Abstract
Archaeological research is radically transforming the view that the Amazon basin and surrounding areas witnessed limited societal development before European contact. Nevertheless, uncertainty remains on the nature of the subsistence systems and the role that aquatic resources, terrestrial mammalian game, and plants had in supporting population growth, geographic dispersal, cultural adaptations and political complexity during the later stages of the pre-Columbian era. This is exacerbated by the general paucity of archaeological human remains enabling individual dietary reconstructions. Here we use stable carbon and nitrogen isotope analysis of bone collagen to reconstruct the diets of human individuals from São Luís Island (Brazilian Amazon coast) dated between ca. 1800 and 1000 cal BP and associated with distinct ceramic traditions. We expanded our analysis to include previously published data from Maracá and Marajó Island, in the eastern Amazon. Quantitative estimates of the caloric contributions from food groups and their relative nutrients using a Bayesian Mixing Model revealed distinct subsistence strategies, consisting predominantly of plants and terrestrial mammals and variably complemented with aquatic resources. This study offers novel quantitative information on the extent distinct food categories of polyculture agroforestry systems fulfilled the caloric and protein requirements of Late Holocene pre-Columbian populations in the Amazon basin.
Similar content being viewed by others
Introduction
Archaeological investigations in lowland South America are radically transforming our understanding of long-term socio-ecological systems in the Neotropics and dismissing the view that the Amazon basin witnessed limited socio-economic development in pre-Columbian times1. Recent studies reveal a complex picture during the Late Holocene, marked by increased archaeological visibility associated with groups producing distinct material cultures and engaging in diversified subsistence strategies1,2 with extensive transformation of soils3, vegetation composition4, and landscapes5,6,7.
During the Late Holocene specialized land-use systems based on agriculture or the management of aquatic resources coexisted with more versatile subsistence strategies founded on polyculture agroforestry8,9, with substantial temporal and spatial variability across the basin2. Evidence from coastal areas is relatively limited, but equally diversified. Plant cultivation with extensive raised fields has been documented in seasonally flooded savannahs of the northern Amazon (The Guianas)10, while fishing-based economies supported by water management systems, complemented with plant gathering and possibly some cultivation, occurred in some areas of the eastern Amazon (Marajó Island)11,12. Numerous shell middens along the coast suggest that food security was also achieved through the exploitation of estuarine and marine resources by groups producing some of the earliest ceramic artefacts in Amazonia13.
Despite the growing interest in the origin and changing nature of pre-Columbian economies8, attempts to comprehend their impacts on individual diets have been comparatively limited12,14,15,16,17. Moreover, while the role of agriculture in supporting population growth and the establishment of sedentary settlements in the recent pre-Columbian era has been the subject of contentious debate2,11, the importance of aquatic resources has received comparatively little attention18. Aquatic resources are often invoked as economically important food items to both coastal and riverine populations7,18,19,20,21,22, but their relative contribution to diet is poorly understood and surrounded by controversial generalizations23,24,25,26. Uncertainties also remain regarding the extent to which plants (cultivated, gathered) and terrestrial animals fulfilled the energy requirements of specialized and mixed subsistence systems. Their potential intensification and role in sustaining dense populations during the later stages of the pre-Columbian era is still a matter of debate2. Stable carbon and nitrogen isotope analysis of bone collagen is a well established approach for deriving dietary information from human, faunal, and plant remains27,28,29 and has been employed to reconstruct pre-Columbian subsistence economies across much of South America30,31,32. However, due to the paucity of archaeological human remains there have been limited applications on past populations in the Amazon basin until now12,14,15,16,17, with most studies lacking direct and robust chronological information, as well as contextual isotopic baselines12,15.
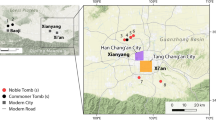
Here we offer a novel contribution to unravel pre-Columbian diets in lowland Amazonia and specifically the role of fish as a source of dietary protein. We analyzed the stable carbon and nitrogen isotope composition of bone collagen from five human individuals from archaeological sites (Panaquatira, Bacanga, and Paço do Lumiar) on São Luís Island, located off the Brazilian Amazon coast (Fig. 1). Four individuals were directly radiocarbon dated (AMS) to between ca. 1800 to 1000 cal BP and all were recovered from archaeological contexts associated with distinct ceramic traditions, along with terrestrial mammal, fish, and shellfish remains. The proportional contributions of fish, terrestrial mammals and plants to diet were estimated using Bayesian Stable Isotope Mixing Models (BSIMMs), which provide quantitative estimates of relative caloric and protein contributions from available food groups. Pre-Columbian human remains are rare in the Amazon basin and the obtained results offer the most robust and chronologically secure information on individual diets in pre-Columbian lowland Amazonia to date.
The study area in the eastern Amazon. (A) The location of São Luís Island and the approximate location of Late Holocene sites at Maracá region (Gruta das Caretas and Gruta do Pocinho) and on Marajó Island (Teso dos Bichos, Monte Carmelo and Matinadas). (B) Detailed location of pre-Columbian sites on São Luís Island, including the sites of Panaquatira, Bacanga and Paço do Lumiar. Maps generated using ArcGIS 10.7 (https://desktop.arcgis.com/en/) and Adobe Illustrator CS6 (https://www.adobe.com/es/products/illustrator.html), on data publicly available from NASA/JPL/NIMA (South American), the Brazilian Institute of Geography and Statistics (IBGE), the National Institute for Space Research (DPI/INPE), and the Brazilian Agricultural Research Corporation (Embrapa) (detailed maps of Amazonia and São Luís Island).
Results
Stable isotope analysis and Bayesian dietary reconstruction
Six human individuals were recovered from the archaeological shell middens of Bacanga (n = 1), Paço do Lumiar (n = 1) and Panaquatira (n = 4). The individuals were fragmented and mostly disarticulated, preventing accurate sex and age identification. Four individuals were directly radiocarbon dated using AMS, with the calibrated ages corrected for the average relative contribution of marine carbon to collagen estimated by the BSIMMs (Table 1; see Methods for details).
