Abstract
The association between sleep apnea (SA) and depression had been reported in a few previous studies. However, whether SA increases the risk of major depressive disorder (MDD) has not been studied comprehensively in a large-scale study. We performed this population-based cohort study to assess the association between SA and MDD. We identified adult patients having SA from the Taiwan National Health Insurance Research Database and excluded those having MDD before SA diagnosis. Thirty control subjects were randomly selected to match to each SA patient by age and sex. Totally, 10,259 SA patients were matched to 102,590 control subjects. The incidence rate and cumulative incidence of MDD were significantly higher in SA patients than in the control subjects (both p < 0.0001). Multivariable Cox regression analysis showed that SA remained an independent risk factor for incident MDD after adjusting for age, sex, residency, income level, and comorbidities (hazard ratio = 2.9 [95% CI 2.8–3.1], p < 0.0001). In summary, SA patients have an increased risk to develop MDD. Physicians caring for SA patients must pay attention to their psychosocial health status.
Similar content being viewed by others
Introduction
Sleep apnea (SA) is the most common sleep disorder, which presents with repetitive cessation of breathing during sleep, usually associated with intermittent hypoxia, sleep fragmentation and daytime sleepiness1,2,3,4,5,6. The prevalence of SA, estimated by Wisconsin Sleep Cohort study, was higher in men than in women (24% and 9%, respectively)7. The diagnosis of SA is usually confirmed by nocturnal polysomnography (PSG), and the severity is usually classified by apnea–hypopnea index (AHI)8. Obstructive SA is the major form of SA, accounting for more than 90% of cases. The risk factors for SA include obesity, nasal congestion, alcohol, smoking, estrogen depletion in menopause, and so on9. SA has been associated with cardiovascular mortality and morbidity, including myocardial infarction, arrhythmia, sudden cardiac death, and heart failure10.
Major depressive disorder (MDD) is one of the most common psychiatric disorders, which may lead to disability and incur significant economic burden. The prevalence of depression is 4.2% in men and 8.9% in women in a German study11. The lifetime prevalence of MDD was found lower (about 1.2%) in Taiwan, while this might be underestimated12,13. In addition to depressed mood, patients may have other symptoms such as difficulty falling asleep, daytime sleepiness, poor appetite, fatigue, and low energy sensation. These symptoms may overlap the symptoms of SA. Because functional impairment from MDD may cause family and social problems, early detection and intervention are needed.
The association between SA and MDD has been reported in some previous studies. Besides the shared symptoms, both SA and depression are associated with systemic diseases such as cardiovascular diseases and diabetes mellitus14,15,16,17. Douglas et al. have reported a high prevalence of depressive symptoms (32–53%) in patients with snoring or SA18. Although previous studies had also demonstrated the association between depression and SA19,20, whether SA increases the risk of MDD has not been well studied in a large-scale study comprehensively. Since SA has been associated with various comorbidities, which might contribute to the development of MDD, we performed a population-based cohort study using Taiwan National Health Insurance (NHI) Research Database (NHIRD) to assess the association between SA and MDD.
Methods
Data sources
Since March, 1995, the Taiwan NHI has covered ambulatory care, inpatient care, dental services, and prescription drugs with a coverage rate of more than 96% of whole population of about 23 million people1,2. The NHIRD, containing information about reimbursement claims in the NHI system, was managed by the Taiwan National Health Research Institutes and was released with encrypted identification of patients and healthcare providers for medical research. In this study, we used Longitudinal Health Insurance Database 2010 (LHID2010), which included one million randomly sampled subjects in the NHI system in 2013 with their associated information until the end of 2010. This study has been approved by the Institutional Review Board in Kaohsiung Medical University Hospital (KMUH-IRB-EXEMPT-20130034 and KMUHIRB-EXEMPT(II)-20150068) and was exempted from obligation to gain inform consent of the study population. All the methods were carried out in accordance with relevant guidelines and regulations.
