional diversity of bird species occurred in temperate forests of North (Canada) and South (Chile) America29. In this southern temperate forest, logging reduced species richness and functional diversity parameters. On the other hand, logging did not affect species richness but reduced functional parameters in the northern temperate forest29. This shows that although some species can persist in these habitats, their ecological functions may be severely affected29. Meanwhile, in subtropical forests of South America, specifically in Argentina, studies were conducted on the effect of logging on functional traits in cavity trees for secondary cavity-nesting birds40, but not on the effects of functional traits in cavity-nesting bird species. The main results of this study showed a negative effect in richness and functional parameters of cavity trees as a result of logging40. Therefore, the importance of carrying out studies that analyze and compare the effect of forest exploitation on taxonomic and functional diversity is crucial33,41,42, especially on cavity-nesting birds in temperate and subtropical forests in the Americas.
In this study, we assess taxonomic richness and functional diversity of cavity-nesting birds at both logged and unlogged Andean subtropical forests of northwestern Argentina. Based on the impact of logging on avian cavity nesters, and the results of prior studies in American temperate forests29, we expect lower taxonomic richness and functional diversity of these bird species in disturbed sites. Finally, we compare how the taxonomic and functional diversity of cavity-nesting birds respond to logging in temperate and subtropical forests of America.
Methods
Study area
This study took place in piedmont rainforest of northwestern Argentina, which belongs to the seasonally dry forests of South America located between 400 and 900 m a. s. l.43. The climate is highly seasonal with annual rainfall between 800 and 1000 mm, concentrated from November to February44. This forest is characterized by the presence of several dominant tree species: Calycophylum multiflorum, Phyllostylon rhamnoides, Myroxylon peruiferum, Amburana cearensis, and Cedrela angustifolia. Over the years, timber extraction on piedmont forests has been mainly focused on 10 tree species: Cedrela balansae, A. cearensis, Anadenanthera colubrina, C. multiflorum, P. rhamnoides, Myracrodruon urundeuva, Tabebuia avellanedae, Myroxylon peruiferum, Cordia trichotoma, and Pterogyne nitens45. The abundance of these species has been widely reduced in logged sites46,47: a volume of 4 m3/ha on overage is extracted, ranging from a minimum of 1 m3/ha to a maximum of 13 m3/ha. The largest volume of the harvested timber belongs to M. peruiferum, A. colubrina, C. multiflorum, P. rhamnoides, C. balansae and Handroanthus impetiginosus48. This logging has led to impoverished and simplified forests, with basal area values less than half of their potential45.
Fieldwork: habitat structure and bird survey
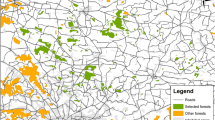
Field sampling was carried out in six sites, three of which have not been logged for more than 45 years, and three with ongoing selective logging. We grouped treatments as ‘unlogged’ and ‘logged’ sites, respectively, for statistical analyses (Fig. 1). At each site, we delimited an area of 100 ha. Habitat characterization was performed using 20 vegetation plots per site, where we measured: diameter at breast height (DBH), tree density (ind/ha, of trees with DBH > 10 cm), basal area, and tree height, as well as the density, DBH, cavity and tree heights of trees with cavities (considered trees with available cavities for secondary cavity-nesting birds).
Location of the study sites in subtropical piedmont forests of northwestern Argentina: unlogged (green) and logged (red) forest sites are depicted on the map. Total tree species and habitat variables measured are also shown. This figure was produced using QGIS version 3.20.2 (https://www.qgis.org/).
We conducted a survey of cavity-nesting birds using point-counts in logged and unlogged sites during two breeding seasons: October to January of 2015–2016 and 2016–2017. For each breeding season, in two unlogged sites, we performed 30 point- counts (site 1 and 2), while at a third site we performed 26 point-counts. We used the same methods at three logged sites as well (Fig. 1). Similarly, at two logged sites, we conducted 30 point-counts (sites 2 and 3), while at the remaining site (site 1) we conducted 26 point counts (Fig. 1). We performed the point-counts every 150 m49, recording every cavity-nesting bird species seen or heard within a 50 m radius for 10 min between 07:00 and 11:00 am. Point-counts in consecutive seasons helped us to reduce possible observer bias in species detections and to average the values of both breeding periods29,50.
