Abstract
This paper reports a deep-water coral framework (a single colonial bush or a larger bioconstruction of coral covering the sea bottom), formed entirely by the scleractinian coral Eguchipsammia fistula (Alcock, 1902) (Dendrophylliidae), in the northern Red Sea waters of Saudi Arabia at a depth of about 640 m. The framework consists of mostly live corals with a total area of about 10 m2 and the length of the individual coral branches range from 12 to 30 cm. Although E. fistula is ubiquitous, this discovery is the second record of a framework formed by this species and the first discovery of a large living reef in the Red Sea. The results of the genetic study indicate the potential existence of a genetic variation of E. fistula in the Red Sea. This discovery implies that the Red Sea has favorable habitats for framework-forming DWC species and highlights the need for conducting more systematic surveys for understanding their distribution, abundance, and ecology.
Similar content being viewed by others
Introduction
Azooxanthellate deep-water corals (DWCs) are reported from the tropics to the polar seas, occurring on continental margins and topographical high points throughout much of the world’s oceans1,2. A majority (around 74%) of the DWCs is solitary in habit, while the remainder (around 26%) are colonial3. Despite its highly saline and warm deep waters, the Red Sea is known for its diverse benthic habitats. Shallow-water coral reefs span the entire coastline of the Red Sea and are known for their high productivity, biodiversity, and endemism4. The occurrence of azooxanthellate deep-water corals has been only seldom reported from the warm, saline, and oxygen- and food-deprived environment of the Red Sea5,6,7,8. Several frame-building corals, including azooxanthellate species, have been reported from the twilight zone (100–210 m) of the Gulf of Aqaba in the Red Sea9,10. Their range of distribution depth is in the transient zone for coral distribution, which includes the upper distribution limits of a few DWCs and the lower distribution limits of several shallower water species11.
Here, a live DWC framework that was observed at a depth of about 640 m (Latitude: 26° 24.124′ N; Longitude: 36° 04.139′ E) and at a distance of ~ 26 km from the nearest shoreline in the northern Red Sea (Fig. 1) is reported for the first time. This live DWC framework was observed during an expedition in June 2014 using the Research Vessel R. V. Aegaeo, belonging to the Hellenic Centre for Marine Research. The framework is exclusively formed by the DWC species Eguchipsammia fistula (Alcock, 190212) (Figs. 2 and 3), which was identified according to the morphological descriptions by Zibrowius13 and Cairns14, and further complemented by molecular data (See Supplementary information). The total area of the observed framework was around 10 m2 and the length of each coral branch ranged from 12 to 30 cm, mostly live with no or only a few instances of dead colonies along the periphery of the framework (Fig. 2). In July 2013, Tempera et al.15 reported a similar framework (reef) formed by Eguchipsammia (Dendrophylliidae) found off the Faial-Pico Channel (Azores, Northeast Atlantic) using the manned submersible Lula 1,000. The discovery reported here is the second record of such a living Eguchipsammia framework worldwide, suggesting that the Red Sea provides a habitat suitable for framework forming DWC species.
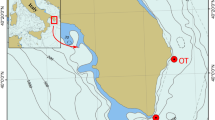
Location map of the Study Area (Basemap: Google) showing the tracks of ROV dives (orange lines) performed and the position of the observed coral framework (pink polygon), and stations (green points) used for water quality studies. Maps were created using ArcGIS 10.2 software by ESRI (www.arcgis.com). ArcGIS and ArcMap are the intellectual property of ESRI.

taken from different angles. (G) Specimen collection using the ROV. (H) Corallum of E. fistula photographed in the laboratory.
