Abstract
SiO2-SO3H, with a surface area of 115 m2/g, pore volumes of 0.38 cm3g−1 and 1.32 mmol H+/g, was used as a 10% w/w catalyst for the preparation of 5-hydroxymethyl-2-furfural (HMF) from fructose. A conversion of 100% was achieved in a microwave reactor during 10 min at 150 °C in DMSO, with 100% selectivity for HMF, at a molar ratio of fructose: DMSO equal to 1:56. The catalyst could be re-used three times.
Similar content being viewed by others
Introduction
5-Hydroxymethyl-2-furfural (HMF), which can be obtained from the dehydration of sugars, has a high potential as renewable raw material for the production of a variety of important molecules containing or derived of the furan ring, including biofuels, solvents, drugs, and biopolymer monomers1,2,3. Thus, several studies have described the synthesis of HMF by catalytic dehydration of fructose4,5,6,7,8,9,10,11,12,13,14,15,16,17,18, Fig. 1. Glucose is less efficient than fructose for the synthesis of HMF19,20,21; according to Kuster22,23 and Zakrzewska et al.24, this results from the difference in stability of the two cyclical sugar structures, which are composed of six atoms in glucose (pyranose form) and five atoms in fructose (furanose form). The five-membered ring apparently facilitates the enolizations responsible for the generation of HMF. The main catalysts used for these conversions are mineral acids in aqueous solutions, such as H2SO4, HCl, or H3PO42,23. However, the utilization of these systems suffers from several drawbacks; apart from the generally non-selectivity of the processes due to degradation of HMF via rehydration and polymerization reactions, the use of mineral acids involves material corrosion, difficulties in the separation of the acid from the reaction mixture, and, of course, the high toxicity of the acids themselves8,25,26,27,28. Therefore, the development of more environmentally friendly and convenient solid acid catalysts to replace the liquid acid catalysts is highly desirable.
The use of inorganic acids25, ionic liquids24, Lewis acids, metallic chlorides, oxides, phosphates, heteropoly acid compounds, mesoporous solids3 and ion exchange resins29,30 have been used as green solid acid catalysts for the formation of HMF. Although H‐form zeolites could be employed as solid acid catalysts for the dehydration of fructose and the reaction can be highly selective (60–90%), their practical use in the conversion of fructose into HMF has been largely limited by the lower conversion efficiency31. Materials such as reticulated polystyrene containing sulfonic groups32,33 and another polymeric heterogeneous catalyst such as polyaniline24,34 have also been studied.
Apart from the catalyst, the choice of solvent for the dehydration reaction is also very important. The commonly applied solvents are water, methanol, DMSO and ionic liquids3,8,27,35; water/organic solvent biphasic systems are also important to increase the conversion rates and selectivity, but the current methods require large amounts of solvents because of the high solubility of HMF in water and the poor partitioning into the organic phase, which sometimes requires additional salting-out techniques for the separation of the product25. In particular, anhydrous DMSO has such a strong tendency to associate with water molecules that it has been used to force transformations such as 2 R-OH + DMSO → R-O-R + DMSO·H2O, and accordingly, d-fructose was converted to HMF in pure DMSO kept at 150 °C for 2 h36.
As a part of an ongoing research on the use of the SiO2–SO3H catalyst for clean synthesis37, we report herein the preparation of HMF from fructose with DMSO as the solvent and the hydrophilic SiO2–SO3H catalyst using microwave irradiation in processes under ambient atmosphere; MW irradiation has been widely used in the production of HMF17, and recently, in its oxidation to value added acid derivatives using solid catalysts containing Ru38 or Ag39.
Experimental
Raw materials and chemicals
All reagents (analytical grade), including dry DMSO and d-Fructose were supplied by Vetec, São Paulo, Brazil.
