Abstract
Children with autism spectrum disorder (ASD) or attention deficit hyperactivity disorder (ADHD) have 2–3 times increased healthcare utilization and annual costs once diagnosed, but little is known about their utilization patterns early in life. Quantifying their early health system utilization could uncover condition-specific health trajectories to facilitate earlier detection and intervention. Patients born 10/1/2006–10/1/2016 with ≥ 2 well-child visits within the Duke University Health System before age 1 were grouped as ASD, ADHD, ASD + ADHD, or No Diagnosis using retrospective billing codes. An additional comparison group was defined by later upper respiratory infection diagnosis. Adjusted odds ratios (AOR) for hospital admissions, procedures, emergency department (ED) visits, and outpatient clinic encounters before age 1 were compared between groups via logistic regression models. Length of hospital encounters were compared between groups via Mann–Whitney U test. In total, 29,929 patients met study criteria (ASD N = 343; ADHD N = 1175; ASD + ADHD N = 140). ASD was associated with increased procedures (AOR = 1.5, p < 0.001), including intubation and ventilation (AOR = 2.4, p < 0.001); and outpatient specialty care, including physical therapy (AOR = 3.5, p < 0.001) and ophthalmology (AOR = 3.1, p < 0.001). ADHD was associated with increased procedures (AOR = 1.41, p < 0.001), including blood transfusion (AOR = 4.7, p < 0.001); hospital admission (AOR = 1.60, p < 0.001); and ED visits (AOR = 1.58, p < 0.001). Median length of stay was increased after birth in ASD (+ 6.5 h, p < 0.001) and ADHD (+ 3.8 h, p < 0.001), and after non-birth admission in ADHD (+ 1.1 d, p < 0.001) and ASD + ADHD (+ 2.4 d, p = 0.003). Each condition was associated with increased health system utilization and distinctive patterns of utilization before age 1. Recognizing these patterns may contribute to earlier detection and intervention.
Similar content being viewed by others
Introduction
Autism spectrum disorder (ASD) and attention deficit hyperactivity disorder (ADHD) affect approximately 1.5%1 and 11%2 of children in the United States, respectively, and ADHD symptoms are present in 40–60% of children with ASD3,4. Both diagnoses lead to higher rates of healthcare utilization5,6,7,8,9 and place considerable financial burden on affected individuals and their families10,11,12,13,14,15,16,17. Rates of hospitalization and outpatient clinic visits among children with ASD are approximately twice that of the average child, contributing to a two- to threefold increase in annual costs18 comparable to other chronic medical diagnoses13. Children with ADHD visit the emergency department (ED) approximately twice as often as those without7, with even higher rates reported among those with comorbid psychiatric conditions19. These factors are compounded in children with co-occurring ASD and ADHD, who incur higher healthcare costs than children with either disorder alone20.
These findings demonstrate that individuals with ASD and ADHD utilize greater healthcare resources once diagnosed, but little is known about their health system utilization early in life, prior to diagnosis. Emerging evidence suggests that diagnosis is preceded by distinct patterns of increased utilization. For example, a study of newly diagnosed ADHD patients in Germany found that healthcare costs were elevated in the year before diagnosis21. More recently, analysis of nationwide registry data from Denmark demonstrated that rates of medical service use were 1.8 times higher in children later diagnosed with ADHD, and co-occurring ASD further increased this difference22. These results suggest that increased utilization may be characteristic of ASD and/or ADHD, and not simply a consequence of recognizing and treating them. However, similar findings have not yet been reported in the United States, and specific health services more commonly used by children later diagnosed with ASD and ADHD have not yet been identified.
Based on known early risk factors for both disorders, distinctive patterns of utilization are likely to be present in the electronic health record (EHR) from an early age. Both ASD and ADHD are associated with lower birth weight23,24,25, preterm birth, low APGAR scores, and other perinatal complications26,27 that may lead to procedures and increase patients’ length of stay. Early ASD-related comorbidities28, such as postnatal hyperbilirubinemia29 and respiratory infections30, also appear in the EHR as possible indicators of increased risk. Problems with crying, sleeping, and feeding more common in infants with ADHD31 and ASD32 may lead parents to seek additional support, leading to higher use of specific health services. Utilization patterns may be unique among children with co-occurring ASD and ADHD, who are often diagnosed with ASD much later than children without ADHD symptoms33,34. While some risk factors for ASD and ADHD are based on physiologic or diagnostic findings that are not always available, others relate to health interactions readily accessed through most EHR systems.
Identifying EHR-based risk factors is a critical step toward earlier referral, diagnosis, and treatment, which in turn lead to markedly improved outcomes in both ASD35,36,37,38,39,40 and ADHD41,42,43 and reduced healthcare costs44. Multiple studies have illustrated the potential of EHR-based prediction models45, which can utilize existing EHR data standards to analyze a very large number of predictors46. This approach can also uncover unknown risk factors to further illuminate the early trajectories and biological bases of neurodevelopmental disorders and other pediatric conditions. Unlike data from Medicaid claims or health maintenance organization registries19,47, EHR data can be immediately analyzed within the hospital system itself to inform provider decision-making during routine care.
In this study, we retrospectively analyzed EHR data collected over a 12.5-year period in a large U.S. health system to compare health system utilization before age 1 between children later diagnosed with ASD and/or ADHD and other children. Analysis focused on hospital admissions, emergency department (ED) visits, outpatient clinic encounters, and procedures across multiple health services. We hypothesized that (a) children later diagnosed ASD and/or ADHD interacted with the health system at higher rates, including increased rates of admissions, ED visits, and non-routine procedures; (b) these children visited specific medical specialties at higher rates, including neurology and gastroenterology in ASD48,49,50; and (c) distinct patterns of interactions were present in each disorder. Our primary aim was to improve our understanding of the early trajectories of children with ASD and ADHD, contributing to earlier, EHR-based risk stratification. A secondary aim was to illustrate a framework that could be used to analyze early healthcare trajectories in other pediatric conditions.