Taxonomic and functional diversity analyses
With all point-counts, we calculated the richness and abundance of cavity-nesting birds for each treatment during the two years. We used the total number of species identified on the point-count to estimate species richness, and the maximum number of individuals detected as abundance (mean ± SE).
Based on Ibarra et al. (2017), functional traits associated with cavity-nesting birds in our analyses included categorical traits (nesting guild, trophic guild, and foraging substrate) and continuous traits (body mass and nest-tree size (DBH)). Categorical traits and body mass data were extracted from different publications15,29,51,52, while nest-tree size was obtained from our own data (Table 1). The nesting guild was divided into three categories: strong excavators (woodpeckers), weak excavators (trogons), and secondary-cavity nesters29. The trophic guild was divided into four categories: frugivore–insectivore, granivore, insectivore and omnivore53,54. Foraging substrate contained six categories: canopy, understory, ground, all forest strata, bark, and air29,53. Body masses were obtained from the Handbook of Birds of the World Alive55, and we averaged minimum and maximum values for each sex of each species. Traits in nest-tree size (DBH) were obtained in the field through nest searching performed at the same sites28,52,56.
The functional diversity parameters included: Functional Richness (FRic), which represents the multivariate space filled by the functional traits in bird species present, regardless of the relative abundance of bird species; Functional Evenness (FEve), which represents the degree to which bird trait values are regularly distributed according to their abundance; and Functional Divergence (FDiv), which indicates the proximity of the most frequent functional trait values in the avian community. FRic and FDiv have no upper limits, whereas FEve varies between 0 and 138,57. We also estimated the Community-Weighted mean (CWM), defined as the average of trait values in the community, weighted by the relative abundance of the species carrying each value, for body mass and nest-tree DBH29,38,57.
After standardization of traits, we estimated the functional diversity parameters for each treatment (logged and unlogged) in R (Version 1.0–11)58, using the dbFD function in package ‘FD’59. The taxonomic and functional diversity parameters were compared between treatments using an ANOVA and the Tukey-HSD posthoc test (F)29.
Finally, we compared our results with those obtained by Ibarra et al. (2017) for temperate forests of Canada and Chile with clearcutting (the authors also included partial logging) in order to compare different American forests. We reported the data obtained in the temperate forests of America along with our results, since the same analyses were carried out, with species richness, abundance, and functional diversity parameters (FRic, FEve, FDiv, CWM-biomass birds and CWM-nest tree size) obtained for both unlogged and logged sites.
Results
Site characteristics
In unlogged forests, we registered 54 tree species, and 22 of them had decay-formed cavities. In logged sites, we found 42 tree species, and 16 of them had decay-formed cavities. Averaged variable values (total trees/ha, basal area, DBH, and tree height) and dominant tree species are detailed in Fig. 1 and tables S1, S2, respectively.
Avian taxonomic diversity
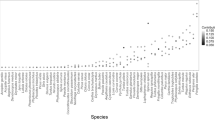
A total of 1673 individuals from 22 cavity-nesting bird species were recorded across the two breeding seasons. We registered 21 bird species at unlogged sites and 21 in ongoing logging sites. Species richness (F = 46.26; p < 0.001) and abundance (F = 64.55; p < 0.001) per point-count were significantly higher at unlogged sites (Fig. 2). Table 1 and S3 show details of cavity-nesting bird species registered and abundance in unlogged and logged sites.
Functional richness and diversity in both temperate forests in Canada and Chile and subtropical forests in Argentina. The number of bird species and species richness for each site is also noted above: distinct letters indicate differences in the mean values between treatments. Functional diversity indices measured in all sites (below) and significant differences in mean values between treatments (*) are also depicted. This figure was produced using INFOSTAT software (//www.infostat.com.ar).
