Abstract
This study aimed to assess whether basal hormonal levels can predict the levels of progesterone (P4) on the day of oocyte retrieval (OR) and examine the impact of P4 levels on the day of OR on the outcome of assisted reproduction. One hundred sixty-four patients that were enrolled in the assisted reproduction procedure were classified according to their P4 levels on the OR day (< 2 ng/ml vs. ≥ 2 ng/ml). Patients who had P4 levels < 2 ng/ml had significantly higher follicle stimulating hormone (FSH) levels and significantly lower anti-Mullerian hormone (AMH) levels. More than half of patients with P4 levels < 2 ng/ml on the OR day got pregnant and delivered healthy infants. There was a significant correlation between lower FSH values and higher P4 values at OR and between higher AMH values and higher P4 values on the day of OR. Regression analysis showed that high FSH levels are the most important factor that can reliably imply lower P4 levels on OR day. Our study confirmed that lower basal FSH levels can predict the levels of P4 on the OR day. Moreover, lower levels of P4 on the day of OR are associated with a positive outcome in assisted reproduction.
Similar content being viewed by others
Introduction
Multiple studies have indicated that some patients have progesterone (P4) elevation on the day of human chorionic gonadotropin (hCG) administration during controlled ovarian stimulation (COS) of assisted reproductive technology (ART)1,2,3. Although the exact mechanism is unclear, this elevation has been shown to negatively impact the outcome of the ART procedure. Researchers believe that preovulatory P4 elevation can be caused by several mechanisms: accumulation of P4 production from multiple developing follicles during COS3, excessive amount of exogenous gonadotropins (GT)4, premature luteinization5, poor ovarian response with increased luteinizing hormone (LH) sensitivity3, total dose of administered GTs4,6, and duration of ovarian stimulation7.
Some studies have shown that premature P4 elevation is a cause of reduced pregnancy rates as it affects endometrial receptivity (i.e., endometrial maturation), leading to asynchrony between the endometrium and the embryo8, while other investigations have found that oocyte quality may be compromised by premature P4 elevation9,10. Despite effective suppression with gonadotropin-releasing hormone (GnRH) analogs, P4 elevation is reported in 5–53% of stimulated cycles3,4,5,6,7.
Several studies have examined the effect of P4 elevation on the day of hCG trigger in cycles with agonists and antagonists, but only a few studies have examined the effect of P4 levels on the day of oocyte retrieval (OR) on the outcome of ART1,2,3,11,12.
Previous studies on ART procedures have shown different relationships between steroid hormones and the outcome of the ART procedures. Basal hormonal status is generally determined before the commencement of ovarian stimulation. Levels of anti-Mullerian hormone (AMH), basal follicle stimulating hormone (FSH), and estradiol (E2) are widely used as a prognostic test for ovarian reserve, while basal P4 levels is used to predict the outcome of the ART procedure3,4,5,6,7.
The study aimed to determine P4 levels on the day of OR, 34–36 h post hCG administration, and investigate whether P4 levels at this time affect the outcome of the ART procedure and subsequently the pregnancy rate. Moreover, since FSH, E2, P4, and AMH are considered predictors of ovarian response and ART outcome and because little is known of the variables that could identify patients at risk of P4 elevation, this study also aimed to determine whether basal hormonal levels can predict the levels of P4 on the day of OR.
Results
Patients’ characteristics and basal hormonal levels according to P4 levels
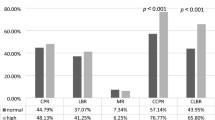
The study evaluated 164 patients undergoing an ART procedure (OR and embryo transfer) in our clinic. Out of them 44.5% were in the P4-low group (< 2 ng/ml) and 55.5% in the P4-high group (≥ 2 ng/ml). The baseline characteristics of the patients are shown in Table 1. Patients on average were 34.66 ± 3.39 years old, the mean BMI was 22.23 ± 2.65, and their infertility generally lasted for 4.86 ± 2.61 years (range 1 to 17 years). The mean P4 values on OR day were 1.26 ± 0.46 ng/ml in the P4-low group, while in the P4-high group the average P4 was 3.74 ± 1.62 ng/ml. No significant differences were found in LH, P4, and E2 levels or in GT dose regarding the P4 values on the day of OR, while patients from the P4-low group had significantly higher FSH levels and significantly lower AMH levels (Table 1).