Instrumentation
HMF content and yield were determined with a GC/MS-QP 2010/AOC 5000 AUTO INJECTOR/Shimadzu Gas Chromatograph/Mass Spectrometer equipped with a 30 m Agilent J&W GC DB-5 MS column. Direct insertion spectra were measured at 70 eV. Quantitative analyses were performed on a Shimadzu GC-2010 gas chromatograph equipped with a flame ionization detector37. 1H- and 13C-NMR spectra were recorded on Bruker Avance 400 Spectrometers37. All reactions were monitored by TLC using Silica Gel 60 F 254 on aluminum. The chromatograms were visualized by UV light or by using the ethanolic vanillin developing agent37. The purification of the products was made by chromatographic column flash chromatography using a mixture of hexane/ethyl acetate in a 9/1 proportion as eluent37. The MW reactions were carried out in 10 mL G-10 vials of an Anton Paar single-mode MW synthesis reactor Monowave 300, powered by an 850 W magnetron, and equipped with temperature sensor and magnetic stirring.
Preparation of the silica gel and sulfonated silica (SiO2–SO3H)
The preparation of silica gel and the sulfonated silica SiO2–SO3H catalyst have been reported previously37.
Typical procedures
Dehydration of fructose using SiO2–SO3H as catalyst in DMSO
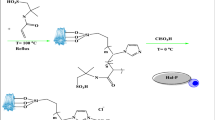
In a 10 mL microwave reactor vial, it was added 0.2604 mg (1.44 mmol) of D-fructose, 8.0 mL (8.80 g; 112.63 mmol) of DMSO and 0.0260 g (10% w/w in relation d-fructose) of SiO2–SO3H. The vial was heated in the microwave reactor at 150 °C for 10 min. The resulting dark liquid obtained after filtration of the solid catalyst was transferred to an extraction funnel and diluted with 30 mL of ethyl acetate and 30 mL of water. The lower fraction containing DMSO was removed and stored for future DMSO extraction and water purification, and the remaining organic solution was partitioned between 30 mL of ethyl acetate and 30 mL saturated NaCl, dried with magnesium sulfate, filtered and evaporated under reduced pressure. The resulting residue was subjected to a GC/MS analysis, which demonstrated the absence of unreacted d-fructose. The residue was then purified by flash column chromatography on silica, using hexane: ethyl acetate (2:1) as the mobile phase to yield HMF as a reddish-brown oil. Alternatively, the mixture of d-fructose, catalyst and DMSO were placed in an 100 mL round bottomed flask equipped with a condenser protected by a CaCl2 tube, and heated in a sand bath for 2 h; the treatment of the resulting dark liquid was identical to the described above.
Results and discussion
In 1983 Musau and Munavu described the synthesis of HMF in 92% yield when d-fructose was dehydrated using DMSO as both a dehydrating agent and as the solvent at 150 °C for 2 h. In their work the optimum conversion occurred at a d-Fructose: DMSO molar ratio of 1:8.5. Under those reaction conditions the condensation of HMF itself becomes important, and 1.2% of oxobis (5-methyl-2-furaldehyde) was also obtained36. Seeking to improve the efficiency of this reaction and reduce the reaction time, we inserted the SiO2–SO3H catalyst37 into the reaction mixture, using both conventional and microwave heating. Scheme 1 summarizes the conditions employed in the dehydration of d-fructose.
Microwave irradiation was used as an energy source for reaction activation because of its advantages over conventional heating methods14,15. As expected, conventional heating of the reaction mixture required longer reaction times and furnished lower yields than microwave heating, except for Entry 7 in Table 1, where no solvent was added to the mixture of catalyst and HMF; Table 1 resumes the principal results of the present work.
In Table 1, the first entry refers to the original work of Musau and Munavu, which used DMSO as the sole dehydrating agent, using conventional heating36. This work shows that, as expected, the use of MW irradiation speeds the overall process, and the excellent mark of 100% HMF conversion could be achieved in only 30 min, Entry 2.