Patients and methods
Cohort identification and data extraction
All results are based on retrospective data analysis approved by the Duke Health Institutional Review Board and conducted at the Duke University School of Medicine (2/1/2019–10/1/2019). Analyses were executed within the Duke Protected Analytics Computing Environment (PACE), a highly protected virtual network space designed for protected health information. Participant consent was waived due to the minimal risk posed by study procedures and infeasibility of obtaining consent in a large retrospective cohort.
Analyses were based on inpatient and outpatient encounters within the Duke University Health System (DUHS), a large academic medical center based in Durham, North Carolina. DUHS is comprised of Duke University Hospital, a large (957 bed) facility that includes a regional emergency/trauma center and major (51 operating rooms) surgery suite; Duke Regional Hospital, a medium (369 bed) facility serving Durham and 5 surrounding counties; Duke Raleigh Hospital, a medium (189 bed) facility serving Raleigh and Wake County; and a large network of outpatient facilities, including 30 Duke Primary Care locations. The surrounding region, called the Research Triangle, includes the cities of Raleigh, Durham, and Chapel Hill, and has an estimated population of 2,079,687 (2019 census). Records were extracted from the current (2013—present) DUHS EHR, which is based on the platform developed by Epic (Verona, Wisconsin), as well as several EHR platforms operating prior to 2013.
Study inclusion criteria were (1) date of birth between 10/1/2006 and 10/1/2016; and (2) ≥ 2 outpatient well-child visits within the DUHS before age 1. Criterion (2) was designed to limit analyses to patients likely to have received routine care through DUHS before age 1. Well-child visits were identified using visit type fields present in current and legacy EHR systems. DUHS encounters between 10/1/2006 and 2/1/2019 were extracted for patients meeting criteria. Children diagnosed with ASD or ADHD before age 1 were excluded to ensure that observed health system interactions were not a consequence of an existing diagnosis.
Diagnosis identification
Five mutually exclusive diagnosis groups were defined based on billing codes from the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) and Tenth Revision, Clinical Modification (ICD-10-CM). Problem list-based diagnoses were excluded due to concerns regarding data quality and availability.
Patients with an ASD or ADHD diagnosis were grouped as ASD (i.e., ASD only), ADHD (i.e., ADHD only), or ASD + ADHD. ASD was defined as ICD-9-CM code 299.00, 299.80, or 299.9051; or ICD-10-CM code F84.0, F84.8, or F84.9. ADHD was defined as ICD-9-CM code 314.0, 314.01, 314.1, 314.2, 314.8, or 314.9; or ICD-10-CM code F90.0, F90.1, F90.2, F90.8, or F90.9. Code selection was guided by literature review and the authors’ knowledge of billing practices.
Patients without an ASD or ADHD diagnosis were grouped as upper respiratory infection (URI) or No Diagnosis. URI was chosen to serve as a comparison group due to its status as a common but non-chronic diagnosis. URI was defined as any ICD-9-CM or ICD-10-CM code categorized as URI by Clinical Classification Software (CCS) developed by the Agency for Healthcare Research and Quality52.
Data preprocessing
Analyses focused on rates of hospital admissions, outpatient clinic visits, ED encounters, and procedures among patients in the five previously defined groups. Hospital admissions were further categorized by discharge service rather than admission service, which was not reported prior to 2013. Similarly, outpatient clinic visits were further categorized by clinic service or specialty. Discharge and clinic services were grouped into clinically meaningful and interpretable service types (eTables 8–9). ED encounters resulting in a hospital admission were counted separately from the admission itself. Procedures were categorized by applying CCS to the associated ICD-9-CM or ICD-10-CM code, then further grouping related CCS categories (eTable 10). The following CCS categories were grouped as routine procedures and omitted from the analysis: “Other diagnostic and therapeutic procedures”, “Vaccinations and inoculations”, and “Circumcision”.
Birth encounters (admission source was “born in hospital” or admission date matched date of birth) were analyzed separately from other admissions. Encounter length was calculated as the time difference between arrival and discharge (non-birth encounters) or birth and discharge (birth encounters).
Medicaid status was determined based on documented insurance status associated with all encounters before age 1. Patients whose status was most commonly documented as “Medicaid”, “NC Medicaid”, or “Medicaid Pending” were grouped as “Medicaid”. This group includes patients receiving assistance through the Innovations Waiver program offered to children with documented developmental disabilities by the Division of Mental Health, Developmental Disabilities and Substance Abuse Services within the North Carolina Department of Health and Human Services. Patients whose insurance status was not documented in any encounter were grouped as “Unknown”. Other patients were grouped as “Non-Medicaid”.
Statistical analysis
Group differences in demographic variables (sex, race, ethnicity, Medicaid status) were assessed via chi-square test. Encounters and procedures before age 1 in each category described previously were counted for each patient. Group differences in encounters and procedures were then assessed by (a) comparing counts by pairwise two-tailed Mann–Whitney U test, and (b) comparing the proportion of patients with at least one occurrence (i.e. rate of occurrence) by two-tailed two-sample proportion test. Differences in length of stay were assessed by two-tailed Mann–Whitney U test. Mann–Whitney U tests (rather than parametric tests) were selected because count and length of stay data are non-negative and long-tailed.
Raw p-values have been reported throughout, but statistical significance was determined by applying Bonferroni correction with an initial threshold of \(\alpha =0.05\). Results with \(p<\alpha /m\), where \(m\) is the number of categories for a given analysis, were considered statistically significant. In the analyses of outpatient clinic services, for example, 9 distinct types of services were examined, therefore results with p < 0.0056 were considered statistically significant.