Functional diversity
We found no significant differences in functional richness (F = 4.50, p = 0.10) and functional divergence (F = 3.63, p = 0.13), but functional evenness was significantly higher in logging sites (F = 10.77; p = 0.03). The Community-Weighted Mean (CWM) showed no significant difference for body mass (F = 0.01; p = 0.93) and nest-tree size (F = 0.11; p = 0.75) (Fig. 2, and Table S4).
Taxonomic and functional diversity in temperate and subtropical forests
Our results were partially similar to those reported for temperate forests (Fig. 2). In Canada, no significant differences were found in species richness between logged and unlogged sites (unlogged mean: 9.19 ± 2.94 vs. logged: 7.98 ± 1.12, p > 0.05), whereas Chile presented significant differences between treatments (unlogged mean: 10.19 ± 2.64 vs logged: 5.15 ± 1.52, p < 0.01) (Fig. 2). Functional richness values in temperate forests were close to 0, with significant differences between treatments (p < 0.01, Fig. 2). In subtropical forests, there were no significant differences between treatments and functional richness values close to 1 were observed (p = 0.10, Fig. 2). Functional evenness, on the other hand, was similar in temperate and subtropical forests (close to 0.75). Only in subtropical forests did we find significant differences regarding both treatments (logging sites presented higher values, p = 0.03). Similar values of functional divergence were also found in our three sites (close to 0.70–0.80), but treatments in Chile were different, being higher in unlogged forest sites (p < 0.05, Fig. 2). CWM-body mass turned out to be lower in temperate forests, with values close to 30 in logged and close to 50 in unlogged forests (p < 0.01, in Canada and Chile). Moreover, CWM-body mass in subtropical forests (value close to 110 g in unlogged and logged) presented no differences between treatments (p = 0.93, Fig. 2). CWM-nest tree size between treatments was significantly different only in temperate forests of Chile (p < 0.01, Fig. 2). In addition, this parameter was lower in Canada (close to 30 cm) compared to Chilean temperate forests and Argentinian subtropical forests, in which values exceeded 50 cm (Fig. 2 and Table S4).
Discussion
Our results show how logging practices affect the richness and abundance of cavity-nesting birds, but maintain avian species composition (Fig. 2). Research conducted in other temperate and subtropical environments has reported a decrease in richness and abundance of birds in logged forests, mainly due to changes in forest structure (e.g. understory and canopy), food availability and suitable nesting sites8,9,29,60. This decrease may be explained by the low availability of nesting cavities7. Additionally, in these subtropical forests, logging has negatively affected the nest density of cavity-nesting birds56. This may also explain the decrease in the abundance of both excavators and secondary-cavity nesters, since logging reduces the availability of different and abundant trees, which may affect cavity-nesting 10,27,47,52.
We found that changes in species richness between treatments (unlogged vs logged) did not affect functional diversity. This indicates that changes in taxonomic richness caused by anthropogenic disturbances do not necessarily translate into changes in functional richness31,34. Thus, available ecological niches (e.g. trees available for nesting) are still being well exploited regardless of location57.
Avian cavity nesters in the studied forests have, therefore, similar ecological functions with equally defined ecological traits34,61,62. For example, in subtropical forests, FRic (unlogged: 0.99 vs. logged: 0.98, p = 0.10) was not affected by the number of species, since this index increased with species richness. Our FRic results differ from the ones reported for the temperate forests of Canada (unlogged: 0.18 vs. logged: 0.12, p < 0.01) and Chile (unlogged: 0.20 vs. logged: 0.08, p < 0.01), where this parameter was found to be significantly lower at subtropical logged sites, causing a decrease in the amount of functional niche volume filled by these bird species29. This supports the idea that in subtropical forests the functional richness observed in logging areas does not result in a decrease in the amount of functional niche volume occupied, and this niche volume is not colonized by generalist or opportunistic cavity-nesting birds29. It has been found that in disturbed forests, opportunistic species rapidly replace forest-specialized bird species, causing a decrease in functional traits51,63.