At the shell midden of Bacanga one infant individual (BC1) was found in direct association with a ceramic vessel from the Mina tradition, faunal remains, and blocks of ferruginous rocks, and was directly dated to 1840–1700 cal BP (2σ). At Paço do Lumiar, one adult individual (PDL2), dated to 1190–990 cal BP (2σ), was found at the base of the shell midden, with ceramic fragments stylistically comparable to Incised Rim and Mina traditions. At Panaquatira (PNQ), a double burial with one adult (PNQ1A) and one infant (PNQ1B) was found within a ceramic funerary urn with Polychrome decoration, surrounded by stones and shells, emerging from a superficial dark soil overlying the shell midden. The adult (PNQ1A) was dated to 1830–1620 cal BP (2σ). Another adult individual (PNQ2) was found disarticulated and buried with shells and stone tools in a funerary urn with Polychrome decoration, in a dark soil covering the shell midden. This individual did not produce collagen for radiocarbon dating or stable isotope analysis. Another adult individual (PNQ3) was found disarticulated at the base of the shell midden in association with ceramic fragments of Mina tradition and shell beads, and was dated to 1710–1540 cal BP (2σ).
Faunal material was collected from the site of Panaquatira (PNQ). The δ13C and δ15N values of terrestrial mammals (n = 12) ranged from − 21.1 to − 19.1 ‰ and from + 4.0 to + 9.4 ‰, respectively (Fig. 2). The analyzed specimens that could be taxonomically identified (paca, cavy, agouti, brocket deer) were predominantly herbivorous and all δ13C values fell within the range associated with environments dominated by C3 plants33, with some amount of water stress that could be expected in the transitional zone between humid and savannah forest34. The δ13C and δ15N values for fish (n = 11) were statistically distinguishable from terrestrial mammals (n = 23, p < 0.01, F = 617.8 for δ13C, p < 0.01, F = 44.52 for δ15N, One-way Anova) and ranged from − 12.3 to − 8.9 ‰ and from + 7.5 to + 12.5 ‰, respectively. Some of the remains could be identified as catfish (Ariidae), which is a common taxa in the region35. The high δ13C values observed are typical of fish from marine ecosystems36 and are also comparable with other marine taxa from the southwestern Atlantic Ocean30,37. Our marine samples show δ13C values considerably higher than modern fish from mangrove environments along the Amazon coast38, suggesting a larger contribution of marine phytoplankton to PNQ’s marine food web. We exclude that fish δ13C values at PNQ could reflect the input of C4 aquatic macrophytes since their contribution to fish diet is negligible in this region39. Fish δ15N values reflect diets based on a mixture of benthic organisms (crustaceans, polychaetes) and detritus38.
Modern plant δ13C and δ15N values from São Luís Island varied considerably within and between taxa (Supplementary information 1). The δ13C values of bulk plant (tuber and kernel) were corrected for the Suess effect (+ 2.13 ‰) (see Methods for details). Maize (Zea mays) had δ13C and δ15N values ranging from − 9.1 to − 8.2 ‰ and from + 2.3 to + 6.3 ‰, respectively. Manioc and macaxeira (both Manihot esculenta) and sweet potato (Ipomoea batatas) had comparable δ13C values, from − 25.3 to − 24.0 ‰, but contrasting δ15N values. A single sweet potato had a δ15N value of + 9.2 ‰, while manioc tubers displayed a larger variability, ranging from − 1.9 to + 6.0 ‰. The reason for the differences in δ15N values among manioc and macaxeira samples is unclear but may arise from the differential use of fertilizers and/or cultivation of macaxeira in soils with high 14N volatilization or mineralization in the context of swidden agriculture40.
The δ13C values for human individuals from São Luís Island ranged from − 16.4 ‰ (PNQ1A and PNQ3) to − 18.3 ‰ (BC1) (Fig. 2). The δ15N values ranged from + 11.1 ‰ (PNQ1B) to + 12.4 ‰ (PNQ3). There were no substantial differences in the δ15N values between adults and infants, and the lack of sex identification prevented further consideration of the sex and gender dimensions of diet. Nevertheless, it is worth noting that δ15N values for the two infants (PNQ1B and BC1) could also reflect some level of breastfeeding effect41, implying that some young-adult lactating women had even lower δ15N values compared to those reported here. Overall the isotope results indicate mixed subsistence strategies with substantial contribution of C3 plants, terrestrial mammalian game, marine fish and possibly maize (although a number of C4 plants occur in the Amazon42).
The stable isotope values for the individuals from São Luís Island show significant differences compared to Late Holocene pre-Columbian groups that inhabited the eastern Amazon basin (Fig. 2). A number of human individuals from the sites of Teso dos Bichos, Monte Carmelo, Matinadas and an unknown site on Marajó Island (n = 7) have collagen δ13C and δ15N values consistent with mixed contributions from freshwater fish, C3, and C4 plant protein12. The precise chronology of these samples is unknown12, but they may be attributed to stratified chiefdom-level societies (Marajoara Phase) which supposedly intensified the exploitation of freshwater resources at ca. 1600–650 BP, a process that supported or catalyzed considerable social complexity around the control of aquatic resources in the region43. Freshwater fish was also frequently consumed by Late Holocene individuals (n = 17) from Gruta das Caretas and Gruta do Pocinho in Maracá, on the north bank of the Amazon river, dating to ca. 500 BP. The bulk collagen δ13C and δ15N values for these individuals were interpreted as resulting from diets based on freshwater resources, together with C3 plants, and possibly also some contributions from C4 or CAM crops15.