Logistic regression was used to control for sex, race (White, Black or African American, Asian, Other Race), ethnicity (Hispanic or Latino, not Hispanic or Latino), and Medicaid status when comparing encounter and procedure rates between groups. Models were implemented in Statsmodels v0.1153 with Python 3.654. An additional binary variable (Epic, not Epic) was included to control for differences in encounter and procedure reporting between the current, Epic-based EHR platform and legacy platforms prior to 2013. Two-tailed t-tests were applied to all model parameters, and those with \(p<\alpha /m\) (as above) were considered statistically significant. Corresponding confidence intervals and adjusted odds ratios (AOR) were based on the Student’s t-distribution.
Institutional review and informed consent
All methods were carried out in accordance with the relevant guidelines and regulations. Study procedures were approved by the Duke Health Institutional Review Board and comply with institutional policies and federal regulations. A waiver of participant consent was approved due to the minimal risk posed by study procedures and infeasibility of obtaining consent in a large retrospective cohort. Analyses were executed within the Duke Protected Analytics Computing Environment (PACE), a highly protected virtual network space designed for protected health information.
Results
Description of cohort
Records for 200,423 unique patients born 10/1/2006–10/1/2016 were initially extracted, and 29,931 had ≥ 2 well-child visits before age 1. T wo patients were excluded due to ADHD diagnosis before age 1, leaving 29,929 patients who met study criteria. Demographics by group and age at diagnosis are presented in Table 1.
Among patients meeting criteria, unadjusted ASD and ADHD prevalence were 1.6% and 4.4%, respectively. Co-occurring ASD + ADHD (N = 140) was identified in 29.0% of patients with ASD and 10.6% of patients with ADHD. Mean age at ASD diagnosis was 1.4 years higher in patients with co-occurring ADHD (5.5y vs. 4.1y), whereas mean age at ADHD diagnosis was 1.3 years lower in patients with co-occurring ASD (5.9y and 7.2y).
Male to female ratios were 3.6 for ASD, 2.5 for ADHD, and 7.2 for ASD + ADHD. ADHD diagnosis was strongly associated with racial (Χ2 = 133.6, dof = 6, p < 0.001) and ethnic status (Χ2 = 37.9, dof = 2, p < 0.001), but ASD diagnosis was not (Χ2 = 10.6, dof = 6, p = 0.102; and Χ2 = 3.4, dof = 2, p = 0.179, respectively). Both diagnoses were associated with Medicaid status (ASD: Χ2 = 14.7, dof = 2, p < 0.001; ADHD Χ2 = 765.3, dof = 2, p < 0.001). Among patients with known insurance status, Medicaid rates were 52.9% and 60.5% in the ASD and ADHD groups, respectively, compared to 42.3% in the No Diagnosis group.
Summary of health system utilization
Most patients (87.5%) were born within DUHS. Before age 1, 8.8% were admitted (non-birth) to a DUHS hospital, 8.8% underwent a procedure, and 30.2% visited an ED. Hospital admission and procedures were more likely in ASD (AOR = 1.30, p = 0.1173; AOR = 1.47, p = 0.0042), although only the latter reached statistical significance, and in ADHD (AOR = 1.61, p < 0.0001; AOR = 1.43, p < 0.0001). In contrast, ED visits were more likely in ADHD (AOR = 1.56, p < 0.0001), but not in ASD (AOR = 0.93, p = 0.5454). Hospital admission, procedures, and ED visits were increased in ASD + ADHD but not statistically significant due to smaller group size (AOR = 1.43, p = 0.1643; AOR = 1.44, p = 0.0703; AOR = 1.48, p = 0.0301). These rates were not increased in URI (Fig. 1, eTable 1).
Adjusted odds ratios and rates of hospital admissions, procedures, and ED visits. Adjusted odds ratios (AORs) (A) and unadjusted occurrence rates (B) in each diagnosis group are shown for non-birth hospital admissions, procedures, and emergency department visits. Error bars indicate 95% confidence intervals for the AOR (A) and the standard error of the proportion (B). AORs were assessed for statistical significance (*) after applying Bonferroni correction to a baseline threshold of α = 0.05. Unadjusted occurrence rates are descriptive only, and were not tested for statistical significance.
Hospital admission and ED visits were more likely in males (AOR = 1.13, p = 0.0034; AOR = 1.13, p < 0.0001), but procedures were not (AOR = 1.08, p = 0.0237). Rates of hospital admissions, procedures, and ED visits were also higher among African American, Hispanic/Latino, and Medicaid patients and lower among White and Asian patients (eTable 2).
All patients had ≥ 2 outpatient clinic encounters for inclusion, but the mean number of these encounters was higher in ASD (14.8, p < 0.001), ADHD (14.7, p < 0.001), and ASD + ADHD (14.6, p < 0.001) compared to No Diagnosis (12.5), whereas it was lower in URI (11.4, p < 0.001).
Outpatient clinic services
Distinct patterns of outpatient clinic encounters were observed in ASD versus ADHD. Visits to medical and surgical specialists were increased (Fig. 2, eTable 1), with the latter being even more likely in ASD + ADHD (AOR = 1.93, p < 0.0011). ASD and ADHD groups were both more likely to visit neonatology (AOR = 2.61, p < 0.0001; AOR = 1.73, p < 0.0001), speech pathology (AOR = 2.52, p < 0.0001; AOR = 1.45, p = 0.0033), and ophthalmology (AOR = 3.45, p < 0.0001; AOR = 1.74, p < 0.0001), but these increases were more pronounced in ASD.
Adjusted odds ratios and rates of hospital admissions by discharge service category. Adjusted odds ratios (AORs) (A,B) and unadjusted occurrence rates (C,D) are shown for nine outpatient clinic visit types (see eTable 8 for definitions). Error bars indicate 95% confidence intervals for the AOR (A,B) and the standard error of the proportion (C,D). AORs were assessed for statistical significance (*) after applying Bonferroni correction to a baseline threshold of α = 0.05. Unadjusted occurrence rates are descriptive only, and were not tested for statistical significance. Child abuse and neglect (B,D) has been plotted with a different y-scale due to its lower rate of occurrence.
