Abstract
The aim of the study was to check if the subtendons of the Achilles tendon can be identified in vivo on MRI in the midportion of the tendon. The relation of the plantaris tendon to the Achilles tendon was also examined. A retrospective study of 200 MRI of ankle joints including the Achilles tendon was conducted. Statistical analysis of the correlation between the possibility of identifying the subtendons and the side, gender, presence of the central soleus tendon and plantaris tendon variation was performed. The inter-observer agreement between two reviewers in their evaluation of the subtendons was assessed using kappa statistics. The subtendon from the lateral head of the gastrocnemius muscle was identified in 65% (k = 0.63) and was located in the anterior part of the Achilles tendon. The subtendon from the soleus muscle was recognized in 12% (k = 0.75) comprising anterior part of the tendon. In 6% the subtendon from the medial head of the gastrocnemius muscle was identified (k = 0.58). The central soleus tendon was identified in 85% of cases. Statistical analysis shows the weak correlation of the presence of the central soleus tendon and the possibility of identifying the subtendon from the soleus muscle. The plantaris tendon was directly related to the insertion of the Achilles tendon in 42.5%. Identification of the subtendons of the Achilles tendon on MRI is challenging, and most often it is only possible to find the subtendon of the lateral head of the gastrocnemius muscle.
Similar content being viewed by others
Introduction
The Achilles tendon is composed of twisted subtendons corresponding to each part of the triceps surae muscle. The internal structure of the muscle and tendon has been studied intensively over the last few years because the incidence of injuries has increased almost three times over the past 30 years (18 vs 55 per 100,000)1,2,3. The fascicular structure of the Achilles tendon is well known and documented on cadavers4, however, there is no corresponding radiological work on large material in magnetic resonance imaging (MRI). The Achilles tendon subtendons initially run vertically, corresponding to the position of the muscle bellies. In the midportion of the tendon, a clear rotation is observed so that subtendon from the lateral head of the gastrocnemius muscle (S-LGC) is found in the anterior part while subtendon from the medial head (S-MGC) in the posterior part of the tendon4,5. The subtendon from the soleus muscle (S-Sol), which is a prolongation of the soleus central tendon (T-Sol)6,7,8,9 is located in the medial and anterior part of the tendon. Subtendons are separated by thin membranes which makes it possible to separate them during dissection10.
The midportion of the Achilles tendon is located about 2–7 cm above the calcaneal tuber and is a typical site for tendinopathy and injury. In most cases, no diagnostic imaging is needed because the clinical examination is sufficient11. However, diagnostic imaging supports the diagnosis of Achilles tendinopathy, which is the most common cause of pain of the Achilles tendon in athletes and the general population8,12. MRI is a very sensitive method for detecting pathological changes in the Achilles tendon because pathological changes can easily be identified in the tendon showing a low signal on T2-weighted and proton density sequences10,13. The use of ultra-short pulse sequences techniques allows an increase in the sensitivity of the detecting pathologies in the enthesis organ14. Ultrasound is a routinely used imaging method of the pathology of the Achilles tendon8, but due to the fact that our work applies to the use of MRI, we will not discuss ultrasound more broadly.
The aim of the study was to evaluate the possibility of identifying subtendons in the midportion of the Achilles tendon in vivo on MRI, assessing the rotation of the subtendons and the relation of the plantaris tendon to the Achilles tendon.
Results
Identification of the subtendons
Identification of the separation between the subtendons of the Achilles tendon was possible in n = 141 cases (70.5%) and occurred on the anterior part of the tendon (Table 1). Identification of the subtendons was possible due to the presence of thin high-signal planes separating the subtendons, which usually occur around the S-LGC (Figs. 1, 2, 4). The highest agreement between reviewers was in the case of the S-Sol (Table 2) while the lowest was seen in identification of the S-MGC. Final determination of the subtendons was made by consensus (Table 1).
MRI right ankle, PD FS at the level of the talus. (a)—The highest section, (d)—the lowest section. A 42-year-old male with ankle sprain without trauma. The arrow marks a narrow septation between the subtendon from the lateral head of the gastrocnemius muscle and the other subtendons of the Achilles tendon.