In general, the δ13C values at São Luís Island were significantly higher compared with those from the Maracá region (n = 22, p < 0.05, Kruskal–Wallis), but not significantly different from those of Marajó Island (n = 12, p > 0.05, F = 0.1734, One-way ANOVA). Differences between São Luís Island and Maracá can be attributed to the consumption of marine resources at São Luís Island. The high δ13C values observed at Marajó Island, instead, could indicate the contribution of maize to dietary proteins12. The δ15N values at São Luís were significantly higher compared to both Maracá (n = 22, p < 0.05, Kruskal–Wallis) and Marajó Island (n = 12, p < 0.05, Kruskal–Wallis), suggesting a greater consumption of terrestrial or aquatic animal protein at São Luís and/or regional isotopic differences in resource baselines44. These mixed dietary regimes, large variations in the macronutrient compositions of foods, and uncertainties in food isotopic values and in diet-to-consumer isotopic offsets limit the use of simple linear mixing models. The multiple sources of uncertainty can be more efficiently handled by employing Bayesian Stable Isotope Mixing Models (BSIMMs) that express dietary contribution probability distributions45,46,47. To estimate the caloric contributions for the São Luís individuals from four food groups (terrestrial C3 plants, maize, terrestrial mammals, and marine fish) and for both Maracá region and Marajó Island (terrestrial C3 plants, maize, terrestrial mammals, and freshwater fish/reptile) we employed the Bayesian mixing model FRUITS45. There is no evidence that individuals from both Maracá region and Marajó Island exploited marine resources, however freshwater fish remains were reported for the site of Teso dos Bichos (Marajó Island), along with plants and a few terrestrial mammals12,15. The model in FRUITS accounts for the uncertainties in consumer and food isotopic values, macronutrient composition, diet-to-consumer isotopic offsets, and dietary routing. The latter accounts for non-protein contributions (carbohydrates and lipids) to consumer δ13C collagen, necessary for higher-accuracy dietary estimates48. Two dietary estimates were generated: the relative caloric contribution from each food group, and the relative caloric contribution from the protein-only component of each food group. These two estimates differ since the macronutrient composition of the food groups varies. Animal food groups typically have a considerably higher protein content when compared to plant foods, thus, caloric contributions may be predominantly from plants while animal sources simultaneously constitute the main source of protein. In the following sections, caloric estimates/contributions refers to food caloric contributions and protein estimates/contributions refers to the caloric contributions from the protein-only component of the food groups (see Material and Methods for model implementation details).
The dietary estimates generated by FRUITS for individuals at São Luís Island indicated that the majority of calories were supplied by terrestrial mammals (median 36%, Q1 19% and Q3 53%) and/or C3 plants (median 31%, Q1 16% and Q3 49%), followed by maize (median 14%, Q1 7% and Q3 25%) and marine fish (median 10%, Q1 4% and Q3 19%) (Fig. 3). There was considerable overlap between caloric output from terrestrial mammals and C3 plants for both individual and group estimates. This lack of estimate resolution is due to insufficient separation of the distribution of isotopic values characterizing the two food groups and to isotopic equifinality, that is, varying combinations of food contributions that may result in the same consumer isotopic values.
Box and whiskers plot summarizing the probability distribution of the relative caloric contributions from distinct food groups to individual diets at Maracá, Marajó Island and São Luís Island. The horizontal lines correspond to the median and the hinges to the 25th (Q1) and 75th (Q3) percentiles. The whiskers extend from the hinge to the smallest or largest observation greater than or equal to − 1.5 * IQR or less than or equal to + 1.5 * IQR, respectively.
The model estimated that terrestrial mammals were the main source of dietary protein for all individuals (median 48%, Q1 28% and Q3 65%), followed by marine fish (median 30%, Q1 15% and Q3 48%), C3 plants (median 8%, Q1 3% and Q3 18%), and then maize (median 5%, Q1 2% and Q3 11%) (Fig. 4). Compared to relative caloric estimates, protein contributions achieved a higher precision (see also Supplementary information 2 and 3). In summary, individuals at São Luís Island relied primarily on terrestrial resources for both calories and proteins.
Box and whiskers plot summarizing the probability distribution of the relative caloric contribution from the protein-only component from distinct food groups to individual diets at Maracá region, Marajó Island and São Luís Island. The horizontal lines correspond to the median and the hinges to the 25th (Q1) and 75th (Q3) percentiles. The whiskers extend from the hinge to the smallest or largest observation greater than or equal to -1.5 * IQR or less than or equal to + 1.5 * IQR, respectively.
For individuals from Marajó Island, FRUITS estimates assigned the majority of dietary calories to terrestrial mammals (median 26%, Q1 13% and Q3 42%), and/or C3 plants (median 25%, Q1 12% and Q3 42%) and/or maize (median 26%, Q1 15% and Q3 39%), with considerable overlap among estimates. Freshwater fish/reptiles offered the smallest contribution of all food groups (median 10%, Q1 4% and Q3 21%). It is worth noting that one individual (MrJ3, from Teso dos Bichos) had maize as the prevailing source of calories (average 34 ± 14%). Terrestrial mammals (median 37%, Q1 18% and Q3 57%) and/or freshwater fish/reptiles (median 34%, Q1 16% and Q3 55%) were the main sources of dietary protein, although with estimates overlapping considerably for most of the individuals. These were then followed by maize (median 11%, Q1 5% and Q3 21%) and C3 plants (median 6%, Q1 2% and 15%).
Similarly, the probability distributions generated by FRUITS for individuals from the Maracá region revealed that the majority of dietary calories were sourced from terrestrial mammals (median 30%, Q1 15% and Q3 48%) and/or C3 plants (median 26%, Q1 12% and Q3 43%), with some overlap in the estimates. These were followed by maize (median 17%, Q1 8% and Q3 28%) and freshwater fish/reptiles (median 15%, Q1 7% and Q3 27%), although for some individuals these two food groups also overlapped considerably. Dietary proteins were obtained from freshwater fish/reptiles (median 42%, Q1 23% and Q3 62%) and/or terrestrial mammals (median 38%, Q1 19% and Q3 59%), followed by contributions from maize (median 6%, Q1 2% and Q3 12%) and C3 plants (median 5%, Q1 2% and Q3 14%).
Discussion
The results from São Luís Island, along with the review of previously published δ13C and δ15N values using a BSIMMs approach, reveal distinct adaptive strategies in the eastern Amazon basin during the Late Holocene. The differential dietary contributions from terrestrial mammal and C3 plants at São Luís Island could not be efficiently resolved by BSIMMs due to the isotopic similarity of these sources and the isotopic equifinality resulting from the mixed contributions from all food groups. However, model estimates adequately distinguished the relative contribution of marine fish from the terrestrial food groups (terrestrial mammals, C3, and maize). Despite the proximity to marine resources and contextual zooarchaeological evidence for fishing, model estimates reveal that terrestrial mammals provided greater amounts of dietary proteins to individuals at São Luís Island compared to marine fish. These results call into question the widespread assumption that aquatic resources (e.g.fish, reptiles) were the main economic component, or source of protein, to pre-Columbian populations living in proximity to productive aquatic environments in lowland Amazonia20,21,22,23,24. However, it is necessary to acknowledge that only a small number of samples were analysed.
