Abstract
Chronic low-grade inflammation plays a role in the pathogenesis of several chronic diseases including cancer. Physical activity (PA) and diet have been supposed to modulate inflammatory markers. We evaluated the effects of a 24-month dietary and/or PA intervention on plasma levels of pro-inflammatory cytokines, a secondary analysis in the DAMA factorial trial. The 234 study participants (healthy postmenopausal women with high breast density, 50–69 years, non smokers, no hormone therapy) were randomised to four arms: (1) isocaloric dietary intervention mainly based on plant-foods; (2) moderate-intensity PA intervention with at least 1 h/week of supervised strenuous activity; (3) both interventions; (4) general recommendations on healthy dietary and PA patterns. Interleukins (IL)-1α, -1β, -6, tumor necrosis factor-α and C-reactive protein were measured at baseline and at the end of the intervention. Intention-to-treat-analyses were carried out using Tobit regression. Although all cytokines tended to increase over time, after 24 months women in the PA intervention (arms 2 + 3) showed lower levels of IL-1α (exp(β) = 0.66; p = 0.04) and IL-6 (exp(β) = 0.70; p = 0.01) in comparison with women in the control group (arms 1 + 4). No effects of the dietary intervention emerged. In healthy postmenopausal women with high breast density a moderate-intensity PA appears to slow the age-related increase of pro-inflammatory cytokines.
Similar content being viewed by others
Introduction
It is widely accepted that chronic low-grade inflammation may play a role in a wide range of age related diseases including cardiovascular diseases, type 2 diabetes and cancer1,2.
A higher level of inflammation, associated to the decline in circulating oestrogens, has been documented among postmenopausal women3,4,5. Also obesity and abdominal adiposity have been associated with a pro-inflammatory profile6.
Cytokines measured as circulating markers of chronic low-grade inflammation include: interleukin 1 (IL-1), interleukin 6 (IL-6), tumor necrosis factor alpha (TNF-α), and C-reactive protein (CRP). IL-6, a multifunctional cytokine involved in the regulation of immune responses, acute-phase responses, ematopoiesis, and inflammation, is one of the most studied cytokines in relation to aging and the development of chronic diseases7,8. TNF-α plays crucial roles in the initiation of the inflammatory cascade9. CRP is a sensitive systemic biomarker of inflammation and tissue damage mainly produced by hepatocytes, under transcriptional control by IL-610,11. IL-1 is made up of two major proteins, IL-1α and IL-1β with similar biological properties of coordination of the innate immune and inflammatory responses12. Several studies support the association of a diet rich in vegetables, fruit, nuts, whole grains and unsaturated fatty acids, and of a Mediterranean Diet pattern, with lower levels of circulating pro-inflammatory cytokines13,14,15,16,17. An anti-inflammatory effect of physical exercise and the association between sedentary habits and chronic low-grade inflammation have been shown18,19,20. A modulation of circulating markers of inflammation through modification of dietary21,22,23 and physical activity (PA) habits21,24,25,26, partially mediated by weight reduction, has been reported.
The DAMA (Diet, physical Activity and MAmmography) study is a factorial randomised trial aimed at investigating whether a dietary and/or a PA intervention were able to reduce mammographic breast density (BD) among postmenopausal women with high BD27 and therefore at higher breast cancer risk28. The results of the DAMA trial on BD, the primary outcome of the trial, have been reported recently29. Here we report the effects of the dietary and PA interventions on plasma levels of five pro-inflammatory cytokines (IL-1α, IL-1β, IL-6, TNF-α, and CRP), as a secondary outcome of the trial.
Materials and methods
The DAMA study (trial registration number: ISRCTN28492718, date of trial registration 17/05/2012) is a single-centre randomised intervention trial with a 2 × 2 factorial design. The trial was approved by the Ethics Committee of the Local Health Authority “Azienda Sanitaria Firenze”, the research was performed in accordance with relevant guidelines/regulations and an informed consent was obtained from all participants.