Clinical features and procedure outcome depending on P4 level
In the overall sample, the most common causes of infertility were unknown factors (30.5%) and the male factor (28%). In the P4-high group of patients, the most common cause of infertility was the male factor, while the ovarian factor was the most frequent cause in patients from the P4-low group. Nevertheless, there was no significant difference in the cause of infertility regarding P4 levels on the day of OR (Table 2). The average number of all retrieved oocytes, as well as the number of mature oocytes and fertilized oocytes, was significantly higher in the P4-high group. In contrast, the average rate of fertilization was significantly lower in the P4-high group. Although the embryos were mostly of grade A quality in the P4-high group on the OR day, the difference in embryo quality was not significant between P4 level groups. Moreover, there were no significant differences in the number of transferred embryos regarding the two groups of patients (Table 2).
Patients on the short stimulation protocol had significantly lower levels of P4 on the OR day than those on a long stimulation protocol. It was noticed that patients from the P4-high group underwent more often a long stimulation agonist protocol (Table 2). The odds for procedure success of subjects from the P4-low group stimulated with a short protocol was almost three times higher than those stimulated with a long protocol (OR = 2.17; 95% CI 1.06–4.42). Patients undergoing IVF or ICSI techniques had lower levels of P4 on the OR day compared to those who underwent the combined technique of insermination (Table 2). We found a statistically significant difference in the levels of P4 on the day of OR in relation to the positive outcome of the ART procedure as well as regarding the outcome of pregnancy. More than half of the patients from the P4-low group on the OR day got pregnant after ART procedure. In addition, 80.7% of all investigated women successfully delivered an infant (Table 2).
Correlation of P4 on the OR day with patient and procedure characteristics, basal hormonal status, and regression analysis
By examining the association between P4 and basal hormone levels, a statistically significant correlation was found between lower FSH values and higher P4 values on OR day both directly (i.e., continuously) and with P4 ≥ 2 ng/ml on the OR day (Table 3). Furthermore, higher AMH values were significantly correlated with higher P4 values on the day of OR. Higher P4 values on OR day were also correlated with long stimulation protocol and the use of the IVF/ICSI combined technique (Table 3).
Univariant regression analysis suggested that basal FSH level could significantly predict P4 levels, as well as combined ART technique and stimulation protocol. Furthermore, preformed multivariate regression analysis showed that high FSH levels on OR day are the most important factor for reliably imply on lower P4 levels. The regression analysis is presented in Table 4.
Discussion
The impact of preovulatory P4 rise on the outcome of ART procedures remains controversial. While some studies have not shown a negative effect on ART outcome11,12, other studies associated unsuccessful ART with elevation of P4 levels1,2,3. Possible explanations for the discrepancies in the findings are the use of retrospective study design, the use of different protocols of controlled ovarian stimulation, and different cutoff levels for P4at the time of data analysis. Some studies imprecisely define references for elevated serum P4 levels, and there is variation in the statistical methods used to estimate specific circulating P4 limit values and in the precision of P4measurements that use different immunoassays12,13. The cutoff of P4 absolute concentrations on the day of hCG administration generally ranges from 0.8 to 2 ng/ml14,15,16. However, in recently published studies, it has often been adjusted to 1.5 ng/mL17. Based on the ROC analysis we performed, 2 ng/mL was confirmed as the P4 cutoff level for achieving pregnancy. Therefore, this P4 level was used for all further analyses.