Study population
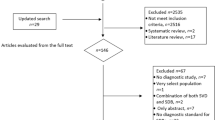
Patients having SA diagnosis between March 1995 and December 2013 were identified initially (Fig. 1). The diagnosis of SA was identified using the International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM) codes of 780.51, 780.53, and 780.571,2,3,4. This method for identifying SA patients in the NHIRD has been validated in previous studies1,21,22,23. The dates of their first SA diagnosis were defined as their index dates. We excluded patients with washout periods (from NHI enrollment to the index date) < 1 year or follow-up periods < 1 year to increase the likelihood of including newly-diagnosed SA cases and to ensure sufficient follow-up periods. Patients with MDD before SA diagnosis were also excluded. Besides, patients with age < 18 years or > 90 years were also excluded. The remaining SA patients (namely “suspected SA patients”) were included for further analyses.
Ten control subjects (namely “control A subjects”) were randomly selected to match to each SA patient by age and sex. The control subjects, who were assigned index dates as their corresponding SA patients, had no MDD diagnosis before their index dates and had sufficient washout periods and follow-up periods as the SA patients. The study arm A consisted of suspected SA patients and control A subjects (Fig. 1).
In order to confirm our findings, patients without SA diagnosis after PSG examination or patients who never received PSG examination and their corresponding control subjects were excluded. In another word, a subgroup of SA patients (namely “probable SA patients”), who remained having SA diagnosis after PSG, and their corresponding control subjects (namely “control B subjects”) were further extracted for another set of analyses (study arm B).
To further confirm our findings, we selected the suspected SA patients who had SA diagnosis in at least three ambulatory claims or one inpatient claim (namely “highly suspected SA patients”) and their corresponding control subjects (namely “control C subjects”) for another set of analyses (study arm C) (Fig. 1). Similarly, we also selected the probable SA patients who had SA diagnosis in at least three ambulatory claims or one inpatient claim (namely “highly probable SA patients”) and their corresponding control subjects (namely “control D subjects”) for another set of analyses (study arm D).
Criteria and definitions of variables
Comorbidities were identified by the presence of any corresponding diagnostic codes in at least three ambulatory claims or one inpatient claim and the first appearance before the index date. Based on the comorbidities, the Charlson Comorbidity Index (CCI) score was calculated24.
Study outcome
The endpoint of this study was incident MDD, defined by the first MDD diagnosis. The diagnosis of MDD was identified with the ICD-9-CM codes of 296.2 and 296.325, while only the patients having MDD diagnosis in at least three ambulatory claims or one inpatient claim were considered having MDD to increase the reliability of the diagnosis.
All subjects were followed from their index dates to either incident MDD or end of the record due to the end of the study period, withdrawal from the NHI, or death, whichever came first.
Statistical analysis
The demographic data and comorbidities were compared between the SA patients and the control subjects using the Student’s t test and Pearson χ2 test and for continuous variables and categorical variables, respectively. The MDD incidence rate was calculated as the number of incident MDD during the follow-up period divided by the total person-year. The MDD incidence rates (IR) were further compared between SA patients and the control subjects by calculating the incidence rate ratio (IRR). Under the assumption that the observed number of incident MDD followed a Poisson probability distribution, a 95% confidence interval (CI) was calculated for each IRR. Stratified analyses, by classifying the subjects with age, sex, residency, income level, or comorbidities, were also performed. Using multivariable analyses adjusting for age, sex, residency, income level, and comorbidities, the adjusted IRRs were calculated. Cumulative incidences of MDD in SA patients and control subjects were calculated and compared using Kaplan–Meier method and log-rank test. Using multivariable Cox regression analyses adjusting for sex, age, residency, income level, and comorbidities, the effect of SA on incident MDD was also assessed. Adjusted hazard ratios (HRs) are presented with 95% CI.