In situ photographs showing the habitat view of the Eguchipsammia fistula framework (distance between two red laser points is 10 cm). (A, B) Overall view of the coral framework recorded by the ROV. (C–F) In situ photographs of the framework
E. fistula is present throughout the Indo-West Pacific 16,17,18,19, and Marenzeller7 first discovered it in the Red Sea during the Pola expedition from 1895 to 1897. Recently, researchers have recorded scattered small colonies of this species again in the Saudi waters of the Red Sea5,6. Subsequent studies have revealed the ability of this coral species to cope with rapid and prolonged environmental changes, indicating a wide physiological plasticity, thereby providing reasons for its widespread distribution20,21. Climate change will alter the water temperature and chemistry of the oceans and negatively impact the shallow water and deep water coral reefs22. The combined effects of high temperatures and low oxygenation limit the proliferation of DWCs in the deep sea environment23. The Red Sea is in a stage of intense and abrupt warming, exhibiting an increase of 0.7 °C in the last decade24 with an increase of 3.45 °C projected over 2010–209925. Cantin et al.26 have reported declining growth and calcification rates of coral colonies in the Red Sea. Considering the potential impacts of climate change, it is necessary to estimate the vulnerability of E. fistula to temperature changes. Seawater temperature affects the distribution of azooxanthellate DWCs, with most of the species living within the temperature range of ∼4–14°C27,28. Unlike most DWCs, these species in the Red Sea live at considerably higher temperatures (> 20 °C). Corals in the family Dendrophylliidae, (Dendrophyllia cornigera, D. ramea, E. fistula etc.) apparently have a natural tolerance to warm conditions29. The average seawater temperature measured in the vicinity of the E. fistula coral framework in this study is 21.5 °C (Table 1) and it is comparable to the temperature recorded at the Red Sea stations where this species were recorded earlier5,6. E. fistula coral specimens demonstrate a wide physiological plasticity and adapt well to basic aquaria systems when transferred from the deep water habitat in the Red Sea into a rearing system20. Deep-sea corals such as E. fistula in the Red Sea are acclimatized to the warm, saline, and low oxygen environment, and compared to their counterparts from other parts of the globe, they are better suited for adapting to climate change.
Taxonomic classification of the specimen was confirmed by the phylogenetic analyses of partial sequences of the mitochondrial 16S ribosomal RNA encoding gene. The 16S sequences determined in this study match (96.51% identical) the 16S sequence of Eguchipsammia fistula deposited in GenBank (JX629250), determined in corals collected from the deep Red Sea5. However, there is a 3.49% (100–96.51%) divergence in the 16S fragments between samples used in this study and that in E. fistula specimens previously sampled in the Red Sea5, indicating a new haplotype of this species and the existence of a genetic variation within the Red Sea populations. An IGR sequence from Eguchipsammia fistula is not available in the GenBank, and the IGR sequence obtained in this study is considered as the first record on this species. The coral E. fistula has a cosmopolitan distribution, and is known to occur in the Indo-Pacific, Australia, and New Zealand30. Most of the DWCs have been reported from the glacial Red Sea and they went extinct in response to unfavourable basin-wide hyperhaline conditions during the Last Glacial Maximum8. However, during the Holocene period, several bathyl taxa including the DWCs, have moved from the western Indian Ocean through the narrow and shallow sill of Bab al Mandab and successfully recolonized the Red Sea basin8. Hence, the E. fistula population in the Red Sea apparently represents a recent immigration from the western Indian Ocean.
The hydrodynamical and hydrographical conditions of the location (Fig. 1) where coral framework was recorded were also studied (Table 1). Relatively strong currents were detected along the entire water column, with a significant decreasing trend from surface (average 36.9 cm/s at 20 m depth) to the bottom (average 8.7 cm s−1 at 500 m depth). The surface currents were flowing towards the northwest direction (up to 100 m), while the bottom currents were in the west-southwest direction (Fig. S1). Under experimental conditions for DWC such as L. pertusa (now Desmophyllum pertusum31), low flow speed currents (< 7 cm s−1) were found optimal for successful prey capture32. Three layers of water masses—a surficial narrow mixed layer (down to 23 m with an average temperature of 29.01 °C), a sharp thermocline (from 23 m down to 200 m with a temperature gradient of 7 °C), and a homogenous bottom layer (from 200 m down to the bottom with a uniform temperature of ~ 21.5 °C) were detected (Fig. 4). Salinity profiles show two high-saline and one low-saline tongues in the subsurface waters between a depth of 20 and 70 m. Dissolved oxygen (DO) content at the surface (6.02 mg L−1) increased to 6.5 mg L−1 at 50 m. This subsurface maximum DO layer extends down to 150 m, followed by a sharp decreasing trend towards the mid layer with a DO of 1.5 mg L−1 at 400 m. A minor increasing trend in the DO level was detected from about 600 m to the seabed (Fig. 4). Yum et al.21, reported very low dissolved oxygen content (0.53 mg L−1) at a depth of 359 m in the central Red Sea from where E. fistula samples were collected and speculated that these corals use mitochondrial hypometabolism and anaerobic glycolysis to manage low oxygen conditions.
Concentrations of all nutrients, except silicon, were in traces within the euphotic zone, while the concentration of nitrate, phosphate, and silicon rapidly increased below the euphotic zone (Table 1). Concentration of Chlorophyll a ranged between 0.15 and 0.2 µg L−1 within the euphotic zone and increased only at a depth of 65 m, and the total suspended solids (TSS) was around 10 mg L−1 irrespective of the depth (Table 1). Chlorophyll maxima (DCM) occur in the deeper layers (> 70 m) with a significantly higher concentration of Chl a than that in the euphotic zone in the area north of 22–24°N33, where DWCs were recorded in this study.
