Abstract
Swimming is an extremely popular sport around the world. The streamlined body position is a crucial and foundational position for swimmers. Since the density of lungs is low, the center of buoyancy is always on the cranial side and the center of gravity is always on the caudal side. It has been reported that the greater the distance between the centers of buoyancy and gravity, the swimmer’s legs will sink more. This is disadvantageous to swimming performance. However, the way to reduce the distance between the centers of buoyancy and gravity is yet to be elucidated. Here we show that swimmers with high gliding performance exhibit different abdominal cavity shapes in the streamlined body position, which causes cranial movement of the abdominal organs. This movement can reduce the distance between the centers of buoyancy and gravity, prevent the legs from sinking, and have a positive effect on gliding performance.
Similar content being viewed by others
Introduction
The streamlined body position is a crucial and foundational position for swimmers; furthermore, gliding ability plays an important role in race performance1. In the streamlined body position, the swimmer places one hand over the other, with their fingers overlapping, raises their arms above their head, straightens their legs, and plantar flexes their feet2,3. Swimming is one of the most challenging locomotion techniques for humans, and achieving a streamlined body position is important for improving swimming performance4,5. The glide phase occurs when the swimmer attempts to travel through the water while maintaining the streamlined body position and taking no other action6. One indicator of gliding performance is the distance traveled during glide intervals6,7. This is significantly affected by drag because swimming is performed in water, which has a greater density than air1,4,8. The drag force is referred to as “passive” if the participant is being towed or is gliding, with no limb movement, whereas it is considered “active drag” when the swimmer is propelling themself5. Hence, to improve gliding performance, passive drag must be reduced.
The streamlined body position is acted upon by two forces in the water: buoyancy and gravity9. Because the chest contains the air-filled lungs, which have a lower density than the surrounding water, the center of buoyancy is located here, on the cranial side of the torso, whereas the center of gravity is always on the caudal side10. When submerged, an object will rotate until the centers of buoyancy and gravity are vertically aligned. In the human body, this rotation in the streamlined body position causes the legs to sink11,12 (Fig. 1a). This has been studied as underwater torque, which is one of the main factors increasing drag in swimming12,13,14. A previous study found that the greater the distance between the centers of buoyancy and gravity, the greater the underwater torque15. Thus, for reducing the effects of drag, investigating ways to reduce the distance between the centers of buoyancy and gravity in the streamlined body position is important. Some swimmers are able to keep their legs afloat in the streamlined body position without any significant action (e.g., kicking) (Supplementary Movie 1). As shown in the video, these swimmers use their abdominal muscles to draw in the belly, which in turn causes the legs to float. This phenomenon can be explained by abdominal contraction reducing the distance between the center of buoyancy and the center of gravity. Abdominal contraction may improve gliding performance because if the legs do not sink, the frontal surface area is reduced. As the frontal surface area decreases, the swimmer’s drag decreases6,16. However, the mechanism underlying the reduced distance between the centers of buoyancy and gravity when the belly is drawn in has not yet been elucidated.
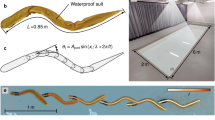
Streamlined body position. (a) Schematic of the streamlined body position in water. The white circle indicates the center of buoyancy, and the black circle indicates the center of gravity. The white arrow indicates buoyancy force, and black arrows indicate gravity force. Since lungs have lower density and are located in the chest, the center of buoyancy is always on the cranial side and the center of gravity is always on the caudal side. An object rotates in water until the center of buoyancy and the center of gravity are aligned vertically. As shown, sinking legs lead to increased frontal surface area. This is disadvantageous to swimming performance. (b) Glide is the phase in which the swimmer attempts to maintain speed in the streamlined body position without using actions to propel the body. (c) Participants were positioned in the prone position on the patient table.
The abdominal cavity is a large body cavity in humans and contains many organs. The average weights of abdominal organs in healthy males (18.5 kg/m2 ≤ body mass index (BMI) < 25 kg/m2) are as follows: liver, 1414 g (range 838–2013 g); right kidney, 121 g (range 84–200 g); left kidney, 129 g (range 93–201 g); and spleen, 127 g (range 53–299 g)17. The abdominal cavity also contains luminal organs, which have weights that are greatly affected by meals. It has been reported that these heavy abdominal organs can change their location in the abdomen depending on posture18,19,20,21. Hence, our hypothesis is that a swimmer with high gliding performance is able to move their abdominal organs to the cranial side of the abdominal cavity, thereby reducing the distance between the centers of buoyancy and gravity.
