Abstract
Hypertrophic and keloid scars result from abnormal wound healing and can have a variable response to a number of available treatment modalities. The evolution of laser treatments in recent years has shown a wide range of clinical applications including their use in the treatment of scars. We investigated the effectiveness of a 1470 nm diode laser using an intralesional optical fibre delivery device in the treatment of hypertrophic and keloid scars. We evaluated its safety and efficacy as a novel and minimally invasive treatment alternative for scar modulation and volume reduction. A prospective cohort study was performed involving 21 patients with hypertrophic scars (HS) (n = 9) and keloids (n = 12) resulting from various aetiology. Patients were treated with one to three treatment sessions. Comprehensive evaluations were performed using the Vancouver Scar Scale, Doppler ultrasound, Cutometer, Mexameter and PeriCam PSI. Scar thickness was reduced by an average of 0.308 ± 0.138 cm (p < 0.001). In particular the two subgroups showed a significant 27.7% and 28.2% reduction in scar thickness of HS and Keloids, respectively. Scar firmness showed a significant improvement of 1.2% (p < 0.05) for HS, though for keloids this was 0.4% (p = 0.26). Keloids had a significant reduction in pigmentation at 21.3%. Blood perfusion had a significant reduction of 29.6% in HS and 22.7% in Keloids. Overall VSS total score improvement of 42% in the HS and at 37.9% in the Keloid subgroup. No adverse events such as hypo/hyperpigmentation, skin infection, or recurrence were reported. This study shows that the intralesional 1470 nm bare-fibre diode laser significantly improved hypertrophic and keloid scars based on both subjective and objective analyses and supports this type of laser therapy as a safe and effective minimally-invasive treatment option.
Similar content being viewed by others
Introduction
Hypertrophic Scars (HS) and Keloids (K) are the result of abnormal wound healing and scar formation. These pathological scars present as a continuous inflammation and histologically are characterized by fibroblasts proliferation, newly formed blood vessels and collagen deposition. HSK are distinguished clinically by the extent of tissue overgrowth with respect to the margins of the original wound. HS typically occur within a few months in areas were scars are under tension, including the sternal region, around joints and can persist along the margins of the original wound. Keloid scars occur more frequently in individuals with darker pigmented skin, can develop up to several years following minor trauma, and as well develop in areas such as earlobes, shoulders and chest. Keloids are more often symptomatic, being associated with pain and itching, and can proliferate well beyond the original limits of the wound1,2. HSK can occur following different injury mechanisms including skin injuries, burns, surgery, injections and dermatitis. Among them, deep burns are reported to be the main cause of HSK3,4.
Various treatments have been implicated in their management, however there is still no completely satisfactory technique for treating these scars. A number of different therapies have been proposed such as silicone sheet and gel application, pressure garments, topical and intralesional injections of agents including corticosteroids, interferon, bleomycin, five-fluorouracil (5-FU), as well as cryotherapy, laser, radiotherapy and surgery5,6,7. Despite the wide range of treatments, the reported recurrence rates for keloids remain high at between 50 to 80% and around 10% for hypertrophic scars8,9.
In the first instance non-surgical treatment options are preferred. However, if failed, surgery, laser and radiation therapies are considered favourable treatment options10.
Although minimally-invasive methods have demonstrated good outcomes for soft and thin scars, there are difficulties for harder textured and thicker scars. Topical medications scarcely penetrate, and injection of therapeutic agents are obstructed by fibrotic tissue, requiring multiple treatments with often limited efficiency11. Traditional laser diffusion has also limited efficacy and penetrance within thicker scars. HSK showed positive response to chemotherapeutic agents but adverse effects include pain, ulceration, burning and hyperpigmentation12. Surgical excision for HSK reduction remains an option but unfortunately long-term results can be poor, with a high risk of scar recurrence. Currently, no single method provides a complete benefit with recent publications showing contradicting results5,6,10. Moreover, the treatment of HSK remains a clinical challenge.
This study is based on our previous positive experience using the 1470 nm diode laser and fibre system to treat inflamed and infected keloids13. We demonstrated that the 1470 nm diode laser was able to deliver through the fibre system a localised heating effect within a narrow range. The fibrotic scar tissue rapidly vaporises, instigating cell lysis, necrosis and solidification, that results in tissue ablation and inflammation control. Furthermore, the 1470 nm laser was able to coagulate blood vessels, reducing blood supply and local tissue vascularization14,15,16.