The study protocol of the DAMA trial, including the objectives of the interventions and the activities performed, was reported in detail in a previous paper27.
Selection of study participants
Participants were recruited between 2009 and 2010 among postmenopausal women, aged 50–69 years, with a negative mammogram performed in the local screening programme showing a BD > 50% according to the Breast Imaging Reporting and Data System (BI-RADS) classification30, no recent (past 12 months) hormone therapy use, never smokers (or former smokers for more than 6 months), without a previous diagnosis of cancer, diabetes, major cardiovascular and neurological diseases or other diseases able to hamper their active participation in the study.
Baseline and follow-up visits
We invited participants for a visit at baseline and 24 (± 3) months after their enrolment into the study. In both visits trained personnel measured weight, height, hip and waist circumference and collected information on dietary and lifestyle habits through validated questionnaires previously used in the frame of the European Prospective Investigation into Cancer and Nutrition (EPIC) study: the Italian Food Frequency Questionnaire31, and the EPIC Lifestyle Questionnaire including a section on PA at work and during leisure time32. A fasting venous blood sample was taken at baseline and at follow-up visit. Within 24 h after collection, blood samples were processed into plasma, red cells and buffy-coat aliquots and stored in the liquid nitrogen biological bank of the project.
Random assignment
After the baseline visit we randomised each woman to one of the four arms of the study through permuted-block randomisation stratified by age (50–59/60–69 years) and Body Mass Index (BMI) category (< 25/ ≥ 25 kg/m2), with a constant block size (n = 4).
Dietary intervention (arm #1)
The aim of the dietary intervention was to change the composition of diet, in an isocaloric context (without specific advice on the quantity of food to be consumed), adopting a diet mainly based on plant food, with a low glycemic load, low in saturated- and trans-fats and alcohol and rich in antioxidants. The intervention objectives were: (a) substitution of refined grains with whole-grains; (b) at least two portions of vegetables at each meal; (c) consumption of fish at least two–three times/week; (d) fresh and processed red meat less than one time/week; (e) at least three-four portions/week of legumes and pulses; (f) two–three portions/day of fruit; (g) a maximum of one portion/week of cakes or desserts; (h) no more than one portion/day of milk or yogurt and two portions/week of cheese; (i) extra-virgin olive oil as the only dressing and cooking fat; (j) no more than one glass of wine per day.
Participants were invited to attend six group meetings and eight cooking classes during the two years of the study.
Physical activity intervention (arm #2)
The aim of PA intervention was to increase moderate daily recreational activities, corresponding to about three MET-hours/day (MET = Metabolic Equivalent), up to one hour/day. Suggested daily activities were walking at moderate pace, biking, etc. Women were also requested to perform at least one hour/week of more strenuous activity (6–10 MET). To achieve this latter objective women were requested to attend weekly one-hour sessions led by qualified personnel in an appropriate fitness facility (97 sessions over the 24-month period). Equipment and schemes for home exercises were also provided. Women were requested to attend six group meetings and six collective walks supervised by the study team.
Dietary + Physical activity intervention (arm #3)
Participants were asked to combine arms #1 and #2 activities. To reduce possible contamination, women in this arm had separate dietary and PA sessions from the women randomised to arms #1 or #2, respectively.
Control arm (arm #4)
Participants received general advice and printed material on healthy dietary and PA patterns according to updated recommendations on cancer prevention33 during a group meeting at the beginning of the study.
Monitoring the adherence to the intervention
Study participants assigned to the intervention arms were requested to keep 5 written 1-week diaries on diet (arm #1), PA (arm #2), or both (arm #3) during the 24-month intervention period. Dietary diaries encompassed all foods and beverages consumed in a 7-day period. In the PA diaries, women were asked to describe their daily non-occupational activities, to report the number of flights of stairs climbed and to record their steps through a digital pedometer provided at the start of the study. The study personnel reviewed the diaries with the aim to monitor the achievement of the study objectives and to counsel the participants who reported specific difficulties.