MR examination for swimmers
Seventeen male college swimmers participated in this study. The participants’ gliding distances in the streamlined body position were measured in a pool and used to divide participants into two groups (Fig. 1b). In the high performance group, the average gliding distance was > 10 m, whereas in the low performance group, the average gliding distance was < 10 m. No significant difference in characteristics with regard to age, height, weight, body mass index (BMI), body surface area (BSA), upper limb length, lower limb length, and shoulder width was noted between participants in the high performance group and those in the low performance group (Table 1). Participants were asked to lie in the prone position and MRI measurements were taken in resting and prescribed streamlined body positions (Fig. 1c). In order to examine the abdominal cavity shape, cross-sectional area (CSA) was measured at three levels along the torso: upper liver level, lower lung level, and umbilical level (Fig. 2).
Measurements of the cross-sectional area of the abdominal cavity at three levels. 1 Axial MR image at the upper liver level showing measurements of cross-sectional area. 2 Axial MR image at the lower lung level showing measurements of cross-sectional area. 3 Axial MR image at the umbilical level showing measurements of cross-sectional area and thickness of the abdominal muscles. 4 Sagittal MR image at the left kidney level showing abdominal cavity, which is marked in yellow. (1) indicates the upper liver level, (2) indicates the lower lung level, and (3) indicates the umbilical level.
Results
CSA within-group comparisons
In the high performance group, CSA increased significantly from the resting to the streamlined body positions at the upper liver and lower lung levels, and decreased significantly at umbilical level (Wilcoxon signed rank test, P < 0.01, P < 0.01, P < 0.01, respectively). In the low performance group, the CSA increased significantly only at the upper liver level, and there were no significant changes at the lower lung and umbilical levels (Wilcoxon signed rank test, P = 0.027, P = 0.65, P = 0.25, respectively) (Fig. 3a) (Table 2). Furthermore, we measured the maximum thickness of the rectus abdominis muscle (RA) and antero-lateral abdominal wall, which is composed of the transversus abdominis muscle (TrA), the external oblique muscle (EO), and internal oblique muscle (IO), at the umbilical level (TrA + EO + IO). In both high and low performance groups, the thickness of the TrA + EO + IO increased significantly from the resting to the streamlined body positions at the umbilical level (Wilcoxon signed rank test, P = 0.016, P = 0.012, respectively). However, in both the high and low performance groups, the thickness of the RA was not significantly altered (Wilcoxon signed rank test, P = 0.35, P = 0.25, respectively) (Table 2).
Representative case presentation and changes in CSA between study groups. (a) The sagittal MR images at the left kidney level show abdominal cavity, which is marked in yellow. (1) indicates the upper liver level, (2) indicates the lower lung level, and (3) indicates the umbilical level. (a-A,A’) Case 1 is a representative case of the high performance group: the years of swimming practice (YSP) 8 years, gliding distance 13 m. From the resting position (A) to the streamlined body position (A’). a-B,B’, Case 2 is the representative case for the low performance group: YSP 1 year, gliding distance 8 m. From the resting position (B) to the streamlined body position (B’). (b–d) Box plot of the changes in CSA between the high performance and low performance groups at three levels: upper liver level (b), lower lung level (c), umbilical level (d). Box plots show the median (center line), interquartile range (hinges), 10th and 90th percentiles (whiskers). CSA in the high performance group (as shown in Case 1: a-A, A’) increased at the upper liver and lower lung levels, and decreased at the umbilical level in the streamlined body position. These changes in CSA are significantly higher than those of the low performance group at all three levels (b–d).