Adjusted odds of physical therapy were increased almost threefold in ASD (AOR = 2.92, p < 0.0001) and twofold in ADHD (AOR = 2.01, p = 0.0306), although only the former was statistically significant. In contrast, adjusted odds of visits related to child abuse and neglect were increased over fivefold in ADHD only (AOR = 5.30, p = 0.0019). Adjusted odds of diagnostic visits (e.g., labs, radiology) were also higher in ADHD (AOR = 2.06, p = 0.0014).
The allocation of visits among medical and surgical specialties differed between groups (eFigure 1). Among medical specialties, cardiology and medicine visits were more common in ADHD (AOR = 1.53, p = 0.0004; AOR = 1.47, p = 0.0008), whereas endocrinology, gastroenterology, and neurology visits were over threefold more likely in ASD (AOR = 3.39, p < 0.0001; AOR = 3.25, p = 0.0001, AOR = 3.15, p < 0.0001) (eFigure 2, eTable 3). Among surgical specialties, general surgery visits were most common in ADHD (AOR = 1.73, p = 0.0006), whereas ear, nose, and throat visits were twice as likely in ASD (AOR = 2.01, p = 0.0031) (eFigure 3, eTable 3).
Statistically significant differences between demographic groups included higher rates of general surgery visits among male (AOR = 2.60, p < 0.0001), African American (AOR = 1.52, p < 0.0001), and Medicaid (AOR = 2.34, p < 0.0001) patients, higher rates of cardiology visits among African American (AOR = 1.41, p = 0.0003), Hispanic/Latino (AOR = 1.46, p = 0.0003), and Medicaid (AOR = 1.46, p = 0.0034) patients, and higher rates of audiology (AOR = 1.79, p < 0.0001) and speech pathology visits (AOR = 1.94, p < 0.0001) among Hispanic/Latino patients (eTable 4). A number of visit types were substantially more likely in Medicaid patients, including physical therapy (AOR = 79.86, p < 0.0001), labs and imaging (AOR = 38.58, p < 0.0001), and urgent care (AOR = 84.13, p < 0.0001).
Procedures
Distinct patterns of procedure occurrence before age 1 were also observed in ASD versus ADHD (Fig. 3, eTable 1). Echocardiograms were more likely in ASD (AOR = 1.60, p = 0.0327), although not statistically significant; ADHD (AOR = 1.59, p = 0.0002); and ASD + ADHD (AOR = 2.36, p = 0.0028). Enteral and parental nutrition were also more likely in all three groups, although not statistically significant in ASD + ADHD (ASD AOR = 2.26, p = 0.0002; ADHD AOR = 2.06, p < 0.0001; ASD + ADHD AOR = 1.79, p = 0.0966).
Adjusted odds ratios and rates of procedures. Adjusted odds ratios (AORs) (A,B) and unadjusted occurrence rates (C,D) are shown for ten categories (see eTable 10 for definitions) of procedures from both birth and non-birth encounters. Error bars indicate 95% confidence intervals for the AOR (A,B) and the standard error of the proportion (C,D). AORs were assessed for statistical significance (*) after applying Bonferroni correction to a baseline threshold of α = 0.05. Unadjusted occurrence rates are descriptive only, and were not tested for statistical significance. Blood transfusion (B,D) has been plotted with a different y-scale due to its lower rate of occurrence.
Intubation and ventilation were twice as likely in ASD (AOR = 2.37, p < 0.0001), but adjusted odds were also increased in ADHD (AOR = 1.59, p = 0.0012). Blood transfusions were over four times more likely in ADHD (AOR = 4.57, p < 0.0001) and in ASD + ADHD (AOR = 6.38, p = 0.0022), but not in ASD (AOR = 1.00, p = 0.9989).
Procedures from birth versus non-birth encounters were also analyzed independently (eTables 5–6).
Admission services and length of stay
Patients were discharged by Pediatrics in 79.7% of all non-birth hospital admissions, therefore adjusted odds of discharge by Pediatrics (eFigure 4) are similar to hospital admission rates (Fig. 1). Whereas higher rates of hospital discharge by Pediatrics were observed in ASD (AOR = 1.51, p = 0.0171) and statistically significant in ADHD (AOR = 1.57, p < 0.0001), higher rates of discharge by Neonatology were found only in ADHD (AOR = 2.06, p = 0.0014), and in ASD + ADHD (AOR = 2.12, p = 0.2025).
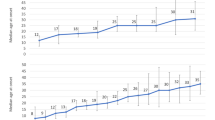
The typical hospital stay (non-birth admission) was over a day longer in ADHD (median = 3.76d, p < 0.0001) and over two days longer in ASD + ADHD (median = 5.09d, p = 0.0032) compared to those in the No Diagnosis group (median = 2.67d). The typical hospital stay after birth was 6.5 h longer in ASD (median = 2.55d, p < 0.0001), 3.8 h longer in ADHD (median = 2.44d, p < 0.0001), and 5.8 h longer in ASD + ADHD (median = 2.52d, p = 0.0310) compared to those in the No Diagnosis group (median = 2.28 days) (Fig. 4, eTable 7).
Length of Hospital Stay by Diagnosis. (A) shows the length of hospital stay after birth for patients in each diagnosis group. (B) shows the length of hospital stay after non-birth admission for patients in each diagnosis group. Box midlines and edges show the median and interquartile range (IQR), respectively. The notches indicate a 95% confidence interval for the median, which can extend beyond the IQR (e.g. ASD and ASD + ADHD in B). Lower and upper whiskers extend to outliers within \(1.5*\mathrm{IQR}\) of first and third quartile, respectively. Outliers have been clipped to show boxplot detail, but were included in the statistical analysis.