The aim of this research study was to evaluate the efficacy and safety of the 1470 nm fibre laser as a novel method for scar volume reduction. Its effectiveness was assessed in the treatment and improvement of HSK.
Methods
Patients
A prospective, single-center cohort study was performed to evaluate the efficacy of a fractional non-ablative diode laser and fibre to treat patients with HSK in various anatomical regions caused by trauma, burns, acne or surgery. Twenty-one patients were observed over a 2-year period between January 2017 and January 2019 treated at the Ninth People's Hospital in Shanghai. All cases were previously treated for more than 1 year with different therapies such as steroid ointments or silicone tapes, but improvements were not observed. Inclusion criteria for the study were a hypertrophic scar or keloid recalcitrant to other therapies with the size of scar more than 10 cm2; caused by burns, trauma, surgery, insect bite or acne.
Exclusion criteria were as follows: patients who had undergone previous scar therapies such as laser or surgery, had heart or kidney failure, received immunosuppressive drugs or had autoimmune disorders or uncontrolled diabetes, and those unable to follow the protocol.
Scar evaluation
Subjective assessment of the scar was evaluated by an experienced independent evaluator pre-operatively and at 6 and 12 months after treatment using the Vancouver Scar Scale (VSS) score. The VSS grading score includes pigmentation (0, normal; 1, hypopigmented; 2, mixed pigmentation; 3, hyperpigmented), vascularity (0, normal; 1, pink; 2, red; 3, purple), pliability (0, normal; 1, supple; 2, yielding; 3, firm; 4, banding; 5, contracture), and height (0, flat; 1, ≤ 2 mm; 2, 2–5 mm; 3, ≥ 5 mm). The score for each parameter was calculated separately, and then all 4 parameters were combined17. A questionnaire was completed to study the impact of the scar on quality of life (QOL)18. Adverse effects associated with the laser treatment including erythema, burning, pain, and itching were collected. Photographs were obtained using identical digital camera settings, lighting conditions, and patient positioning at every consultation.
Objective evaluations of each scar were collected before and after every treatment through the following instruments: [A] Scar thickness using a doppler ultrasound DP-6600 digital ultrasonic diagnostic imaging system (Mindray, Redmond, WA) with a 10 MHz frequency probe; [B] Scar firmness was assessed with a Cutometer dual MPA 580 (Courage Khazaka Electronic GmbH, Germany) mounted with a 6 mm handheld probe. The Cutometer uses a principle based on suction and elongation measurements, generating a negative pressure (set at 500 mbar for 1 s suction and relaxation times) which draws skin into the probe’s central aperture and estimates skin penetration depth using an optical measuring system19; [C] Scar pigmentation with a Mexameter MX 18 connected to the Multiprobe Adapter System (Courage Khazaka Electronic GmbH, Köln, Germany). The device measures pigmentation of the scar expressed by Melanin index19; [D] Blood supply of the scar were examined using a blood perfusion imager PeriCam PSI System (Perimed, Järfälla, Sweden) and Mexameter MX 18 (Courage Khazaka Electronic GmbH, Germany)20. Both were applied to detect changes of scar vascularity. The PeriCam PSI System is a blood perfusion imager 70-mW system based on Laser Speckle Contrast Analysis (LASCA) technology utilizing a laser wavelength of 785 nm20. The Mexameter MX 18 uses an erythema index to represent the quantity of haemoglobin at the detection site19. The Cutometer and Mexameter measurements were measured over three areas which were repeated three times. The first point was measured at the proximal 1/3 along the axis of the keloid or hypertrophic scars. The second point was measured in the middle of the scar and the final measurement was taken at the distal 1/3 of the scar. The mean average of the three detection points were then calculated to achieve an overall mean result.