Higher Functional Evenness (FEve) values at subtropical logged sites (unlogged: 0.65 vs. logged: 0.72, p = 0.03) may be linked to habitat change and low tree availability, where birds are exploring new functional niches to reproduce and feed38,57. In temperate forests, no differences were revealed in FEve between logged and unlogged forests, also indicating that the functional niche volume filled by bird species can be relatively resistant to tree removal29,38,51. These high values of functional evenness in logged temperate and subtropical forest (≈ 0.70) are associated with more efficient resource use by cavity-nesting birds, indicating that abundance in traits drives ecosystem processes independent of taxonomic richness64.
Functional Divergence (FDiv) was not significantly different between treatments in subtropical forests (unlogged: 0.70vs. logged: 0.78, p = 0.13), indicating that subtropical forests lack birds with dominant functional traits (e.g. bird species that utilize the largest available trees for nesting in unlogged sites)29,57. The same results were obtained for Canadian temperate forests (≈ 0.80 in unlogged and logged), whilst in Chile, this parameter was significantly higher in unlogged sites (unlogged: 0.67 vs. logged: 0.53, p < 0.05) (Fig. 2). Thus, in temperate forests of South America, logging practices may cause differentiation of functional traits in bird species (e.g. highly values of DBH nest-tree used in unlogged forests) (Fig. 2). In subtropical and north temperate forests, however, logging may not reduce nest-tree availability and characteristic38,40,57.
The community weighted mean (CWM) for body mass presented no differences between treatments in subtropical forests, revealing that the species composition and body mass are maintained3,38,57. In contrast to our findings, the two temperate forests presented significant differences between treatments with lower bird body mass (≈30–50 g in temperate and ≈100–110 g in subtropical forests, Fig. 2). Despite the higher body mass of cavity nesters in subtropical forests, species were not excluded and this variable remained constant29, which may be explained by habitat type. Subtropical forests have a higher diversity of tree species available for nesting and foraging, thus, decreasing competition and maintaining bigger species51,65. CWM for nest-tree size used by birds in subtropical forests also appeared to be unaffected between treatments. This result possibly relates to bird usage of tall tree species for nesting (DBH > 50 cm, Fig. 2), despite the lower availability of trees40. Similar results were found in Canada, but not in Chile, where significant differences were reported. This may be due to in Chile, trees used for nesting had higher DBH values in unlogged sites, whereas in Canada, birds used trees of similar size in both treatments (Fig. 2 and Table S4). Besides this, in Canada, coniferous trees cannot form as many decayed cavities as deciduous trees, resulting in secondary-cavity nesters that depend mostly on cavities created by woodpeckers15. Secondary-cavity nesters in Chile, however, depend on large, decomposing trees, which are generally removed in logging sites29. Interestingly, in subtropical forests, these bird species use cavities generated by tree decay processes in different species of living trees28,52. This may allow birds to have more niches available for exploration, even in logged sites. This indicates that birds are using trees of similar size for nesting in both logged and unlogged sites in temperate forests of North America and subtropical forests of Argentina.
Conclusion
Evidence from this study suggests that the ecological niches (e.g. nesting trees) are being well explored by birds in disturbed sites. This is an interesting finding since the analysis of richness and functional traits in trees available for secondary-cavity nesters (e.g. DBH, cavity height) showed lower values in logged sites40. Thus, despite the lower availability of tree species and the significant differences in the functional diversity of cavity trees, only trees with distinctive traits (such as DBH > 50 cm) were used in both sites, logged and unlogged. This is relevant for the management and conservation of logging sites in subtropical forests, which should consider the retention of large trees.
Finally, we highlight the importance of incorporating functional analyses under different forest disturbances, since changes in taxonomic richness do not always translate into changes in functional traits31. These analyses help in the clarification of the mechanisms and relationships between taxonomic richness and ecosystem functions57.
References
Newbold, T. et al. Global effects of land use on local terrestrial biodiversity. Nature 520, 45–50 (2015).
Chaudhary, A., Burivalova, Z., Koh, L. P. & Hellweg, S. Impact of forest management on species richness: global meta-analysis and economic trade-offs. Sci. Rep. 6, 23954 (2016).
Edwards, F. A., Edwards, D. P., Hamer, K. C. & Davies, R. G. Impacts of logging and conversion of rainforest to oil palm on the functional diversity of birds in Sundaland. Ibis 155, 313–326 (2013).