One might argue that the ICD-9-CM code of 296.3 (Major depressive disorder, recurrent episode) should not be used to identify incident MDD, because, by definition, a past depressive episode prior to the index episode is required to qualify for this diagnostic code. We therefore performed a set of sensitivity analyses, taking only ICD-9-CM code of 296.2 (major depressive disorder, single episode) for the outcome (incident MDD) (in another word, 296.3 was not considered as the outcome in the sensitivity analyses).
Extraction, computation, linkage, processing, and sampling, of data and statistical analyses were performed using SAS, Version 9.4 of the SAS System for Windows (SAS Institute Inc., Cary, NC, USA). A two-sided p value of < 0.05 was taken as the criterion for statistical significance.
Results
Through the algorithm (Fig. 1), 10,259 “suspected SA” patients, including 4150 “probable SA” patients, were identified and matched to 102,590 “control A” subjects, including 41,500 “control B” subjects. Table 1 presented the baseline characteristics of the study cohorts. The mean (± standard deviation) ages of the study population were 47.1 (± 14.7) and 47.2 (± 13.3) years in study arms A and B, respectively. In study arm A, 64% of the study subjects were male; 79% of the study subjects were male in study arm B. Compared with the corresponding control subjects, SA patients had better economic status and more comorbidities (Table 1).
The incidence rates of MDD in suspected SA patients and probable SA patients are significantly higher than the corresponding control subjects (suspected SA patients vs. control A subjects: 4.4 vs. 1.3 per 1000 patient-year, adjusted IRR [95% CI] 2.9 [2.8–3.1], p < 0.0001; probable SA patients vs. control B subjects: 3.1 vs. 1.1 per 1000 patient-year, adjusted IRR [95% CI] 2.1 [2.0–2.3], p < 0.0001) (Table 2). In the stratified analyses of the study population stratified by age, sex, residency, income level, or the presence of any comorbidity, SA patients had a significantly higher MDD incidence rate compared with the corresponding control subjects in all strata (Table 2).
The cumulative MDD incidence are significantly higher in suspected SA patients and probable SA patients than in the corresponding control subjects (both p < 0.0001) (Fig. 2a,b). In the strata classified with sex or age, SA patients had a significantly higher cumulative MDD incidence compared with the corresponding control cohorts (all p < 0.0001) (Fig. 2c–j).
The cumulative incidences of major depressive disorder (MDD). The red continuous lines and blue dashed lines show the cumulative incidence of MDD for the sleep apnea patients and the control subjects, respectively. (a, c, e, g, i) suspected sleep apnea patients vs. control A subjects; (b, d, f, h, j) probable sleep apnea patients vs. control B subjects; (a, b) all eligible subjects; (c, d) female subjects; (e, f) male subjects; (g, h) subjects ≤ 50 years old; (i, j) subjects > 50 years old.
On multivariable Cox proportional hazards regression analyses adjusted for age, sex, residency, income, and comorbidities, SA was an independent risk factor for incident MDD (study arm A: adjusted HR [95% CI] 2.9 [2.6–3.4], p < 0.0001; study arm B: adjusted HR [95% CI] 2.1 [1.6–2.8], p < 0.0001) (Fig. 3). Stratified analyses revealed that SA was associated with a higher risk for developing MDD in nearly all strata (Fig. 3).
Stratified analyses of multivariable Cox regression analyses assessing the effect of sleep apnea on incident major depressive disorder. The results are presented with adjusted HRs (95% CI) of sleep apnea, which are adjusted for sex, age, residency, income level and the presence of various comorbidities (except for the variable used for stratification). (a) Study arm A (suspected sleep apnea patients and control A subjects); (b) study arm B (probable sleep apnea patients and control B subjects). *SA sleep apnea, CCI Charlson Comorbidity Index, HR hazard ratio, CI confidence interval. †Due to small sample size, hazard ratio cannot be estimated.