Laser technique
A fractional non-ablative laser LASEmaR 1500 (EUFOTON, Trieste, Italy) with a semiconductor gallium arsenide (GaAs) emitting 1470 nm wavelengths of light, was adopted in this study. The energy was delivered through a disposable optical bare fibre with a 300-micron diameter. The power output was set according to scar hardness in order to penetrate the scar with minimal resistance, generally starting from 3 W and gradually increasing to a maximum power output of 6 W if necessary, with a maximum fluence 999.9 kJ/cm2. The optical fibre was initiated with the laser to penetrate and deliver the energy through 1 to 4 passes of the scar. Entry points with a distance of 2–4 mm were made surrounding the scar (Video 1). The procedure was repeated once every one to two months until satisfactory results were achieved for the clinician and patient.
Statistical analysis
All data were initially entered into an Excel database (Microsoft, Redmond, Washington—United States) and the analysis was performed using the Statistical Package for the Social Sciences Windows, version 15.0 (SPSS, Chicago, Illinois, USA).
The paired t-test was used to compare the changes in scar thickness, scar firmness, scar pigmentation, and scar blood supply before and after the laser treatment.
Subjective assessment before and after laser treatment were compared using Chi-square test.
Descriptive statistics consisted of the mean ± standard deviation (SD) for continuous parameters with normal distributions (after confirmation with histograms and the Kolmogorov–Smirnov test).
Comparison among groups was performed with the Student T-test for paired data or non-parametric Chi-Square test or Fisher’s exact test (if cells < 5) for categorical data. A p value < 0.05 was considered statistically significant. To evaluate the minimum sample size, a “power analysis” was conducted, which considered a downward difference of 25% from the average parameter of HS Thickness between pre and post treatment. Moreover, a level of 95% (alpha = 5%) for a 1-tailed test, a power of 82% was accepted with a sample size of 9 patients.
Ethics
The study was conducted according to the criteria set by the declaration of Helsinki (1964) and successive modifications. The study protocol was approved by the local institutional ethics review committee (Jiao Tong University-Ninth People's Hospital, Shanghai). Clinical trial registration number: ChiCTR2000038092 (http://www.chictr.org/en/), last update 10/09/2020. Written informed consent was obtained from each patient. Informed consent from a parent and/or legal guardian was obtained for patients under the age of 18 to participate in this study.
Results
Patient characteristics
A total of twenty-one patients (mean age 28.4 years, range 3–66) with HSK were enrolled in this study which we divided in two subgroups. The first subgroup contained nine patients who had HS whilst the second subgroup contained twelve patients with keloids. Fourteen patients were male and seven females. Locations of the scars were chest (9), back (5), flank (4), thigh (4), shoulder (3), dorsal foot (2), arm (2), and other areas (6). The average scar size was 207.19cm2 (range 18.00–1063.50 cm2). The aetiology of scars was trauma (7 patients), burns (6 patients), acne scarring (4 patients), furuncle (3 patients), and previous surgery (1 patient). The average number of treatments was 1.38 (range 1–3). The mean single treatment energy was 4486.76 J (range 388–17536 J) and mean single treatment power was 4 W (range 3–6 W). Average follow-up was 7.2 months (range 6–12 months) (Table 1).
Objective assessment
The scar thickness was assessed at each visit and the doppler showed a decrease of 0.308 ± 0.138 cm (p < 0.05) at 6 months. Scar thickness significantly reduced compared to the baseline (0.942 ± 0.377 cm vs. 0.633 ± 0.306 cm). In particular, the two subgroups showed a 27.7% and 28.2% reduction in scar thickness of HS and Keloid scar.
The scar firmness was evaluated at each visit and the Cutometer tester (R0 and Q0 parameters of hardness are inversely proportional to its numerical size) measured a change respectively of − 0.023 ± 0.008 (p = 0.007) and − 2.616 ± 1.169 Units (p = 0.029). The scar firmness was improved (R0: 1.816 ± 0.055 Units, Q0: 365.629 ± 13.113 Units vs. R0:1.838 ± 0.057 Units, Q0:368.244 ± 12.442 Units). Respectively, the two subgroups showed a significant improvement of 1.2% (p < 0.05) in HS and an improvement of 0.4% (p = 0.26) in keloids which was not statistically significant.
Regarding the scar pigmentation parameters, from the two subgroups combined there was no significant improvement in scar pigmentation. The Melanin Index before the treatment was 251.413 ± 157.716 Units, and after 6 months was 234.349 ± 90.708 Units with a difference of 17.063 ± 131.33 Units (p = 0.308). However, the keloid group independently showed a 21.3% (p < 0.01) improvement in scar pigmentation.