Study subjects characteristics
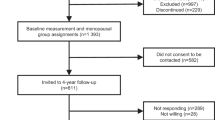
We recruited and randomised 234 women of 728 eligible (32.1%). Analyses included 230 women with information and blood samples collected at baseline and follow-up available (Fig. 1). The mean interval between the two visits was 2.0 years (SD 0.1).
The main characteristics of the study participants at baseline were compared by treatment. The mean age of participants was 58.7 year (SD 5.2), with about 70% reporting a high school degree. Most participants were never smokers and normal weight (BMI < 25). Among the 132 (57.4%) women with a paid work, 60.6% reported a sedentary work. The treatment and control groups were similar at baseline for these and other listed variables (Table 1).
Outcome measures
Concentrations of pro-inflammatory cytokines were measured in plasma samples collected at baseline and at the end of the intervention. Paired samples were blinded and analysed in the same batch in the Department of Experimental and Clinical Medicine laboratory (University of Florence, Florence, Italy). Samples were tested for IL-1α, IL-1β, IL-6, and TNF-α levels by MILLIPLEX MAP kit (EMD Millipore, Massachusetts, USA) and for CRP level by Affymetrix ProcartaPlex Multiplex Immunoassays (Invitrogen, California, USA), based on Luminex xMAP technology, according to the manufacturer’s instructions. The upper and lower limits of quantification (ULOQ-LLOQ) for each tested analyte were as follows: 10,000–9.4 pg/ml for IL-1α, 10,000–0.8 pg/ml for IL-1β, 10,000–0.9 pg/ml for IL-6, 10,000–0.7 pg/ml for TNF-α and 9–0.02 mg/L for CRP. Cytokines were detected using the fluorescent imager MAGPIX System (Luminex, Texas, USA) and data were acquired and analysed by xPONENT 4.2 software. The concentration of the samples was calculated by plotting the expected concentration of the standards against the mean fluorescence intensity generated by each standard. A four-parameter logistic algorithm was used for the best curve fit.
Statistical analyses
Plasma cytokines concentrations falling below the LLOQ were replaced with one-half the respective LLOQ for descriptive purposes34,35. No subject had values above the ULOQ. Cytokines concentration values were log-transformed in order to normalize the distribution and baseline and follow-up geometric means and standard deviations were estimated.
The factorial design allowed to evaluate the effect of dietary or PA interventions separately.
In order to evaluate the effect of the dietary intervention we compared subjects randomised to arms #1 and #3 versus arms #2 and #4. To evaluate the effect of the PA intervention we compared subjects randomised to arms #2 and #3 versus arms #1 and #4.
The analysis was performed on an intention-to-treat basis. We applied Tobit regression models for left censored variables36,37 to evaluate the effect of the dietary and PA interventions on follow-up log-transformed cytokine levels in comparison with the respective control groups. Analyses were adjusted for baseline log-transformed level of each cytokine and randomisation block variables (age and BMI). We also run the analyses adding to the model the weight change occurred during the intervention. Tobit regression coefficients (β), and relative 95% confidence intervals (95% CI), were then back transformed in the exponential form (exp(β)). An exp(β) value less than 1.0 indicates a lower cytokine level in treatment group compared to control group after the 24-month intervention. We also tested the interaction between the two treatments38.
The analyses were performed using Stata 14.0 statistical software (StataCorp LLC, College Station, TX).
Results
Concentrations of pro-inflammatory cytokines in plasma samples collected at baseline and at the end of the 24-month study period are reported in Table 2 together with the number of subjects with baseline cytokine level under LLOQ. No differences between treatment groups emerged in baseline levels of the cytokines considered. Cytokine levels under LLOQ were equally distributed among treatment groups at baseline (all P values for χ2 test > 0.05).