Discussion
This is the first study reporting health system utilization among individuals with ASD, ADHD, and co-occurring ASD + ADHD in their first year of life, prior to diagnosis. Analysis drew on records from over 200,000 unique patients visiting DUHS over a 12.5-year period, almost 30,000 of whom met study criteria. Results corroborate studies from Europe demonstrating increased utilization prior to ADHD diagnosis, extend this finding to ASD and co-occurring ASD + ADHD, and highlight distinct interaction patterns underlying the increased rates of utilization observed in both disorders. We also highlight the demographics of co-occurring ASD + ADHD, which was associated with a 1.4 year delay in ASD diagnosis33,34 and a higher male to female ratio (7.2) than either disorder alone. More broadly, this work demonstrates that ASD and ADHD have profound effects on health and health system resources prior to diagnosis. Thus, EHR platforms provide an opportunity to monitor health interactions early in life to stratify patients’ risk of developing ASD and ADHD, and may offer a unique window into the early trajectory and biological bases of these conditions.
Many of these findings are not surprising given current knowledge of characteristics and comorbidities associated with ASD and ADHD. Higher rates of clinic follow-up, hospital admission, and intubation may result from the association with perinatal complications found in both disorders26,27, for example, but our results show that these associations have measurable impact on a health system level. Additionally, many other findings were unexpected. In ASD, increased visits to gastroenterology and neurology were expected, but increased visits to ophthalmology and endocrinology were not; this finding suggests that these comorbidities28 also have substantial impact on early utilization. In ADHD, increased odds of blood transfusion and visits related to child abuse and neglect are consistent with known associations with ADHD55, including problems in infancy31 and increased parental stress previously observed in infants with ADHD12, but are striking nevertheless.
The size and number of statistically significant effects suggest that early health interactions contain valuable information about ASD and ADHD risk, implying that health services data from the EHR could inform risk stratification. Given the importance of prompt ASD and ADHD diagnosis, further work is needed to determine how this information might be leveraged to inform clinician decision-making and/or health system policy. Risk monitoring systems could be implemented within the EHR itself, for example, in order to automatically alert the families and/or providers of children whose ASD or ADHD risk is high. Follow-up work should not only implement and validate risk monitoring, but also explore the effectiveness of a range of interventions aimed at families, individual providers, and the health system as a whole. Moreover, this paradigm could be applied to promote earlier identification and intervention in a wide range of pediatric conditions.
The current findings demonstrate that patterns of health system utilization associated with ASD and ADHD, such as longer hospital stays after birth, emerge in the first few days of life, and many more emerge within the first year. Increases in specific forms of utilization, such as ED visits, are not limited to ASD or ADHD, therefore additional work is needed to determine which utilization patterns—or combinations of patterns—have high predictive value. Subsequent work will focus on the development of predictive models that can be deployed and iteratively evaluated in clinical workflow.
Limitations
Results are observational and associative, and should be interpreted with a degree of caution due to possible confounding effects. Analyses controlled for sex, race, ethnicity, insurance status, and an institutional transition between EHR systems that affected the reporting of some encounter types. Socioeconomic status and other potentially relevant covariates, such as family characteristics, were not consistently available. Importantly, our analysis did account for a potentially impactful confounder, namely, the presence of a diagnosis after age 1. By including the URI comparison group, we demonstrate that selecting children with a non-chronic diagnosis who remain engaged with the health system does not produce similar effects to those observed in ASD and ADHD. Future work should focus on comparisons to other chronic health conditions (e.g., asthma, congenital heart disease), and on understanding differences between demographic groups, including disadvantaged groups, in greater detail.
Analysis was based on all patients born 10/1/2006–10/1/2016 who received routine care within DUHS prior to age 1. Our observation window (10/1/2006–2/1/2019) implies that patients’ ages ranged from 2.3 to 12.3 years at the end of the observation. Patients in our cohort may have been (or still be) diagnosed with ASD or ADHD subsequent to observation, or at facilities outside of DUHS. Thus, analyses should be viewed as comparing patients with known ASD and ADHD diagnoses to all other patients, including those with other medical and psychiatric conditions, rather than a cohort of typically developing children. Results demonstrate that children later diagnosed with ASD and ADHD utilize health system resources at higher rates than the average child.
EHR-based analyses are affected by known data quality issues, including unreliable race and ethnicity reporting. Our analysis groups do not reflect gold standard diagnoses, and identification of ADHD by diagnosis codes was 70–75% accurate across 10 institutions56. Further, analysis was based on DUHS encounters only, whereas patients may have received additional care outside of DUHS. Study criteria (e.g., ≥ 2 well-child visits) were designed to identify patients receiving routine care within DUHS, but their effectiveness is uncertain. These limitations are consistent with our overarching goal to identify EHR-based predictors of ASD and ADHD, but results may not generalize to health systems and/or geographic regions with different demographic makeup compared to DUHS and its surrounding area; or to other sources of health utilization data, particularly non-EHR data sources.
Conclusion
ASD and ADHD are associated with increased health system utilization in the first year of life, prior to diagnosis. Moreover, the two disorders are associated with distinct patterns of early health interactions that could be monitored through the EHR to stratify patients’ risk of developing ASD and ADHD. Subsequent work will focus on developing predictive models that could be deployed within a health system to inform provider decision-making, contributing to earlier diagnosis and treatment.