Cycles using GnRH agonists (GnRHa) have been the most studied, while the effect of P4 levels on the day of hCG trigger or at other critical points during stimulation with GnRH antagonists (GnRHant) has been less examined. An increase in P4 levels was observed in GnRHa protocols despite down-regulation of the pituitary gland, while slightly lower P4 levels in patients treated with GnRHant were explained by lower steroidogenic activity of granulose cells or half-life of the antagonist (about 20 h), which leads to rapid recovery of pituitary suppression5,18.
In this study, a short protocol with GnRH antagonists and a long protocol with GnRH agonists were used, and although the number of patients with a long stimulation protocol was smaller, we found that an elevation in P4 levels on the day of OR was more frequent in the long stimulation protocol. Our findings can also be explained to some extent by the fact that the long protocol was more commonly used in younger patients where we received more oocytes. Moreover, although we found that higher P4 levels on the day of OR were more prevalent in the younger patients, these findings were not significant. This is also consistent with the findings of a meta-regression analysis by Venetis et al.19, which showed that the protocol with GnRH antagonists was associated with lower rates of P4 rise compared to protocols with GnRH agonists, independent of the P4 threshold. However, a study by Huang et al.2, which examined elevated P4 in both the long and short protocols, showed that P4 levels were higher in the short protocol than in the long stimulation protocol. They explained these differences in the findings based on the fact that the characteristics of patients in the short and long protocol differed; more poor responders were on the short stimulation protocol, the cycle characteristics of the two protocols differed, and the number of patients on the short protocol was higher, which could have been confusing2.
Some studies have not found an adverse effect of P4 levels on the outcome of ART procedures and on pregnancy rates12,15; however, there are several publications that have found a significant negative association between P4 levels on the hCG trigger day and the success of ART procedures4,20,21. In a study of 2566 patients undergoing the ART procedure, Huang et al.2 found a negative correlation between elevated P4 and live birth rate (the cutoff value was 1.2 ng/ml in long protocols and 2.0 ng/ml in short protocols). Another meta-analysis by Venetis et al.19 showed that P4 elevation on the day of hCG administration was associated with significantly reduced pregnancy rates after a fresh embryo transfer. A recent publication showed that the live birth rates were significantly lower in patients with both low and high P4levels on the day of hCG administration22.
All of these studies measured P4 levels on the day of hCG administration, but a study by Niu et al.23 examined P4 levels on the day of OR in cycles with GnRH agonists (long and short protocol) and found that P4 levels correlated with the number of oocytes and embryos, but can’t predict the outcome of the pregnancy. In a prospective study of 186 women, Nayak et al.24 examined the prediction of ART success depending on P4 level on the day of OR in patients on a short stimulation protocol with GnRHant. There was no correlation of basal P4 andP4 levels on the day of antagonist initiation with P4 on the day of hCG administration. Patients with a P4 rise on the day of OR had lower pregnancy and implantation rates in their study24. In this study, a statistically significant difference in the level of P4 on the day of OR compared to pregnancy rates was found. Patients with P4 levels < 2 ng/ml had higher pregnancy rates. In terms of pregnancy outcome, there were fewer miscarriages in this group and the delivery rate was higher. This study demonstrates the predictive effect of lower P4 values on OR day and the positive outcome of the ART procedure.
In terms of basal hormones, we found lower AMH levels in women with lower P4 (P4 < 2 ng/ml) on OR day as well as a direct correlation between these parameters. However, AMH has not been shown to predict P4 rise on the day of oocyte aspiration. Our finding is consistent with a recent study by Kavoussi et al.25, who also did not find a predictive value of AMH in P4 rise. Studies have shown that AMH can predict the response to ovarian stimulation as well as the number of achieved oocytes and embryos; however, its correlation with P4 rise has not been extensively studied25.
