Abstract
Documenting post-bleaching trajectories of coral reef communities is crucial to understand their resilience to climate change. We investigated reef community changes following the 2015/16 bleaching event at Aldabra Atoll, where direct human impact is minimal. We combined benthic data collected pre- (2014) and post-bleaching (2016–2019) at 12 sites across three locations (lagoon, 2 m depth; seaward west and east, 5 and 15 m depth) with water temperature measurements. While seaward reefs experienced relative hard coral reductions of 51–62%, lagoonal coral loss was lower (− 34%), probably due to three-fold higher daily water temperature variability there. Between 2016 and 2019, hard coral cover did not change on deep reefs which remained dominated by turf algae and Halimeda, but absolute cover on shallow reefs increased annually by 1.3% (east), 2.3% (west) and 3.0% (lagoon), reaching, respectively, 54%, 68% and 93% of the pre-bleaching cover in 2019. Full recovery at the shallow seaward locations may take at least five more years, but remains uncertain for the deeper reefs. The expected increase in frequency and severity of coral bleaching events is likely to make even rapid recovery as observed in Aldabra’s lagoon too slow to prevent long-term reef degradation, even at remote sites.
Similar content being viewed by others
Introduction
Climate change-induced coral bleaching events are increasing in frequency and severity, threatening the persistence of coral reef ecosystems worldwide. Global warming reduced the time frames between bleaching events from 27 years in the early 1980s to 5.9 years in 20161 and recovery windows are predicted to shorten even further as severe bleaching events are expected to occur annually on 90% of the world’s coral reefs by 20552. In this context, assessing post-disturbance reef trajectories is crucial to understand which conditions favour reef recovery3,4.
The metric most widely used to assess reef recovery is the return time of coral cover to pre-disturbance values, hereafter ‘coral recovery’5,6. Coral recovery rates are influenced by various environmental and physical factors (e.g. water depth7,8, light intensity7, nutrient levels9, water flow10, temperature variability11,12). Furthermore, as bleaching susceptibility and rebound potential of corals varies on multiple levels (e.g. among13,14 and within taxa15,16, across coral growth forms17, among symbiont type18,19), coral recovery is typically faster than the return to pre-disturbance coral community composition, hereafter ‘reassembly’6. Reassembly is a critical aspect of reef recovery, ensuring that the coral community’s traits and functions are restored6. Speed of coral recovery and reassembly varies substantially among reefs6,20,21 and regions5, underlining the complex nature of reef recovery processes.
Reef recovery relies on the growth and propagation of surviving colonies and coral recruitment3,22,23 which is influenced by abiotic and biotic conditions and anthropogenic disturbance. High wave exposure, for example, limits coral growth and coral larvae settlement24, while coral recruitment and survival can be enhanced by herbivores that control algal turf and fleshy macroalgae and promote crustose coralline algae (CCA) growth25,26,27. These natural drivers of recovery may be disrupted by direct human stressors such as overfishing of herbivores and/or nutrient enrichment, favouring algal proliferation and impeding or preventing reef recovery. This implies that, despite substantial variation in coral recovery and reassembly, both can be promoted through targeted management of direct anthropogenic disturbance28.
Consequently, coral reefs removed from direct human stressors serve as a baseline to assess the natural recovery potential in the face of the exacerbating effects of climate change and diminishing time frames for reef recovery. Understanding the variation in bleaching impact and recovery trajectories at such sites can provide crucial information for regional marine spatial planning and climate policies29. Aldabra Atoll in the Western Indian Ocean (WIO) offers the opportunity to examine bleaching impacts and subsequent reef trajectory dynamics under minimal direct human disturbance30,31. Designated as a Special Reserve, the highest level of national protection, under Seychelles’ legislation in 1981, and inscribed as a UNESCO World Heritage Site in 1982, Aldabra’s marine ecosystem has been protected from commercial fishing pressure for almost 40 years, and human-driven nutrient inputs are absent. Nevertheless, coral bleaching events have caused high coral mortality at Aldabra in 1998/1999 (38% and 66% on the seaward reef at 10 m and 20 m water depth, respectively32) and in 2015/2016 (35% in the lagoon at 2 m water depth; 54% at 5 m and 55% at 15 m water depth on the seaward reef33).
Here we utilise a unique 5-year data set that covers the aftermath of a major global coral bleaching event at a remote reef system with minimal local human impact. Following the findings of Cerutti et al.33 that bleaching induced coral mortality was lower at Aldabra’s lagoon than at the seaward reef after the 2015/16 coral bleaching (35% vs. 55% loss), we examine early post-bleaching reef trajectories at Aldabra Atoll in the context of spatial variations in bleaching impact by: (1) assessing changes of benthic communities across locations between 2014 (pre-bleaching) and 2016 (post-bleaching) and quantifying daily water temperature variability to explore whether this links to spatial differences in bleaching impact12, and (2) evaluating the post-bleaching trajectories (recovery/stability/degradation) of the benthic communities at these locations between 2016 and 2019. We use our results to outline expected future prospects for Aldabra’s reefs and the implications for remote reefs elsewhere.
Results
Benthic community change directly after bleaching
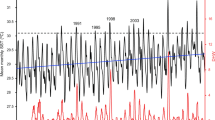
Overall, between Dec 2014 and Dec 2016, Aldabra’s reefs experienced 53% and 92% reductions of hard and soft corals, respectively. Hard coral reduction, however, was only significant on the seaward reefs, where losses were substantially higher than inside the lagoon (lagoon: − 34%, west shallow: − 56%; east shallow: − 62%, west and east deep; − 51%; Fig. 1a, Supplementary Table S1). In the lagoon, mean daily water temperature range was more than three times higher than the ranges recorded at the shallow seaward reefs (Table 1, Fig. 2). Soft coral cover declined by 91–92% at all locations (Fig. 1b,g), but absolute losses were considerably lower inside the lagoon as soft coral cover there was already < 2% in 2014 (compared to 7–26% at the seaward reefs). At the lagoon and west, CCA and turf algae increased (CCA, lagoon: 5–14%, shallow west: 4–12%, deep west: 1–9%; turf algae, lagoon: 46–57%, shallow west: 48–59%, deep west: 30–62%), together comprising 70% of the benthos at these locations in 2016 (Fig. 1c–i). While calcareous green alga Halimeda only increased at the shallow west and the lagoon (from ca. 1 to 6% cover), it remained most abundant at the east (25–29% cover in 2016) and together with CCA and turf algae (i.e. all algae groups combined) comprised 70–81% of the benthos there in 2016 (Fig. 1c–j).
Mean cover of major benthic groups at shallow (a–e) and deep locations (f–j) between 2014 and 2016 at Aldabra (transect sections: lagoon n = 6, west n = 10 per depth, east n = 8 per depth). Bars represent back-transformed estimates of GEE analysis with 95% confidence interval. Significant differences across years are indicated with asterisks (*p < 0.5; **p < 0.01; ***p < 0.001).