The subtendons from the S-LGC was clearly identified in n = 130 cases (Figs. 1, 2, 3). The S-Sol was recognized in n = 24 cases, while the S-MGC in n = 12 cases (Fig. 4). Rarely could only the S-Sol (n = 5) or S-MGC (n = 1) be identified.
MRI right ankle, PD FS at the level of the talus (a–c), (a)—the highest section, (d)—the lowest section and at the level of the deep calcaneal bursa just above the calcaneus (d). A 17-year-old female 1 week after ankle sprain, pain posteriorly to the lateral malleolus. 1—S-Sol, 2—S-LGC, 3—the plantaris tendon (type 4).
Identification of the subtendons of the Achilles tendon was possible in the midportion of the tendon (Figs. 1, 2, 3, 4, 5, 6, 7, 8) whereas septation between the subtendons was not visible in the insertion in any of the cases (Figs. 7D, 8F).
The anterior outline of the Achilles tendon is always comprised by two subtendons, one from the lateral head of the gastrocnemius and the other from the soleus muscle, with dominance of the first one in n = 114 cases (57%) (Figs. 2, 3, 4, 5, 7, 8). Less often, the anterior outline is comprised mainly by the subtendon originating from the soleus muscle (Fig. 6). The posterior outline of the Achilles tendon is composed of the S-MGC (Figs. 6, 8).
The posterior outline of the Achilles tendon is composed by the S-MGC, the medial outline by the S-Sol while the anterior by the S-LGC and S-Sol. The lateral outline of the Achilles tendon is formed by the S-LGC and S-MGC.
The presence of the T-Sol was found in n = 170 cases (85%) (Figs. 9, 10), however, only a weak statistically significant correlation between the presence of the T-Sol (Fig. 9) and the possibility of identifying the S-Sol within the Achilles tendon was demonstrated (r = 0.15, P > 0.05). No statistically significant correlation was found between the side or gender of the patient and the possibility of identifying the subtendons within the Achilles tendon.
MRI right Achilles tendon, PD FS. The words medial and lateral are given for better orientation. (A,B) coronal picture of the right Achilles tendon. (C,D) transverse section at level shown on (B). 1—fibers from the soleus muscle, 2—fibers from the lateral head of the gastrocnemius muscle, 3—fibers from the medial head of the gastrocnemius muscle, 4—the plantaris tendon.
Connection of the plantaris tendon to the Achilles tendon
We identified five variations of the plantaris tendon (Table 1, Figs. 4, 5, 6, 7, 8, 11). The direct connection of the plantaris tendon to the Achilles tendon was seen in type 2 and 4, n = 89 cases (42.5%). Weak statistically significant positive correlation is visible between the possibility of visualizing the subtendon from the lateral head of the gastrocnemius muscle and types 3 and 4 of the plantaris tendon (Table 3). In contrast, a negative weak correlation occurs between type 5 or the absence of the plantar tendon.
The final consensus of the possibility of identification of the subtendons of the Achilles tendon, the central tendon of the soleus muscle and variation of the plantaris is presented in Table 1.
Rotation of the subtendons in the Achilles tendon
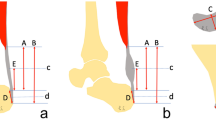
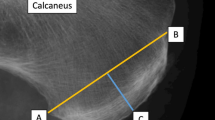
The angles between the long axis of the tibia and long axes of each subtendon were measured on axial and coronal sections (Figs. 1, 2, 3, 4, 5, 6, 7, 8, Table 4). The Achilles tendon was visible on 2 (n = 49 cases) or 3 (n = 151) coronal slides. The mean angles of the long axis of the subtendons are given in Table 4. R Pearson values near to zero were noted in correlation with the subtendon’s angles in correlation to gender and side, consequently no explicit differences were noted (Table 4).