Overall, we observed a tendency to an increase in the concentration of each cytokine at the end of the study period. However, when we compared the follow-up values of each cytokine by treatment groups in adjusted models, women randomised to PA intervention (arms #2 + #3) showed lower levels of each cytokine in comparison with women not randomised to this intervention (arms #1 + #4), with exp(β) below 1.00, except for CRP and TNF-α for which we observed exp(β) values around 1. In particular the effect of PA intervention emerged for IL-1α (exp(β) 0.66; 95% CI 0.44–0.98; P = 0.04) and IL-6 (exp(β) 0.70; 95% CI 0.53–0.93; P = 0.01). For IL-1β the effect was less evident (exp(β) 0.84; 95% CI 0.66–1.06; P = 0.14).
No differences in cytokine levels at follow-up emerged in women randomised to the dietary intervention (arms #1 + #3) in comparison with women not randomised to that intervention (arms #2 + #4). All exp(β) were close to 1 (Table 2).
Overall, participants experienced a 0.34 kg weight loss (SD 2.9). The weight loss was more evident in the dietary intervention arm (0.48 kg; SD 3.3) and less evident in the control arm (0.16 kg; SD 2.5). When the analyses were further adjusted for weight change occurred during the intervention, results were materially unchanged (data not shown).
No interaction effect emerged between dietary and PA treatments for any cytokine (Table 2).
Compliance with the proposed interventions
The baseline and end of study reported dietary and PA habits are shown in Table 3 by treatment. No differences were evident at baseline except for some differences in reported consumption of cheese and cakes and in estimated energy intake by PA treatment. A higher vegetables, legumes and extra virgin olive oil consumption and a lower red meat consumption emerged at the end of the study among participants randomised to the dietary intervention group in comparison with the control group. We also found an increase in reported MET-hours/week of recreational activities in the PA intervention group in comparison with the control group (Table 3).
Discussion
In this secondary analysis of the DAMA 24-month trial in healthy postmenopausal women, we observed that at the end of the intervention, women randomised to the PA treatment showed lower plasma levels of pro-inflammatory cytokines IL-1α and IL-6 in comparison with control women, while no significant effect emerged for TNF-α, IL-1β and CRP levels. No effect of the isocaloric dietary intervention on cytokine levels emerged. All these results were confirmed after further adjustment for weight change occurred during intervention. No evidence of interaction emerged between the dietary and the PA intervention thus supporting the independent effect of the PA on the cytokine plasma levels.
Data collected through questionnaires completed at baseline and at the end of the study period showed a change in dietary and physical activity habits in agreement with the objective of the treatment assigned. In particular, women randomised to physical activity treatment showed at the end of the study a significant increase in recreational activities in comparison with the control group. An increase in the consumption of specific foods, namely total vegetables, nuts and seeds, legumes and extra-virgin olive oil and a decrease in the consumption of red meat emerged in women randomised to the dietary intervention.
To our knowledge most of the studies investigating the association between diet and/or physical exercise and circulating levels of cytokines in postmenopausal women were focused on women with metabolic syndrome or overweight, two conditions that are associated with baseline higher level of CRP and other inflammatory markers39,40,41.
Some randomised trials23,24,26,42 were focused on overweight or obese postmenopausal women. The ALPHA trial24 and the trial by Campell et al.26 reported a reduction in CRP levels in women randomised to a 1-year long PA intervention while no effect emerged for IL-6 and other cytokines examined. The CRP reduction appeared dependent on fat loss and proportional to the exercise volume. In a trial in which a six-month intervention focused on sedentary, overweight/obese women with elevated blood pressure was applied, no effects on CRP plasma level emerged in women randomised to one of three exercise groups characterized by increasing energy expenditure while a reduction in CRP emerged in women who experienced a weight loss > 2.6 kg irrespective of the intervention group assignment42. In the NEW trial a reduction in plasma level of CRP and IL-6 was observed in women randomised to a caloric restriction diet with or without exercise, while no effect on biomarkers emerged in women randomised to exercise23.