Mean in-situ water temperature ranges (°C) at three representative sites in the lagoon and at the seaward east and west of Aldabra. Solid lines indicate daily mean ranges calculated from temperature records obtained in 30-min intervals between February 2015 and November 2018 (see “Methods”). Dashed lines represent mean daily temperature range within the study period. Inset shows the same data as histogram, representing the number of days during which a given temperature range was recorded at each site.
Cover of all coral taxa declined significantly between 2014 and 2016 (except for ‘other hard corals’ and Isopora palifera, the latter of which slightly increased at the shallow east during 2014–2016) but absolute losses varied across locations (Supplementary Table S2, Supplementary Fig. S1). Acropora and Montipora were most affected at the shallow west and east, dropping from 2 to 5% cover in 2014 to ≤ 0.7% cover in 2016, a relative reduction of 84–99%. Although relative losses of 83–95% were also recorded for both taxa at the deep locations and the lagoon, absolute losses there were lower, as cover of these taxa was already low in 2014 (deep locations: 0.4–1.7%; lagoon: 0.6–0.9%). Branching Porites experienced a relative decline of 83–99% at the seaward reefs (1–4% cover in 2014; < 0.1% cover in 2016), but retained 50% of pre-bleaching cover at the lagoon (6% in 2014, 3% in 2016). Of all taxa, the highest absolute losses were recorded for Rhytisma at the seaward west where it reduced from 8 and 26% cover (shallow and deep, respectively) in 2014 to < 0.1% cover at both depths in 2016.
Post-bleaching trajectories
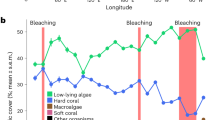
Except for soft coral cover, which did not change at any location between 2016 and 2019, trajectories of benthic groups varied across locations (Fig. 3, Supplementary Table S3). By 2019, mean hard coral cover had increased at all shallow locations to 13%, 23% and 30% at the east, west and lagoon, respectively (Fig. 3a), equating to 54% (east), 68% (west) and 93% (lagoon) of the pre-bleaching hard coral cover. The absolute annual rate of change in hard coral cover was 1.3%, 2.3% and 3.0% at the east, west and lagoon, respectively (Table 2). From 2019, the projected time until hard coral cover has fully recovered to pre-bleaching levels (2014) is 0.7, 4.8 and 8.5 years at the lagoon, west and east, respectively (Table 2). These are conservative estimates as hard coral cover gains until 2019 were non-linear and accelerated over time (e.g. 1.6–2.0 times higher from 2018 to 2019, than from 2017 to 2018, and 2016 to 2017). Simultaneously with hard coral cover increase, turf algae reduced to below pre-bleaching levels by 2018 (east: 28–18%, west: 64–40%, lagoon: 60–38%; Fig. 3d). However, while benthic communities at the shallow west and lagoon were no longer turf algae-dominated by 2018, and CCA cover remained unchanged during 2016–2019, CCA cover dropped at the shallow east during 2016–2019 (27–11%), with turf algae and Halimeda covering 18–21% and 45–50% of the shallow eastern benthos in 2017, 2018 and 2019, respectively (Fig. 3c–e).
Mean cover of major benthic groups at shallow (a–e) and deep locations (f–j) between 2016 and 2019 at Aldabra (transect sections: lagoon n = 9, west n = 15 per depth, east n = 12 per depth). Bars represent back-transformed estimates of GEE analysis with 95% confidence interval. Significant differences across years are indicated with asterisks (*p < 0.5; **p < 0.01).
At the deep locations hard coral cover did not change between 2016 and 2019 (Fig. 3f). Although turf algae cover decreased between 2016 and 2018, it still covered 51% of the western benthos in 2019 (Fig. 3i), 70% more than pre-bleaching. At the east, turf algae cover remained below pre-bleaching levels but, similar to the shallow east, Halimeda and turf algae combined dominated the benthos at 53–61% and 9–23% cover between 2017 and 2019, respectively (Fig. 3i,j).
Montipora was the only hard coral genus that increased significantly between 2016 and 2019 (Supplementary Table S4, Supplementary Fig. S3). Although the absolute cover of Montipora was relatively low in 2019 at all shallow locations (< 3.5%), its increase contributed ca. 39% to the overall hard coral cover increase at the shallow west and east during 2016–2019 (Supplementary Table S5). The increase of ‘other hard corals’, albeit statistically not significant, contributed 38% (shallow east) and 29% (shallow west) to overall hard coral cover increase. In the lagoon, branching Porites and ‘other hard corals’ contributed most to overall hard coral cover increase between 2016 and 2019 (36% and 33%, respectively) although increases were not statistically significant. Although overall soft coral cover did not increase at any location, Rhytisma covered 7.0% of the benthos at the deep west in 2019 (vs. not being recorded in 2016), while remaining < 1.5% at all other locations (Supplementary Fig. S3).
In terms of coral community composition (Fig. 4), in the lagoon, where bleaching-induced coral mortality was lowest and the magnitude of hard coral recovery was highest, coral community composition transitioned back to pre-bleaching assemblages between 2016 and 2019. Less pronounced than in the lagoon, western communities also showed returning transitions towards pre-bleaching assemblages, even at the deep reefs.
Non-metric multidimensional scaling (nMDS) of coral communities between 2014 and 2019 at Aldabra’s shallow (a) and deep (b) locations. Vectors connecting years display directional change of coral community composition at each location. Benthic and coral (italics) groups driving differences among locations and years displayed in grey. Scaled points indicate mean percent hard coral cover per location per year. Note that ‘other hard corals’ and ‘other soft corals’ contain different taxa than in the GEE analysis.
Discussion
Spatial variation in coral mortality following bleaching
Spatial variation in the extent of bleaching-induced hard coral mortality was clearly evident at Aldabra (see also Cerutti et al.33). Hard coral loss on the seaward reefs ranged between 51 and 62% and was only marginally lower at 15 m compared to 5 m water depth, indicating that corals in deeper water were as susceptible to heat stress as those in the shallows. This contrasts with other studies in which shallow coral communities suffered substantially higher post-bleaching mortality than deeper ones (e.g. Marshall and Baird13), but appears similar to observations in the Chagos Archipelago following the 1998 bleaching event34. In Aldabra’s lagoon, hard coral loss (34%) was considerably lower than on seaward reefs (51–62%). A lower reduction of hard coral cover inside Aldabra’s lagoon was also observed following the 1998/1999 bleaching event35, although no data is available for comparison. Within our study’s time frame, mean daily temperature variability was three-fold higher in the lagoon than on the seaward reefs, which has been proposed to result in inherently higher heat stress tolerance of hard corals and thus lower bleaching mortality11,12,36. Lagoon corals may also have been protected from UV radiation as a result of UV-absorbing leachate from seagrass leaves being steeped inside the lagoon37 or due to light attenuation from suspended particles38,39. Seagrass beds can be found in the vicinity of the lagoonal study reefs40 and (in comparison to the seaward reefs) turbidity in the lagoon was noted to be relatively high during surveys at slack high tide (pers. obs. Dec 2016, 2017, 2018, A.K.). Both mechanisms could result in reduced irradiance intensity experienced by corals, acting as additional buffers against thermal stress (but see Fisher et al.41). Furthermore, the high amount of suspended particulate material may allow corals to derive nutrients by heterotrophic feeding, contributing to higher bleaching survival when symbiont-acquired nutrients are low or entirely lacking9,42.