To confirm our findings, we selected the suspected SA patients and probable SA patients who had SA diagnosis in at least three ambulatory claims or one inpatient claim (namely “highly suspected SA patients” and “highly probable SA patients”, respectively) and their corresponding control subjects (namely “control C subjects” and “control D subjects”, respectively) for another sets of analyses (study arms C and D) (Fig. 1, Supplementary Table S1 online). The study arms C and D showed consistent results as the findings in the study arms A and B (Supplementary Figs. S1, S2 and Table S2 online).
The sensitivity analyses, taking only ICD-9-CM code of 296.2 (major depressive disorder, single episode) for the outcome (incident MDD), showed similar findings as those in the previous analyses (Supplementary Figs. S3–S6 and Tables S3–S4 online).
Discussion
In this large population-based cohort study, we found that patients with SA had a significantly higher MDD incidence than the subjects without SA. Multivariable analyses adjusting for age, sex, and comorbidities showed that SA remained an independent risk factor for developing MDD. In further investigation, the patients having SA diagnosis after PSG also had higher incidence of MDD.
A few previous studies have used NHIRD to investigate the association between SA and mood disorder. Chen et al. used data from Longitudinal Health Insurance Database 2000 (LHID2000) to compare the risk of subsequent depressive disorder during one-year follow-up in 2818 patients being diagnosed with SA after PSG and 14,090 matched control subjects26. They used ICD-9-CM codes 296.2, 296.3, 300.4, and 311 to identify the diagnosis of depressive disorder, whereas we used only 296.2 and 296.3 to identify pure major depressive disorder. As in our study, they showed SA as an independent risk factor for subsequent depressive disorder. Another study using LHID2000 by Lu et al. showed an increased risk of mood disorder, especially MDD and bipolar disorder, in SA patients than subjects in the comparison group25. They mixed MDD with bipolar disorders and unspecified episodic mood disorders, whereas we focused on MDD. Furthermore, a study using Longitudinal Health Insurance Database 2005 (LHID2005) by Pan et al. also found a bidirectional association between SA and depression27. As Chen et al.26 they used ICD-9-CM codes 296.2, 296.3, 300.4, and 311 to identify the diagnosis of depression27. Both Lu et al.25 and Pan et al.27 used only diagnostic codes to identify SA patients, whereas we performed comprehensive analyses using both the cohort with SA patients identified by diagnostic codes and a subtracted cohort with SA patients diagnosed after PSG. Although some differences existed in the study design, these studies consistently showed increased risk of depression in SA patients, as shown in our study.
Other studies involving different study population showed similar results as our findings. In a prospective study involving 447 people diagnosed with OSA in a German sleep center, the prevalence of depression according to ICD-10 was 21.5% in patients with AHI > 9 (n = 303)28. Heinzer et al. performed a general population-based study including 2121 people with polysomnography data in Switzerland. The prevalence of moderate-to-severe sleep-disordered breathing (≥ 15 events per hour) was 23.4% in women and 49.7% in man. Multivariable analysis showed the upper quartile for the apnea–hypopnea index (> 20.6 events per hour) was associated independently with depression (odds ratio = 1.92, p = 0.029)29. A population-based study in Australia showed that people with OSA and daytime sleepiness had a strong association with depression (mild–moderate apnea: adjusted odds ratio [95% CI] 3.86 [1.87–7.95]; severe apnea: adjusted odds ratio [95% CI] 4.82 [1.42–16.35])30.
Although some previous studies have shown the association between SA and depression, the definite mechanisms have not been fully understood31. Sleep fragmentation and hypoxemia might contribute to the incident MDD in SA patients. Sleep fragmentation and sleep deprivation result in excess daytime sleepiness and may worsen the cognitive function and mood32,33. The SA patients with excess daytime sleepiness are more likely to have depressive symptoms34. Elra et al. also found that daytime sleepiness, sleep medications, and initial insomnia were independently related to depression in SA patients, whereas SA severity was not35. Although some studies showed that intermittent hypoxia was not significantly associated with depressive symptoms31, others still demonstrated the possible association between hypoxia and depression. Bardwell et al. conducted a randomized controlled trial of using continuous positive airway pressure (CPAP) treatment or oxygen supplementation in SA patients with depression and found improvement in psychological symptoms in the patients receiving oxygen supplementation, but not those using CPAP treatment36. This finding might suggest that hypoxemia played an important role in SA-associated depression, while the actual mechanisms remained uncertain. The IR of MDD appeared lower in probable SA patients than suspected SA patients. Although information about long-term oxygen supplementation or CPAP treatment was not available in the NHIRD, we believe those having SA diagnosis after PSG might have higher chance to be adequately treated, which ameliorated the effect of SA in increasing the risk of MDD.