Abbreviations
- ASD:
-
Autism spectrum disorder
- ADHD:
-
Attention deficit hyperactivity disorder
- URI:
-
Upper respiratory infection
- ED:
-
Emergency department
- AOR:
-
Adjusted odds ratio
- EHR:
-
Electronic health records
- DUHS:
-
Duke University Health System
- CCS:
-
Clinical classification software
References
Baio, J. et al. Prevalence of autism spectrum disorder among children aged 8 years—autism and developmental disabilities monitoring network, 11 sites, United States, 2014. MMWR Surveill. Summ. 67(6), 1–23. https://doi.org/10.15585/mmwr.ss6706a1 (2018).
Danielson, M. L. et al. Prevalence of parent-reported ADHD diagnosis and associated treatment among U.S. children and adolescents, 2016. J. Clin. Child Adolesc. Psychol. 47(2), 199–212. https://doi.org/10.1080/15374416.2017.1417860 (2018).
Salazar, F. et al. Co-occurring psychiatric disorders in preschool and elementary school-aged children with autism spectrum disorder. J. Autism Dev. Disord. 45(8), 2283–2294. https://doi.org/10.1007/s10803-015-2361-5 (2015).
Rommelse, N. N. J., Franke, B., Geurts, H. M., Hartman, C. A. & Buitelaar, J. K. Shared heritability of attention-deficit/hyperactivity disorder and autism spectrum disorder. Eur. Child Adolesc. Psychiatry 19(3), 281–295. https://doi.org/10.1007/s00787-010-0092-x (2010).
Zerbo, O. et al. Health care service utilization and cost among adults with autism spectrum disorders in a U.S. integrated health care system. Autism Adulthood 1(1), 27–36. https://doi.org/10.1089/aut.2018.0004 (2018).
Cidav, Z., Lawer, L., Marcus, S. C. & Mandell, D. S. Age-related variation in health service use and associated expenditures among children with autism. J. Autism Dev. Disord. 43(4), 924–931. https://doi.org/10.1007/s10803-012-1637-2 (2013).
Cuffe, S. P., Moore, C. G. & McKeown, R. ADHD and health services utilization in the national health interview survey. J. Atten. Disord. 12(4), 330–340. https://doi.org/10.1177/1087054708323248 (2009).
Cummings, J. R. et al. Health services utilization among children with and without autism spectrum disorders. J. Autism Dev. Disord. 46(3), 910–920. https://doi.org/10.1007/s10803-015-2634-z (2016).
Liu, G., Pearl, A. M., Kong, L., Leslie, D. L. & Murray, M. J. A profile on emergency department utilization in adolescents and young adults with autism spectrum disorders. J. Autism Dev. Disord. 47(2), 347–358. https://doi.org/10.1007/s10803-016-2953-8 (2017).
Doshi, J. A. et al. Economic impact of childhood and adult attention-deficit/hyperactivity disorder in the United States. J. Am. Acad. Child Adolesc. Psychiatry 51(10), 990-1002.e2. https://doi.org/10.1016/j.jaac.2012.07.008 (2012).
Horlin, C., Falkmer, M., Parsons, R., Albrecht, M. A. & Falkmer, T. The cost of autism spectrum disorders. PLoS ONE 9(9), e106552. https://doi.org/10.1371/journal.pone.0106552 (2014).
Raphael, J. L., Zhang, Y., Liu, H. & Giardino, A. P. Parenting stress in US families: implications for paediatric healthcare utilization. Child Care Health Dev. 36(2), 216–224. https://doi.org/10.1111/j.1365-2214.2009.01052.x (2010).
Chan, E., Zhan, C. & Homer, C. J. Health care use and costs for children with attention-deficit/hyperactivity disorder: national estimates from the medical expenditure panel survey. Arch. Pediatr. Adolesc. Med. 156(5), 504–511. https://doi.org/10.1001/archpedi.156.5.504 (2002).
Buescher, A. V. S., Cidav, Z., Knapp, M. & Mandell, D. S. Costs of autism spectrum disorders in the United Kingdom and the United States. JAMA Pediatr. 168(8), 721–728. https://doi.org/10.1001/jamapediatrics.2014.210 (2014).
Lavelle, T. A. et al. Economic burden of childhood autism spectrum disorders. Pediatrics 133(3), e520–e529. https://doi.org/10.1542/peds.2013-0763 (2014).
Guevara, J., Lozano, P., Wickizer, T., Mell, L. & Gephart, H. Utilization and Cost of Health Care Services for Children With Attention-Deficit/Hyperactivity Disorder. Pediatrics 108(1), 71–78. https://doi.org/10.1542/peds.108.1.71 (2001).
Zhao, X. et al. Family burden of raising a child with ADHD. J. Abnorm. Child Psychol. 47(8), 1327–1338. https://doi.org/10.1007/s10802-019-00518-5 (2019).
Croen, L. A., Najjar, D. V., Ray, G. T., Lotspeich, L. & Bernal, P. A comparison of health care utilization and costs of children with and without autism spectrum disorders in a large group-model health plan. Pediatrics 118(4), e1203–e1211. https://doi.org/10.1542/peds.2006-0127 (2006).
DeBar, L. L., Lynch, F. L. & Boles, M. Healthcare use by children with attention deficit/hyperactivity disorder with and without psychiatric comorbidities. J. Behav. Health Serv. Res. 31(3), 312–323. https://doi.org/10.1007/BF02287293 (2004).
Peacock, G., Amendah, D., Ouyang, L. & Grosse, S. Autism spectrum disorders and health care expenditures: the effects of co-occurring conditions. J. Dev. Behav. Pediatr. 33(1), 2–8. https://doi.org/10.1097/DBP.0b013e31823969de (2012).
Klora, M., Zeidler, J., Linder, R., Verheyen, F. & von der Schulenburg, J.-M.G. Costs and treatment patterns of incident ADHD patients—a comparative analysis before and after the initial diagnosis. Health Econ. Rev. https://doi.org/10.1186/s13561-015-0078-y (2015).