Across all locations, Acropora and Montipora consistently exhibited the highest susceptibility and suffered extensive losses following the bleaching event. Both genera are amongst the most susceptible hard corals to bleaching13,43,44 while massive Porites, as found elsewhere45,46, appeared resistant. However, the atoll-wide resistance of I. palifera and the location-specific resistance of branching Porites are striking (see also Cerutti et al.33). Whilst I. palifera remained abundant at the shallow western and lagoonal reefs, branching Porites (at Aldabra e.g. P. cylindrica, P. harrisoni, P. monticulosa, P. profundus and P. rus, which can exhibit branching or digitate growth forms31) suffered substantially lower mortality in the lagoon and remained relatively abundant there post-bleaching. Although it is currently unknown whether different species of branching Porites were present at Aldabra’s seaward reefs and in the lagoon, it is reasonable to suggest that adaptation to the variable lagoon environment was conducive to lower bleaching susceptibility and mortality.
Post-bleaching trajectories
Aldabra’s reef trajectories in the three post-bleaching years varied substantially across locations. In 2019, hard coral cover at the shallow west (23%) and the lagoon (30%) reached 68% and 93% of the pre-bleaching cover, respectively (vs. 15% and 21% hard coral cover in 2016, i.e. 44% and 65% of pre-bleaching cover, respectively), and by 2018, benthic communities were no longer dominated by turf algae. Although coral recovery also occurred at the shallow east (from 9% hard coral cover, i.e. 38% of pre-bleaching cover in 2016 to 13% hard coral cover, i.e. 54% of pre-bleaching cover in 2019), Halimeda remained the single most dominant benthic group. There was no coral recovery at the deep locations, with reefs remaining dominated by turf algae (west) and Halimeda (east) between 2016 and 2019.
Coral recovery was particularly fast inside Aldabra’s lagoon, almost reaching pre-bleaching levels within 3 years. It is possible that a large proportion of hard corals in the lagoon experienced only partial mortality and were able to rapidly regrow post-bleaching. This seems especially likely because the coral community composition also nearly reassembled to pre-bleaching levels by 2019. Survival and growth of remnant coral colonies is an important component in reef recovery, specifically in the first few years post-disturbance when the reproductive capacity of corals may be low23,47,48. This process is particularly important for isolated reefs that rely on coral recruitment from local sources as was reported from the remote Scott Reef, Australia23 and the Inner Seychelles3. In both cases, recovery following the 1998/1999 bleaching event was slow for 7–10 years but then increased exponentially with increasing recruitment capacity. Given that recovery of Aldabra’s seaward reefs following the 1998/1999 bleaching event was slow until 200549 (after which no further records are available), but coral cover had reached high levels by 2014, coral recruitment from local sources is likely important. The coral community in Aldabra’s lagoon may be critical for the long-term recovery of Aldabra’s reef system, with the potential to boost recovery at the seaward reefs. Nevertheless, information on the connectivity within Aldabra’s reefs as well as to other reefs in the region is limited (but see Crochelet et al.50) and research is needed in this area.
At both the lagoon and shallow west, the trajectories of turf algae and CCA following the bleaching event indicate a critical component of reef recovery44. Although the post-bleaching increase in CCA cover may be an artefact of the two-dimensional survey method employed here (i.e. where the loss of canopy forming corals may simply have increased the visibility of CCA underneath51), its stable cover in all post-bleaching years may benefit recovery processes. CCA can promote reef recovery by stabilising the reef framework52 and enhancing coral larvae settlement26,27, whereas turf algae, often the first benthic group to grow over dead coral substrate following disturbance20,23,53,54, can aggressively compete with adult corals through smothering and allelopathy52, and can inhibit coral larval settlement26,55 and the survival of coral recruits56. The rapid proliferation of turf algae following bleaching events can thus be detrimental to hard coral recovery. At Aldabra’s shallow reefs, however, turf algae cover was reduced to pre-bleaching levels within 2 years after bleaching (by 2018). This rapid reduction and the general lack of fleshy macroalgae across Aldabra’s reefs indicate high grazing pressure. Indeed, Aldabra hosts the highest biomass of herbivorous fish in the Seychelles31 and herbivory may actually have facilitated an increase in CCA52 following the bleaching event. Both herbivory and CCA abundance may be positively influenced by nutrient input of the guano from Aldabra’s numerous seabird populations57, as was recently shown at the Chagos Archipelago58.
At the shallow east and the deep reefs, recovery is likely restricted by abiotic conditions. Robinson et al.4 found that coral recovery in the Inner Seychelles following the 1997/1998 bleaching event was prolonged with increasing water depth and wave exposure which was attributed to lower coral growth rates, e.g. due to increased light attenuation at depth59,60. In terms of wave exposure, coral community composition is naturally shaped along wave energy gradients24,61,62. Branching (i.e. usually fast-growing) hard corals are less likely to occur where wave energy is high, which ultimately prolongs the time for hard coral recovery in those areas4. These abiotic conditions likely reflect the lack of hard coral recovery at Aldabra’s deep reefs (depth) and the lower magnitude of recovery at the shallow east (wave exposure). Although Aldabra’s deep western reefs remain dominated by turf algae, hard coral recovery can still occur in the long term. However, as already noted by Drew63 and Stobart et al.35, there is a prominent gradient of decreasing coral cover and increasing Halimeda cover from the western towards the eastern seaward reefs, attributed to an increase in hydrodynamic energy. Halimeda was already present in high abundance on Aldabra’s eastern reefs pre-bleaching and remained unchanged until 2016. However, by 2017 it became the single most dominant benthic category in the east, reaching up to 61% of overall benthic cover by 2018, potentially further constraining coral recovery there.
Hard coral recovery following disturbance is often driven by Acropora and Pocillopora4,64,65,66,67,68, but also other taxa such as Porites, Montipora, Isopora, Galaxea and Pavona are named in the literature64,67,69,70,71. At Aldabra’s lagoon, hard coral cover increase was largely due to the contributions of branching Porites and ‘other hard corals’. Many taxa that exhibit branching growth forms are characterised by life history traits that favour fast growth and wide dispersal, and are therefore often the first to recover at recently disturbed habitats. However, at the seaward reefs, most taxa that exhibit branching growth forms remained scarce during 2016–2019, with encrusting Montipora and ‘other hard corals’ contributing most to the early overall hard coral recovery. Previously, fast recovery of encrusting Montipora was associated with higher wave exposure20, which may explain why it is mainly found at Aldabra’s seaward locations.