Other trials were focused on postmenopausal women with metabolic syndrome. Kemmler et al. evaluated the effect of a 12-months exercise intervention including both high-intensity strength and endurance exercises and no differences in CRP levels emerged between women randomised to exercise and the control group in which a very low intensity weekly exercise was proposed43. In a randomised factorial trial, in which the effects of PA and of a dietary intervention aimed to reduce total and saturated fats and cholesterol intake on the changes in CRP levels were evaluated, the authors reported a reduction of inflammatory biomarkers only in women randomised to the dietary intervention or dietary + PA intervention arms. This intervention was not specifically aimed to reduce the weight of participants and analyses were adjusted for weight change21. Another study carried out in subjects with metabolic syndrome showed, after two years, a reduction in serum concentration of CRP, IL-6, IL-7 and IL-8 together with a reduction in metabolic syndrome components including body weight, in the intervention arm in which a Mediterranean style diet was proposed in comparison with the control arm in which only general advice was offered44.
Differences in the targeted population, duration of the intervention, and protocols adopted make difficult to compare studies, however, in our trial a lower level of IL-6 at FU emerged in the PA treatment group compared to the control group in agreement with other studies. Interestingly, mean IL-6 level among our participants is consistent with expected plasma concentration among healthy subjects. The plasma IL-6 level in resting healthy subjects was about 1 pg/ml, while a chronic low level increase (< 10 pg/ml) was associated with several pathologic conditions and with low PA levels. Moreover, a negative association was documented between IL-6 plasma levels and the amount of regular PA level. IL-6 seems indeed to be down-regulated by PA training45. On the other hand we did not find a reduction in CRP plasma values. Again, in our study on healthy women, CRP plasma levels were below the value of 3 mg/L usually considered the threshold level for “normal” values10 and we did not register a weight loss as reported in other trials.
All participants in the trial were characterized by a high BD, a well known breast cancer risk factor. However scanty data are available on the possible association between inflammatory cytokines and high BD. In a study where the expression of inflammatory markers in normal breast tissue of breast cancer patients was evaluated in immunohistochemical stained slides, women having higher expression of pro-inflammatory cytokines had higher BD while anti-inflammatory markers were associated with lower BD46. However, in a large cross-sectional study in postmenopausal women no association emerged between plasma levels of IL-6, TNF-α and CRP and BD47. An inverse association emerged in a recent study carried out in Chinese women48.
Strengths of the DAMA trial include an approach aimed to obtain a general modification of dietary and PA habits in the daily life for a relatively long period through practical activities, group meetings and individual counselling. We monitored the compliance of participants, although the use of self reported dietary and PA information represents a limit of this study in comparison to more objective measures.
Participants in the DAMA trial were healthy women not affected by metabolic syndrome and mainly non overweight or obese thus with low inflammatory markers levels at baseline. These characteristics represent at the same time a strength and a limitation of the study. The strength is represented by the possibility to evaluate the effect of general modifications of dietary and PA habits among healthy individuals, while in previous studies participants were mainly obese or affected by metabolic syndrome. The limitation is represented by the general low inflammatory markers level that make it difficult to detect further decreases. Actually, we observed a slowdown in the inflammatory markers level increase in women randomised to the PA treatment.
We observed a lack of effect of our Mediterranean style isocaloric dietary intervention on all the examined cytokines. We carried out a dietary intervention aimed to a comprehensive modification of diet, providing indication on food choices and frequency of consumption. No indications were provided concerning the amount of foods to be consumed with the aim to avoid a weight change, being aware of the effect of body fat on BD measurements, the main outcome of the study29. The lack of results of our dietary intervention is probably due to this isocaloric protocol while, in Mediterranean countries dietary modifications probably must lead to some weight loss to achieve results in terms of biomarkers changes.