Studies have shown that the level of basal P4 can influence the outcome of the ART procedure26,27. The association of basal P4 with P4 elevation on the day of hCG was also examined previously28. Although this study did not confirm the adequate predictive value of basal P4 on P4 rise on the day of OR, patients with lower P4 on OR day had slightly lower basal P4 values, while patients with higher P4 on OR day (≥ 2 ng/ml) had higher basal P4 levels but not in a statistically significant amount. Venetis et al. showed that ART cycles with higher basal P4 concentrations are more likely to show P4 rise after stimulation19. Such a finding could indicate that a jump in P4 on hCG administration days may not be related to stimulation alone; it may be adrenal29,30,31. We also did not find a correlation with GT dose, so we recommend that further studies regarding the progesterone source are needed.
During the menstrual cycle, FSH and LH promote steroid biosynthesis among which is P4 biosynthesis. As FSH and LH act on granulose cells, they, as well as theca cells, produce P49. We found that FSH is a predictor for P4 levels. Huang et al.32 found that patients that are younger have lower FSH levels and a longer duration of P4 elevation, so prolonged duration of P4 elevation may occur easily in patients with a better ovarian reserve32. In another study that investigated the association of basal levels of E2 and FSH with elevated P4, authors Park et al.33 found that this association was not significant.
Different characteristics, such as age and weight, are closely associated with the hormonal status of women and therefore impact fertility34,35. However, in our study, none of the tested socio-demographic parameters were confirmed as factors that could be used for prediction of P4 levels on the OR day. These findings are in accordance with other performed multivariate analyses in which, after adjusting for female age, body mass index, smoking status, and previous parity, only sperm quality and P4 levels were significant predictors of ART outcomes36,37.
The key finding of our study was that high basal FSH levels can be used as predictors of low P4 levels on the OR day, which is necessary to improve the chances of successful ART. Consequently, our findings are useful when planning ART in clinical practice in general. In the case of either inadequately low basal FSH or high P4 on the OR day, to achieve better fertility rates, the embryo can be frozen and another embryo transfer can be planned for one of the following cycles.
There are some limitations to our study. Firstly, the number of patients in the study could have been larger. Furthermore, the sample sizes of the long and short protocols vary, as there were more patients in the short GnRH antagonist protocol. However, the study included all patients who had the ART procedure at the clinic during a one-year period. Also, as in all centers/clinics, the value and confidence intervals vary with different P4assay kits.
In conclusion, this study found a significant correlation between the levels of P4 on the day of OR and a positive outcome for the ART procedure. Patients with lower levels of P4 (< 2 ng/ml) were more likely to both achieve pregnancy and deliver a vital infant. By examining the association between P4 and basal hormone levels, a significant positive correlation was found between higher AMH values and higher P4 values at OR day, and an inverse correlation was found between lower FSH values and higher P4 values. High basal FSH levels were found to be an important predictor of lower P4 levels (< 2 ng/ml) on OR day.
Materials and methods
Patients
A prospective observational study was conducted at the Clinic for Gynecology and Obstetrics Clinical Center of Serbia from January 1st to December 31st, 2015. The study included all patients that were enrolled in the ART procedure at the Clinic during the study period and that fulfilled the inclusion criteria. The inclusion criteria included being 18 to 40 years old and independent (not having a legal guardian); having a BMI of 18 to 30 kg/m2, a regular menstrual cycle (25 to 32 days), an established diagnosis of infertility and agreeing to participate in the study. All types of female infertility patients were included in the study. The exclusion criteria included azoospermic male patients or female patients that do not fulfill the inclusion criteria. Infertility was diagnosed and classified according to European Society of Human Reproduction and Embryology (ESHRE) guidelines38. For all patients finally included in the study sample socio-demographic data as well as a thorough medical history were taken.
All patients were thoroughly informed about the research and all booked ART procedures and signed the informed consents for inclusion in the study as well as the ART itself. All procedures were carried out in accordance with relevant guidelines and regulations. The recruitment of patients was performed in accordance with the Helsinki declaration. The research was approved by the Ethics Committee of the Faculty of Medicine in Belgrade (25138). ClinicalTrials.gov Identifier: NCT04447677, date of registration 25.06.2020.