Due to different bleaching susceptibility and recovery potential of coral taxa, overall hard coral recovery can be accompanied by a shift in coral community composition6,72 (but examples for reassembly also exist6,23,72). Such community shifts can alter the ecological functions of a reef and their response to future disturbance6. At Aldabra, coral communities in the lagoon recovered and reassembled almost completely within 3 years and also the western communities showed reassembly trajectories until 2019. The lower post-bleaching mortality of branching Porites in the lagoon and its relative contribution to overall hard coral recovery possibly indicates location-specific resilience, which may also have been important for the rapid return to pre-bleaching coral community composition there. As reef recovery is ongoing at Aldabra’s seaward reefs, however, it is possible that further taxa emerge which could dominate hard coral recovery, particularly if coral recruitment speeds up coral recovery3,23.
Furthermore, in contrast to the lagoon, pre-bleaching coral communities at the seaward reefs were not only characterized by hard corals. Soft corals also constituted an important component at the seaward reefs prior to the bleaching event and community reassembly there also depends on soft coral recovery. Overall soft coral recovery was negligible during 2016–2019, however, Rhytisma covered 7% of the benthos at the deep west in 2019, albeit not being recorded in 2016. This is similar to observations at Aldabra following the 1998 bleaching event where Rhytisma increased at 10 m water depth from zero to 8% cover by 2002, i.e. during the same time frame as studied here49,73. The subsequent increase to 26% cover by 2004 and the slow hard coral recovery caused reason to suggest Aldabra’s reefs had undergone a shift from hard to soft coral dominance32. It is possible that Rhytisma rapidly re-gains its previous abundance, with potential negative implications for hard coral recovery, but unlike implied previously32,74, the high abundance of Rhytisma has been restricted to Aldabra’s deep western reefs and does not affect the entire reef system.
Aldabra’s reef recovery in context
Overall annual rate of change in absolute hard coral cover at Aldabra’s shallow reefs was 2.2% over 3 years (Supplementary Table S6), similar to values reported for reefs within and outside no-take marine protected areas in Kenya (2.3% over 3 years), the Maldives (2.3% over 4 years) and Palau (2.1% over 8 years). Higher annual increases during similar recovery time frames were reported from Alphonse Atoll, Seychelles (2.9% over 2 years) and the Lakshadweep Islands (3.5% over 3 years), both of which are unprotected. In the Inner Seychelles, overall hard coral cover increased annually by 1.8% over 9 years (including reefs within and outside no-take marine protected areas) and at the remote Chagos Archipelago, cover increased annually by 2.6% over 11 years. However, looking at individual reefs regardless of location and level of protection, 56% of the annual rates of change provided in Supplementary Table S6 fall within the range reported for Aldabra’s reefs (1.3–3.0%), with 21% and 23% of the values falling below or above that range, respectively (Supplementary Figure S4). Hard coral cover increase at Aldabra’s shallow reefs, in terms of annual rate of change, was therefore not exceptionally low or high in comparison to reefs elsewhere.
Graham et al.5 reviewed global recovery rates following various acute disturbances and found a mean annual rate of 3.56% coral cover increase, with a range from 0.13 to 12.49%. However, rates were lowest in fully protected areas (0.8% vs. 4.0% in open areas) and varied depending on post-disturbance coral cover (between 2 and 4%). It was proposed that because protected areas promote higher coral cover (provided they are undisturbed), they are more susceptible to disturbances such as coral bleaching and may lose a higher proportion of corals than unprotected areas (the authors noted however, that enforcement or compliance could not be standardised among the assessed protected areas). Arguing that recovery dynamics are likely nonlinear and may be faster where disturbances have opened up more space for coral recruitment, recovery from high to medium coral loss (in their study 6–20% post-disturbance cover) may be faster than from extreme (< 5% post-disturbance cover) and small losses (21–30% post-disturbance cover)5. Our results do not match these findings (i.e. at Aldabra, recovery rate was highest where coral loss was lowest), probably because the early recovery at Aldabra observed here, particularly in the lagoon, is likely driven by the growth of remnant coral colonies rather than coral recruitment. Furthermore, in contrast to the studies used by Graham et al.5, full recovery is not realised at all of Aldabra’s locations due to the short recovery time frame studied here. These discrepancies show that post-disturbance recovery measured by hard coral cover increase alone misses the complexities of this process5 and reduces the meaningfulness of comparisons if no context is provided.
The return of coral cover and coral community composition to pre-disturbance values can provide this context and are useful indicators. For example, Aldabra’s lagoon nearly completely recovered its pre-bleaching hard coral cover within 3 years, matched by only two other reefs in our comparison within a similar time frame for recovery (Vipingo and Kanamai in Kenya, both unprotected; Supplementary Table S6). However, despite these often being the best indicators at data scarce locations (if available at all) it is important to acknowledge that longer-term effects of reef degradation may be masked and that reef state immediately pre-disturbance is likely not the ideal baseline (i.e. shifting baselines75).
Future prospects for Aldabra’s reefs
Despite its remoteness and strict protection, Aldabra’s reefs were significantly impacted by the 2015/2016 bleaching event, joining other remote reef systems such as the Chagos Archipelago54 and the northern Great Barrier Reef76 that suffered extensive bleaching-induced coral mortality. Nevertheless, we show that Aldabra’s lagoon rapidly recovered its pre-bleaching coral cover and reassembled to its pre-bleaching coral community composition within 3 years. While at the shallow seaward reefs, coral recovery is predicted to take ca. 5–9 more years (if there is no major bleaching event), it is likely prolonged at the deep reefs.
Our results add to the work of Cerutti et al.33 in providing locally important baseline information for ongoing coral reef research, ultimately feeding into Aldabra’s management and corroborating its protective status. On the global level, our study adds to previous work conducted at remote reefs (e.g. Gilmour et al.23, Sheppard et al.34), advancing the knowledge of coral bleaching impact and recovery in the absence of direct human disturbance. With an expected increase in the magnitude and frequency of mass bleaching events2, intervals between major events will become too short for adequate reef recovery, and reef locations where bleaching impact has been low so far, are likely to become more vulnerable to temperature stress. Our research underlines the need for drastic measures to reduce greenhouse gas emissions, alongside continued reduction and management of local human disturbance to conserve the world’s coral reefs.
Methods
Study site
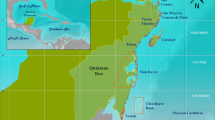
Aldabra (46° 20′ E, 9° 24′ S), managed by a Public Trust, the Seychelles Islands Foundation (SIF), since 1979, is an elevated coral atoll in the southwest of the Seychelles archipelago spanning 34 × 14.5 km (Fig. 5). Two distinct seasons govern Aldabra’s climate; the south-east trade winds from April to November create a dry and cooler climate whilst the north-west monsoon from November/December to March generates wet and warmer conditions. Aldabra’s large lagoon (196 km2) is encircled by four main islands and subject to a 2–3 m tidal range77. The north-east, east and south-east parts of Aldabra are exposed to strong winds and high wave energy, whilst the north-west and west are relatively sheltered78. Because of this contrasting level of exposure, we divided Aldabra’s reefs into three locations: seaward western, seaward eastern and lagoonal reefs (contrasting with Cerutti et al.33 who did not make the distinction between west and east at the seaward reef).