The scar vascularity was measured by PeriCam PSI blood perfusion imager that indicated a decrease of blood perfusion volume in the scar of 33.645 ± 15.667 Units and by the Mexameter that showed the Erythema index decreasing 17.349 ± 8.959 Units. This indicates that the laser significantly reduces the blood supply of the scar (PeriCam PSI: 117.888 ± 44.593 vs. 84.243 ± 33.244; p < 0.05. Erythema Index: 437.714 ± 96.771vs. 420.365 ± 96.475; p < 0.05). Furthermore, the two subgroups independently showed a significant reduction of blood perfusion at 29.6% and erythema index at 4% in HS and a reduction of blood perfusion at 22.7% and erythema index at 3.1% in keloids.
The overall results are summarized in Table 2, whereas the independent findings of each subgroups are showed in Table 3.
Subjective assessment
Pigmentation, Height, Vascularity and Pliability were assessed according to the VSS score. The overall findings are reported in Table 2, whilst independent analysis of each subgroup is summarized in Table 3. Results showed that the laser therapy can significantly reduce the blood supply, decrease thickness and enhance pliability of HSK. In particular, there was an improvement in the total VSS score of 42% in the HS subgroup and 37.9% in the keloid subgroup.
Photographs of representative patients are shown in Figs. 1, 2, 3, 4, 5 and 6.
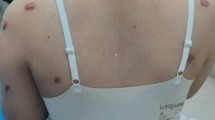
A 9-year-old boy [case 3] with a hypertrophic scar on his back prior laser treatment (A); 6 months post two laser therapy sessions (B); the scar blood perfusion showed 140.86 Units prior to treatment (C), a 32.64% reduction of the blood supply to the scar post laser treatment (D); the average thickness was 1.05 cm before the operation (E); a 63.81% decrease of scar thickness post laser treatment (F).
A 49-years-old woman [case 19] sustained a post-traumatic hypertrophic scar over 2-years prior to treatment (A); after 6 months follow-up and 2 laser sessions (B); the scar blood perfusion was significantly decreased: prior the operation was 104.29 Units (C), post operation there was a reduction of 28.46% (D); the scars thickness prior to the operation was 0.73 cm (E), post operation was decreased 43.84% (F).
A 66-years-old man [case 21] sustained a keloid on his chest caused by acne over 10 years period (A); after a 6 months follow-up and 2 treatments using intralesional 1470 nm bare-fibre diode laser (B); the scar blood perfusion showed 98.79 Units before the treatment (C), which decreased to 38.92% after two sessions of laser treatment (D); the average thickness of the scar was 0.79 cm (E), resulting in a decreased of 45.57% after the laser treatment (F).
Incidence of postoperative pain was 85.71%, duration was 2.19 ± 0.75 days, pain degree score was 4.19 ± 1.23Units, local swelling rate was 100% and the duration was 7.00 ± 1.10 days. The rate of partial scar tissue necrosis was 28.57%, healing time was 24.83 ± 12.05 day. None of the patients after laser procedure showed hypertrophy during the 6 to 12 months of follow-up period. None reported heavy pigmentation > 3 months, hypopigmentation or skin infection. This data is summarized in Table 4. The percentage of patients who were very satisfied or satisfied was over 85%.
Discussion
Several innovative strategies have been introduced for the treatment of HSK over the past few decades11,12,13,14,15,16. The evolution of laser treatment has shown a wide range of clinical and cosmetic applications including its use in the treatment of different types of scars.
In the 1980’s, Castro et al.21 reported first experiments analysing the effects of the Nd:YAG laser on human skin fibroblasts. Despite encouraging early reports, subsequent studies showed limited efficacy and a high incidence of side effects22. Other authors used the continuous wave CO2 laser (10,600 nm) to selectively inhibit collagen production23. However, initial results were poor and further studies showed that keloid formation was not inhibited after 1 year22,23,24. Thanks to the introduction of selective photothermolysis on the microvasculature of the scars, pulsed lasers were used to provide targeted selectivity with limited thermal injury25.