Protocols of stimulation
A detailed explanation of the stimulation protocol and patient monitoring were presented in a previous study13. Briefly, patients on a long stimulation protocol with GnRH agonists received Triptoreline (Diphereline, Ipsen Pharma Biotech, France) starting in the mid-luteal phase of the cycle prior to the study. Following pituitary suppression, 2–3 days of the next cycle, ovarian stimulation with gonadotropins (GTs)—recombined FSH (Gonal-F, Serono, Switzerland) was initiated depending on patient age, ovarian reserve, and ovarian response in the previous procedures. Patients on a short stimulation protocol with GnRH antagonists received the same stimulation starting on the 2nd or 3rd day of the cycle. GnRH antagonists (Cetrorelix [Cetrotide, Merk Serono, Germany]) were added when the leading follicle reached a diameter of 14 mm and were administered until the day of hCG trigger. In both COS protocols the ovarian response was followed by sequential transvaginal ultrasound examinations and determination of estradiol levels in the blood. When two or more follicles were > 18 mm in diameter, hCG (Pregnyl, Organon, The Netherlands) was administered.
ART procedure
The insemination methods were in vitro fertilization (IVF), intracytoplasmic sperm injection (ICSI), or the combined method. Whether fertilization occurred or not was ascertained 16–20 h after insemination by the presence of two pronuclei. In assessing obtained embryo quality, the criteria of the Istanbul Consensus of Clinical Embryologists were used. The criteria include evaluation of the degree of fragmentation and symmetry of the blastomere. Embryos are divided into class A (solid, with 1–10% or without fragmentation, perfect symmetry), class B (medium, 11–25% fragmentation, moderate asymmetry), class C (poor, > 25% fragmentation, pronounced asymmetry)39. All evaluations were performed together by our team of three embryologists. Finally, on the second or third day after OR, an embryo transfer was performed under the control of a transabdominal ultrasound. According to current recommendations, we transferred 1–3 embryos in all patients regardless of the P4 group (Table 2). The luteal phase was supported with P4 depo intramuscular administration.
Hormone measurements
We measured basal serum levels of estradiol (E2), progesterone (P4), follicle-stimulating hormone (FSH), luteinizing hormone (LH), and anti-Mullerian hormone (AMH) for all patients on the 2nd or 3rd day of their cycle, which we always do before stimulation commencement according to classic protocols. The referral range in our laboratory for basal FSH was 3.5 to 12.5 IU/L, for LH 2.4 to 12.6 IU/L, for E2 0 to 160 pg/mL, and for P4 0 to 1.14 ng/mL, while for AMH it was 0.19 to 9.13 ng/ml. Levels of P4 were also measured on the day of OR. Blood samples were taken using Vacutainer tubes (BD Vacutainer Systems) and centrifuged. The measurement methods are explained in our previous study13. Briefly, the AMH value in the serum was measured using enzyme-linked immunosorbent assay (ELISA), and other hormones (FSH, LH, E2, and P4) were analyzed using chemiluminescent immunoassay. Intra- and inter-assay precision at concentrations most relevant to the study were 5.9% and 4.7% for FSH, 7.3% and 4.5% for LH, 5.2% and 3.2% for E2, and 4.1% and 2.3% for P4. In our study, we used the Receiver Operating Characteristics (ROC) analysis to estimate the effect of P4 levels on achieving clinical pregnancy. We determined that a cutoff of 2 ng/mL represented the value where the event frequency changes abruptly (sensitivity = 47%; specificity = 69%) (Fig. 1). The obtained cutoff was in accordance with previous literature data (14–16). Therefore, the level of P4 on the OR day was assessed as continuous as well as low (< 2 ng/ml) and high/elevated (≥ 2 ng/ml). Consequently, patients were classified into two groups depending on their P4 levels on the OR day: 1) low P4 (< 2 ng/ml) and 2) P4-high (≥ 2 ng/ml).