The comparison between MW and conventional heating becomes more interesting when one considers the reactions involving the synergic dehydrating effects of the solvent DMSO and of the hydrophylic catalyst SiO2–SO3H, which had already been successfully used to dehydrate benzylic alcohol to dibenzyl ether37. Apparently, under conventional heating (Entry 3, Table 1) the combined effects are small, and the % results of the dehydration of fructose in the presence of the catalyst are comparable to Musau’s report. However, it is interesting to note that with the use of DMSO alone, Musau and Monavu observed the formation of 1.2% of the HMF condensation product (Entry 1), which was not observed in the same reaction performed in the presence of the sulfurous catalyst (Entry 3). It is possible that the catalyst increases the specificity of the DMSO towards the dehydration of the hexose.
Entry 4 in Table 1 suggests that the synergic DMSO/ SiO2–SO3H hexose dehydrating effect may be at its optimum level under MW irradiation, by showing that the excellent mark of 100% fructose conversion can be achieved again, but within only 10 min irradiation, with no products other than HMF being formed (by CG-MS analysis). This result compares very favorably with that of Watanabe et al.40, who found a very good 94% fructose conversion but a smaller 73% HMF yield, obtained by the catalytic dehydration of fructose in acetone–water mixtures in the presence of a sulfonic acid resin catalyst, at the same temperature of 150 °C, and under MW irradiation.
The specificity and dehydrating capacity of the DMSO/ SiO2-SO3H system was further tested increasing the contact times of the fructose conversion reactions, and Entries 5 and 6 in Table 1 show that condensation to oxobis(5-methyl-2-furaldehyde) indeed occurs after the hexose had been 100% converted to HMF. No other side-product was observed. The augment of the yield of the bis-furaldehyde ether from 1.2% (pure DMSO, conventional heating, ref.36) to 5% (DMSO/SiO2–SO3H/MW irradiation, this work) may indicate a possible, clean route for that interesting ether, but this route was not investigated further.
Conclusions
The use of microwave irradiation decreased fourfold the time required for the production of HMF by dehydration of fructose with DMSO at 150 °C and allowed 100% conversion and 100% selectivity. Addition of the hydrophylic catalyst SiO2–SO3H to that system reduced the reaction time a further threefold, maintaining the excellent mark of 100% fructose conversion and 100% selectivity to HMF. The catalytic system seems to be promising for the preparation of oxobis (5-methyl-2-furaldehyde) ether from HMF.
References
Takagaki, A. Production of 5-hydroxymethylfurfural from glucose in water by using transition metal-oxide nanosheet aggregates. Catalysts 9, 818 (2019).
Crisci, A. J., Tucker, M. H., Dumesic, J. A. & Scott, S. L. Bifunctional solid catalysts for the selective conversion of fructose to 5-hydroxymethylfurfural. Top. Catal. 53, 1185–1192 (2010).
van Putten, R.-J. et al. Hydroxy methyl furfural, A versatile platform chemical made from renewable resources. Chem. Rev. 113, 1499–1597 (2013).
Takagaki, A., Ohara, M., Nishimura, S. & Ebitani, K. A one-pot reaction for biorefinery: Combination of solid acid and base catalysts for direct production of 5-hydroxymethylfurfural from saccharides. Chem. Commun. 41, 6276–6278 (2009).
Dutta, S., De, S., Saha, B. & Alam, M. I. Advances in conversion of hemicellulosic biomass to furfural and upgrading to biofuels. Catal. Sci. Technol. 2, 2025–2036 (2012).
Luque, R. et al. Biodiesel as feasible petrol fuel replacement: A multidisciplinary overview. Energy Environ. Sci. 3, 1706–1721 (2010).
Tian, X. et al. Sulfonic acid-functionalized mesoporous carbon/silica as efficient catalyst for dehydration of fructose into 5-hydroxymethylfurfural. RSC Adv. 6, 101526–101534 (2016).
Román-Leshkov, Y., Chheda, J. N. & Dumesic, J. A. Phase modifiers promote efficient production of hydroxyl methyl furfural from fructose. Science 312, 1933–1937 (2006).
Asghari, F. S. & Yoshida, H. Acid-catalyzed production of 5-hydroxymethyl furfural from D-fructose in subcritical water. Ind. Eng. Chem. Res. 45, 2163–2173 (2006).