Recent advances in the use of fractionated lasers, based on the concept of using fractional photothermolysis showed faster epidermal tissue repair26. Thus, using columns of coagulation in microthermal zones has no destructive effect on the epidermis, leaving the skin intact between the small cores of coagulation, preserving the integrity of the epidermis27.
New tools were developed based on these innovative devices, to deliver the laser energy. A laser diode system LASEmaR 1500, at the wavelength of 1470 nm, was used as the laser energy source, near a peak in the water absorption spectrum. Laser energy was delivered through a specific type of optical bare fibre with 300-micron diameter. The fibre, with directional laser irradiation, enables penetration through tissue to a depth of 2–3 mm, enabling deeper penetration of the non-ablative fractional laser when treating thicker scars, an issue inherent with this type of laser in comparison to longer wavelength ablative fractional lasers28. The heat produced from this laser is emitted in a narrow field, so water quickly vaporizes in the surrounding tissue, resulting in selected tissue ablation and coagulation, with localised cell lysis and tissue necrosis. These kinds of fibre laser are widely adopted in vascular surgery for the treatment of endovenous ablation of incompetent saphenous veins and varicose veins29,30. The use of fibre laser has rapidly expanded in other disciplines such as urology, for the treatment of prostate hyperplasia14,15, otorhinolaryngology, for the treatment turbinate hypertrophy31, colorectal surgery for the treatment of anorectal fistula32, plastic surgery, for body contouring and lymphedema treatment33,34.
To our knowledge, we were the first in adopting this technique for scar therapy13, and have now evaluated its efficacy and safety in a larger cohort of patients for the treatment of HSK.
Our study showed that the 1470 nm laser can limit inflammation and decrease perfusion therefore suppressing the development of aberrant scars by effectively reducing the thickness (almost 30%) and inflammation of HSK (p < 0.05).
Different studies have demonstrated that the denaturation temperature of collagen is 60–70 °C, and the carbonization temperature is above 200 °C35. The temperature of the tissue surrounding the optical fibre tip measures between 100 and 200 °C, allowing the breakdown of collagen deposits and the extracellular matrix. The collagen of cicatricial tissue degenerates and shrinks whilst diffused heat leads to protein denaturation and tissue coagulation36. Albergel et al.37, demonstrated that HSK produce more collagen and an extracellular matrix, increasing scar tissue density and collagen type I and III, forming fibrotic cords that increase firmness and decrease elasticity. Furthermore, Miles et al.38 analysed the collagen composition highlighting the triple helical structure that is stabilized by 4-hydroxyproline which forms a hydrogen bond. This triple helical structure is strongly associated with the stability of the collagen. When the collagen is heated, the triple helix structure changes into amorphous random coils, which causes the collagen contraction by up to one-third and modifies the collagen tensile strength and viscoelasticity39.
Our study showed that the scar firmness index was reduced after fibre laser treatment, leading to softer tissue, therefore improving elasticity of the scar (p < 0.05). The HS subgroup showed a significant improvement of 1.2% (p < 0.05) while the keloid subgroup an improvement of 0.4% (p = 0.26) was not statistically significant. This suggests a modification of the scar tissue texture. Thermal effects change cross-linkage between scar collagen and modify the three helical collagen structures, leading to collagen contraction and degeneration.
Post-operative hyperpigmentation, discoloration or prolonged erythema are the most common adverse effect after scar laser therapy22,27,40. Our study found that the 1470 nm fibre laser therapy does not intensify the melanin deposition in the keloids scar, therefore cannot create hyperpigmentation. In particular keloids had a significant reduction of pigmentation at 21.3%. This indicates that melanin is not absorbed by the 1470 nm wavelength. Moreover, the fibre laser may destroy melanin deposits, reducing the pigmentation of the scar.
Different authors reported that the proliferation of fibroblasts, collagen synthesis, cell activities and metabolism is enhanced in HSK, stimulating new capillaries that transport nutrients. Ogawa, et al.11,41 noted that angiogenic cytokine VEGF levels and vascular density in HSK were significantly higher than normal tissue. This assumes that the blood supply is the most important factor for the nutrients of the scar tissue. Investigating changes on scar vascular supply through erythema index and PeriCam PSI, we report that the scar blood perfusion was significantly decreased (p < 0.001). The erythema of the scar, as assessed by colorimetry, showed a significant difference in comparison to the preoperative status. In particular, blood perfusion had a reduction of 29.6% in HS and 22.7% in keloids; the erythema index also has an improvement of 4% in HS and 3.1% in keloids. This is in contrast to the controlled study of Wittenberg et al.42, where HSK treated by pulsed dye laser tended to lose the redness over a period of 1-year post operation. A possible explanation is that the fibre laser reduces the inflammatory period of the scar hence accelerates its maturation phase.