Outcome measures
The main outcome measures included procedure success and pregnancy outcome. Pregnancy was confirmed by a positive finding of serum β-hCG (> 25 MIU/ml) 14 days after embryo transfer. Clinical pregnancies were confirmed by transvaginal ultrasound findings of a gestational sac with a vital embryo at the 6th gestational week. All patients with confirmed pregnancy were regularly followed-up according to current protocols throughout the pregnancy. Based on the gestational week of the pregnancy, end-of-pregnancy outcomes were classified as either miscarriage or delivery (live born infant ≥ 24 weeks of gestation).
Statistical analysis
Results were presented as a mean ± standard deviation for variables with normal distribution and as a median and interquartile range for variables for which the distribution was not normal. Categorical variables are presented as absolute and relative frequencies. Receiver Operating Characteristics (ROC) analysis was applied to determine the P4 cutoff level that would be used for classification of patient groups (low and high P4). Comparison of the mean values of independent groups of data was performed using ANOVA. For parameters without normal distribution, testing of the significance between groups was performed using the Kruskal–Wallis test. Correlation analysis (Pearson/Spearman) was used to investigate the association of patient and procedure characteristics with P4 levels on OR day. The potential predictive value of tested variables on P4 on OR day was assessed using Univariate and Multivariate logistic regression analyses. A significance of 0.05 was required for a variable to be included in the multivariate model, whereas 0.1 was the cutoff value for exclusion. Odd ratios with the corresponding 95% confidence intervals were estimated. All analysis was performed using the Statistical Package for the Social Sciences 22.0 (SPSS, Inc, Chicago, IL), and differences were considered statistically significant at a probability level of less than 0.05.
Abbreviations
- IVF:
-
In vitro fertilization
- ART:
-
Assisted reproductive technologies
- OR:
-
Oocyte retrieval
- ICSI:
-
Intracytoplasmic sperm injection
- BMI:
-
Body mass index
- GnRH:
-
Gonadotropin-releasing hormone
- GT:
-
Gonadotropin
- COS:
-
Controlled ovarian stimulation
- LH:
-
Luteinizing hormone
- FSH:
-
Follicle stimulating hormone
- E2:
-
Estradiol
- P4:
-
Progesterone
References
Al-Azemi, M. et al. Elevated progesterone during ovarian stimulation for IVF. Reprod Biomed. Online 24, 381–388 (2012).
Huang, R., Fang, C., Xu, S., Yi, Y. & Liang, Y. Premature progesterone rise negatively correlated with live birth rate in IVF cycles with GnRH agonist: An analysis of 2566 cycles. Fertil. Steril. 98, 664–670 (2012).
Kilicdag, E. B., Haydardedeoglu, B., Cok, T., Hacivelioglu, S. O. & Bagis, T. Premature progesterone elevation impairs implantation and live birth rates in GnRH-agonist IVF/ICSI cycles. Arch. Gynecol. Obstet. 281, 747–752 (2010).
Bosch, E. et al. Circulating progesterone levels and ongoing pregnancy rates in controlled ovarian stimulation cycles for in vitro fertilization: Analysis of over 4000 cycles. Hum. Reprod. 25, 2092–2100 (2010).
Hugues, J. N. et al. Impact of endogenous luteinizing hormone serum levels on progesterone elevation on the day of human chorionic gonadotropin administration. Fertil. Steril. 96, 600–604 (2011).
Younis, J. S., Matilsky, M., Radin, O. & Ben-Ami, M. Increased progesterone/estradiol ratio in the late follicular phase could be related to low ovarian reserve in in vitro fertilization–embryo transfer cycles with a long gonadotropinreleasing hormone agonist. Fertil Steril. 76, 294–299 (2001).
Bosch, E. et al. Premature luteinization during gonadotropin-releasing hormone antagonist cycles and its relationship with in vitro fertilization outcome. Fertil. Steril. 80, 1444–1449 (2003).