Bicknell, J. E., Struebig, M. J. & Davies, Z. G. Reconciling timber extraction with biodiversity conservation in tropical forests using reduced-impact logging. J. Appl. Ecol. 52, 379–388 (2015).
Tews, J. et al. Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures: animal species diversity driven by habitat heterogeneity. J. Biogeogr. 31, 79–92 (2004).
Robles, H. et al. Sylvopastoral management and conservation of the middle spotted woodpecker at the south-western edge of its distribution range. For. Ecol. Manag. 242, 343–352 (2007).
Aleixo, A. Effects of selective logging on a bird community in the brazilian atlantic forest. Condor 101, 537–548 (1999).
Robles, H., Ciudad, C. & Matthysen, E. Tree-cavity occurrence, cavity occupation and reproductive performance of secondary cavity-nesting birds in oak forests: the role of traditional management practices. For. Ecol. Manag. 261, 1428–1435 (2011).
Burivalova, Z. et al. Avian responses to selective logging shaped by species traits and logging practices. Proc. R. Soc. B. 282, 20150164 (2015).
Wiebe, K. L. Nest sites as limiting resources for cavity-nesting birds in mature forest ecosystems: a review of the evidence. J. Field Ornithol. 82, 239–248 (2011).
Politi, N., Hunter, M. & Rivera, L. Assessing the effects of selective logging on birds in Neotropical piedmont and cloud montane forests. Biodivers. Conserv. 21, 3131–3155 (2012).
Bergner, A. et al. Influences of forest type and habitat structure on bird assemblages of oak (Quercus spp.) and pine (Pinus spp.) stands in southwestern Turkey. For. Ecol. Manag. 336, 137–147 (2015).
van der Hoek, Y., Gaona, G. V. & Martin, K. The diversity, distribution and conservation status of the tree-cavity-nesting birds of the world. Divers. Distrib. 23, 1120–1131 (2017).
Aitken, K. E. H. & Martin, K. The importance of excavators in hole-nesting communities: availability and use of natural tree holes in old mixed forests of western Canada. J. Ornithol. 148, 425–434 (2007).
Cockle, K. L., Martin, K. & Wesołowski, T. Woodpeckers, decay, and the future of cavity-nesting vertebrate communities worldwide. Front. Ecol. Environ. 9, 377–382 (2011).
Schaaf, A. A. et al. Tree use, niche breadth and overlap for excavation by woodpeckers in subtropical piedmont forests of Northwestern Argentina. Acta Ornithol. 55 (2020).
Sekercioglu, C. H. Effects of forestry practices on vegetation structure and bird community of Kibale National Park, Uganda. Biol. Conserv. 12 (2002).
Stratford, J. A. & Robinson, W. D. Gulliver travels to the fragmented tropics: geographic variation in mechanisms of avian extinction. Front. Ecol. Environ. 3, 85–92 (2005).
Moore, R. P., Robinson, W. D., Lovette, I. J. & Robinson, T. R. Experimental evidence for extreme dispersal limitation in tropical forest birds. Ecol. Lett. 11, 960–968 (2008).
Woltmann, S. Bird community responses to disturbance in a forestry concession in lowland Bolivia. 16.
Strubbe, D. & Matthysen, E. Experimental evidence for nest-site competition between invasive ring-necked parakeets (Psittacula krameri) and native nuthatches (Sitta europaea). Biol. Conserv. 142, 1588–1594 (2009).
Rivera, L., Politi, N. & Bucher, E. H. Nesting habitat of the Tucuman Parrot Amazona tucumana in an old-growth cloud-forest of Argentina. Bird Conserv. Int. 22, 398–410 (2012).
Schaaf, A. A., Tallei, E., Politi, N. & Rivera, L. Cavity-tree use and frequency of response to playback by the Tropical Screech-Owl in northwestern Argentina. NBC 14, 99–107 (2019).
Schepps, J., Lohr, L. & Martin, T. E. Does tree hardness influence nest-tree selection by primary cavity nesters?. Auk 116, 658–665 (1999).