Location of Aldabra Atoll in the Western Indian Ocean (inset) with its four main islands, 12 survey sites at the seaward west (five sites), east (four sites) and in the lagoon (three sites) and three temperature logger sites (modified from Cerutti et al.33).
During the 2014–2017 Global Scale Coral Bleaching Event79, Aldabra’s reefs experienced continuous bleaching risk between December 2015 and June 2016. Satellite derived sea surface temperature reached a maximum of 30.7 °C in March 2016, resulting in a peak degree heating weeks (DHW80) value of 3.4 °C-weeks33 (but note that the NOAA Aldabra virtual station is located ca. 55 km north-east of Aldabra: 46° 50′ E, 9° 00′ S).
Data collection
Benthic data was collected from 12 permanent study sites (Fig. 5) once prior to bleaching, and four times post-bleaching. We refer to the survey periods as follows: (1) 2014 (pre-bleaching; data collected Dec 2014–Jan 2015), (2) 2016 (1st year post-bleaching; Dec 2016), (3) 2017 (2nd year post-bleaching; Dec 2017), (4) 2018 (3rd year post-bleaching; Dec 2018–Jan 2019) and (5) 2019 (4th year post-bleaching, Nov 2019–Jan 2020).
Of the 12 study sites, permanently marked transects are located at 5 and 15 m water depth at nine seaward survey sites (n = 18 seaward transects) and at ca. 2 m water depth at three sites in the lagoon (n = 3 lagoonal transects) (Fig. 5). All permanent transects are 50 m long, follow the depth contour, and are parallel to the shoreline. In 2014, two transect sections at 0–10 m and 20–30 m were surveyed on each permanent transect. During all post-bleaching surveys, increased availability of resources allowed an additional section to be surveyed on each permanent transect at 40–50 m. Along each section, benthic photoquadrats were collected on both sides of the tape measure with a GoPro camera attached to a 70 × 50 cm PVC frame at 70 cm height (pre-bleaching: GoPro Hero-3 Silver, 11 megapixels; post-bleaching: GoPro Hero-4 Silver and GoPro Hero-5 Black, 12 megapixels).
Water temperature data was obtained from three Onset loggers (HOBO U22-001) deployed between 2015 and 2018 at three of the permanent sites representing conditions in the lagoon (Site 9, 2 m water depth), shallow west (Site 1, 5 m) and shallow east (Site 5, 5 m; Fig. 5). Water temperature was recorded every 30 min with an accuracy of ± 0.2°C81.
Data processing and statistical analysis
R version 3.6.182 was used for statistical analysis. All graphs were created with the ggplot2 package83.
Benthic photos were analysed using Coral Point Count with Excel extensions (CPCe)84 by identifying the benthos at 16 randomly assigned points per image as described by Cerutti et al.33. This yielded a total of 179,247 points (excluding points on photoquadrat frame, transect tape or shadow) across all survey years. Five major categories were assigned for benthic cover analysis: hard coral, soft coral, turf algae, CCA and Halimeda spp. (hereafter ‘Halimeda’), which was by far the most dominant macroalga on Aldabra’s reefs. Fleshy macroalgae was not included as a category in the analysis as its mean cover was low across all locations and years (0–1.5%), except in 2018 at the deep eastern reefs (9.8%) where a seasonal Caulerpa racemosa bloom was recorded in Dec 2018 which had subsided by Feb 2019 (Supplementary Table S7), For the coral community, nine taxonomic categories were chosen following Cerutti et al.33 whereby the category ‘Acroporidae (excluding Isopora palifera)’ was replaced by Acropora and Montipora for more detailed analysis: Acropora (branching, plating and encrusting growth forms combined), Montipora (encrusting), I. palifera, Merulinidae, branching Porites (also includes digitate growth forms), massive Porites, other hard corals, Rhytisma, and other soft corals. To assess changes of the selected categories across locations between 2014 and 2016 (bleaching impact), the mean percent benthic cover of two transect sections was used as a response variable due to no third transect sections being conducted in 2014. To evaluate changes at these locations between 2016 and 2019 (post-bleaching trajectories), the mean percent benthic cover of three transect sections was included as a response variable.
Generalised Estimating Equations (GEE—‘geeglm’ function, geepack package85) with auto-regressive correlation structure were used to test for differences in the benthic and coral cover categories across time (bleaching impact: 2014, 2016; post-bleaching trajectory: 2016, 2017, 2018, 2019) and location (lagoon, west, east) at shallow (2 m, 5 m) and deep (15 m) water depth. Fixed explanatory variables included year, location, and their interactive effect (i.e. model structures: Year × Location; Location + Year; Location; Year). Survey site was set as a random factor to correct for pseudo-replication (transects sections). To correct for non-normality of the response variable (percentage benthic cover), we used different error distributions and link functions that best fitted the models and depended on the nature of the data (see Supplementary Tables S1–S4 for further details). We validated the models by running Generalised Linear Models first (‘glm’ function, R stats package82) and checking the residual distribution to see if the assumption of homogeneity of variance, normality and leverage were met. We then also checked the Pearson residual distribution for the GEE models. We used a post-hoc analysis based on least square means with Bonferroni adjustment (‘lsmean’ function, lsmean package86) to identify pairwise differences between the variables in significant interactive models (Year × Location). Due to many low values of Rhytisma (shallow and deep) and all coral categories at deep locations between 2016 and 2019, models of post-bleaching trajectories had a poor fit and these categories could not be tested statistically. All summaries of the GEE model outputs are provided in Supplementary Table S8 (separate file).
To further assess reef recovery, we calculated: (1) the annual rate of change in absolute hard coral cover increase87 and (2) an estimate of years remaining (from 2019) until hard coral cover reaches pre-bleaching levels (see Eqs. 1 and 2 in Supplementary Material). Acknowledging the latter as an extremely simplified projection that assumes a linear increase in hard coral cover, we applied both calculations only to those locations where hard coral cover had increased significantly between 2016 and 2019 (i.e. according to GEE analysis).
To assess which hard coral categories contributed most to overall hard coral cover increase (where it was significant according to GEE analysis), we calculated the absolute change in percentage cover between 2016 and 2019 for each hard coral category. Based on this, we calculated the contribution of each coral category to overall hard coral cover increase.
To visualise coral community trajectories, non-metric multidimensional scaling (nMDS) based on Bray–Curtis dissimilarity matrices of the coral community (using percent cover) was performed (‘metaMDS’ function, vegan package88). Only coral categories covering ≥ 5% of any transect section were included in the analysis. All other categories were combined into ‘other hard corals’ and ‘other soft corals’ (note that these categories contain different taxa to ‘other hard corals’ and ‘other soft corals’ in the GEE analysis). To display which coral and major benthic categories correlated with the community differences, significant correlation vectors were overlaid on nMDS plots (‘envfit’ function).