Papanikolaou, E. G. et al. Progesterone rise on the day of human chorionic gonadotropin administration impairs pregnancy outcome in day 3 single-embryo transfer, while has no effect on day 5 single blastocyst transfer. Fertil. Steril. 91, 949–952 (2009).
Long, X., Pen, C. & Lu, G. Isolation and identification of genes differentially expressed in premature luteinization granulosa cell during controlled ovarian hyperstimulation. Fertil. Steril. 92, 1767–1771 (2009).
Yovel, I. et al. High progesterone levels adversely affect embryo quality and pregnancy rates in in vitro fertilization and oocyte donation programs. Fertil. Steril. 64, 128–131 (1995).
Andersen, C. Y., Bungum, L., Andersen, A. N. & Humaidan, P. Preovulatory progesterone concentration associates significantly to follicle number and LH concentration but not pregnancy rate. Reprod. Biomed. Online. 23, 187–195 (2011).
Venetis, C. A. et al. Is progesterone elevation on the day of human chorionic gonadotrophin administration associated with the probability of pregnancy in in vitro feritlization? A systematic review and meta-analysis. Hum. Reprod. Update. 13, 343–355 (2011).
Tulic, L. et al. Oxidative stress markers in GnRH agonist and antagonist protocols in IVF. J. Med. Biochem. 36, 1–8 (2017).
Edelstein, M. C. et al. Progesterone levels on the day of human chorionic gonadotropin administration in cycles with gonadotropin-releasing hormone agonist suppression are not predictive of pregnancy outcome. Fertil. Steril. 54, 853–857 (1990).
Ubaldi, F. et al. Subtle progesterone rise after the administration of the gonadotrophin-releasing hormone antagonist cetrorelix in intracytoplasmic sperm injection cycles. Hum. Reprod. 1, 1405–1407 (1996).
Givens, C. R., Schriock, E. D., Dandekar, P. V. & Martin, M. C. Elevated serum progesterone levels on the day of human chorionic gonadotropin administration do not predict outcome in assisted reproduction cycles. Fertil. Steril. 62, 1011–1017 (1994).
Van Vaerenbergh, I. et al. Progesterone rise on HCG day in GnRH antagonist/rFSH stimulated cycles affects endometrial gene expression. Reprod. Biomed Online. 22, 263–271 (2011).
The Ganirelix Dose-finding Study Group. A double-blind, randomized, dose-finding study to assess the efficacy of the gonadotrophin-releasing hormone antagonist ganirelix (Org 37462) to prevent premature luteinizing hormone surges in women undergoing ovarian stimulation with recombinant follicle stimulating hormone (Puregon). Hum. Reprod. 13, 3023–3031 (1998).
Venetis, C. A., Kolibianakis, J. K., Bosdou, J. K. & Tarlatzis, B. B. Progesterone elevation and probability of pregnancy after IVF: A systematic review and meta-analysis of over 60000 cycles. Hum. Reprod. Update. 19, 433–457 (2013).
Wu, Z. et al. Progesterone elevation on the day of hCG trigger has detrimental effect on live birth rate in low and intermediate ovarian responders, but not in high responders. Sci. Rep. 9, 5127. https://doi.org/10.1038/s41598-019-41499-1 (2019).
Kolibianakis, E. M., Venetis, C. A., Bontis, J. & Tarlatzis, B. C. Significantly lower pregnancy rates in the presence of progesterone elevation in patients treated with GnRH antagonists and gonadotrophins: A systematic review and meta-analysis. Curr. Pharm. Biotechnol. 13, 464–470 (2012).
Arvis, P., Lehert, P. & Guivarc’h-Levêque, A. Both high and low HCG day progesterone concentrations negatively affect live birth rates in IVF/ICSI cycles. Reprod. Biomed Online. 39, 852–859 (2019).
Niu, Z., Yun, F., Aijun, Z., Yijuan, S. & Huiqin, Z. Progesterone levels on oocyte retrieval day can predict the quantity of viable embryos but not pregnancy outcome of intracytoplasmic sperm injection. Gynecol. Endocrinol. 24, 452–458 (2008).