Rudolph, D. C., Conner, R. N. & Turner, J. Competition for red-cockaded woodpecker roost and nest cavities: effects of resin age and entrance diameter. Wilson Bull. 102(1), 23–36 (1990).
Drever, M. C. & Martin, K. Response of woodpeckers to changes in forest health and harvest: implications for conservation of avian biodiversity. For. Ecol. Manag. 259, 958–966 (2010).
Styring, A. R. & Hussin, M. Z. Effects of logging on woodpeckers in a Malaysian rain forest: the relationship between resource availability and woodpecker abundance. J. Trop. Ecol. 20, 495–504 (2004).
Ruggera, R. A., Schaaf, A. A., Vivanco, C. G., Politi, N. & Rivera, L. O. Exploring nest webs in more detail to improve forest management. For. Ecol. Manag. 372, 93–100 (2016).
Ibarra, J. T., Martin, M., Cockle, K. L. & Martin, K. Maintaining ecosystem resilience: functional responses of tree cavity nesters to logging in temperate forests of the Americas. Sci. Rep. 7, 4467 (2017).
Dı́az, S., Cabido, M. Vive la différence: plant functional diversity matters to ecosystem processes. Trends Ecol. Evolut. 16, 646–655 (2001).
Córdova-Tapia, F. & Zambrano, L. Functional diversity in community ecology. ECOS 24, 78–87 (2015).
Leaver, J., Mulvaney, J., Ehlers Smith, D. A., Ehlers Smith, Y. C. & Cherry, M. I. Response of bird functional diversity to forest product harvesting in the Eastern Cape, South Africa. For. Ecol. Manag. 445, 82–95 (2019).
Georgiev, K. B. et al. Salvage logging changes the taxonomic, phylogenetic and functional successional trajectories of forest bird communities. J. Appl. Ecol. 57, 1103–1112 (2020).
Hooper, D. U. et al. Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol. Monogr. 75, 3–35 (2005).
Kassen, R. The experimental evolution of specialists, generalists, and the maintenance of diversity: experimental evolution in variable environments. J. Evolut. Biol. 15, 173–190 (2002).
Scherer-Lorenzen, M. Biodiversity and ecosystem functioning: basic principles. Struct. Funct. 10 (2005).
Devictor, V., Julliard, R. & Jiguet, F. Distribution of specialist and generalist species along spatial gradients of habitat disturbance and fragmentation. Oikos 117, 507–514 (2008).
Villéger, S., Mason, N. W. H. & Mouillot, D. New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 89, 2290–2301 (2008).
Laliberté, E. & Legendre, P. A distance-based framework for measuring functional diversity from multiple traits. Ecology 91, 299–305 (2010).
Schaaf, A. A. et al. Functional diversity of tree cavities for secondary cavity-nesting birds in logged subtropical Piedmont forests of the Andes. For. Ecol. Manag. 464, 118069 (2020).
Lindenmayer, D. B., Margules, C. R. & Botkin, D. B. Indicators of biodiversity for ecologically sustainable forest management. Conserv. Biol. 14, 941–950 (2000).
Gregory, R. D. et al. The generation and use of bird population indicators in Europe. Bird Conserv. Int. 18, S223–S244 (2008).
Prado, D. E. Seasonally dry forests of tropical South America: from forgottenecosystems to a new phytogeographic unit. Edinb. J. Bot. 57, 437–461 (2000).
Arias, M. Estadísticas climatológicas de la Provincia de Salta. Dirección de Medio Ambiente y Recursos Naturales, Provincia de Salta, Estación Experimental Agropecuaria Salta, Inta. (1996).
Brown, A. D. & Malizia, L. R. Las Selvas Pedemontanas de las Yungas. Ciencia hoy 14, 52–63 (2004).
Politi, N., Hunter, M. Jr. & Rivera, L. Nest selection by cavity-nesting birds in subtropical montane forests of the andes: implications for sustainable forest management. Biotropica 41, 354–360 (2009).
Politi, N., Hunter, M. & Rivera, L. Availability of cavities for avian cavity nesters in selectively logged subtropical montane forests of the Andes. For. Ecol. Manag. 260, 893–906 (2010).