To assess daily water temperature regimes, overlapping time series of temperature data logged at the three monitored sites (Sites 1, 5 and 9) were selected (Feb 2015 and Nov 2018). Due to technical issues there were data gaps of 1-week (2–9 Apr 2015) and 5-months (12 Dec 2016–5 May 2017) for Site 1 and a data gap of 3-weeks for Site 9 (10 Dec 2016–1 Jan 2017). At Site 5, data was only available from Feb 2015 to Apr 2017 due to logger loss. Across the entire period, a minimum of 809 days remained for each logger (Site 9: 1353; Site 1: 1248, Site 5: 809) from which annual mean daily temperature (mean, maximum, minimum) and temperature variability (range, coefficient of variation) was calculated. To test for differences in these measures between sites, two-sided t-tests were conducted for each combination (i.e. Site 1 vs. Site 9; Site 1 vs. Site 5; Site 5 vs. Site 9) with Bonferroni adjustment applied to p values to correct for multiple comparisons.
Aldabra’s coral recovery in global context
To gain perspective on how Aldabra’s reef recovery fits into the global context, a table of studies reporting reef recovery was compiled (Supplementary Table S6) and the annual rate of change in absolute hard coral cover (Eq. 1 in Supplementary Material)87 was calculated for each reef. Studies were drawn from Baker et al.89 and Graham et al.5 and supplemented by more recent literature. Only studies reporting uninterrupted recovery from bleaching events were included; i.e. where no additional acute disturbance (e.g. bleaching, storm, Acanthaster outbreak) was reported within the recovery period.
Data availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
References
Hughes, T. P. et al. Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 359, 80–83 (2018).
Van Hooidonk, R., Maynard, J. A., Manzello, D. & Planes, S. Opposite latitudinal gradients in projected ocean acidification and bleaching impacts on coral reefs. Glob. Chang. Biol. 20, 103–112 (2014).
Graham, N., Jennings, S., MacNeil, M. A., Mouillot, D. & Wilson, S. K. Predicting climate-driven regime shifts versus rebound potential in coral reefs. Nature 518, 94–97 (2015).
Robinson, J. P. W., Wilson, S. K. & Graham, N. Abiotic and biotic controls on coral recovery 16 years after mass bleaching. Coral Reefs 38, 1255–1265 (2019).
Graham, N., Nash, K. L. & Kool, J. T. Coral reef recovery dynamics in a changing world. Coral Reefs 30, 283–294 (2011).
Johns, K. A., Osborne, K. O. & Logan, M. Contrasting rates of coral recovery and reassembly in coral communities on the Great Barrier Reef. Coral Reefs 33, 553–563 (2014).
Mumby, P. et al. Unprecedented bleaching-induced mortality in Porites spp. at Rangiroa, Atoll French Polynesia. Mar. Biol. 139, 183–189 (2001).
Furby, K. A., Bouwmeester, J. & Berumen, M. L. Susceptibility of central Red Sea corals during a major bleaching event. Coral Reefs 32, 505–513 (2013).
Grottoli, A. G., Rodrigues, L. J. & Palardy, J. E. Heterotrophic plasticity and resilience in bleached corals. Nature 440, 1186–1189 (2006).
Nakamura, T. & Van Woesik, R. Water-flow rates and passive diffusion partially explain differential survival of corals during the 1998 bleaching event. Mar. Ecol. Prog. Ser. 212, 301–304 (2001).
Oliver, T. & Palumbi, S. R. Do fluctuating temperature environments elevate coral thermal tolerance?. Coral Reefs 30, 429–440 (2011).
Safaie, A. et al. High frequency temperature variability reduces the risk of coral bleaching. Nat. Commun. 9, 1671. https://doi.org/10.1038/s41467-018-04074-2 (2018).
Marshall, P. A. & Baird, A. H. Bleaching of corals on the Great Barrier Reef: Differential susceptibilities among taxa. Coral Reefs 19, 155–163 (2000).
Stimson, J., Sakai, K. & Sembali, H. Interspecific comparison of the symbiotic relationship in corals with high and low rates of bleaching-induced mortality. Coral Reefs 21, 409–421 (2002).
Edmunds, P. J. Evidence that reef-wide patterns of coral bleaching may be the result of the distribution of bleaching-susceptible clones. Mar. Biol. 121, 137–142 (1994).
Mizerek, T. L., Baird, A. H. & Madin, J. S. Species traits as indicators of coral bleaching. Coral Reefs 37, 791–800 (2018).
Darling, E. S., McClanahan, T. R. & Côté, I. M. Life histories predict coral community disassembly under multiple stressors. Glob. Chang. Biol. 19, 1930–1940 (2013).
Sampayo, E. M., Ridgway, T., Bongaerts, P. & Hoegh-Guldberg, O. Bleaching susceptibility and mortality of corals are determined by fine-scale differences in symbiont type. Proc. Natl. Acad. Sci. USA 105, 10444–10449. https://doi.org/10.1073/pnas.0708049105 (2008).
Rowan, R. Thermal adaptation in reef coral symbionts. Nature 430, 742 (2004).
Gouezo, M. et al. Drivers of recovery and reassembly of coral reef communities. Proc. R. Soc. B. 286, 20182908 (2019).
Adjeroud, M. et al. Recovery of coral assemblages despite acute and recurrent disturbances on a South Central Pacific reef. Sci. Rep. 8, 9680 (2018).
Chong-Seng, K., Graham, N. & Pratchett, M. Bottlenecks to coral recovery in the Seychelles. Coral Reefs 33, 449–461 (2014).
Gilmour, J., Smith, L. D., Heyward, A. J., Baird, A. H. & Pratchett, M. Recovery of an isolated coral reef system following severe disturbance. Science 340, 69–71 (2013).
Gove, J. M. et al. Coral reef benthic regimes exhibit non-linear threshold responses to natural physical drivers. Mar. Ecol. Prog. Ser. 522, 33–48 (2015).
Smith, J., Hunter, C. L. & Smith, C. M. The effects of top-down versus bottom-up control on benthic coral reef community structure. Oecologia 163, 497–507 (2010).
Johns, K. A. et al. Macroalgal feedbacks and substrate properties maintain a coral reef regime shift. Ecosphere 9, e02349. https://doi.org/10.1002/ecs2.2349 (2018).
Yadav, S., Rathod, P., Alcoverro, T. & Arthur, R. “Choice” and destiny: The substrate composition and mechanical stability of settlement structures can mediate coral recruit fate in post-bleached reefs. Coral Reefs 35, 211–222 (2016).
Anthony, K. R. N. et al. Operationalizing resilience for adaptive coral reef management under global environmental change. Glob. Chang. Biol. 21, 48–61 (2015).
Sandin, S. A. et al. Baselines and degradation of coral reefs in the Northern Line Islands. PLoS One 3, e1548. https://doi.org/10.1371/journal.pone.0001548 (2008).
Stobart, B., Teleki, K., Buckley, R., Downing, N. & Callow, M. Coral recovery at Aldabra Atoll, Seychelles: Five years after the 1998 bleaching event. Philos. Trans. R. Soc. A. 363, 251–255 (2005).