Discussion
The distribution of the subtendons of the midportion the Achilles tendon found in our study is the following the S-Sol is located antero-medially, the S-LGC anteriorly while the S-MGC posteriorly. The posterior outline of the Achilles tendon is formed by the S-MGC, the medial outline by the S-Sol, the anterior by the S-LGC and S-Sol. The lateral outline is formed by the S-LGC and S-MGC. The internal structure and position of the subtendons of the normal Achilles tendon has not yet been studied on MRI in larger material before. Most of the previous studies assessing the internal structure of the Achilles tenon were conducted on cadavers4,5,16,17,18,19,20 and very few in ultrasound8. We think that our study is necessary due to the fact that MRI is a modern method of imaging the musculoskeletal and an excellent tool for assessing the Achilles tendon anatomy4,5,21. We noticed that the possibility of identifying the subtendons on MRI is not as clear as during an anatomical dissection and classical anatomy of the Achilles tendon is significantly different from then radiological anatomy. Identification of the subtendons of the Achilles tendon on MRI depends on the presence of narrow high-signal septations between the subtendons21 and the ability to identify is partly dependent on the reviewer. No large-scale MRI studies such as this have been previously reported. We studied the Achilles tendon between the lower outline of the soleus muscle belly to the level of the calcaneal tuber, which includes the zone where tendinopathy and ruptures most often occur2,8,22. Active tracking of the subtendons from the myotendinous junction, distal to the calcaneus, showed the same twisted structure of the Achilles tendon as revealed previously5,10,17,18,20,23. Most often it was possible to determinate the subtendon originating from the lateral head of the gastrocnemius muscle. The revealed subtendon’s location described in previous studies4,17,20 was consistent with our MRI findings. The localization of the subtendons revealed in our study is the following: the subtendon from the lateral head of the gastrocnemius muscle is located in the anterior part of the Achilles tendon, the subtendon from the soleus muscle is located medially to this, while the posterior part of the Achilles tendon is comprised of the subtendon from the medial head of the gastrocnemius muscle. We found intermediate to good interobserver agreement values (k = 0.58–0.75). We do not think that different MRI sequences or other cross-section or section thicknesses would improve agreement but that would presumably improve the assessment of the angles.
In contrast to anatomical studies4,17,18,19,20 we were not able to distinguish the subtendons at the level of bone attachment because the tendon structure is quite compact and the position of the subtendons at the insertion can be only presumed on MRI16,17. The tight packing of the fascicles of the Achilles tendon at the insertion may explain why the subtendons could not be identified. The Achilles tendon shows symptoms of diseases affecting surrounding structures, e.g. retrocalcaneal bursitis or Haglund deformity. This area is also quite sensitive to partial injuries causing isolated laxity of the tendon22. The largely uniform structure of the Achilles tendon is only apparent because the nonuniform function of the Achilles tendon occurs during movement and exercise23 and internal tendon tears are longitudinal in direction24.
In our study, we revealed that the anterior outline of the Achilles tendon is formed laterally by the subtendon originating from the lateral head of the gastrocnemius muscle while the subtendon of the soleus muscle is located anteriorly and medially. This structure is similar to the results of previous anatomical examinations4,17,18,20. Usually, dominance of the subtendon from the lateral gastrocnemius muscle was observed which corresponds to type II according to Edama17,18. In no case was the subtendon from the medial head of the gastrocnemius muscle divided into parts as was previously suggested4. This difference may be due to the fact that the MRI examination showed poor fiber rotation within the subtendon, whereas the configuration of superficial fibers is clearly visible on gross anatomy specimens. This is probably also the reason for the inability to compare with the results of previous anatomical studies19,20,21. Several anatomical studies have shown different degrees of the Achilles tendon twist structure, but there are no studies assessing the tendon twist on MRI. Previous anatomical studies assessing the rotation of the tendon fibers give very divergent results from 5 to 90 degrees17,18,19,20. On MRI, it is not possible to distinguish between fibers in the subtendons, but this is not difficult during anatomical dissection. The appropriate method of angle measuring should consider the three-dimensional nature of the Achilles tendon19. The angles measured in our study were estimated based on the axial sections and two or three coronal sections, which is a projection of the twisted tendon surface to a flat view plane which is a limitation of this study. Our results also differ from most anatomical works because we measured the angle of the subtendon’s axis, not the angle of the superficial fibers or the fiber rotation as in most previous anatomical studies. On the other hand, counting only superficial fibers orientation may lead to misinterpretation of the tendon rotation. The results of our study are comparable only with the work published Prosenz et al.21, where the internal structure of the subtendons was examined. A solution could be the use of ultra-thin MRI sections of the Achilles tendon, which is not routinely used clinically.
In occasional anatomical studies, variability in the degree of tendon torsion was observed, separating the types of torsion variability19,21. In our material we did not find cases where only one subtendon formed the anterior outline of the Achilles tendon, which was revealed by some authors17. Only a variable size ratio between the subtendons originating from the soleus and the lateral head of the gastrocnemius muscles was observed.