Eliano, P. M., Badinier, C. & Malizia, L. R. Manejo forestal sustentable en Yungas: protocolo para el desarrollo de un plan de manejo forestal e implementación en una finca piloto. Ediciones del Subtrópico, San Miguel de Tucumán (2009).
Ralph, C. J., Droege, S. & Sauer, J. R. Managing and monitoring birds using point counts: standards and applications 1: 3-8 (1995).
Hill, D. Handbook of biodiversity methods: survey, evaluation and monitoring (Cambridge University Press, 2005).
Ibarra, J. T. & Martin, K. Biotic homogenization: loss of avian functional richness and habitat specialists in disturbed Andean temperate forests. Biol. Conserv. 192, 418–427 (2015).
Schaaf, A. A. et al. Identification of tree groups used by secondary cavity-nesting birds to simplify forest management in subtropical forests. J. For. Res. 31, 1417–1424 (2020).
Blendinger, P. G. & Álvarez, M. E. Aves de la Selva Pedemontana de las Yungas australes. In: Selva Pedemontana de las Yungas. Historia Natural, Ecología y Manejo de un Ecosistema en Peligro. (Eds AD Brown, A. D et al.) 233–272 (2009).
Wilman, H. et al. EltonTraits 1.0: Species-level foraging attributes of the world’s birds and mammals: Ecol. Arch. Ecol. 95, 2027–2027 (2014).
del Hoyo, J. A., Sargatal, J., Christie, D. A. & de Juana, E. Handbook of the Birds of the World Alive. (Lynx Edicions, 2017).
Schaaf, A. A. et al. Influence of logging on nest density and nesting microsites of cavity-nesting birds in the subtropical forests of the Andes. For. Int. J. For. Res. https://doi.org/10.1093/forestry/cpab032 (2021).
Mason, N. W. H., Mouillot, D., Lee, W. G. & Wilson, J. B. Functional richness, functional evenness and functional divergence: the primary components of functional diversity. Oikos 111, 112–118 (2005).
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org/ (2016).
Laliberté, E., Legendre, P. & Shipley, B. FD: measuring functional diversity (FD) from multiple traits, and other tools for functional ecology. http://cran.r-project.org/web/packages/FD (2011).
Ghadiri Khanaposhtani, M., Kaboli, M., Karami, M., Etemad, V. & Baniasadi, S. Effects of logged and unlogged forest patches on avifaunal diversity. Environ. Manag. 51, 750–758 (2013).
Tilman, D. The influence of functional diversity and composition on ecosystem processes. Science 277, 1300–1302 (1997).
Mouchet, M. A., Villéger, S., Mason, N. W. H. & Mouillot, D. Functional diversity measures: an overview of their redundancy and their ability to discriminate community assembly rules: functional diversity measures. Funct. Ecol. 24, 867–876 (2010).
Mackey, B. et al. Policy options for the world’s primary forests in multilateral environmental agreements. Conserv. Lett. 8, 139–147 (2015).
Petchey, O. L. & Gaston, K. J. Functional diversity: back to basics and looking forward. Ecol. Lett. 9, 741–758 (2006).
Azeria, E. T. et al. Differential response of bird functional traits to post-fire salvage logging in a boreal forest ecosystem. Acta Oecol. 37, 220–229 (2011).
Acknowledgements
We thank the reviewers and the editor for the useful comments which greatly improved the manuscript. We thank the assistants involved in the fieldwork. We thank National Park Administration of Argentina for allowing us to work in Calilegua National Park. This study was funded by CONICET, Rufford small grant, Optic for the tropic, Bergstrom Award, and Idea Wild. AAS, DG, ET, and ET are postdoctoral fellows at CONICET; RAR is researchers at CONICET, Argentina.
Author information
Authors and Affiliations
Contributions
A.A.S, E.T, CGV and R.A.R conceived the study and conducted the fieldwork; D.G analyzed data. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schaaf, A.A., Gomez, D., Tallei, E. et al. Responses of functional traits in cavity-nesting birds to logging in subtropical and temperate forests of the Americas. Sci Rep 11, 24309 (2021). https://doi.org/10.1038/s41598-021-03756-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-03756-0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.