Changes in CSA between study groups
In the high performance group, changes in CSA from resting to streamlined body positions were: at upper liver level 36.8 cm2 (median, interquartile range 28.7 to 67.3), at lower lung level 23.2 cm2 (17.3–44.2), at umbilical level − 37.4 cm2 (− 47.4 to − 24.9). In the low performance group, the following changes occurred in the CSA when transitioning from the resting to streamlined body positions: 16.4 cm2 (9.3–22.2) at the upper liver level, − 5.1 cm2 (− 15.4 to 11.6) at the lower lung level, and − 1.2 cm2 (− 18.1 to 0.75) at the umbilical level. The changes in the CSA were significantly greater in the high performance group than in the low performance group at all three levels: upper liver level, lower lung level, umbilical level (Mann–Whitney U test, P = 0.036, P < 0.01, P < 0.01, respectively) (Fig. 3b–d) (Table 3). These change in abdominal cavity shape can cause cranial side movement of abdominal organs.
Representative case presentation
In order to compare the characteristics of the high and low performance groups, we present two cases. Case 1 is the representative case for the high performance group: height 1.80 m, weight 79 kg, the years of swimming practice (YSP) 8 years, and gliding distance 13 m. From the resting position to the streamlined body position, CSA at the upper liver level changed from 354.3 to 437.9 cm2, CSA at the lower lung level changed from 318.9 to 326.2 cm2, CSA at the umbilical level changed from 122.8 to 71.7 cm2 (Fig. 3a-A,A'). Case 2 is the representative case for the low performance group; height 1.67 m, weight 60 kg, YSP 1 year, and gliding distance 8 m. From the resting position to the streamlined body position, CSA at the upper liver level changed from 303.9 to 313.2 cm2, CSA at the lower lung level changed from 230.6 to 245.1 cm2, and CSA at the umbilical level changed from 113.8 to 112.5 cm2 (Fig. 3a-B,B'). Swimmers with high performance group (as shown in Case 1) exhibit different abdominal cavity shapes in the streamlined body position compared to low performance group (as show in Case 2), which can cause cranial-side movement of abdominal organs.
Discussion
Cranial-side movement of abdominal organs
To our knowledge, this is the first study to analyze the change in abdominal cavity shape in the streamlined body position by MRI. Results indicated that the high performance group experienced significant changes in CSA in the streamlined body position compared to those of the low performance group. Specifically, CSA of the high performance group increased at the upper liver and lower lung levels, and decreased at the umbilical level in the streamlined body position. This change in abdominal cavity shape may cause cranial-side movement of abdominal organs (Fig. 3a). When these abdominal organs move to the cranial side in the streamlined body position, the center of gravity accordingly shifts to the cranial side.
Center of gravity
The center of gravity is an imaginary point through which the gravitational force acts on an object22. In order to objectively measure the movement of the center of gravity from the resting to the streamlined body positions, we further recruited 10 from the 17 participants who underwent MR imaging. Five participants were the high performance group, and other five participants were the low performance group. No significant differences in characteristics with regard to height, the length between the tip of the longest finger and the soles of the feet, and BMI were observed between participants in the high performance group and those in the low performance group (Table 4). The center of gravity was measured using the reaction board method10,23, and ground reaction force data were acquired using 90 × 60 cm force plates (Anima Corporation, Chofu, Tokyo, Japan)24. The participants were placed on the balance board in the supine position, and measurements were performed in both the streamlined body position and resting position. For the streamlined body position, participants were instructed to hold a streamlined position as if underwater. For the resting position, participants were instructed to raise their arms above their head, like in the streamlined body position, and to relax their body.
The measurement point was at the tip of the finger, and the pivot point was at the soles of the feet (Supplementary Fig. 1). The center of gravity was calculated as follows:
where CG is the distance from the feet to the participant’s center of gravity, L is the length between the tip of the longest finger and the soles of the feet, RF is the reaction force at the tip of the finger (without the weight of the balance board itself), and W is the participant’s weight. The movement of CG from the resting position to the streamlined body position (CGx) was calculated as follows:
where CG1 is the center of gravity in the streamlined body position, CG2 is the center of gravity in the resting position, RF1 is the center of gravity in the streamlined body position, and RF2 is the center of gravity in the resting position. To perform comparisons between the participants, CGx was expressed as a percentage (CGx%) of each participant’s length between the tip of the longest finger and the soles of the feet as follows:
The median CGx% was 0.334 (interquartile range 0.184–0.359) in the high performance group and 0.079 (0.048–0.170) in the low performance group. CGx% was significantly greater in the high performance group compared with the low performance group (Mann–Whitney U test, P = 0.032) (Table 4). The CGx% was a positive value, which means that the center of gravity moved cranially when the participants were instructed to assume the streamlined body position. Furthermore, the movement of the center of gravity was significantly greater in the high performance group. These results are consistent with our MRI findings and underpin our hypothesis. In other words, the cranial movement of the center of gravity is caused by the cranial movement of the abdominal organs and activated abdominal muscles. This movement of center of gravity reduces the distance between the centers of buoyancy and gravity, causing decreased underwater torque.