Overall we observed a tendency to an increase of each cytokine in study subjects over the 24-months of the study irrespective of the treatment assigned. This suggests that even in healthy people with low baseline cytokine levels, a detectable although modest increase in inflammatory markers might occur in a relatively short time period, thus supporting the hypothesis of the age related modification of the cytokine expression pattern that has been reported as an underlying molecular mechanism for a series of chronic diseases including cancer49,50. Lifestyle habits such as PA and diet and their changes across the life span may play a role in modifying this process.
Chronic low-grade inflammatory state, is a condition in which inflammatory processes are chronically stimulated at low levels affecting the entire organism. The link between chronic low-grade inflammation and various chronic diseases is now evident and the control of this condition is probably important for the prevention of the most common diseases in the general population1,2.
In conclusion this study support the effectiveness of PA, even at moderate intensity, in reducing the increase of inflammatory markers over time and the possibility to reduce the risk of chronic diseases in postmenopausal women. Primary prevention activities aimed to the modification of lifestyle habits are becoming increasingly important in reducing chronic low-grade inflammatory processes.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to participants privacy protection but are available from the corresponding author on reasonable request.
References
Couzin-Frankel, J. Inflammation bares a dark side. Science 330, 1621 (2010).
Furman, D. et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 25, 1822–1832 (2019).
Cioffi, M. et al. Cytokine pattern in postmenopause. Maturitas 41, 187–192 (2002).
Pfeilschifter, J., Köditz, R., Pfohl, M. & Schatz, H. Changes in proinflammatory cytokine activity after menopause. Endocr. Rev. 23, 90–119 (2002).
Malutan, A. M., Dan, M., Nicolae, C. & Carmen, M. Proinflammatory and anti-inflammatory cytokine changes related to menopause. Prz Menopauzalny 13, 162–168 (2014).
Iyengar, N. M., Gucalp, A., Dannenberg, A. J. & Hudis, C. A. Obesity and cancer mechanisms: tumor microenvironment and inflammation. J. Clin. Oncol. 34, 4270–4276 (2016).
Maggio, M., Guralnik, J. M., Longo, D. L. & Ferrucci, L. Interleukin-6 in aging and chronic disease: a magnificent pathway. J. Gerontol. A Biol. Sci. Med. Sci. 61, 575–584 (2006).
Li, H., Liu, W. & Xie, J. Circulating interleukin-6 levels and cardiovascular and all-cause mortality in the elderly population: a meta-analysis. Arch. Gerontol. Geriatr. 73, 257–262 (2017).
Popa, C., Netea, M. G., van Riel, P. L., van der Meer, J. W. & Stalenhoef, A. F. The role of TNF-alpha in chronic inflammatory conditions, intermediary metabolism, and cardiovascular risk. J. Lipid Res. 48, 751–762 (2007).
Pepys, M. B. & Hirschfield, G. M. C-reactive protein: a critical update. J. Clin. Investig. 111, 1805–1812 (2003).
Kushner, I., Rzewnicki, D. & Samols, D. What does minor elevation of C-reactive protein signify?. Am. J. Med. 119(166), e17-166.e28 (2006).
Akdis, M. et al. Interleukins, from 1 to 37, and interferon-γ: receptors, functions, and roles in diseases. J. Allergy Clin. Immunol. 127, 701–721 (2011).
Giugliano, D., Ceriello, A. & Esposito, K. The effects of diet on inflammation: emphasis on the metabolic syndrome. J. Am. Coll. Cardiol. 48, 677–685 (2006).
Schwingshackl, L. & Hoffmann, G. Mediterranean dietary pattern, inflammation and endothelial function: a systematic review and meta-analysis of intervention trials. Nutr. Metab. Cardiovasc. Dis. 24, 929–939 (2014).
Casas, R., Sacanella, E. & Estruch, R. The immune protective effect of the Mediterranean diet against chronic low-grade inflammatory diseases. Endocr. Metab. Immune Disord. Drug Targets 14, 245–254 (2014).
Gotsis, E. et al. Health benefits of the Mediterranean diet: an update of research over the last 5 years. Angiology 66, 304–318 (2015).