In this study we solely utilized a 1470 nm fibre laser to treat HSK and we achieved satisfactory results in terms of scar volume reduction, improving scar elasticity, decreasing erythema, pigmentation and scar vascularity. The penetration depth of the 1470 nm laser can only reach up to 2–3 mm. Thus, to treat scars that protrude the skin more than 2 mm multiple passes and treatments are necessary. Consequently, this laser method may be combined with other lasers or agents28,43,44,45,46.
There are a few limitations to this study which include a small sample size and the lack of a control group. Caution is warranted when interpreting subjective results as improvements can be seen as highly variable due to research observer bias. The scar measurements method can also be a limitation of the study. This is due to a reduction in diameter of the scar that could lead to the exact spot not being remeasured each time, leading to variability in the results. The therapy was shown to be safe with no adverse reactions or recurrence, although moderate pain was experienced for an average of two days by most patients. Finally, follow-up numbers and different timing of interventions between patients may have also had an impact on the reported results.
Future studies should address the optimal timing of treatment, the optimal laser parameters and settings and the combination of complimentary therapies to achieve higher satisfactory and long-term results28.
Conclusion
The present study shows that the 1470 nm fibre laser could be a promising treatment modality for HSK. We have demonstrated that it is a safe and effective method, based on both subjective and objective analyses. It is an effective minimally invasive scar reduction therapy that may aid as an additional powerful tool in the surgeon's armamentarium. However, to confirm these results, additional investigations on the fibre laser mechanisms including immuno-histopathological studies are necessary.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Meymandi, S. S., Moosazadeh, M. & Rezazadeh, A. Comparing Two methods of cryotherapy and intense pulsed light with triamcinolone injection in the treatment of keloid and hypertrophic scars: a clinical trial. Osong Public Health Res. Perspect. 7, 313–319 (2016).
Robles, D. T. & Berg, D. Abnormal wound healing: keloids. Clin. Dermatol. 25, 26–33 (2007).
Atiyeh, B. S. Nonsurgical management of hypertrophic scars: evidence-based therapies, standard practices, and emerging methods. Aesthet. Plast. Surg. 31, 468–494 (2007).
Berman, B., Maderal, A. & Raphael, B. Keloids and hypertrophic scars: pathophysiology, classification, and treatment. Dermatol. Surg. 43(Suppl 1), S3–S18 (2017).
Wolfram, D., Tzankov, A., Pülzl, P. & Piza-Katzer, H. Hypertrophic scars and keloids—a review of their pathophysiology, risk factors, and therapeutic management. Dermatol. Surg. 35, 171–181 (2009).
Gauglitz, G. G., Korting, H. C., Pavicic, T., Ruzicka, T. & Jeschke, M. G. Hypertrophic scarring and keloids: pathomechanisms and current and emerging treatment strategies. Mol. Med. 17, 113–125 (2011).
Bellew, S. G., Weiss, M. A. & Weiss, R. A. Comparison of intense pulsed light to 595-nm long-pulsed pulsed dye laser for treatment of hypertrophic surgical scars: a pilot study. J. Drugs Dermatol. 4, 448–452 (2005).
Stewart, C. E. IV. & Kim, J. Y. Application of mitomycin-C for head and neck keloids. Otolaryngol. Head Neck Surg. 135, 946–950 (2006).
Kang, S., Hur, J. K. & Kim, D. Advances in diagnostic methods for keloids and biomarker-targeted fluorescent probes. Analyst 11(144), 1866–1875 (2019).
Bouzari, N., Davis, S. C. & Nouri, K. Laser treatment of keloids and hypertrophic scars. Int. J. Dermatol. 46, 80–88 (2007).
Ogawa, R., Mitsuhashi, K., Hyakusoku, H. & Miyashita, T. Postoperative electron-beam irradiation therapy for keloids and hypertrophic scars: retrospective study of 147 cases followed for more than 18 months. Plast. Reconstr. Surg. 111, 547–555 (2003).