Underwater torque
Underwater torque is one of the main effects of increasing the frontal surface area in the streamlined body position25. As increased frontal surface area leads to increased drag, swimmers aim to reduce their underwater torque26. The center of buoyancy in the human body is always on the cranial side, and the center of gravity is always on the caudal side10. The distance between the centers of buoyancy and gravity varies from individual to individual and depends on factors such as age, gender, and body composition9,16,26. Usually, the distance is greater for males than females12 and for adults compared with children14,27. In body composition studies, the density of the fat component is assumed to be approximately 0.9007 g/cm3 and that of fat-free muscle is approximately 1.066 g/cm3 28. Because water density is 1.00 g/cm3, fat will float and muscle will sink. When the fat component is concentrated in the lower body, the distance between the centers of buoyancy and gravity decreases15. The use of buoyancy tools such as pull buoys (which are widely used as swimming tools around the world) also affects underwater torque. Swimmers place the pull buoy between their thighs or their ankles while swimming and the pull buoy reduces underwater torque by providing improved flotation support for hips and legs15. However, swimmers are not allowed to use support tools in competitions. It is impossible to change the age of a swimmer, and impractical to alter gender, and body composition cannot be changed within a short period of time. Hence, it is imperative that swimmers find other techniques to reduce underwater torque.
Swimmer’s drag and body surface area
Underwater torque increases the frontal surface area, which causes the swimmer’s drag to increase. In addition to the increasing frontal surface area due to underwater torque, the BSA is also an important factor affecting a swimmer’s drag3,4. Frictional drag, which is one type of swimmers’ drag, can be calculated from the density of the water, the gliding velocity, and the BSA by numerical simulations (computer fluid dynamics, CFD)5,29. The BSA was calculated using Du Bois formulas30 based on height and weight. In our study, there was no significant difference in the BSA between participants in the high performance group and those in the low performance group.
Abdominal drawing-in maneuver
In our study, the thickness of the antero-lateral abdominal wall was increased and CSA was decreased at the umbilical level in the streamlined body position. This action is similar to the abdominal drawing-in maneuver (ADIM), a body trunk exercise. The ADIM is a method which increases abdominal pressure by pulling the abdominal walls to the inside that the TrA and oblique abdominal muscles are contracted31. Because lumbar trunk stability is effectively accomplished through the increase in abdominal pressure, the ADIM has been reported to be important in the rehabilitation of lower back pain32,33. The action of the abdominal wall muscles during ADIM has been studied using ultrasound imaging, MRI, and electromyography34,35. The muscle bellies of the antero-lateral abdominal wall are observed to thicken and shorten in length during the ADIM, whereas the RA muscle does not thicken during the ADIM34,36. These muscle thickening responses were also observed in our study. From the resting position to the streamlined body position, the thickness of the antero-lateral abdominal wall muscle increased significantly, whereas the thickness of the RA muscle did not change significantly. A previous study also reported that the CSA of the trunk (including the abdominal cavity and trunk muscles but excluding subcutaneous tissue) at the L3–L4 disc decreased with the ADIM32,34. This CSA differed from our study in the point of measurement range and measurement height; however, the observed decrease in the CSA at the umbilical level was also observed in our study. Therefore, despite being instructed to only hold the streamlined position, it appears that these participants also unconsciously made similar movements to the ADIM. Since abdominal contraction, similar to ADIM, occurred unconsciously, it was impossible to assess the gliding distance both with and without abdominal contraction.