Friedlander, A. et al. Biodiversity and Ecosystem Health of the Aldabra Group, Southern Seychelles: Scientific report to the Government of Seychelles (National Geographic Pristine Seas, 2015)
Norström, A. V., Nyström, M., Lokrantz, J. & Folke, C. Alternative states on coral reefs: Beyond coral–macroalgal phase shifts. Mar. Ecol. Prog. Ser. 376, 293–306 (2009).
Cerutti, J. et al. Impacts of the 2014–2017 global bleaching event on a protected remote atoll in the Western Indian Ocean. Coral Reefs 39, 15–26 (2020).
Sheppard, C., Harris, A. & Sheppard, A. Archipelago-wide coral recovery patterns since 1998 in the Chagos Archipelago, central Indian Ocean. Mar. Ecol. Prog. Ser. 362, 109–117 (2008).
Stobart, B. et al. Aldabra: Monitoring the path to recovery in Coral Degradation in the Indian Ocean: Status Report 2002 (eds. Lindén, O., Souter, D., Wilhelmsson, D. & Obura, D.) 232–247 (CORDIO, Department of Biology and Environmental Science, University of Kalmar, 2002).
Donner, S. D. An evaluation of the effect of recent temperature variability on the prediction of coral bleaching events. Ecol. Appl. 21, 1718–1730 (2011).
Iluz, D., Vago, R., Chadwick, N. E., Hoffman, R. & Dubinsky, Z. Seychelles lagoon provides corals with a refuge from bleaching. Res. Lett. Ecol. 2008, 281038. https://doi.org/10.1155/2008/281038 (2008).
Woesik, R. et al. Climate-change refugia in the sheltered bays of Palau: Analogs of future reefs. Ecol. Evol. 2, 2474–2484 (2012).
Morgan, K. M., Perry, C. T., Johnson, J. A. & Smithers, S. G. Nearshore turbid-zone corals exhibit high bleaching tolerance on the Great Barrier Reef following the 2016 ocean warming event. Front. Mar. Sci. 4, 224 (2017).
Hamylton, S., Hagan, A., Bunbury, N., Fleischer-Dogley, F. & Spencer, T. Mapping the lagoon at Aldabra Atoll, Western Indian Ocean. Atoll Res. Bull. 619, 45–59 (2018).
Fisher, R., Bessell-Browne, P. & Jones, R. J. Synergistic and antagonistic impacts of suspended sediments and thermal stress on corals. Nat. Commun. 10, 2346. https://doi.org/10.1038/s41467-019-10288-9 (2019).
Palardy, J. E., Rodrigues, L. J. & Grottoli, A. G. The importance of zooplankton to the daily metabolic carbon requirements of healthy and bleached corals at two depths. J. Exp. Mar. Bio. Ecol. 367, 180–188 (2008).
Hoey, A. et al. Recent advances in understanding the effects of climate change on coral reefs. Diversity 8, 1–22 (2016).
Fox, M. D. et al. Limited coral mortality following acute thermal stress and widespread bleaching on Palmyra Atoll, central Pacific. Coral Reefs 38, 701–712 (2019).
Loya, Y. et al. Coral bleaching: The winners and the losers. Ecol. Lett. 4, 122–131 (2001).
Pratchett, M., McCowan, D., Maynard, J. A. & Heron, S. F. Changes in bleaching susceptibility among corals subject to ocean warming and recurrent bleaching in Moorea, French Polynesia. PLoS One 8, e70443. https://doi.org/10.1371/journal.pone.0070443 (2013).
Diaz-Pulido, G. et al. Doom and boom on a resilient reef: Climate change, algal overgrowth and coral recovery. PLoS One 4, e5239. https://doi.org/10.1371/journal.pone.0005239 (2009).
Golbuu, Y. et al. Palau’s coral reefs show differential habitat recovery following the 1998-bleaching event. Coral Reefs 26, 319–332 (2007).
Buckley, R., Stobart, B., Downing, N. & Teleki, K. Aldabra Marine Programme Phase V. Report on the 2004 Aldabra Marine Programme Research. Cambrige Coastal Research Unit Report (University of Cambridge, 2004)
Crochelet, E. et al. A model-based assessment of reef larvae dispersal in the Western Indian Ocean reveals regional connectivity patterns—potential implications for conservation policies. Reg. Stud. Mar. Sci. 7, 159–167 (2016).
Goatley, C. H. R. & Bellwood, D. R. The roles of dimensionality, canopies and complexity in ecosystem monitoring. PLoS One 6, e27307. https://doi.org/10.1371/journal.pone.0027307 (2011).
Smith, J. E. et al. Re-evaluating the health of coral reef communities: Baselines and evidence for human impacts across the central Pacific. Proc. R. Soc. B. 283, 1–9 (2016).
Diaz-Pulido, G. & McCook, L. The fate of bleached corals: Patterns and dynamics of algal recruitment. Mar. Ecol. Prog. Ser. 232, 115–128 (2002).
Head, C. et al. Coral bleaching impacts from back-to-back 2015–2016 thermal anomalies in the remote central Indian Ocean. Coral Reefs 38, 605–618 (2019).
Ford, A. K. et al. Evaluation of coral reef management effectiveness using conventional versus resilience-based metrics. Ecol. Indic. 85, 308–317 (2017).
Arnold, S. N., Steneck, R. & Mumby, P. Running the gauntlet: Inhibitory effects of algal turfs on the processes of coral recruitment. Mar. Ecol. Prog. Ser. 414, 91–105 (2010).
Šúr, M., Bunbury, N. & Van De Crommenacker, J. Frigatebirds on Aldabra Atoll: Population census, recommended monitoring protocol and sustainable tourism guidelines. Bird Conserv. Int. 23, 214–220 (2013).
Benkwitt, C. E., Wilson, S. K. & Graham, N. Seabird nutrient subsidies alter patterns of algal abundance and fish biomass on coral reefs following a bleaching event. Glob. Chang. Biol. 25, 2619–2632 (2019).
Huston, M. Variation in coral growth rates with depth at Discovery Bay, Jamaica. Coral Reefs 4, 19–25 (1985).
Pratchett, M. et al. Spatial, temporal and taxonomic variation in coral growth—implications for the structure and function of coral reef ecosystems. In Oceanography and Marine Biology: An Annual Review Vol 53 (eds Hughes, R. N. et al.) 215–295 (CRC Press, Boca Raton, 2015).
Done, T. J. Patterns in the distribution of coral communities across the central Great Barrier Reef. Coral Reefs 1, 95–107 (1982).
Williams, G., Gove, J., Eynaud, Y., Zgliczynski, B. J. & Sandin, S. Local human impacts decouple natural biophysical relationships on Pacific coral reefs. Ecography 38, 751–761 (2015).
Drew, E. A photographic survey down the seaward reef-front of Aldabra Atoll. Atoll Res. Bull. 193, 1–17 (1977).
Hagan, A. B. & Spencer, T. Reef resilience and change 1998–2007, Alphonse Atoll, Seychelles in Proceedings of the 11th International Coral Reef Symposium (eds. Riegl, B. & Dodge, R.) 388–392 (Nova Southeastern University National Coral Reef Institute, 2008).