Jones, C. D., Guiot, L., Samy, M., Gorman, M. & Tehrani, H. The use of chemotherapeutics for the treatment of keloid scars. Dermatol. Rep. 21(7), 5880 (2015).
Li, K. et al. The 1470 nm diode laser with an intralesional fiber device: a proposed solution for the treatment of inflamed and infected keloids. Burns Trauma 15(7), 5 (2019).
Seitz, M. et al. The diode laser: a novel side-firing approach for laser vaporisation of the human prostate–immediate efficacy and 1-year follow-up. Eur Urol. 52, 1717–1722 (2007).
Seitz, M. et al. Ex vivo and in vivo investigations of the novel 1,470 nm diode laser for potential treatment of benign prostatic enlargement. Lasers Med. Sci. 24, 419–424 (2009).
Shaydakov, E., Ilyukhin, E. & Rosukhovskiy, D. Blood absorption during 970 and 1470 nm laser radiation in vitro. Int. Angiol. 34, 475–482 (2015).
Durani, P., McGrouther, D. A. & Ferguson, M. W. J. Current scales for assessing human scarring: a review. J. Plast. Reconstr. Aesth. Surg. 62, 713–720 (2009).
Reinholz, M. et al. The dermatology life quality index as a means to assess life quality in patients with different scar types. J. Eur. Acad. Dermatol. Venereol. 29, 2112–2119 (2015).
Seo, S. R., Kang, N. O., Yoon, M. S., Lee, H. J. & Kim, D. H. Measurements of scar properties by SkinFibroMeter, SkinGlossMeter, and Mexameter and comparison with Vancouver Scar Scale. Skin Res. Technol. 23, 295–302 (2017).
Hu, Z. et al. High-precision, non-invasive anti-microvascular approach via concurrent ultrasound and laser irradiation. Sci. Rep. 11(7), 40243 (2017).
Castro, D. J. et al. Effects of the Nd:YAG laser on DNA synthesis and collagen production in human skin fibroblast cultures. Ann. Plast. Surg. 11, 214–222 (1983).
Haedersdal, M. Cutaneous side effects from laser treatment of the skin: skin cancer, scars, wounds, pigmentary changes, and purpura—use of pulsed dye laser, copper vapor laser, and argon laser. Acta Derm. Venereol. Suppl. (Stockh). 207, 1–32 (1999).
Kirschner, R. A. Cutaneous plastic surgery with the CO2 laser. Surg. Clin. North Am. 64, 871–883 (1984).
Avram, M. M., Tope, W. D., Yu, T., Szachowicz, E. & Nelson, J. S. Hypertrophic scarring of the neck following ablative fractional carbon dioxide laser resurfacing. Lasers Surg. Med. 41, 185–188 (2009).
Reiken, S. R. et al. Control of hypertrophic scar growth using selective photothermolysis. Lasers Surg. Med 21, 7–12 (1997).
Nicoli, F. et al. Correction of cicatricial ectropion using non-ablative fractional laser resurfacing. Lasers Med. Sci. 34, 79–84 (2019).
Rivera, A. E. Acne scarring: a review and current treatment modalities. J. Am. Acad. Dermatol. 59, 659–676 (2008).
Seago, M. et al. Laser treatment of traumatic scars and contractures: 2020 international consensus recommendations. Lasers Surg. Med. 52, 96–116 (2020).
Pannier, F., Rabe, E. & Maurins, U. First results with a new 1470-nm diode laser for endovenous ablation of incompetent saphenous veins. Phlebology 24, 26–30 (2009).
Park, J. A. et al. The 1,470-nm bare-fiber diode laser ablation of the great saphenous vein and small saphenous vein at 1-year follow-up using 8–12 W and a mean linear endovenous energy density of 72 J/cm. J. Vasc. Interv. Radiol. 25, 1795–1800 (2014).
Sroka, R., Rösler, P., Janda, P., Grevers, G. & Leunig, A. Endonasal laser surgery with a new laser fiber guidance instrument. Laryngoscope 110(2 Pt 1), 332–334 (2000).