Neurotransmitters have important roles in both depression and SA. The majority of evidence supports under-activation of serotonin system in depression37. Reduced serotonin 1A receptor binding was also associated with the pathophysiological changes of depression38. Alteration of serotonin system affects both respiratory and sleep-awake cycle, which may contribute to SA. Loss of serotonergic inputs to laryngeal motor neurons in nucleus ambiguus compromises upper airways patency39. An animal study showed that serotonin 1A receptor had an important role in neural control of upper airway patency40.
On the other hand, inflammatory cytokines are also important mediators between SA and depression. In one meta-analysis, the levels of inflammatory markers, such as interleukin 6, interleukin 8, and tumor necrosis factor alpha were higher in SA patients41. Chronic exposure to these inflammatory cytokines may lead to psychiatric disorder and depression. Cytokines activate inflammatory signaling pathways in the brain and result in changes of glutamate, monoamine, and neuropeptide systems, contributing to the development of depression42.
In the current study, the IR of MDD were higher among female subjects than the male subjects. The HR and IRR of SA appeared higher in female subjects than in male subjects, suggesting that the effect of SA on incident MDD might be more prominent in the female. The female had greater proinflammatory responses of monocytes induced by sleep loss43. The enhanced inflammatory responses might contribute to the development of mood disorders in SA patients, and the female patients might have greater effects from SA.
Out study still had some limitations. Firstly, the diagnosis of SA and MDD based on diagnostic codes might include misclassified cases. Therefore, we performed further analyses of SA patients diagnosed after PSG and found consistent results. In addition, the method using diagnostic codes to identify SA and MDD in the NHI database has been validated and used in previous studies21,22,23,25. Secondly, the symptoms, severity and treatment (CPAP or oxygenation therapy) for SA were not adequately available in the NHI database. Previous studies suggested daytime sleepiness and hypoxemia might lead to the development of MDD, so treatment of SA may theoretically decrease the risk of MDD. We believed that a proportion of SA patients included in our study had received treatment, so the risk of developing MDD might be underestimated. Nevertheless, we still found significantly increased MDD incidence in SA patients. Thirdly, some risk factors for MDD, such as alcoholism, were not available in the NHIRD, and we did not control for the use of medications, such as hypnotics, in the current study. The interpretation of our findings should be careful to account for the possible impacts from these factors. Fourthly, the index dates of SA patients and the dates of first MDD diagnosis might not be the actual onset dates. As we have no chance to identify individuals’ intervals of diagnostic delay, the interpretation of our findings should therefore be careful.
In conclusion, this large nationwide population-based cohort study confirmed SA as an independent risk factor for incident MDD. Clinicians should pay attention to any signs of MDD while seeing patients with SA.
Abbreviations
- AHI:
-
Apnea–hypopnea index
- CI:
-
Confidence interval
- CPAP:
-
Continuous positive airway pressure
- ICD-9-CM:
-
International Classification of Diseases, 9th Revision, Clinical Modification
- ICD-10:
-
International Classification of Diseases, 10th Revision
- HR:
-
Hazard ratio
- IQR:
-
Interquartile range
- IR:
-
Incidence rate
- IRR:
-
Incidence rate ratio
- MDD:
-
Major depressive disorder
- NHI:
-
National Health Insurance
- NHIRD:
-
National Health Insurance Research Database
- PSG:
-
Polysomnography
- SA:
-
Sleep apnea
- SD:
-
Standard deviation
References
Wu, K. L. et al. CHADS(2), CHA(2)DS(2)ASc, and new ABCD scores predict the risk of peripheral arterial disease in patients with sleep apnea. J. Clin. Med. https://doi.org/10.3390/jcm8020188 (2019).