Minihane, A. M. et al. Low-grade inflammation, diet composition and health: current research evidence and its translation. Br. J. Nutr. 114, 999–1012 (2015).
Mathur, N. & Pedersen, B. K. Exercise as a mean to control low-grade systemic inflammation. Mediat. Inflamm. 2008, 6 (2008) (article ID 109502).
Gleeson, M. et al. The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat. Rev. Immunol. 11, 607–615 (2011).
Walsh, N. P. et al. Position statement. Part one: immune function and exercise. Exerc. Immunol. Rev. 17, 6–63 (2011).
Camhi, S. M., Stefanick, M. L., Ridker, P. M. & Young, D. R. Changes in C-reactive protein from low-fat diet and/or physical activity in men and women with and without metabolic syndrome. Metabolism 59, 54–61 (2010).
Mena, M. P. et al. Inhibition of circulating immune cell activation: a molecular anti inflammatory effect of the Mediterraneandiet. Am. J. Clin. Nutr. 89, 248–256 (2009).
Imayama, I. et al. Effects of a caloric restriction weight loss diet and exercise on inflammatory biomarkers in overweight/obese postmenopausal women: a randomized controlled trial. Cancer Res. 72, 2314–2326 (2012).
Friedenreich, C. M. et al. Inflammatory marker changes in a yearlong randomized exercise intervention trial among postmenopausal women. Cancer Prev. Res. (Phila) 5, 98–108 (2012).
Friedenreich, C. M. et al. Inflammatory marker changes in postmenopausal women after a year-long exercise intervention comparing high versus moderate volumes. Cancer Prev. Res. (Phila) 9, 196–203 (2016).
Campbell, P. T. et al. A yearlong exercise intervention decreases CRP among obese postmenopausal women. Med. Sci. Sports Exerc. 41, 1533–1539 (2009).
Masala, G. et al. The DAMA trial: a diet and physical activity intervention trial to reduce mammographic breast density in postmenopausal women in Tuscany, Italy. Study protocol and baseline characteristics. Tumori 100, 377–385 (2014).
McCormack, V. A. & dos Santos, S. I. Breast density and parenchymal patterns as markers of breast cancer risk: a meta-analysis. Cancer Epidemiol. Biomark. Prev. 15, 1159–1169 (2006).
Masala, G. et al. Can dietary and physical activity modifications reduce breast density in postmenopausal women? The DAMA study, a randomized intervention trial in Italy. Cancer Epidemiol. Biomark. Prev. 28, 41–50 (2019).
American College of Radiology. Breast Imaging Reporting and Data System (BI-RADS®) 4th edn. (American College of Radiology, Reston, VA, 2003).
Pala, V. et al. Diet in the Italian EPIC cohorts: presentation of data and methodological issues. Tumori 89, 594–607 (2003).
Masala, G. et al. Physical activity and mammographic breast density in a Mediterranean population: the EPIC Florence longitudinal study. Int. J. Cancer 124, 654–1661 (2009).
World Cancer Research Fund, American Institute for Cancer Research. Food, Nutrition and the Prevention of Cancer: a Global Perspective (AICR, Washington, DC, 2007).
Hewett, P. & Ganser, G. H. A comparison of several methods for analyzing censored data. Ann. Occup. Hyg. 51, 611–632 (2007).
Hornung, R. W. & Reed, L. D. Estimation of Average Concentration in the Presence of Non detectable Values. Appl. Occup. Environ. Hyg. 5, 46–51 (1990).
Lubin, J. H. et al. Epidemiologic evaluation of measurement data in the presence of detection limits. Environ. Health Perspect. 112, 1691–1696 (2004).
Ballenberger, N., Lluis, A., von Mutius, E., Illi, S. & Schaub, B. Novel statistical approaches for non-normal censored immunological data: analysis of cytokine and gene expression data. PLoS ONE 7, e46423 (2012).