Sheppard, C. & Sheppard, A. British Indian Ocean Territory (Chagos Archipelago). In World Seas: An Environmental Evaluation. Volume II: The Indian Ocean to the Pacific (ed. Sheppard, C.) 237–252 (Academic Press, New York, 2019).
Arthur, R., Done, T. J., Marsh, H. & Harriott, V. Local processes strongly influence post-bleaching benthic recovery in the Lakshadweep Islands. Coral Reefs 25, 427–440 (2006).
Bruno, J. F., Siddon, C., Witman, J., Colin, P. & Toscano, M. E. Niño related coral bleaching in Palau, Western Caroline Islands. Coral Reefs 20, 127–136 (2001).
Wilson, S. K., Robinson, J. P. W., Chong-Seng, K., Robinson, J. & Graham, N. Boom and bust of keystone structure on coral reefs. Coral Reefs 38, 625–635 (2019).
McClanahan, T. R., Maina, J., Starger, C. J., Herron-Perez, P. & Dusek, E. Detriments to post-bleaching recovery of corals. Coral Reefs 24, 230–246 (2005).
Brown, B. E. & Suharsono, S. Damage and recovery of coral reefs affected by El Niño related seawater warming in the Thousand Islands, Indonesia. Coral Reefs 8, 163–170 (1990).
Guzman, H. M. & Cortés, J. Reef recovery 20 years after the 1982–1983 El Niño massive mortality. Mar. Biol. 151, 401–411 (2007).
Burt, J., Bartholomew, A. & Usseglio, P. Recovery of corals a decade after a bleaching event in Dubai, United Arab Emirates. Mar. Biol. 154, 27–36 (2008).
Stobart, B., Teleki, K., Buckley, R., Downing, N. & Callow, M. Coral recovery at Aldabra Atoll, Seychelles: Five years after the 1998 bleaching event. Philos. Trans. R. Soc. A 363, 251–255 (2005).
Ateweberhan, M. et al. Climate change impacts on coral reefs: Synergies with local effects, possibilities for acclimation, and management implications. Mar. Pollut. Bull. 74, 526–539 (2013).
Hughes, T. P. et al. Shifting base-lines, declining coral cover, and the erosion of reef resilience: Comment on Sweatman et al. (2011). Coral Reefs 30, 653–660 (2011).
Hughes, T. P. et al. Global warming transforms coral reef assemblages. Nature 556, 492–496 (2018).
Farrow, G. E. & Brander, K. M. Tidal studies on Aldabra. Philos. Trans. R. Soc. B 260, 93–121 (1971).
Taylor, J. D. Intertidal zonation at Aldabra Atoll. Philos. Trans. R. Soc. B 260, 173–213 (1971).
Eakin, C. M., Sweatman, H. P. A. & Brainard, R. E. The 2014–2017 global-scale coral bleaching event: Insights and impacts. Coral Reefs 38, 539–545 (2019).
Liu, G. et al. NOAA Technical Report NESDIS 143. NOAA Coral Reef Watch 50 km satellite sea surface temperature-based decision support system for coral bleaching management (National Oceanic and Atmospheric Administration, 2013).
Onset Computer Corporation. HOBO Water Temp Pro v2 (U22-001) Manual (2012).
R Core Team. R: A Language and Environment for Statistical Computing (2019).
Wickham, H. ggplot2—Elegant graphics for data analysis (2nd edition). J. Stat. Softw. 77, 3–5 (2016).
Kohler, K. E. & Gill, S. M. Coral Point Count with Excel extensions (CPCe): A visual basic program for the determination of coral and substrate coverage using random point count methodology. Comput. Geosci. 32, 1259–1269 (2006).
Halekoh, U., Højsgaard, S. & Yan, J. The R package geepack for generalized estimating equations. J. Stat. Softw. 15, 1–11 (2006).
Lenth, R. V. Least-squares means: The R package lsmeans. J. Stat. Softw. 69, 1–33 (2016).
Coté, I. M., Gardner, T. A., Hutchinson, D. J. & Watkinson, A. R. New approaches to estimating recent ecological change on coral reefs. In Coral Reef Conservation (eds Coté, I. M. & Reynolds, J. R.) 293–313 (Cambridge University Press, Cambridge, 2006).
Oksanen, J. et al. Package vegan: Community ecology package 2.5-1 (2013).
Baker, A. C., Glynn, P. W. & Riegl, B. Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery trends and future outlook. Estuar. Coast. Shelf Sci. 80, 435–471 (2008).
Acknowledgements
A special thanks goes to the Seychelles Islands Foundation and staff for their work and support. Specifically we thank Jude Brice, April Burt, Daig Romain, Rebecca Filippin, Sheril De Commarmond, Samuel Basset, Terence Mahoune, Ella Nancy, Joel Bonne, Marvin Roseline, Leeroy Estrale, Ronny Marie, Jennifer Appoo, Trevor Henriette, Jake Letori, Lorraine Cook, Selwyn Laurence, Albert Belmont, Jeremy Raguain, Janske van de Crommenacker, Germano Soru, Diane Ernesta, Matthew Waller, Luke A'Bear, Maria Bielsa, Mikael Esparon, Sophie Adams and Julio Agricole for data collection and support in the field. We further thank the Global Environment Facility for funding diving and research equipment and the development of the Aldabra Reef Monitoring programme. Thanks also go to Phillip Haupt as well as Anthony Bernard and Nick Riddin from the South African Institute for Aquatic Biodiversity and SIF staff for establishing the permanent benthic transects; and Rebecca Klaus for advice on benthic transect set-up and photoquadrat methodology. Finally, we thank Rowana Walton for the identification of pre-bleaching data. We also want to thank the editor and two anonymous reviewers for their constructive feedback.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
A.K., C.W. and N.B. initiated the study. A.K. and C.S. collected data. A.K. and V.M. analysed the data and created figures and tables. A.K. drafted the manuscript and prepared the supplementary material, with significant input from N.B., A.F. and C.W. All authors reviewed the manuscript and gave final approval of the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Koester, A., Migani, V., Bunbury, N. et al. Early trajectories of benthic coral reef communities following the 2015/16 coral bleaching event at remote Aldabra Atoll, Seychelles. Sci Rep 10, 17034 (2020). https://doi.org/10.1038/s41598-020-74077-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-74077-x
This article is cited by
-
A functional perspective on the meaning of the term ‘herbivore’: patterns versus processes in coral reef fishes
Coral Reefs (2024)
-
Impact of petroleum hydrocarbon and heavy metal pollution on coral reefs and mangroves: a review
Environmental Chemistry Letters (2024)
-
Growth rate and projected age at sexual maturity for immature hawksbill turtles and green turtles foraging in the remote marine protected area of Aldabra Atoll, Seychelles
Marine Biology (2023)
-
Spatial and temporal patterns in the coral assemblage at Clipperton Atoll: a sentinel reef in the Eastern Tropical Pacific
Coral Reefs (2022)
-
Rapid recovery of coral communities from a mass bleaching event in the summer of 2016, observed in Amitori Bay, Iriomote Island, Japan
Marine Biology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.