Chen, C. M. et al. Erectile dysfunction in patients with sleep apnea: a nationwide population-based study. PLoS ONE 10, e0132510. https://doi.org/10.1371/journal.pone.0132510 (2015).
Chou, K. T. et al. Sleep apnea and risk of deep vein thrombosis: a non-randomized, pair-matched cohort study. Am. J. Med. 125, 374–380. https://doi.org/10.1016/j.amjmed.2011.07.003 (2012).
Chou, P. S. et al. Increased risk of benign prostate hyperplasia in sleep apnea patients: A nationwide population-based study. PLoS ONE 9, e93081. https://doi.org/10.1371/journal.pone.0093081 (2014).
Veasey, S. C. & Rosen, I. M. Obstructive sleep apnea in adults. N. Engl. J. Med. 380, 1442–1449. https://doi.org/10.1056/NEJMcp1816152 (2019).
Tzeng, N. S. et al. obstructive sleep apnea in children and adolescents and the risk of major adverse cardiovascular events: A nationwide cohort study in Taiwan. J. Clin. Sleep Med. 15, 275–283. https://doi.org/10.5664/jcsm.7632 (2019).
Young, T. et al. The occurrence of sleep-disordered breathing among middle-aged adults. N. Engl. J. Med. 328, 1230–1235. https://doi.org/10.1056/NEJM199304293281704 (1993).
Myers, K. A., Mrkobrada, M. & Simel, D. L. Does this patient have obstructive sleep apnea? The Rational Clinical Examination systematic review. J. Am. Med. Assoc. 310, 731–741. https://doi.org/10.1001/jama.2013.276185 (2013).
Young, T., Peppard, P. E. & Gottlieb, D. J. Epidemiology of obstructive sleep apnea: A population health perspective. Am. J. Respir. Crit. Care Med. 165, 1217–1239 (2002).
Gottlieb, D. J. et al. Prospective study of obstructive sleep apnea and incident coronary heart disease and heart failure: The sleep heart health study. Circulation 122, 352–360. https://doi.org/10.1161/CIRCULATIONAHA.109.901801 (2010).
Maske, U. E. et al. Prevalence and correlates of DSM-IV-TR major depressive disorder, self-reported diagnosed depression and current depressive symptoms among adults in Germany. J. Affect. Disord. 190, 167–177. https://doi.org/10.1016/j.jad.2015.10.006 (2016).
Hwu, H. G., Chang, I. H., Yeh, E. K., Chang, C. J. & Yeh, L. L. Major depressive disorder in Taiwan defined by the Chinese diagnostic Interview Schedule. J. Nerv. Ment. Dis. 184, 497–502 (1996).
Liao, S. C. et al. Low prevalence of major depressive disorder in Taiwanese adults: Possible explanations and implications. Psychol. Med. 42, 1227–1237. https://doi.org/10.1017/S0033291711002364 (2012).
Reichmuth, K. J., Austin, D., Skatrud, J. B. & Young, T. Association of sleep apnea and type II diabetes: A population-based study. Am. J. Respir. Crit. Care Med. 172, 1590–1595. https://doi.org/10.1164/rccm.200504-637OC (2005).
Sweileh, W. M., Abu-Hadeed, H. M., Al-Jabi, S. W. & Zyoud, S. H. Prevalence of depression among people with type 2 diabetes mellitus: A cross sectional study in Palestine. BMC Public Health 14, 163. https://doi.org/10.1186/1471-2458-14-163 (2014).