Previous studies17 have not shown statistically significant differences in the twisted structure between the sexes and sites examined, however, comparison with our work on larger material and another method is significantly limited. In our study, statistical analysis showed virtually no correlation between torsion of tendon and gender or side.
We decided to assess the location of the plantaris tendon in relation to the Achilles tendon due to its potential role in the medial Achilles tendinopathy8,15. The connection of the plantar tendon with the Achilles tendon was seen relatively often (type 2 and 4) most often as a common insertion to the calcaneal tuber or as a contribution to the medial outline of the Achilles tendon. Considering the methodology of our study, we decided to use the plantaris tendon classification according to Olewnik et al.15, which is a good compromise between the classification according to Cummins and Anson25 and one according to Sterkenburg et al.26. The reason is that MRI has a lower sensitivity than dissection for the visualization of small tendon connections which is described in some rare variants15,26.
There was no correlation between the possibility of subtendon identification and the side, gender, presence of the central tendon of the soleus muscle or the anatomical variant of the plantaris tendon. According to our knowledge, no studies of similar relationships were carried out either on anatomical specimens or MRI.
The limitations of our study were: its retrospective character and no anatomical-surgical correlation.
Classical and radiological anatomy of the Achilles tendon are different. Identification of the individual subtendons in a healthy Achilles tendon on MRI is possible above the calcaneus, although it is challenging. Most often only the subtendon from the lateral head of the gastrocnemius muscle can be identified, which, together with the subtendon from the soleus muscle, forms the anterior outline of the Achilles tendon. The posterior outline of the Achilles tendon is formed by the subtendon from the medial head of the gastrocnemius muscle. In most cases, the plantaris tendon has a direct connection to the medial part of the Achilles tendon. The application of the results of our study allows a more accurate association of the subtendons with individual parts of the triceps surae muscle. This is very valuable knowledge in prevention the development of overuse, tendinopathy and Achilles tendon ruptures.
Methods
We conducted the retrospective study of 200 ankle MRI of athletes (112 males and 88 females; the average age 29 (range 16–47) years; 101 right ankles and 99 left ones) without history of trauma or obvious abnormality in relation to the Achilles tendon. Only patients without tendinopathy of the Achilles tendon and with complete clinical data were included.
MRI examinations were performed at our institution over six months (between November 2018 and April 2019), about 2–4 weeks after the ankle injury (mean duration 2.8 weeks) for clinical purposes. All examinations were performed using a 3.0-T MRI scanner. The systematic re-evaluation was conducted by two independent observers. We used Cohen’s Kappa coefficient to determine inter-observer agreement. A coefficient was calculated for each subtendon, the central soleus tendon and each type of soleus. The kappa coefficient ranges from − 1 to + 1, agreements may be qualified as: excellent (> 0.81), good (0.61–0.8), intermediate (0.21–0.60), or poor (< 0.21)27. Final determination of the subtendons was made by consensus.
Due to the known internal structure of the Achilles tendon4,20, T2-weighted or PD (proton density) images on the frontal and transverse planes were used. On the coronal plane, it was assessed whether the rotation of the subtendons could be identified, then the position of the soleus central tendon was identified.
The angles between the long axis of the tibia and long axes of each subtendon were measured. Active tracking enables the determination of the geometric center of each subtendon at the highest possible cross-section (called X1). Then each subtendon was tracked on subsequent lower axial sections for the S-MGC and S-LGC and on coronal sections for T-Sol. Afterwards, a point X1 of the subtendon was connected with its point X2 to define the axis of the subtendon.
On the transverse plane, we assessed whether the subtendons described in the literature could be distinguished on MRI and whether their location was consistent with the previously described Achilles tendon structure. At a later stage, the relationship of the plantaris tendon to the Achilles tendon was determined, using the plantaris tendon classification according Olewnik et al.15.
The methods were carried out in accordance with the relevant guidelines and regulations, and informed consent was obtained from all participants, and for participants under 18, from a parent and/or legal guardian.
Ethics approval
Ethical approval was waived by the Institutional Ethics Committee at Medical University of Warsaw, Poland in view of the retrospective nature of the study and that all the procedures being performed were part of routine care. The Institutional Ethics Committee at Medical University of Warsaw, Poland was informed about the ongoing retrospective study and the Committee stated there was no need for its approval (AKBE/258/2019).