Freshwater fish/reptiles and/or terrestrial mammals provided the majority of dietary protein to populations located at the mouth of the Amazon river (Marajó Island and Maracá), but uncertainties remain on the relative contributions from these two food groups. According to Roosevelt12, freshwater fish dominated faunal remains at the site of Teso dos Bichos on Marajó Island, while remains of terrestrial mammalian game were rare. However, for most of the sites on Marajó Island and in the Maracá region there is no available information on faunal remains15. This lack of contextual faunal and botanical data, along with uncertainties associated with the local isotope baselines, limit the possibility of obtaining precise dietary estimates. Model estimates, however, do indicate that the majority of caloric contributions were from terrestrial mammals and plants, and not from freshwater resources. While fish may have been a dominant source of protein, our results suggest that considerable investments were allocated to hunting, forest management, and plant cultivation, as shown by other lines of evidence across the Amazon basin1,4,10,49.
Maize, a widespread C4 crop in the Amazon by the Late Holocene50, was not a staple for the sampled individuals, but was likely consumed within the broad spectrum of resources exploited by distinct agroforestry systems. The relatively high consumption of C4 plants (possibly maize) by some individuals at Marajó Island is significant though, and may reopen a contentious debate on the role that plant cultivation had in supporting the emergence of political complexity during the Marajoara Phase11,12. While we recognize that dietary estimates can be further refined with increasing control of contextual faunal and plant baselines51 and isotopic analyses of single amino acids29, the results presented herein constitute the most robust source of information on individual diets in the pre-Columbian Amazon and corroborate the growing consensus that diversified subsistence economies fuelled cultural, demographic and environmental transformations in the eastern Amazon basin during the Late Holocene.
Methods
Geographic and archaeological contexts
São Luís Island, in the state of Maranhão, is approximately 832 km2 and is part of the Maranhense Gulf geological-geomorphological feature which forms a large and complex estuarine system delimited by the bays of São Marcos and São José (Fig. 1). Lying in the transition between Amazon and Northeastern vegetation52, the region has a tropical climate with dry (January to June) and rainy (June to December) seasons, with species from the Amazon forest, caatinga, and cerrado biomes, and extensive restinga and mangrove ecosystems along the coast which are important nurseries for aquatic organisms53. The coastal environment is highly dynamic and shows relatively high biological productivity and biomass with limited seasonal variability due to the combination of semidiurnal macro-tidal regimes, mangrove vegetation, and freshwater input (notably in São Marcos Bay)35. This has favoured fish and shellfish exploitation by groups on the island since at least the Middle and Late Holocene54, during the spread of ceramic artefacts associated with the Mina tradition. The archaeological shell midden of Bacanga contains faunal evidence of fishing in marine and estuarine environments at that time, including taxa of different trophic positions (e.g. sharks, catfish). Marine resources were also exploited by groups using ceramic vessels similar to the Amazonian Incised Rim and possibly the Polychrome tradition during the Late Holocene, the latter associated with dark soils visibly similar to Amazonian Dark Earths55. Evidence of fishing and shellfish collection at such sites are better represented at the shell middens of Paço do Lumiar and Panaquatira55. Fishing is well documented historically among local Tupinambá groups in the seventeenth century56,57,58 and currently has major socio-economic and cultural importance on the Island59.
Sample preparation for stable isotopic analysis
Archaeological human and faunal samples from São Luís Island were provided by Brandi e Bandeira Consultoria Cultural and the permits for stable isotope analyses were obtained from the Instituto do Patrimônio Histórico e Artístico Nacional (IPHAN, protocol no. 01506.00407/2012-14). Rib bones were used for collagen extraction from all six human individuals. The regional faunal isotopic baseline was established with bone collagen of fish (n = 11) and terrestrial mammal (n = 12) remains selected from the dark soil overlying the shell midden of PNQ. Faunal samples were from a range of different skeletal elements and only a few terrestrial species could be taxonomically identified, including agouti (Dasyprocta punctata), brocket deer (Mazama gouazoubira), lowland paca (Cuniculus paca), and cavy (Kerodon rupestris). These are predominantly herbivorous animals, feeding on leaves, fruits, tubers, flowers, and bark. Only a few fish remains could be identified as catfish (Ariidae) (Supplementary information 1).
Modern plants (n = 11) were sampled and analyzed for their stable carbon and nitrogen isotope compositions to complement the faunal isotopic baseline. Major neotropical crops including manioc (Manihot esculenta, with the local variants macaxeira and mandioca) and sweet potato (Ipomoea batatas) (all C3 plants), and maize (Zea maize, a C4 plant) were acquired from a local small scale market by A. Bandeira at São Luís and Baixada Maranhense in 2018. Before sending the samples for isotope analysis at the University of York, the samples were registered in the Sistema Nacional de Gestão do Patrimônio Genético e do Conhecimento Tradicional Associado (SisGen, protocol no. A7BC812) according to article 22 of Decreto nº 8.772, of 11 May 2016. These crops are known to have been cultivated by pre-Columbian groups in lowland Amazonia60 and may have constituted a substantial source of dietary energy to populations on São Luís Island (Supplementary information 1).
Collagen was extracted at the BioArCh facilities of the University of York (UK) following the procedure reported in61 using a modified Longin method62. Bones were cleaned manually and sherds (200–600 mg) demineralized in a 0.6 M HCl solution at 4ºC for 12—72 h. Samples were then rinsed with deionized water (milli-Q®) and immersed in 0.001 M HCl at 80 °C for 48 h. For most samples the supernatant containing the collagen was filtered using Polyethylene Ezee filters (Elkay Laboratories Ltd., 9 mL, pore size 60–90 μm). The samples were then filtered using 30 kDa Amicon® Ultra-4 centrifugal filter units (Millipore, MA, USA), frozen for 24–48 h at − 20ºC, lyophilized, and weighed into tin capsules (1 mg) for stable isotopic analysis.
Plant samples, including tubers of manioc and macaxeira (n = 5) and sweet potato (n = 1), and maize kernels (n = 5), were rinsed in deionized water (milli-Q®), dried and frozen for several hours at − 20 ºC. Samples were lyophilized, homogenized using an agate pestle and mortar, and weighed into tin capsules (2 mg) for bulk stable carbon and nitrogen isotopic analysis.