Limitations
There are several limitations to this study. First, the streamlined body position in MRI may not be exactly the same as the streamlined body position in water. However, it is impossible to take MR images in water because of the interference caused by water. To ameliorate this, we used a soft cushion on the ventral side so that participants could hold the streamlined body position easily, and checked the body position through the monitor during MRI scans. Second, while swimming, swimmers do not hold their breath but breath out normally under water. This differs from the glide phase performed in our study. We measured the maximum gliding distance and had the participants hold their breath while gliding. Thus, our study results based on this glide may not be completely applicable to normal swimming. Third, although gliding distance is mainly influenced by drag, other factors such as physique, push-off characteristics, or swimsuits also may have an effect37,38,39. In this sense, some precaution should be taken when interpreting gliding performance because it can have some biases40. Our participants were college swimmers, so they were not beginner and had a certain amount of swimming ability. There was no physical difference between the two groups. They all wore standard swimsuits, rather than high-performance swimsuits. Therefore, we assumed that these factors are comparable between the two groups in this study.
Conclusion
Swimmers with high gliding performance exhibit different abdominal cavity shapes in the streamlined body position compared to low performance group, which causes cranial movement of abdominal organs. This movement may reduce underwater torque, prevent the legs from sinking during swimming, and have a positive effect on gliding performance.
Methods
This prospective study was conducted at Tohoku University Hospital in Japan. The study was approved by the ethics committee of Tohoku University (approval number: 15263), and all procedures were performed in accordance with the approved guidelines. Written informed consent was obtained from all participants. Written informed consent was obtained from the swimmer in Supplementary Movie 1 for publication in an online open-access publication.
Participants
In this prospective cohort study, we consecutively recruited healthy, male college swimmers between 20 and 30 years old, with BMI of 18–25. Swimmers were excluded if they experienced any pain in the streamlined body position, had neurological or respiratory disorders, had contraindications to MRI, such as claustrophobia, or were deemed by doctors as inappropriate for study participation. Seventeen male college swimmers participated in this study.
Measurements on land
The height, weight, upper extremity length, lower extremity length, and shoulder width of each participant was measured by an experienced physiotherapist. Upper extremity length was measured from the acromion process to the tip of the radial styloid process. Lower extremity length was measured from the anterior superior iliac spine to the medial malleolus. Shoulder width was measured between the acromion processes. BSA was calculated using Du Bois formulas30 based on height and weight.
Gliding distance
The gliding distance in the streamlined body position was measured for each participant in an indoor pool (Fig. 1b). Gliding distance was defined as the maximum distance they could cover in the streamlined body position without any arm strokes or kicks. The pool was 25 m long, and the depth of the pool was 1.3 m. Participants used standard swimsuits. They began by standing on the pool floor, then submerged and maximally pushed off the wall in the streamlined body position. The distance between the pool wall and the tip of the participant’s hand was accurately measured when gliding ceased. This measurement was performed five times for each participant and the average measurement was used for sorting participants into two groups for comparison: the high performance group, in which average gliding distance was > 10 m, and the low performance group, in which average gliding distance was < 10 m.
MR examination
MR examinations were performed using a 3.0-T whole-body MR scanner (Ingenia 3.0 T, Philips Medical Systems, Best, the Netherlands) with a 32-channel torso coil. Participants were placed in the prone position with their hands up and overlapping, head between the extended arms, feet together and plantar flexed (Fig. 1c). A soft cushion was used on the ventral side so that the prone position could be held without difficulty. MR scans were performed in both the resting and streamlined body positions.
Streamlined body position Participants were instructed to hold the streamlined body position as if underwater, inhale, and hold their breath.
Resting position Participants were instructed to relax their body, inhale the same amount of air as they had in the streamlined body position and hold their breath.
The volume of air-intake was determined by the participants, provided that air intake in the resting position was equal to that in the streamlined body position using auto voice. Two-dimensional (2D) turbo-spin echo T2-weighted images were acquired in sagittal orientations with repetition time (TR) = 2441 ms, echo time (TE) = 135 ms, flip angle (FA) = 90°, slice thickness = 4 mm, acquisition matrix = 380 × 225, field of view = 38 cm × 38 cm. Three-dimensional (3D) T1-weighted fast-field echo (enhanced T1 high-resolution isotropic volume excitation [eTHRIVE, Philips Medical Systems]) images were acquired in coronal orientations with TR = 3.7 ms, TE = 1.8 ms, FA = 10°, slice thickness = 4 mm, field of view = 50 cm × 50 cm. 2D T2-weighted and 3D T1-weighted images were acquired to cover the body trunk in two stations. Each MRI scan was performed within 20 s, during which the participants held their breath. The multi-station images were combined into a single full-field view on the MR console (MR MobiView, Philips Medical Systems).