Maeder, M. T., Schoch, O. D. & Rickli, H. A clinical approach to obstructive sleep apnea as a risk factor for cardiovascular disease. Vasc. Health Risk Manag. 12, 85–103. https://doi.org/10.2147/VHRM.S74703 (2016).
Seldenrijk, A. et al. Depression, anxiety and 6-year risk of cardiovascular disease. J. Psychosom. Res. 78, 123–129. https://doi.org/10.1016/j.jpsychores.2014.10.007 (2015).
Douglas, N. et al. Prevalence of depression in patients referred with snoring and obstructive sleep apnoea. Intern. Med. J. 43, 630–634. https://doi.org/10.1111/imj.12108 (2013).
Enright, P. L. et al. Prevalence and correlates of snoring and observed apneas in 5,201 older adults. Sleep 19, 531–538 (1996).
Aloia, M. S. et al. Examining the construct of depression in obstructive sleep apnea syndrome. Sleep Med. 6, 115–121. https://doi.org/10.1016/j.sleep.2004.09.003 (2005).
Lee, Y. C. et al. Sleep apnea and the risk of chronic kidney disease: A nationwide population-based cohort study. Sleep 38, 213–221. https://doi.org/10.5665/sleep.4400 (2015).
Shiao, T. H. et al. Sleep apnea and risk of peptic ulcer bleeding: a nationwide population-based study. Am. J. Med. 126, 249–255. https://doi.org/10.1016/j.amjmed.2012.08.017 (2013).
Su, V. Y. et al. Sleep apnea and risk of panic disorder. Ann. Fam. Med. 13, 325–330. https://doi.org/10.1370/afm.1815 (2015).
Charlson, M. E., Pompei, P., Ales, K. L. & MacKenzie, C. R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 40, 373–383. https://doi.org/10.1016/0021-9681(87)90171-8 (1987).
Lu, M. K. et al. Sleep apnea is associated with an increased risk of mood disorders: A population-based cohort study. Sleep Breath. 21, 243–253. https://doi.org/10.1007/s11325-016-1389-x (2017).
Chen, Y. H., Keller, J. K., Kang, J. H., Hsieh, H. J. & Lin, H. C. Obstructive sleep apnea and the subsequent risk of depressive disorder: A population-based follow-up study. J. Clin. Sleep Med. 9, 417–423. https://doi.org/10.5664/jcsm.2652 (2013).
Pan, M. L. et al. Bidirectional association between obstructive sleep apnea and depression: A population-based longitudinal study. Medicine 95, e4833. https://doi.org/10.1097/MD.0000000000004833 (2016).
Acker, J. et al. Obstructive sleep apnea (OSA) and clinical depression-prevalence in a sleep center. Sleep Breath. 21, 311–318. https://doi.org/10.1007/s11325-016-1411-3 (2017).
Heinzer, R. et al. Prevalence of sleep-disordered breathing in the general population: The HypnoLaus study. Lancet 3, 310–318. https://doi.org/10.1016/S2213-2600(15)00043-0 (2015).
Lang, C. J. et al. Associations of undiagnosed obstructive sleep apnea and excessive daytime sleepiness with depression: An Australian population study. J. Clin. Sleep Med. 13, 575–582. https://doi.org/10.5664/jcsm.6546 (2017).
Chirinos, D. A. et al. Depressive symptoms in patients with obstructive sleep apnea: Biological mechanistic pathways. J. Behav. Med. 40, 955–963. https://doi.org/10.1007/s10865-017-9869-4 (2017).
Pesonen, A. K. et al. REM sleep fragmentation associated with depressive symptoms and genetic risk for depression in a community-based sample of adolescents. J. Affect. Disord. 245, 757–763. https://doi.org/10.1016/j.jad.2018.11.077 (2019).
Schwarz, J. et al. Mood impairment is stronger in young than in older adults after sleep deprivation. J. Sleep Res. 1, e12801. https://doi.org/10.1111/jsr.12801 (2018).