Nayak, S. et al. Progesterone level at oocyte retrieval predicts in vitro fertilizatiojn success in short–antagonist protocol: A prospective cohort study. Fertil. Steril. 101, 676–682 (2014).
Kavoussi, S. K. et al. Serum Antimüllerian hormone does not predict elevated progesterone levels among women who undergo controlled ovarian hyperstimulation for in vitro fertilization. Reprod. Biol. Endocrinol. 17, 35. https://doi.org/10.1186/s12958-019-0477-8 (2019).
Kolibianakis, E. M. et al. Elevated progesterone at initiation of stimulation is associated with a lower ongoing pregnancy rate after IVF using GnRH antagonists. Hum. Reprod. 19, 1525–1529 (2004).
Hadmine, O. et al. Elevated early follicular progesterone levels and in vitro fertilization outcomes: A prospective intervention study and meta-analysis. Fertil. Steril. 102, 448–454 (2014).
Venetis, C. A. et al. Basal serum progesterone and history of elevated progesterone on the day of hCG administration are significant predictors of late follicular progesterone elevation in GnRH antagonist IVF cycles. Hum. Reprod. 31, 1859–1865 (2016).
Eldar Geva, T. et al. The origin of serum progesterone during the follicular phase of menotropin-stimulated cycle. Hum. Reprod. 13, 9–14 (1998).
Fanchin, R. et al. Premature plasma progesterone and androgen elevation are not prevented by adrenal suppression in vitro fertilization. Fertil. Steril. 67, 115–119 (1997).
De Geyter, C., De Geyte, R. M. & Huber, P. R. Progesterone serum levels during the follicular phase of the menstrual cycle originate from the crosstalk between the ovaries and the adrenal corte. Hum. Reprod. 17, 933–939 (2002).
Huang, C. C. et al. The duration of pre-ovulatory serum progesterone elevation before HCG administration affects the outcome of IVF/ICSI cycles. Hum. Reprod. 27, 2036–2045 (2012).
Park, J. H., Jee, B. C. & Kim, S. H. Factors influencing serum progesterone level on triggering day in stimulated in vitro fertilization cycles. Clin. Exp. Reprod. Med. 42, 67–71 (2015).
Leeners, B., Geary, N., Tobler, P. N. & Asarian, L. Ovarian hormones and obesity. Hum. Reprod. Update. 23, 300–321 (2017).
Steiner, A. Z. et al. Association between biomarkers of ovarian reserve and infertility among older women of reproductive age. JAMA 318, 1367–1376 (2017).
Boynukalin, F. K. et al. Measuring the serum progesterone level on the day of transfer can be an additional tool to maximize ongoing pregnancies in single euploid frozen blastocyst transfers. Reprod. Biol. Endocrinol. 17, 102. https://doi.org/10.1186/s12958-019-0549-9 (2019).
Vanni, V. S. et al. Top quality blastocyst formation rates in relation to progesterone levels on the day of oocyte maturation in GnRH antagonist IVF/ICSI cycles. PLoS ONE 12, e0176482. https://doi.org/10.1371/journal.pone.0176482 (2017).
ESHRE—European Society of Human Reproduction and Embryology. https://www.eshre.eu.
Alpha Scientists in Reproductive Medicine and ESHRE Special Interest Group of embryology. The Istanbul Consensus workshop of embryo assessment: Proceedings of an expert meeting. Hum Reprod. 26, 1270–1283 (2011).
Author information
Authors and Affiliations
Contributions
T.L. and T.I. conceived the experiment, T.L., B.J, L.S.M and K.I conducted the experiment, N.LJ. was in charge for biochemistry. D.J analysed the results. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tulic, L., Tulic, I., Bila, J. et al. Correlation of progesterone levels on the day of oocyte retrieval with basal hormonal status and the outcome of ART. Sci Rep 10, 22291 (2020). https://doi.org/10.1038/s41598-020-79347-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-79347-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.