MR image analysis
The MR images were analyzed using a commercially available workstation (Ziostation2; Ziosoft, Tokyo, Japan). The acquired T1-weighted images were reconstructed into axial images. In order to evaluate the cranial movement of abdominal organs, CSA of the abdominal cavity was measured at three levels: upper liver level, lower lung level, and umbilical level. In this study, retroperitoneal space was included in CSA, but the aorta and inferior vena cava were excluded because they were relatively fixed. Furthermore, we measured the maximum thickness of the RA and antero-lateral abdominal wall, composed of the TrA and the EO and IO, at umbilical level (TrA + EO + IO). Thicknesses of muscles were measured on both right and left sides, and an average value for each was calculated (Fig. 2).
Statistical analysis
All statistical analyses were performed with EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan)41, which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). More precisely, it is a modified version of the R commander designed to add statistical functions frequently used in biostatistics. All continuous variables were presented as medians with interquartile ranges. The Mann–Whitney U test was used for between-group comparisons, and the Wilcoxon signed rank test was used for within-group comparisons. All P-values of < 0.05 were considered statistically significant.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Chatard, J. C., Lavoie, J. M., Bourgoin, B. & Lacour, J. R. The contribution of passive drag as a determinant of swimming performance. Int. J. Sports Med. 11, 367–372. https://doi.org/10.1055/s-2007-1024820 (1990).
Vilas-Boas, J. P. et al. Determination of the drag coefficient during the first and second gliding positions of the breaststroke underwater stroke. J. Appl. Biomech. 26, 324–331. https://doi.org/10.1123/jab.26.3.324 (2010).
Zamparo, P., Gatta, G., Pendergast, D. & Capelli, C. Active and passive drag: The role of trunk incline. Eur. J. Appl. Physiol. 106, 195–205. https://doi.org/10.1007/s00421-009-1007-8 (2009).
Barbosa, T. M., Yam, J. W., Lum, D., Balasekaran, G. & Marinho, D. A. Arm-pull thrust in human swimming and the effect of post-activation potentiation. Sci. Rep. 10, 8464. https://doi.org/10.1038/s41598-020-65494-z (2020).
Barbosa, T. M., Ramos, R., Silva, A. J. & Marinho, D. A. Assessment of passive drag in swimming by numerical simulation and analytical procedure. J. Sports Sci. 36, 492–498. https://doi.org/10.1080/02640414.2017.1321774 (2018).
Naemi, R., Easson, W. J. & Sanders, R. H. Hydrodynamic glide efficiency in swimming. J. Sci. Med. Sport 13, 444–451. https://doi.org/10.1016/j.jsams.2009.04.009 (2010).
Starling, R. D. et al. Effect of swimming suit design on the energy demands of swimming. Med. Sci. Sports Exerc. 27, 1086–1089. https://doi.org/10.1249/00005768-199507000-00019 (1995).
Scurati, R., Gatta, G., Michielon, G. & Cortesi, M. Techniques and considerations for monitoring swimmers’ passive drag. J. Sports Sci. 37, 1168–1180. https://doi.org/10.1080/02640414.2018.1547099 (2019).
Zamparo, P. et al. Effects of body size, body density, gender and growth on underwater torque. Scand. J. Med. Sci. Sports 6, 273–280. https://doi.org/10.1111/j.1600-0838.1996.tb00470.x (1996).
Kjendlie, P. L., Stallman, R. K. & Stray-Gundersen, J. Passive and active floating torque during swimming. Eur. J. Appl. Physiol. 93, 75–81. https://doi.org/10.1007/s00421-004-1165-7 (2004).
Yanai, T. Rotational effect of buoyancy in frontcrawl: Does it really cause the legs to sink?. J. Biomech. 34, 235–243. https://doi.org/10.1016/s0021-9290(00)00186-x (2001).
McLean, S. P. & Hinrichs, R. N. Sex differences in the centre of buoyancy location of competitive swimmers. J. Sports Sci. 16, 373–383. https://doi.org/10.1080/02640419808559365 (1998).