Bicker, M., Hirth, J. & Vogel, H. Dehydration of fructose to 5-hydroxymethylfurfural in sub- and supercritical acetone. Green Chem. 5, 280–284 (2003).
Bicker, M., Endres, S., Ott, L. & Vogel, H. Catalytical conversion of carbohydrates in subcritical water: A new chemical process for lactic acid production. J. Mol. Catal. A Chem. 239, 151–157 (2005).
Huber, G. W., Chheda, J. N., Barrett, C. J. & Dumesic, J. A. Production of liquid alkanes by aqueous-phase processing of biomass-derived carbohydrates. Science 308, 1446–1450 (2005).
Cottier, L. & Descotes, G. 5-Hydroxymethylfurfural syntheses and chemical transformations. Trends Heterocycl. Chem. 233, 233–248 (1991).
Lansalot-Matras, C. & Moreau, C. Dehydration of fructose into 5-hydroxymethylfurfural in the presence of ionic liquids. Catal. Commun. 4, 517–520 (2003).
Moreau, C., Durand, R., Rouxand, A. & Tichit, D. Isomerization of glucose into fructose in the presence of cation-exchanged zeolites and hydrotalcites. Appl. Catal. A 193, 257–264 (2000).
Zhao, H. B., Holladay, J. E., Brown, H. & Zhang, Z. C. Metal chlorides in ionic liquid solvents convert sugars to 5-hydroxymethylfurfural. Science 316, 1597–1600 (2007).
Delbecq, F. & Len, C. Recent Advances in the microwave-assisted production of hydroxymethyl furfural by hydrolysis of cellulose derivatives—A review. Molecules 23(8), 1973–1989 (2018).
Mouarrawis, V., Plessius, R., Ivar van der Vlugt, J. & Reek, J. N. H. Confinement effects in catalysis using well-defined materials and cages. Front. Chem. 6, 146–174 (2018).
Sidhpuria, K. B., Silva, A. L. D., Trindade, T. & Coutinho, J. A. P. Supported ionic liquid silica nanoparticles (SILnPs) as an efficient and recyclable heterogeneous catalyst for the dehydration of fructose to 5-hydroxymethylfurfural. Green Chem. 13, 340–349 (2011).
Moreau, C., Finiels, A. & Vanoye, L. Dehydration of fructose and sucrose into 5-hydroxymethylfurfural in the presence of 1-H-3-methyl imidazolium chloride acting both as solvent and catalyst. J. Mol. Catal. A Chem. 253, 165–169 (2006).
Hu, L. et al. Catalytic conversion of biomass-derived carbohydrates into fuels and chemicals via furanic aldehydes. RSC Adv. 2, 11184–11206 (2012).
Kuster, B. F. M. & Temmink, H. M. G. The influence of pH and weak-acid anions on the dehydration of D-fructose. Carbohyd. Res. 54, 185–191 (1977).
Kuster, B. F. M. 5-Hydroxymethylfurfural (HMF)—A review focusing on its manufacture. Starch 42, 314–321 (1990).
Zakrzewska, M. E., Bogel-Łukasik, E. & Bogel-Łukasik, R. Ionic liquid-mediated formation of 5-hydroxymethylfurfural—A promising biomass-derived building block. Chem. Rev. 111, 397–417 (2011).
Gomes, F. N. D. C., Pereira, L. R., Ribeiro, N. F. P. & Souza, M. M. V. M. Production of 5-hydroxymethylfurfural (HMF) via fructose dehydration: Effect of solvent and salting-out. Braz. J. Chem. Eng. 32, 119–126 (2015).
Corma, A., Iborra, S. & Velty, A. Chemical routes for the transformation of biomass into chemicals. Chem. Rev. 107, 2411–2502 (2007).
Chheda, J. N., Roman-Leshkov, Y. & Dumesic, J. A. Production of 5-hydroxymethylfurfural and furfural by dehydration of biomass-derived mono and poly-saccharides. Green Chem. 9, 342–350 (2007).