Oztürk, E. & Gülcü, B. Laser ablation of fistula tract: a sphincter-preserving method for treating fistula-in-ano. Dis. Colon Rectum 57, 360–364 (2014).
Leclère, F. M. et al. Laser-assisted lipolysis for arm contouring in Teimourian grades I and II: a prospective study of 45 patients. Lasers Med. Sci. 30, 1053–1059 (2015).
Nicoli, F. et al. Free lymph node flap transfer and laser-assisted liposuction: a combined technique for the treatment of moderate upper limb lymphedema. Lasers Med. Sci. 30, 1377–1385 (2015).
Matsumoto, K., Suzuki, H., Usami, Y., Hattori, M. & Komoro, T. Histological evaluation of artifacts in tongue tissue produced by the CO2 laser and the electrotome. Photomed. Laser Surg. 26, 573–577 (2008).
Havel, M., Betz, C. S., Leunig, A. & Sroka, R. Diode laser-induced tissue effects: in vitro tissue model study and in vivo evaluation of wound healing following non-contact application. Lasers Surg. Med. 46, 449–455 (2014).
Abergel, R. P. et al. Biochemical composition of the connective tissue in keloids and analysis of collagen metabolism in keloid fibroblast cultures. J. Investig. Dermatol. 84, 384–390 (1985).
Miles, C. A., Burjanadze, T. V. & Bailey, A. J. The kinetics of the thermal denaturation of collagen in unrestrained rat tail tendon determined by differential scanning calorimetry. J. Mol. Biol. 27(245), 437–446 (1995).
Zeeman, R. et al. Successive epoxy and carbodiimide cross-linking of dermal sheep collagen. Biomaterials 20(921–31), 30 (1999).
Manuskiatti, W., Triwongwaranat, D., Varothai, S., Eimpunth, S. & Wanitphakdeedecha, R. Efficacy and safety of a carbon-dioxide ablative fractional resurfacing device for treatment of atrophic acne scars in Asians. J. Am. Acad. Dermatol. 63, 274–283 (2010).
Ogawa, R. & Akaishi, S. Endothelial dysfunction may play a key role in keloid and hypertrophic scar pathogenesis - Keloids and hypertrophic scars may be vascular disorders. Med. Hypotheses 96, 51–60 (2016).
Wittenberg, G. P. et al. Prospective, single-blind, randomized, controlled study to assess the efficacy of the 585-nm flash lamp pumped pulsed-dye laser and silicone gel sheeting in hypertrophic scar treatment. Arch. Dermatol. 135, 1049–1055 (1999).
Giudice, G. et al. Use of the stromal vascular fraction in intermediate-deep acute burns: a case with its own control. J. Burn Care Res. 17(39), 846–849 (2018).
Fioramonti, P., Maruccia, M., Ruggieri, M. & Onesti, M. G. A rare case of lymphangioma in the gluteal region: surgical treatment combined with sclerotherapy and laser therapy. Aesthet. Plast. Surg. 37, 960–964 (2013).
Balzani, A. et al. Efficacy of a novel optimized pulsed light source (MaxG) for the treatment of facial vascular lesions. Photomed. Laser Surg. 35, 12–17 (2017).
Spiekman, M. et al. The power of fat and its adipose-derived stromal cells: emerging concepts for fibrotic scar treatment. J. Tissue Eng. Regen. Med. 11, 3220–3235 (2017).
Author information
Authors and Affiliations
Contributions
K.L., F.N., C.C.: these authors equally contributed to this work. Drafted the manuscript, designed the study and wrote original draft. A.A-M., L.N.: were involved in writing, review and editing the manuscript and language. A.B.: was involved in conceptualization, planning and supervised the work. WJ.X., Z.Z., R.S.: performed the measurements, collect data and pictures, processed the experimental data, performed the analysis. Y.T. and Y.Z.: aided in interpreting the results, conducted experiments, deal with project administration and supervised the manuscript and participants. K.L. and Y.Z.: took photographs. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary video.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, K., Nicoli, F., Cui, C. et al. Treatment of hypertrophic scars and keloids using an intralesional 1470 nm bare-fibre diode laser: a novel efficient minimally-invasive technique. Sci Rep 10, 21694 (2020). https://doi.org/10.1038/s41598-020-78738-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-78738-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.