Gagnon, M. & Montpetit, R. Technological development for the measurement of the center of volume in the human body. J. Biomech. 14, 235–241. https://doi.org/10.1016/0021-9290(81)90068-3 (1981).
Zamparo, P., Capelli, C., Termin, B., Pendergast, D. R. & di Prampero, P. E. Effect of the underwater torque on the energy cost, drag and efficiency of front crawl swimming. Eur. J. Appl. Physiol. 73, 195–201. https://doi.org/10.1007/bf02425476 (1996).
Cortesi, M., Fantozzi, S., Di Michele, R., Zamparo, P. & Gatta, G. Passive drag reduction using full-body swimsuits: the role of body position. J. Strength Condit. Res. 28, 3164–3171. https://doi.org/10.1519/jsc.0000000000000508 (2014).
Zamparo, P. et al. The interplay between propelling efficiency, hydrodynamic position and energy cost of front crawl in 8 to 19-year-old swimmers. Eur. J. Appl. Physiol. 104, 689–699. https://doi.org/10.1007/s00421-008-0822-7 (2008).
Molina, D. K. & DiMaio, V. J. Normal organ weights in men: Part II-the brain, lungs, liver, spleen, and kidneys. Am. J. Forens. Med. Pathol. 33, 368–372. https://doi.org/10.1097/PAF.0b013e31823d29ad (2012).
Hayes, A. R., Gayzik, F. S., Moreno, D. P., Martin, R. S. & Stitzel, J. D. Abdominal organ location, morphology, and rib coverage for the 5(th), 50(th), and 95(th) percentile males and females in the supine and seated posture using multi-modality imaging. Ann. Adv. Automot. Med. 57, 111–122 (2013).
Hayes, A. R., Gayzik, F. S., Moreno, D. P., Martin, R. S. & Stitzel, J. D. Comparison of organ location, morphology, and rib coverage of a midsized male in the supine and seated positions. Comput. Math. Methods Med. 2013, 419821. https://doi.org/10.1155/2013/419821 (2013).
Deukmedjian, A. R., Le, T. V., Dakwar, E., Martinez, C. R. & Uribe, J. S. Movement of abdominal structures on magnetic resonance imaging during positioning changes related to lateral lumbar spine surgery: A morphometric study: Clinical article. J. Neurosurg. Spine 16, 615–623. https://doi.org/10.3171/2012.3.spine1210 (2012).
Howes, M. K., Hardy, W. N. & Beillas, P. The effects of cadaver orientation on the relative position of the abdominal organs. Ann. Adv. Automot. Med. 57, 209–224 (2013).
Hochstein, S., Baumgart, M., Müller, R. & Blickhan, R. Determine the center of mass position in human undulatory swimming: A static approach. Int. J. Sports Sci. Phys. Educ. 1, 21–27. https://doi.org/10.11648/j.ijsspe.20160102.12 (2016).
Watanabe, Y., Wakayoshi, K. & Nomura, T. New evaluation index for the retainability of a swimmer’s horizontal posture. PLoS ONE 12, e0177368. https://doi.org/10.1371/journal.pone.0177368 (2017).
Sekiguchi, Y. et al. Ankle-foot orthosis with dorsiflexion resistance using spring-cam mechanism increases knee flexion in the swing phase during walking in stroke patients with hemiplegia. Gait Post. 81, 27–32. https://doi.org/10.1016/j.gaitpost.2020.06.029 (2020).
Capelli, C. et al. Bioenergetics and biomechanics of front crawl swimming. J. Appl. Physiol. 78, 674–679. https://doi.org/10.1152/jappl.1995.78.2.674 (1995).
Zamparo, P., Capelli, C. & Pendergast, D. Energetics of swimming: A historical perspective. Eur. J. Appl. Physiol. 111, 367–378. https://doi.org/10.1007/s00421-010-1433-7 (2011).
Kjendlie, P. L., Ingjer, F., Stallman, R. K. & Stray-Gundersen, J. Factors affecting swimming economy in children and adults. Eur. J. Appl. Physiol. 93, 65–74. https://doi.org/10.1007/s00421-004-1164-8 (2004).
Prior, B. M. et al. Muscularity and the density of the fat-free mass in athletes. J. Appl. Physiol. 90, 1523–1531. https://doi.org/10.1152/jappl.2001.90.4.1523 (2001).