McAlister, F. A., Straus, S. E., Sackett, D. L. & Altman, D. G. Analysis and reporting of factorial trials: a systematic review. JAMA 289, 2545–2553 (2003).
Devaraj, S., Singh, U. & Jialal, I. Human C-reactive protein and the metabolic syndrome. Curr. Opin. Lipidol. 20, 182–189 (2009).
Festa, A. et al. Chronic subclinical inflammation as part of the insulin resistance syndrome: the Insulin Resistance Atherosclerosis Study (IRAS). Circulation 102, 42–47 (2000).
Choi, J., Joseph, L. & Pilote, L. Obesity and C-reactive protein in various populations: a systematic review and meta-analysis. Obes. Rev. 14, 232–244 (2013).
Stewart, L. K., Earnest, C. P., Blair, S. N. & Church, T. S. Effects of different doses of physical activity on C-reactive protein among women. Med. Sci. Sports Exerc. 42, 701–707 (2010).
Kemmler, W., Von Stengel, S., Engelke, K. & Kalender, W. A. Exercise decreases the risk of metabolic syndrome in elderly females. Med. Sci. Sports Exerc. 41, 297–305 (2009).
Esposito, K. et al. Effect of a mediterranean-style diet on endothelial dysfunction and markers of vascular inflammation in the metabolic syndrome: a randomized trial. JAMA 292, 1440–1446 (2004).
Fischer, C. P. Interleukin-6 in acute exercise and training: what is the biological relevance?. Exerc. Immunol. Rev. 12, 6–33 (2006).
Hanna, M. et al. Association between expression of inflammatory markers in normal breast tissue and mammographic density among premenopausal and postmenopausal women. Menopause 24, 524–535 (2017).
Reeves, K. W., Weissfeld, J. L., Modugno, F. & Diergaarde, B. Circulating levels of inflammatory markers and mammographic breast density among postmenopausal women. Breast Cancer Res. Treat. 127, 555–563 (2011).
Fang, C. Y. et al. Inflammation and breast density among female Chinese immigrants: exploring variations across neighborhoods. Cancer Causes Control 187, 705–716 (2019).
Rea, I. M. et al. Age and age-related diseases: role of inflammation triggers and cytokines. Front. Immunol. 9, 586 (2018).
Bektas, A., Schurman, S. H., Sen, R. & Ferrucci, L. Aging, inflammation and the environment. Exp. Gerontol. 105, 10–18 (2018).
Aknowledgments
The authors wish to thank all the participants of the DAMA Trial and the staff personnel involved in the different phases of the study. Members and volunteers of a local consumers cooperative (UNICOOP-Firenze) are also acknowledged for their support to the study cooking activities. This work was supported by the Istituto Toscano Tumori–ITT (Grant 2009-2012 to GM); the Ministry of Health (Programma Integrato Oncologia 2006 coordinated by DP); and the Associazione Italiana per la Ricerca contro il Cancro (AIRC) Milan, Italy (IG 2012 Grant Number 3236 and IG 2015 Grant Number 17146 to DP).
Author information
Authors and Affiliations
Contributions
G.M. and D.P. conceived and designed the study. D.O., M.C., I.E., B.B., C.S. and S.C. contributed to the trial activity, data acquisition and sample retrieval. M.M.D’E., C.D.B. and S.T. performed the laboratory analyses. G.M., M.A. and B.B. managed and analyzed the data. G.M., B.B., M.M.D’E. and D.P. wrote, revised and approved the manuscript. All the authors contributed to the results interpretation and read and approved the submitted version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Masala, G., Bendinelli, B., Della Bella, C. et al. Inflammatory marker changes in a 24-month dietary and physical activity randomised intervention trial in postmenopausal women. Sci Rep 10, 21845 (2020). https://doi.org/10.1038/s41598-020-78796-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-78796-z
This article is cited by
-
Adipocytokine plasma level changes in a 24-month dietary and physical activity randomised intervention trial in postmenopausal women
European Journal of Nutrition (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.