Laugesen, B. et al. Attention deficit hyperactivity disorder in childhood: healthcare use in a Danish birth cohort during the first 12 years of life. J. Pediatr. 197, 233–240. https://doi.org/10.1016/j.jpeds.2018.01.078 (2018).
Momany, A. M., Kamradt, J. M. & Nikolas, M. A. A meta-analysis of the association between birth weight and attention deficit hyperactivity disorder. J. Abnorm. Child Psychol. 46(7), 1409–1426. https://doi.org/10.1007/s10802-017-0371-9 (2018).
Galéra, C. et al. Early risk factors for hyperactivity-impulsivity and inattention trajectories from age 17 months to 8 years. Arch. Gen. Psychiatry 68(12), 1267–1275. https://doi.org/10.1001/archgenpsychiatry.2011.138 (2011).
Gardener, H., Spiegelman, D. & Buka, S. L. Perinatal and neonatal risk factors for autism: a comprehensive meta-analysis. Pediatrics 128(2), 344–355. https://doi.org/10.1542/peds.2010-1036 (2011).
Getahun, D. et al. Association of perinatal risk factors with autism spectrum disorder. Am. J. Perinatol. 07(3), 295–304. https://doi.org/10.1055/s-0036-1597624 (2017).
Buchmayer, S. et al. Can association between preterm birth and autism be explained by maternal or neonatal morbidity?. Pediatrics 124(5), e817–e825. https://doi.org/10.1542/peds.2008-3582 (2009).
Doshi-Velez, F., Ge, Y. & Kohane, I. Comorbidity clusters in autism spectrum disorders: an electronic health record time-series analysis. Pediatrics 133(1), e54–e63. https://doi.org/10.1542/peds.2013-0819 (2014).
Maimburg, R. D. et al. Neonatal jaundice: a risk factor for infantile autism?. Paediatr. Perinat. Epidemiol. 22(6), 562–568. https://doi.org/10.1111/j.1365-3016.2008.00973.x (2008).
Wang, C., Geng, H., Liu, W. & Zhang, G. Prenatal, perinatal, and postnatal factors associated with autism. Medicine (Baltimore) https://doi.org/10.1097/MD.0000000000006696 (2017).
Bilgin, A. et al. Early crying, sleeping, and feeding problems and trajectories of attention problems from childhood to adulthood. Child Dev. https://doi.org/10.1111/cdev.13155 (2020).
Werner, E., Dawson, G., Munson, J. & Osterling, J. Variation in early developmental course in autism and its relation with behavioral outcome at 3–4 years of age. J. Autism Dev. Disord. 35(3), 337–350. https://doi.org/10.1007/s10803-005-3301-6 (2005).
Miodovnik, A., Harstad, E., Sideridis, G. & Huntington, N. Timing of the diagnosis of attention-deficit/hyperactivity disorder and autism spectrum disorder. Pediatrics 136(4), e830–e837. https://doi.org/10.1542/peds.2015-1502 (2015).
Yee, M. M. & Millichap, J. G. Relationship between age at diagnosis of ADHD and ASD. Pediatr. Neurol. Briefs 29(10), 78. https://doi.org/10.15844/pedneurbriefs-29-10-5 (2015).
Jocelyn, L. J., Casiro, O. G., Beattie, D., Bow, J. & Kneisz, J. Treatment of children with autism: a randomized controlled trial to evaluate a caregiver-based intervention program in community day-care centers. J. Dev. Behav. Pediatr. 19(5), 326–334. https://doi.org/10.1097/00004703-199810000-00002 (1998).
Smith, T., Groen, A. D. & Wynn, J. W. Randomized trial of intensive early intervention for children with pervasive developmental disorder. Am. J. Ment. Retard. 105(4), 269–285. https://doi.org/10.1352/0895-8017(2000)105%3c0269:RTOIEI%3e2.0.CO;2 (2000).
Sallows, G. O. & Graupner, T. D. Intensive behavioral treatment for children with autism: four-year outcome and predictors. Am. J. Ment. Retard. 110(6), 417–438. https://doi.org/10.1352/0895-8017(2005)110[417:IBTFCW]2.0.CO;2 (2005).
Anderson, D. K., Liang, J. W. & Lord, C. Predicting young adult outcome among more and less cognitively able individuals with autism spectrum disorders. J. Child Psychol. Psychiatry 55(5), 485–494. https://doi.org/10.1111/jcpp.12178 (2014).
Dawson, G. et al. Randomized, controlled trial of an intervention for toddlers with autism: the Early Start Denver Model. Pediatrics 125(1), e17-23. https://doi.org/10.1542/peds.2009-0958 (2010).
Rogers, S. J. et al. A multisite randomized controlled two-phase trial of the early start denver model compared to treatment as usual. J. Am. Acad. Child Adolesc. Psychiatry 58(9), 853–865. https://doi.org/10.1016/j.jaac.2019.01.004 (2019).
Wolraich, M. et al. ADHD: clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics 128(5), 1007–1022. https://doi.org/10.1542/peds.2011-2654 (2011).
Biederman, J., Wilens, T., Mick, E., Spencer, T. & Faraone, S. V. Pharmacotherapy of attention-deficit/hyperactivity disorder reduces risk for substance use disorder. Pediatrics 104(2), e20. https://doi.org/10.1542/peds.104.2.e20 (1999).
Dalsgaard, S., Østergaard, S. D., Leckman, J. F., Mortensen, P. B. & Pedersen, M. G. Mortality in children, adolescents, and adults with attention deficit hyperactivity disorder: a nationwide cohort study. Lancet 385(9983), 2190–2196. https://doi.org/10.1016/S0140-6736(14)61684-6 (2015).
Cidav, Z. et al. Cost offset associated with early start denver model for children with autism. J. Am. Acad. Child Adolesc. Psychiatry 56(9), 777–783. https://doi.org/10.1016/j.jaac.2017.06.007 (2017).