The collagen samples of four human individuals (PNQ1A, PNQ1B, PDL2, BC1) were analyzed in duplicate by EA-IRMS on a Thermo Finnigan Delta Plus XL in the Department of Archaeological Sciences of the University of Bradford (UK). The collagen sample of PNQ3 and the faunal remains, along with the bulk plant samples, were analyzed in duplicate on a Sercon GSL elemental analyser coupled to a 20–22 mass spectrometer (Sercon, Crewe, UK) at the University of York (UK). At both York and Bradford, accuracy was determined by measurements of international standard reference materials within each analytical run. The obtained values were corrected from the isotopic ratio of the international standards, Vienna Pee Dee Belemnite (VPDB) for carbon and air (AIR) for nitrogen, according to the following equation: δ13C, δ15N = (Rsample/Rstandard)-163, where R = 13C/12C and 15N/14 N. At York the overall uncertainties on the measurements of each sample were calculated based on the method of Kragten64 by combining uncertainties in the values of the international reference materials and those determined from repeated measurements of samples and reference materials (these are expressed as one standard deviation). Caffeine (IAEA-600), ammonium sulphate (IAEA-N-2), and cane sugar (IA-Cane) international standards were used as reference material in each analytical run. In addition, a homogenised bovine bone was extracted and analysed within the same batch. The in-house collagen standard (bovine control) was also exchanged between laboratories (University of York and University of Bradford) to ensure accuracy.
Collagen was successfully extracted from five human individuals and 23 faunal samples (Table 2 and Supplementary information 1). The extracted collagen had atomic characteristics (C:N ratios, wt%C and wt%N) typical of well-preserved collagen65,66,67. One fish sample had wt%N and wt%C values outside of the acceptable range proposed by Szpak68 but had an acceptable C:N ratio and isotope values comparable to the other fish remains, thus it was considered reliable.
Statistical analysis and Bayesian stable isotope mixing models
Comparisons of δ13C and δ15N values between samples were performed using one-way ANOVA or Kruskal–Wallis tests (α = 0.05), after checking for normal distribution with Shapiro–Wilk test for normality (α = 0.05); both tests were performed in PAST 3.x69. The relative contributions of different food groups to human diet were estimated using Bayesian Stable Isotope Mixing Models (BSIMMs) in FRUITS 3.145 based on dietary proxies (bone collagen δ13C and δ15N values) and food groups (terrestrial mammals, fish and plants) to account for multiple dietary sources, different macronutrient fractions, metabolic routing and uncertainties for all of these parameters. For each food group the average δ13C and δ15N values of the nutrient fraction (protein, carbohydrates, lipids) were estimated using offsets between edible fractions and food remains reported in the literature48,70. In the case of individuals from São Luís Island, the δ13C and δ15N values of the dietary macronutrients (protein and energy) were estimated from the average δ13C and δ15N values of terrestrial mammals (n = 12) and fish (n = 11) found at PNQ, using the following offsets—terrestrial mammals: − 2 ‰ (∆13Cprotein-collagen), − 8 ‰ (∆13Clipids-collagen) and + 2 ‰ (∆15Nprotein-collagen); fish: − 1 ‰ (∆13Cprotein-collagen), − 7 ‰ (∆13Clipids-collagen) and + 2 ‰ (∆15Nprotein-collagen). For plants, bulk δ13C values were first corrected for the Suess effect. A linear regression using δ13C values of atmospheric CO2 between 1962 and 200871 was used to estimate the atmospheric δ13C value for 2018 (− 8.6 ‰), when the plants were collected. The difference between 2018 and the value for 1861 (− 6.48 ‰) was then used to correct the bulk plant δ13C values (+ 2.13 ‰). Plant δ13C values were then corrected for the offsets of − 2 ‰ (∆13Cbulk-protein) and + 0.5 ‰ (∆13Cbulk-carbohydrate), while assuming that the δ15N value of plant protein was the same as the average bulk plant δ15N value. A conservative uncertainty of 1 ‰ was used for most of the δ13C offsets, except for the average δ13C values of fish protein and energy (1.5 ‰). Large uncertainties were used for protein δ15N values of terrestrial C3 plants (6 ‰), terrestrial mammals (2 ‰), maize (2 ‰) and fish (2 ‰) to take into account the isotope variation observed between some of these samples (Supplementary information 1).
Bayesian estimates were also obtained for previously published collagen δ15N and δ13C values of 24 human individuals from the Maracá region (Gruta das Caretas: GdC, and Gruta do Pocinho: GdP)15 and Marajó Island (MrJ: unknown site, Teso dos Bichos, Monte Carmelo and Matinadas)12, at the mouth of the Amazon River. The model used for these sites differed from São Luís Island in regards to the faunal isotope baseline. Due to the lack of contextual isotopic baselines, their dietary macronutrients were estimated on the δ13C and δ15N values of modern terrestrial mammals (n = 8) and freshwater resources (fish and reptiles; n = 27) sampled from Guiana, Lower Orinoco and Upper Amazon15,72,73, and from the modern plants collected at São Luís Island (Supplementary information 2). The δ13C values of modern fauna were first corrected for the Suess effect using the procedure reported above for their year of capture, and then both δ13C and δ15N values were corrected for isotopic fractionation between tissue (muscle and bone collagen) and macronutrients using the aforementioned offsets (Supplementary information 1).
A concentration-dependent and routed model was selected for the Bayesian estimations49, where lipids and carbohydrates were aggregated as energy in the model input. The nitrogen isotopes were assumed to be sourced only from proteins (100%), but the carbon isotopes could have derived from carbohydrates and lipids with de novo synthesis of non-essential amino acids28. We assumed that dietary proteins contributed to 74 ± 4% of bulk collagen carbon, while lipids and carbohydrates provided the remaining 26%74. Diet-to-collagen δ13C offset (+ 4.8 ± 2 ‰) and δ15N offset (+ 5.5 ± 2 ‰) were taken from Fernandes et al.74. Dietary estimations were constrained for a conservative acceptable range for protein intake of > 5% and < 45% of the total calories according to physiological studies45. The model inputs and parameters can be found in the Supplementary information 2.
Box and whiskers plots summarizing the posteriors of dietary estimates generated using FRUITS were created using the R statistical software environment (version 4.0.2) and the package ggplot2 (version 3.3.2)75, with McGill et al.76 variations of Box Plots. For these, the horizontal lines correspond to the median and the hinges to the 25th (Q1) and 75th (Q3) percentiles. The whiskers extend from the hinge to the smallest or largest observation greater than or equal to − 1.5 * IQR (interquartile range) or less than or equal to + 1.5 * IQR, respectively; outliers were excluded. Summary statistics of the dataset were performed using dplyr version 1.0.1 and reported in the Supplementary information 3.