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ganestam, A., Kallemose, T., Troelsen, A. & Barfod, K. W. Increasing incidence of acute Achilles tendon rupture and a noticeable decline in surgical treatment from 1994 to 2013. A nationwide registry study of 33,160 patients. Knee Surg. Sports Traumatol. Arthrosc. 24, 3730–3737. https://doi.org/10.1007/s00167-015-3544-5 (2016).
Karlsson, J., Westin, O., Carmont, M. & Nilsson-Helander, K. in Sports Injuries of the Foot and Ankle Ch. Chapter 33, 369–376 (2019).
Lantto, I., Heikkinen, J., Flinkkila, T., Ohtonen, P. & Leppilahti, J. Epidemiology of Achilles tendon ruptures: Increasing incidence over a 33-year period. Scand. J. Med. Sci. Sports 25, 133–138. https://doi.org/10.1111/sms.12253 (2015).
Szaro, P., Witkowski, G., Smigielski, R., Krajewski, P. & Ciszek, B. Fascicles of the adult human Achilles tendon—An anatomical study. Ann. Anat. 191, 586–593. https://doi.org/10.1016/j.aanat.2009.07.006 (2009).
Winnicki, K., Ochala-Klos, A., Rutowicz, B., Pekala, P. A. & Tomaszewski, K. A. Functional anatomy, histology and biomechanics of the human Achilles tendon—A comprehensive review. Ann. Anat. 229, 151461. https://doi.org/10.1016/j.aanat.2020.151461 (2020).
Balius, R. et al. The soleus muscle: MRI, anatomic and histologic findings in cadavers with clinical correlation of strain injury distribution. Skelet. Radiol. 42, 521–530. https://doi.org/10.1007/s00256-012-1513-3 (2013).
Bolsterlee, B. et al. Three-dimensional architecture of the whole human soleus muscle in vivo. PeerJ 6, e4610. https://doi.org/10.7717/peerj.4610 (2018).
Counsel, P., Comin, J., Davenport, M. & Connell, D. Pattern of Fascicular involvement in midportion Achilles tendinopathy at ultrasound. Sports Health 7, 424–428. https://doi.org/10.1177/1941738115595226 (2015).
Hatzantonis, C., Agur, A., Naraghi, A., Gautier, S. & McKee, N. Dissecting the accessory soleus muscle: A literature review, cadaveric study, and imaging study. Clin. Anat. 24, 903–910. https://doi.org/10.1002/ca.21188 (2011).
Pierre-Jerome, C., Moncayo, V. & Terk, M. R. MRI of the Achilles tendon: A comprehensive review of the anatomy, biomechanics, and imaging of overuse tendinopathies. Acta Radiol. 51, 438–454. https://doi.org/10.3109/02841851003627809 (2010).
Garras, D. N., Raikin, S. M., Bhat, S. B., Taweel, N. & Karanjia, H. MRI is unnecessary for diagnosing acute Achilles tendon ruptures: Clinical diagnostic criteria. Clin. Orthop. Relat. Res. 470, 2268–2273. https://doi.org/10.1007/s11999-012-2355-y (2012).
Couppe, C., Svensson, R. B., Josefsen, C. O., Kjeldgaard, E. & Magnusson, S. P. Ultrasound speckle tracking of Achilles tendon in individuals with unilateral tendinopathy: A pilot study. Eur. J. Appl. Physiol. 120, 579–589. https://doi.org/10.1007/s00421-020-04317-5 (2020).
Han, M., Larson, P. E., Liu, J. & Krug, R. Depiction of achilles tendon microstructure in vivo using high-resolution 3-dimensional ultrashort echo-time magnetic resonance imaging at 7 T. Investig. Radiol. 49, 339–345. https://doi.org/10.1097/RLI.0000000000000025 (2014).
Robson, M. D., Benjamin, M., Gishen, P. & Bydder, G. M. Magnetic resonance imaging of the Achilles tendon using ultrashort TE (UTE) pulse sequences. Clin. Radiol. 59, 727–735. https://doi.org/10.1016/j.crad.2003.11.021 (2004).