Ishman, S. L., Cavey, R. M., Mettel, T. L. & Gourin, C. G. Depression, sleepiness, and disease severity in patients with obstructive sleep apnea. Laryngoscope 120, 2331–2335. https://doi.org/10.1002/lary.21111 (2010).
Bjornsdottir, E. et al. The prevalence of depression among untreated obstructive sleep apnea patients using a standardized psychiatric interview. J. Clin. Sleep Med. 12, 105–112. https://doi.org/10.5664/jcsm.5406 (2016).
Bardwell, W. A. et al. Effects of 2-week nocturnal oxygen supplementation and continuous positive airway pressure treatment on psychological symptoms in patients with obstructive sleep apnea: A randomized placebo-controlled study. Behav. Sleep Med. 5, 21–38. https://doi.org/10.1080/15402000709336724 (2007).
Ressler, K. J. & Nemeroff, C. B. Role of serotonergic and noradrenergic systems in the pathophysiology of depression and anxiety disorders. Depress. Anxiety 12(Suppl 1), 2–19. https://doi.org/10.1002/1520-6394(2000)12:1+%3c2::AID-DA2%3e3.0.CO;2-4 (2000).
Wang, L. et al. Serotonin-1A receptor alterations in depression: A meta-analysis of molecular imaging studies. BMC Psychiatry 16, 319. https://doi.org/10.1186/s12888-016-1025-0 (2016).
Hilaire, G. et al. The role of serotonin in respiratory function and dysfunction. Respir. Physiol. Neurobiol. 174, 76–88. https://doi.org/10.1016/j.resp.2010.08.017 (2010).
Besnard, S. et al. Differential respiratory control of the upper airway and diaphragm muscles induced by 5-HT1A receptor ligands. Sleep Breath. 16, 135–147. https://doi.org/10.1007/s11325-010-0466-9 (2012).
Nadeem, R. et al. Serum inflammatory markers in obstructive sleep apnea: A meta-analysis. J. Clin. Sleep Med. 9, 1003–1012. https://doi.org/10.5664/jcsm.3070 (2013).
Felger, J. C. & Lotrich, F. E. Inflammatory cytokines in depression: neurobiological mechanisms and therapeutic implications. Neuroscience 246, 199–229. https://doi.org/10.1016/j.neuroscience.2013.04.060 (2013).
Irwin, M. R., Carrillo, C. & Olmstead, R. Sleep loss activates cellular markers of inflammation: Sex differences. Brain Behav. Immun. 24, 54–57. https://doi.org/10.1016/j.bbi.2009.06.001 (2010).
Acknowledgements
The authors thank the help from the Statistical Analysis Laboratory, Department of Internal Medicine and the Statistical Analysis Laboratory, Department of Medical Research, Kaohsiung Medical University Hospital. This work was supported by grants from Kaohsiung Medical University Hospital (Grant No.: KMUH108-8R13 and KMUH109-9R14) and Kaohsiung Medical University (Grant No.: KMU-Q108005). This study is based on data from the National Health Insurance Research Database provided by the National Health Insurance Administration, Ministry of Health and Welfare and managed by National Health Research Institutes. The interpretation and conclusions contained herein do not represent those of National Health Insurance Administration, Ministry of Health and Welfare or National Health Research Institutes (NHRI). Due to legal and ethical restrictions, researchers should contact NHRI (http://nhird.nhri.org.tw/index.htm) for access of the data after approved by institutional review board.
Author information
Authors and Affiliations
Contributions
C.M.C., C.Y.K., M.N.W., J.Y.H., C.Y.H., M.J.T. contribute the conceptualization of the study. C.Y.K., M.J.T. were responsible for data collection and analysis. C.M.C., M.J.T. wrote the main manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, CM., Kuo, CY., Wu, MN. et al. Increased risk of major depressive disorder in sleep apnea patients in Taiwan. Sci Rep 11, 765 (2021). https://doi.org/10.1038/s41598-020-80759-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-80759-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.