Finally, we assumed that maize, a plant using a C4 photosynthetic pathway, was consumed by groups at São Luís Island and at the mouth of the Amazon River during the Late Holocene. This assumption is reasonably supported by the presence of maize in the Amazon basin since the Middle Holocene50,77, and by increasing evidence of maize in archaeological and lake sedimentary records in the central and northern Amazon during the Late Holocene10,78.
The radiocarbon dates from São Luís Island, obtained directly from human individuals using Accelerator Mass Spectrometry (AMS), were calibrated (BP) using a combination of the marine (Marine1379) and terrestrial (SHCal1380) curves in OxCal v4.381, taking into account the average relative contribution of marine carbon to collagen estimated by the BSIMMs. We estimated the local marine radiocarbon reservoir correction value (∆R) at São Luís Island using data from the Marine Reservoir Correction Database82. The ∆R point data from the Marine Reservoir Correction Database was used to generate a smooth surface distribution of marine ∆R values using the Bayesian model AverageR developed within the Pandora & IsoMemo initiatives (https://isomemoapp.com). AverageR is a generalized additive mixed model that employs a thin plate regression spline. Further details on the model can be found in Cubas et al.83. The local ∆R (10 ± 41 14C years) was estimated for a point centred at São Luís Island and a radius of 50 km. Calibrated dates were rounded to 10.
Data availability
All the data presented and discussed in this paper is provided in the tables and supplementary information.
References
Clement, C. R. et al. The domestication of Amazonia before European conquest. Proc. Biol. Sci. 282, 20150813 (2015).
Neves, E. G. Was agriculture a key productive activity in pre-colonial Amazonia? The stable productive basis for social equality in the Central Amazon. In Human-Environment Interactions: Current and Future Directions (eds. Brondízio, E. S. & Moran, E. F.) 371–388 (Springer, Netherlands, 2013).
McMichael, C. H. et al. Predicting pre-Columbian anthropogenic soils in Amazonia. Proc. Biol. Sci. 281, 20132475 (2014).
Levis, C. et al. Persistent effects of pre-Columbian plant domestication on Amazonian forest composition. Science 355, 925–931 (2017).
McKey, D. et al. Pre-Columbian agricultural landscapes, ecosystem engineers, and self-organized patchiness in Amazonia. Proc. Natl. Acad. Sci. USA 107, 7823–7828 (2010).
Watling, J. et al. Impact of pre-Columbian ‘geoglyph’ builders on Amazonian forests. Proc. Natl. Acad. Sci. 114, 1868–1873 (2017).
Blatrix, R. et al. The unique functioning of a pre-Columbian Amazonian floodplain fishery. Sci. Rep. 8, 5998 (2018).
Heckenberger, M. & Neves, E. G. Amazonian archaeology. Annu. Rev. Anthropol. 38, 251–266 (2009).
de Souza, J. G. et al. Climate change and cultural resilience in late pre-Columbian Amazonia. Nat. Ecol. Evol. 3, 1007–1017 (2019).
Iriarte, J. et al. Fire-free land use in pre-1492 Amazonian savannas. Proc. Natl. Acad. Sci. USA 109, 6473–6478 (2012).
Schaan, D. P. The nonagricultural Chiefdoms of Marajó Island. In The Handbook of South American Archaeology (eds. Silverman, H. & Isbell, W. H.) 339–357 (Springer, New York, 2008).
Roosevelt, A. C. Moundbuilders of the Amazon: Geophysical Archaeology on Marajo Island, Brazil (Academic Press, San Diego, 1991).
Pugliese, F. A., Augusto Zimpel Neto, C. & Neves, E. G. What do Amazonian shellmounds tell us about the long-term indigenous history of South America? In Encyclopedia of Global Archaeology 1–25 (Springer, Berlin 2018).
van der Merwe, N. J., Roosevelt, A. C. & Vogel, J. C. Isotopic evidence for prehistoric subsistence change at Parmana Venezuela. Nature 292, 536 (1981).
Hermenegildo, T., O’Connell, T. C., Guapindaia, V. L. C. & Neves, E. G. New evidence for subsistence strategies of late pre-colonial societies of the mouth of the Amazon based on carbon and nitrogen isotopic data. Quat. Int. 448, 139–149 (2017).
Roosevelt, A. Resource management in Amazonia before the conquest: Beyond ethnographic projection. Adv. Econ. Bot. 7, 30–62 (1989).
Roosevelt, A. C. The Lower Amazon: A Dynamic Human Habitat. In Imperfect Balance: Landscape Transformations in the Precolumbian Americas (ed. Lentz, D. L.) 455–491 (2000).
Prestes-Carneiro, G., Béarez, P., Bailon, S., Rapp Py-Daniel, A. & Neves, E. G. Subsistence fishery at Hatahara (750–1230 CE), a pre-Columbian central Amazonian village. J. Archaeol. Sci.: Rep.8, 454–462 (2016).
Erickson, C. L. An artificial landscape-scale fishery in the Bolivian Amazon. Nature 408, 190–193 (2000).
Schaan, D. P. The Camutins chiefdom: rise and development of social complexity on Marajó Island, Brazilian Amazon. (University of Pittsburgh, 2004).
Garson, A. G. Comment upon the economic potential of fish utilization in riverine environments and potential archaeological biases. Am. Antiq. 45, 562–567 (1980).
Prestes-Carneiro, G., Béarez, P., Shock, M. P., Prümers, H. & Jaimes Betancourt, C. Pre-hispanic fishing practices in interfluvial Amazonia: Zooarchaeological evidence from managed landscapes on the Llanos de Mojos savanna. PLoS ONE 14, e0214638 (2019).
Gross, D. R. Protein capture and cultural development in the amazon basin. Am. Anthropol. 77, 526–549 (1975).
Lathrap, D. W. The ‘Hunting’ Economics of the Tropical Forest Zone of South America: An Attempt at Historical Perspective (1968).
Beckerman, S. The abundance of protein in Amazonia: a reply to gross. Am. Anthropol. 81, 533–560 (1979).
Gragson, T. L. Fishing the waters of Amazonia: native subsistence economies in a tropical rain forest. Am. Anthropol. 94, 428–440 (1992).
Ambrose, S. H. & Norr, L. Experimental evidence for the relationship of the carbon isotope ratios of whole diet and dietary protein to those of bone collagen and carbonate, In Prehistoric Human Bone - Archaeology at the Molecular Level 1–37 (Springer, Berlin 1993).