Barbosa, T. M. et al. A comparison of experimental and analytical procedures to measure passive drag in human swimming. PLoS ONE 10, e0130868. https://doi.org/10.1371/journal.pone.0130868 (2015).
Du Bois, D. & Du Bois, E. F. A formula to estimate the approximate surface area if height and weight be known 1916. Nutrition 5, 303–311 (1989).
Park, S. D. & Yu, S. H. The effects of abdominal draw-in maneuver and core exercise on abdominal muscle thickness and Oswestry disability index in subjects with chronic low back pain. J. Exerc. Rehab. 9, 286–291. https://doi.org/10.12965/jer.130012 (2013).
Hides, J. et al. An MRI investigation into the function of the transversus abdominis muscle during “drawing-in” of the abdominal wall. Spine 31, E175-178. https://doi.org/10.1097/01.brs.0000202740.86338.df (2006).
Kaping, K., Ang, B. O. & Rasmussen-Barr, E. The abdominal drawing-in manoeuvre for detecting activity in the deep abdominal muscles: Is this clinical tool reliable and valid?. BMJ Open 5, e008711. https://doi.org/10.1136/bmjopen-2015-008711 (2015).
Hides, J. A., Boughen, C. L., Stanton, W. R., Strudwick, M. W. & Wilson, S. J. A magnetic resonance imaging investigation of the transversus abdominis muscle during drawing-in of the abdominal wall in elite Australian Football League players with and without low back pain. J. Orthopaed. Sports Phys. Therapy 40, 4–10. https://doi.org/10.2519/jospt.2010.3177 (2010).
Chon, S. C., Chang, K. Y. & You, J. S. Effect of the abdominal draw-in manoeuvre in combination with ankle dorsiflexion in strengthening the transverse abdominal muscle in healthy young adults: A preliminary, randomised, controlled study. Physiotherapy 96, 130–136. https://doi.org/10.1016/j.physio.2009.09.007 (2010).
Badiuk, B. W., Andersen, J. T. & McGill, S. M. Exercises to activate the deeper abdominal wall muscles: the Lewit: A preliminary study. J. Strength Condit. Res. 28, 856–860. https://doi.org/10.1519/JSC.0b013e3182aac3f3 (2014).
Chatard, J. C. & Wilson, B. Effect of fastskin suits on performance, drag, and energy cost of swimming. Med. Sci. Sports Exerc. 40, 1149–1154. https://doi.org/10.1249/MSS.0b013e318169387b (2008).
Mollendorf, J. C., Termin, A. C. 2nd., Oppenheim, E. & Pendergast, D. R. Effect of swim suit design on passive drag. Med. Sci. Sports Exerc. 36, 1029–1035. https://doi.org/10.1249/01.mss.0000128179.02306.57 (2004).
Roberts, B. S., Kamel, K. S., Hedrick, C. E., McLean, S. P. & Sharp, R. L. Effect of a FastSkin suit on submaximal freestyle swimming. Med. Sci. Sports Exerc. 35, 519–524. https://doi.org/10.1249/01.mss.0000053699.91683.cd (2003).
Barbosa, T. M. et al. How informative are the vertical buoyancy and the prone gliding tests to assess young swimmers’ hydrostatic and hydrodynamic profiles?. J. Hum. Kinet. 32, 21–32. https://doi.org/10.2478/v10078-012-0020-x (2012).
Kanda, Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant 48, 452–458. https://doi.org/10.1038/bmt.2012.244 (2013).
Acknowledgements
We thank Professor Kei Takase as an advisor to establish this study. This work was supported by the Advanced Graduate Program for Future Medicine and Health Care, Tohoku University.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
N.Y. conceived the idea for this study and wrote the manuscript. N.Y., H.O., and S.H. designed the study and analyzed the data. H.O., S.H., and T.K. were responsible for executing the MR examination. Y.S., T.K., and H.S. were responsible for preparing and executing the physical measurement. H.O., S.-I.I., and M.K. assisted in the preparation and editing of the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Video 1.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yoshida, N., Ota, H., Higuchi, S. et al. Gliding performance is affected by cranial movement of abdominal organs. Sci Rep 10, 21430 (2020). https://doi.org/10.1038/s41598-020-78609-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-78609-3
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.