Olewnik, L., Wysiadecki, G., Polguj, M. & Topol, M. Anatomic study suggests that the morphology of the plantaris tendon may be related to Achilles tendonitis. Surg. Radiol. Anat. 39, 69–75. https://doi.org/10.1007/s00276-016-1682-1 (2017).
Ballal, M. S., Walker, C. R. & Molloy, A. P. The anatomical footprint of the Achilles tendon: A cadaveric study. Bone Joint J. 96, 1344–1348. https://doi.org/10.1302/0301-620X.96B10.33771 (2014).
Edama, M. et al. The twisted structure of the human Achilles tendon. Scand. J. Med. Sci. Sports 25, 497–503. https://doi.org/10.1111/sms.12342 (2015).
Edama, M. et al. Structure of the Achilles tendon at the insertion on the calcaneal tuberosity. J. Anat. 229, 610–614. https://doi.org/10.1111/joa.12514 (2016).
Pekala, P. A. et al. The twisted structure of the Achilles tendon unraveled: A detailed quantitative and qualitative anatomical investigation. Scand. J. Med. Sci. Sports 27, 1705–1715. https://doi.org/10.1111/sms.12835 (2017).
van Gils, C. C., Steed, R. H. & Page, J. C. Torsion of the human achilles tendon. J. Foot Ankle Surg. 35, 41–48. https://doi.org/10.1016/s1067-2516(96)80011-1 (1996).
Prosenz, J., Rath, C., Hadrovic-Avdic, M. & Hirtler, L. The twist of the achilles tendon—Associations of torsions in the lower extremity. Clin. Anat. 31, 1085–1091. https://doi.org/10.1002/ca.23247 (2018).
Mahan, J. et al. Achilles tendon complex: The anatomy of its insertional footprint on the calcaneus and clinical implications. J. Orthop. 17, 221–227. https://doi.org/10.1016/j.jor.2019.06.008 (2020).
Handsfield, G. G. et al. A 3D model of the Achilles tendon to determine the mechanisms underlying nonuniform tendon displacements. J. Biomech. 51, 17–25. https://doi.org/10.1016/j.jbiomech.2016.11.062 (2017).
Chan, O. et al. Intratendinous tears of the Achilles tendon—A new pathology? Analysis of a large 4-year cohort. Muscles Ligam. Tendons J. 7, 53–61. https://doi.org/10.11138/mltj/2017.7.1.053 (2017).
Cummins Ej Fau-Anson, B. J. et al. The structure of the calcaneal tendon (of Achilles) in relation to orthopedic surgery, with additional observations on the plantaris muscle. Surg. Gynecol. Obstet. 83, 107–116 (1946).
van Sterkenburg, M. N., Kerkhoffs, G. M., Kleipool, R. P. & van Dijk, C. N. The plantaris tendon and a potential role in mid-portion Achilles tendinopathy: An observational anatomical study. J. Anat. 218, 336–341. https://doi.org/10.1111/j.1469-7580.2011.01335.x (2011).
Landis, J. R. & Koch, G. G. The measurement of observer agreement for categorical data. Biometrics https://doi.org/10.2307/2529310 (1977).
Funding
Open Access funding provided by Gothenburg University Library.
Author information
Authors and Affiliations
Contributions
Protocol/project development: P.S. and B.C. Data collection and management: P.S., W.C.R., S.B. and A.B. Data analysis: M.P. and P.S. Manuscript writing/editing: P.S., S.B., M.P., B.C., W.C.R. and A.B.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Szaro, P., Cifuentes Ramirez, W., Borkmann, S. et al. Distribution of the subtendons in the midportion of the Achilles tendon revealed in vivo on MRI. Sci Rep 10, 16348 (2020). https://doi.org/10.1038/s41598-020-73345-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-73345-0
This article is cited by
-
The correlations between dimensions of the normal tendon and tendinopathy changed Achilles tendon in routine magnetic resonance imaging
Scientific Reports (2021)
-
Classification by degree of twisted structure of the fetal Achilles tendon
Surgical and Radiologic Anatomy (2021)
-
The anatomical variant of high soleus muscle may predispose to tendinopathy: a preliminary MR study
Surgical and Radiologic Anatomy (2021)
-
The twisted structure of the fetal calcaneal tendon is already visible in the second trimester
Surgical and Radiologic Anatomy (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.