Goldstein, B. A., Navar, A. M., Pencina, M. J. & Ioannidis, J. P. A. Opportunities and challenges in developing risk prediction models with electronic health records data: a systematic review. J. Am. Med. Inform. Assoc. 24(1), 198–208. https://doi.org/10.1093/jamia/ocw042 (2017).
Rajkomar, A. et al. Scalable and accurate deep learning with electronic health records. NPJ Digit. Med. 1(1), 1–10. https://doi.org/10.1038/s41746-018-0029-1 (2018).
Ruble, L. A., Heflinger, C. A., Renfrew, J. W. & Saunders, R. C. Access and service use by children with autism spectrum disorders in medicaid managed care. J. Autism Dev. Disord. 35(1), 3–13. https://doi.org/10.1007/s10803-004-1026-6 (2005).
Lee, M. et al. Association of autism spectrum disorders and inflammatory bowel disease. J. Autism Dev. Disord. 48(5), 1523–1529. https://doi.org/10.1007/s10803-017-3409-5 (2018).
Neuhaus, E., Bernier, R. A., Tham, S. W. & Webb, S. J. Gastrointestinal and psychiatric symptoms among children and adolescents with autism spectrum disorder. Front Psychiatry https://doi.org/10.3389/fpsyt.2018.00515 (2018).
Vargason, T., Frye, R. E., McGuinness, D. L. & Hahn, J. Clustering of co-occurring conditions in autism spectrum disorder during early childhood: a retrospective analysis of medical claims data. Autism Res. 12(8), 1272–1285. https://doi.org/10.1002/aur.2128 (2019).
Lingren, T. et al. Electronic health record based algorithm to identify patients with autism spectrum disorder. PLoS ONE 11(7), e0159621. https://doi.org/10.1371/journal.pone.0159621 (2016).
Elixhauser, A., Steiner, C. & Palmer, L. Clinical Classification Software (CCS), 2014. US Agency for Healthcare Research and Quality. (2014).
Seabold, S. & Perktold, J. Statsmodels: econometric and statistical modeling with python. In: 9th Python in Science Conference; (2010).
Python Software Foundation. The Python Language Reference—Python 3.6.9 Documentation. https://docs.python.org/3.6/reference/index.html. Accessed 4th October 2019.
Stern, A. et al. Associations between abuse/neglect and ADHD from childhood to young adulthood: a prospective nationally-representative twin study. Child Abuse Negl. 81, 274–285. https://doi.org/10.1016/j.chiabu.2018.04.025 (2018).
Daley, M. F. et al. Accuracy of electronic health record-derived data for the identification of incident ADHD. J. Atten. Disord. 21(5), 416–425. https://doi.org/10.1177/1087054713520616 (2017).
Acknowledgements
This work was supported by the Forge, Duke University’s center for health data science, and by the C. Keith Conners Fellowship in Digital Health founded by a donation from Multi-Health Systems, Inc. The analysis was executed within the Duke Protected Analytics Computing Environment (PACE), which is supported by Duke’s Clinical and Translational Science Award (CTSA) grant (UL1TR002553), and by the Duke University Health System. The CTSA initiative is led by the National Center for Advancing Translational Sciences at the National Institutes of Health. The authors would also like to thank Victoria Christian, Ursula Rogers, and Amy Herring for their ongoing support of this project.
Funding
Funding support was provided by NICHD (P50HD093074), NIMH (R01MH121329), and NIDA (K24DA023464).
Author information
Authors and Affiliations
Contributions
M.E. completed all analyses, drafted the initial manuscript, and revised the manuscript. S.B., J.G., R.H., and A.O. provided feedback on analyses and reviewed and revised the manuscript. S.R. coordinated and supervised data acquisition, provided feedback on analyses, and reviewed and revised the manuscript. G.D. and S.K. conceptualized the study, provided feedback on analyses, and reviewed and revised the manuscript. All authors contributed to the study design and concept. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
Geraldine Dawson is on the Scientific Advisory Boards of Janssen Research and Development, Akili, Inc., LabCorp, Inc., Tris Pharma, and Roche Pharmaceutical Company, a consultant for Apple, Inc, Gerson Lehrman Group, Guidepoint, Inc., Teva Pharmaceuticals, and Axial Ventures, and is CEO of DASIO, LLC. Dawson has developed technology that has been licensed and Dawson and Duke University have benefited financially. Dawson receives royalties from Guilford Press, Springer, and Oxford University Press. Scott Kollins reports grants, personal fees and non-financial support from Akili Interactive; grants, personal fees and non-financial support from Arbor Pharmaceuticals; grants, personal fees and non-financial support from Bose; grants, personal fees and non-financial support from Otsuka Pharmaceuticals; grants, personal fees and non-financial support from Rhodes; grants, personal fees and non-financial support from Shire Pharmaceuticals; and grants, personal fees and non-financial support from Tris Pharmaceuticals outside the submitted work. Matthew Engelhard, Samuel Berchuck, Jyotsna Garg, Ricardo Henao, Andrew Olson, and Shelley Rusincovitch have no financial relationships or potential conflicts of interest relevant to this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Engelhard, M.M., Berchuck, S.I., Garg, J. et al. Health system utilization before age 1 among children later diagnosed with autism or ADHD. Sci Rep 10, 17677 (2020). https://doi.org/10.1038/s41598-020-74458-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-74458-2
This article is cited by
-
Health Condition Co-Morbidities in Children with Autism and Their Association with Challenging Behavior
Advances in Neurodevelopmental Disorders (2024)
-
A tablet-based game for the assessment of visual motor skills in autistic children
npj Digital Medicine (2023)
-
Age of Diagnosis for Co-occurring Autism and Attention Deficit Hyperactivity Disorder During Childhood and Adolescence: a Systematic Review
Review Journal of Autism and Developmental Disorders (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.