Roman-Leshkov, Y. & Dumesic, J. A. Solvent effects on fructose dehydration to 5-hydroxymethylfurfural in biphasic systems saturated with inorganic salts. Top. Catal. 52, 297–303 (2009).
Shimizu, K., Uozumi, R. & Satsuma, A. Enhanced production of hydroxymethylfurfural from fructose with solid acid catalysts by simple water removal methods. Catal. Commun. 10, 1849–1853 (2009).
Rosatella, A. A., Simeonov, S. P., Fradea, R. F. M. & Afonso, C. A. M. 5-Hydroxymethylfurfural (HMF) as a building block platform: Biological properties, synthesis and synthetic applications. Green Chem. 13, 754–793 (2011).
Ordomsky, V. V., van der Schaaf, J., Schouten, J. C. & Nijhuis, T. A. The effect of solvent addition on fructose dehydration to 5-hydroxymethylfurfural in biphasic system over zeolites. J. Catal. 287, 68–75 (2012).
Whitaker, M. R., Parulkar, A. & Brunelli, N. A. Selective production of 5-hydroxymethylfurfural from fructose in the presence of an acid-functionalized SBA-15 catalyst modified with a sulfoxide polymer. Mol. Syst. Des. Eng. Issue 5, 257–268 (2020).
Dai, J. et al. Sulfonated polyaniline as solid organocatalyst for dehydration of fructose into 5-hydroxymethylfurfural. Green Chem. 19, 1932–1939 (2017).
Cao, X. et al. Enzyme mimic ammonium polymer as a single catalyst for glucose dehydration to 5-hydroxymethylfurfural. Green Chem. 17, 2348–2352 (2015).
Boisen, A. et al. Process integration for the conversion of glucose to 2,5-furandicarboxylic acid. Chem. Eng. Res. Des. 87, 1318–1327 (2009).
Musau, R. M. & Munavu, R. M. The preparation of 5-hydroxymethyil-2-furaldehyde (HMF) from D-fructose in the presence of DMSO. Biomass 13, 67–74 (1987).
Barbosa, S. L. et al. Benzyl benzoate and dibenzyl ether from of benzoic acid and benzyl alcohol under microwave irradiation using a SiO2-SO3H catalyst. Catal. Commun. 68, 97–100 (2015).
Zhao, D. et al. microwave-assisted oxidation of hydroxymethyl furfural to added-value compounds over a ruthenium-based catalyst. ACS Sustain. Chem. Eng. 8, 3091–3102 (2020).
Zhao, D., Rodriguez-Padron, D., Luque, R. & Len, C. Insights into the selective oxidation of 5-hydroxymethylfurfural to 5-hydroxymethyl-2-furancarboxylic acid using silver oxide. ACS Sustain. Chem. Eng. 8, 8486–8495 (2020).
Qi, X., Watanabe, M., Aida, T. M. & Smith, R. L. Jr. Catalytic dehydration of fructose into 5-hydroxymethylfurfural by ion-exchange resin in mixed-aqueous system by microwave heating. Green Chem. 10, 799–805 (2008).
Acknowledgements
The authors acknowledge the support by the SENAI CIMATEC and PRPPG/UFVJM in response to Resolução 15/2019 (001).
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Barbosa, S.L., de S. Freitas, M., dos Santos, W.T.P. et al. Dehydration of d-fructose to 5-hydroxymethyl-2-furfural in DMSO using a hydrophilic sulfonated silica catalyst in a process promoted by microwave irradiation. Sci Rep 11, 1919 (2021). https://doi.org/10.1038/s41598-020-80285-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-80285-2
This article is cited by
-
Efficient and Highly Selective Production of 5-Hydroxymethylfurfural (HMF) from Fructose Using Hybrid Ce-Al Oxide Catalysts: Probing the Role of Calcination Temperature and Crystallinity of Catalyst
Catalysis Letters (2024)
-
Recent Advances in the Processing of Agri-food By-products by Subcritical Water
Food and Bioprocess Technology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.