Jim, S., Jones, V., Ambrose, S. H. & Evershed, R. P. Quantifying dietary macronutrient sources of carbon for bone collagen biosynthesis using natural abundance stable carbon isotope analysis. Br. J. Nutr. 95, 1055–1062 (2006).
Webb, E. C. et al. The influence of varying proportions of terrestrial and marine dietary protein on the stable carbon-isotope compositions of pig tissues from a controlled feeding experiment. STAR: Sci. Technol. Archaeol. Res.3, 36–52 (2017).
Colonese, A. C. et al. Long-term resilience of late holocene coastal subsistence system in Southeastern South america. PLoS ONE 9, e93854 (2014).
Pezo-Lanfranco, L. et al. Middle Holocene plant cultivation on the Atlantic Forest coast of Brazil?. R. Soc. Open Sci. 5, 180432 (2018).
Tafuri, M. A. et al. Dietary resilience among hunter-gatherers of Tierra del Fuego: isotopic evidence in a diachronic perspective. PLoS ONE 12, e0175594 (2017).
O’Leary, M. H. Carbon isotopes in photosynthesis. Bioscience 38, 328–336 (1988).
Martinelli, L. A. et al. The use of carbon and nitrogen stable isotopes to track effects of land‐use changes in the Brazilian Amazon Region. In Terrestrial Ecology vol. 1, 301–318 (Elsevier, 2007).
Silva, M. H. L. et al. Fish assemblage structure in a port region of the Amazonic coast. Iheringia, Sér. Zool.108, (2018).
Schoeninger, M. J. & Deniro, M. J. Carbon isotope ratios of bone apatite and animal diet reconstruction (reply). Nature 301, 178 (1983).
Garcia, A. M., Hoeinghaus, D. J., Vieira, J. P. & Winemiller, K. O. Isotopic variation of fishes in freshwater and estuarine zones of a large subtropical coastal lagoon. Estuar. Coast. Shelf Sci. 73, 399–408 (2007).
Giarrizzo, T., Schwamborn, R. & Saint-Paul, U. Utilization of carbon sources in a northern Brazilian mangrove ecosystem. Estuar. Coast. Shelf Sci. 95, 447–457 (2011).
Oliveira, A. C. B., Soares, M. G. M., Antonio. Martinelli, L. & Zacarias. Moreira, M. Carbon sources of fish in an Amazonian floodplain lake. Aquat. Sci.68, 229–238 (2006).
Szpak, P. Complexities of nitrogen isotope biogeochemistry in plant-soil systems: implications for the study of ancient agricultural and animal management practices. Front. Plant Sci. 5, 288 (2014).
Fuller, B. T., Fuller, J. L., Harris, D. A. & Hedges, R. E. M. Detection of breastfeeding and weaning in modern human infants with carbon and nitrogen stable isotope ratios. Am. J. Phys. Anthropol. 129, 279–293 (2006).
Medina, E., Martinelli, L. A., Barbosa, E. & Victoria, R. L. Natural abundance of 13C in tropical grasses from the INPA, Instituto Nacional de Pesquisas da Amazônia, herbarium. Braz. J. Bot. 22, 44–51 (1999).
Schaan, D. Long-term human induced impacts on Marajó Island landscapes Amazon Estuary. Diversity 2, 182–206 (2010).
Ometto, J. P. H. B. et al. The stable carbon and nitrogen isotopic composition of vegetation in tropical forests of the Amazon Basin, Brazil. In Nitrogen Cycling in the Americas: Natural and Anthropogenic Influences and Controls (eds. Martinelli, L. A. & Howarth, R. W.) 251–274 (Springer, The Netherlands, 2006).
Fernandes, R., Millard, A. R., Brabec, M., Nadeau, M.-J. & Grootes, P. Food reconstruction using isotopic transferred signals (FRUITS): a Bayesian model for diet reconstruction. PLoS ONE 9, e87436 (2014).
Parnell, A. C. et al. Bayesian stable isotope mixing models. Environmetrics10, (2013).
Phillips, D. L. et al. Best practices for use of stable isotope mixing models in food-web studies. Can. J. Zool. 92, 823–835 (2014).
Fernandes, R. A simple(R) model to predict the source of dietary carbon in individual consumers. Archaeometry 58, 500–512 (2016).
Fausto, C. & Neves, E. G. Was there ever a Neolithic in the Neotropics? Plant familiarisation and biodiversity in the Amazon. Antiquity 92, 1604–1618 (2018).
Kistler, L. et al. Multiproxy evidence highlights a complex evolutionary legacy of maize in South America. Science 362, 1309–1313 (2018).
Casey, M. M. & Post, David M. The problem of isotopic baseline: reconstructing the diet and trophic position of fossil animals. Earth-Sci. Rev.106, 131–148 (2011).
Rêgo, J. C. L., Soares-Gomes, A. & da Silva, F. S. Loss of vegetation cover in a tropical island of the Amazon coastal zone (Maranhão Island, Brazil). Land Use Policy 71, 593–601 (2018).
de Menezes, M. P. M., Berger, U. & Mehlig, U. Mangrove vegetation in Amazonia: a review of studies from the coast of Pará and Maranhão States, north Brazil. Acta Amazon. 38, 403–420 (2008).
Bandeira, A. M., Chahud, A., Ferreira, I. C. P. & Pacheco, M. L. A. F. Mobilidade, subsistência e apropriação do ambiente: contribuições da zooarqueologia sobre o sambaqui do Bacanga, São Luís, Maranhão. Boletim do Museu Paraense Emílio Goeldi. Ciências Humanas11, 467–480 (2016).
Bandeira, A. M. Os sambaquis na Ilha de São Luís – MA: processo de formação, cultura material cerâmica e cronologia. Rev. Memorare 5, 315–360 (2018).
Bandeira, A. M. Os Tupis na Ilha de São Luís-Maranhão: Fontes Históricas e a Pesquisa Arqueológica. História Unicap 2, 79–98 (2015).
D’Abbeville, C. História da missão dos padres capuchinhos na Ilha do Maranhão e circunvizinhanças (São Paulo, Siciliano, 2002).
de D’Évreux, Y. Viagem ao norte do Brasil feita nos anos de 1613 a 1614 (São Paulo, Siciliano, 2002).
Cantanhêde, L. G. et al. Environmental perception of fishermen: use and conservation of fisheries resources. Biota Neotrop.18, (2018).
Maezumi, S. Y. et al. New insights from pre-columbian land use and fire management in Amazonian dark earth forests. Front. Ecol. Evol. 6, 111 (2018).
Craig, O. E. et al. Stable isotope analysis of Late Upper Palaeolithic human and faunal remains from Grotta del Romito (Cosenza) Italy. J. Archaeol. Sci. 37, 2504–2512 (2010).
Brown, T. A., Nelson, E. E., Vogel, S. J. & Southon, J. R. Improved collagen extraction by modified longin method. Radiocarbon 30, 171–177 (1988).
Coplen, T. B. Guidelines and recommended terms for expression of stable-isotope-ratio and gas-ratio measurement results. Rapid Commun. Mass Spectrom. 25, 2538–2560 (2011).
Kragten, J. Tutorial review. Calculating standard deviations and confidence intervals with a universally applicable spreadsheet technique. Analyst119, 2161–2165 (1994).
DeNiro, M. J. Postmortem preservation and alteration of in vivo bone collagen isotope ratios in relation to palaeodietary reconstruction. Nature 317, 806–809 (1985).
Van Klinken, G. J. Bone collagen quality indicators for palaeodietary and radiocarbon measurements. J. Archaeol. Sci. 26, 687–695 (1999).
Ambrose, S. H. Preparation and characterization of bone and tooth collagen for isotopic analysis. J. Archaeol. Sci. 17, 431–451 (1990).
Szpak, P. Fish bone chemistry and ultrastructure: implications for taphonomy and stable isotope analysis. J. Archaeol. Sci. 38, 3358–3372 (2011).
Hammer, Ø, Harper, D. & Ryan, P. D. PAST: paleontological statistics software package for education and data analysis palaeontol. Electronica 4, 1–9 (2001).
Fernandes, R., Grootes, P., Nadeau, M.-J. & Nehlich, O. Quantitative diet reconstruction of a Neolithic population using a Bayesian mixing model (FRUITS): the case study of Ostorf (Germany). Am. J. Phys. Anthropol. https://doi.org/10.1002/ajpa.22788 (2015).
Hellevang, H. & Aagaard, P. Constraints on natural global atmospheric CO2 fluxes from 1860 to 2010 using a simplified explicit forward model. Sci. Rep. 5, 17352 (2015).
van der Merwe, N. J. & Medina, E. The canopy effect, carbon isotope ratios and foodwebs in amazonia. J. Archaeol. Sci. 18, 249–259 (1991).
Watson, L. C., Stewart, D. J. & Teece, M. A. Trophic ecology of Arapaima in Guyana: giant omnivores in Neotropical floodplains. Neotrop. Ichthyol. 11, 341–349 (2013).
Fernandes, R., Nadeau, M.-J. & Grootes, P. M. Macronutrient-based model for dietary carbon routing in bone collagen and bioapatite. Archaeol. Anthropol. Sci. 4, 291–301 (2012).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer, New York, 2016).
Mcgill, R., Tukey, J. W. & Larsen, W. A. Variations of box plots. Am. Stat. 32, 12–16 (1978).
Maezumi, S. Y. et al. The legacy of 4500 years of polyculture agroforestry in the eastern Amazon. Nat. Plants 4, 540–547 (2018).
Rostain, S. Where the Amazon River meets the Orinoco River: Archaeology of the Guianas. Amazônica – Rev. de Antropol. 4, 10–28 (2012).
Reimer, P. J. et al. IntCal13 and marine13 radiocarbon age calibration curves 0–50,000 years cal BP. Radiocarbon 55, 1869–1887 (2013).
Hogg, A. G. et al. SHCal13 southern hemisphere calibration, 0–50,000 years cal BP. Radiocarbon 55, 1889–1903 (2013).
Ramsey, C. B. Bayesian analysis of radiocarbon dates. Radiocarbon 51, 337–360 (2009).
Reimer, P. J. & Reimer, R. W. A marine reservoir correction database and on-line interface. Radiocarbon 43, 461–463 (2001).
Cubas, M. et al. Latitudinal gradient in dairy production with the introduction of farming in Atlantic Europe. Nat. Commun. 11, 2036 (2020).
Acknowledgements
The authors are grateful to Jose Iriarte for his helpful discussion and information about early food production in the lowland Amazon. The authors are grateful to Mírian Forancelli Pacheco for identifying some of the faunal remains. All necessary permits were obtained for the described study, which complied with all relevant regulations of the Instituto do Patrimônio Histórico e Artístico Nacional (IPHAN, Protocol No. 01506.00407/2012-14) and Sistema Nacional de Gestão do Patrimônio Genético e do Conhecimento Tradicional Associado for modern plants (SisGen, Protocol No. A7BC812). Archaeological materials used in this study are stored at the Universitat Autònoma de Barcelona (ICTA). This research was partially funded by the EU Marie-Curie Action: “Intra-European fellowships for career development” (Coastal resources and South American hunter-gatherers: biochemical perspectives from Brazilian sambaquis—COREBRAS, Ref. 273734). This work was partially funded by the ERC Consolidator project TRADITION, which has received funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme under grant agreement No 817911.
Author information
Authors and Affiliations
Contributions
A.C.C., R.W., A.B., R.F., S.S., K.M., designed and performed the research; A.B., R.B., T.F., contributed contextual information to aid interpretation; A.C.C., R.W., R.F., S.S., M.V., K.M., analyzed data; A.C.C., R.W., A.B., R.F., wrote the paper; all authors were involved in reviewing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Colonese, A.C., Winter, R., Brandi, R. et al. Stable isotope evidence for dietary diversification in the pre-Columbian Amazon. Sci Rep 10, 16560 (2020). https://doi.org/10.1038/s41598-020-73540-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-73540-z
This article is cited by
-
South American Archaeological Isotopic Database, a regional-scale multi-isotope data compendium for research
Scientific Data (2024)
-
The role of farming and fishing in the rise of social complexity in the Central Andes: a stable isotope perspective
Scientific Reports (2024)
-
Chemical analysis of pottery reveals the transition from a maritime to a plant-based economy in pre-colonial coastal Brazil
Scientific Reports (2023)
-
Fishing intensification as response to Late Holocene socio-ecological instability in southeastern South America
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.