The DWCs typically feed on common food items encountered in the deep water habitats, such as zooplankton, phytoplankton, and particulate organic carbon (POC)34,35. Sinking surface water in the northern Red Sea flows back to the south as deep-water currents, which may act as a food supplying mechanism for these deep-water corals. Zonal eddies have been reported in the central and northern Red Sea, and these eddies may play a significant role in the injection of nutrients into the water column. The presence of an anticylconic eddy centered around 25°N, with an east-flowing current to the north of 26°N (area of the present study) and a west-flowing current to the south has been reported36. The growth of a higher level of phytoplankton in the peripheries of anticyclonic eddies may propagate zooplankton biomass, resulting in high rates of marine snow fall from the periphery of anticyclonic eddies to the DWC framework. Cyanobacterial cells are dominant in the top layers (to a depth of about 20–30 m) of the northern Red Sea, whereas nanoplankton and picoeukaryotes are dominant at depths greater than 30 m and down to the depth of the DCM framework33. A study of the isotopic signatures of E. fistula collected from the Red Sea by Roder et al.5 indicates that E. fistula does not exclusively feed on particulate organic matter (POM), and other food sources such as microbes may contribute to the diet of the corals. Qurban et al.37 studied the primary production in the northern Red Sea and have speculated that the microbial loop plays a greater role in the trophic dynamics of the Red Sea. Hence, the combined effect of favorable current flow, copious supply of food, and moderate nutrient levels can provide the conditions necessary for the survival and growth of some DWCs in the highly demanding habitat of the deep Red Sea. These DWCs thrive in the deep-sea environment by maximizing the available resources and minimizing the metabolic demands5.
Apart from climate change, anthropogenic activities such as bottom trawling, hydrocarbon exploration and/or production, deep sea mining, cable and pipeline installation, pollution, waste disposal, coral exploitation and trade, and destructive scientific sampling threaten the global survival and distribution of DWCs22,38. Shipping, marine pollution, oil exploration, offshore infrastructure development for tourism, and aquaculture using floating cages are some of the activities with the potential to negatively impact the DWCs in the Red Sea. Thus, the highest priority regarding these sensitive ecosystems is to locate, map, and protect them by creating marine protected areas (MPAs), similar to the first MPA established off Florida in 1984 for the protection of a cold-water coral habitat38. Internationally, DWCs come under the purview of the Vulnerable Marine Ecosystems (VMEs) listed by the United Nations in resolution 61/105 that called upon States to protect VMEs. They also meet the criteria for Ecologically and Biologically Significant Marine Areas (EBSAs) laid down by the Conference of the Parties to the Convention on Biological Diversity (CBD)38. Even though recent surveys and studies5,6,20,21 have given a fresh impetus to the conservation of deep water corals in the Red Sea, the findings of these studies are yet to receive the attention of local government agencies, conservation organisations, and policy makers. Hopefully, the discovery of framework forming DWCs in the Red Sea and the previously conducted studies on DWCs from 20135,6,20,21 will receive the due recognition of the local policy makers, resulting in well-defined action plans for the conservation and protection of DWC habitats in the Red Sea.
Methods
An area (26.3843°–26.4335° N; 36.0358°–36.0888° E) located between Al Wajh and Duba in the Saudi waters of the Red Sea (Fig. 1) was randomly chosen for the deep-sea survey. The study area is approximately 30 km from the coast with a depth ranging from 636 to 959 m. A video survey was conducted within a 3 km × 3 km area.
Video survey and analysis
The survey was conducted on June 30, 2014 using the Research Vessel R. V. Aegaeo, belonging to the Hellenic Centre for Marine Research (HCMR). The ROV Max Rover (DSSI, USA) available onboard was well equipped for conducting sea bottom surveys and sampling6, and it was used for the video survey according to the methods described in Qurban et al6. A total of around 12 h of video was recorded during the day of the survey, along the track provided in Fig. 1. Coral samples were collected using the ROV, preserved and brought to the laboratory for further analyses.
The video footage was visually analyzed in the lab to assess the approximate area of coverage of the coral framework. The position of the coral framework was used to develop an ArcGIS distribution map. The coral species was identified based on the specimens collected with the help of taxonomists13,14 and DNA analysis.
DNA analysis
The DNA was extracted from the DWC samples using the Qiagen DNeasy Plant Mini Kit (Qiagen, Hilden, Germany) according to the instructions of the manufacturer. More than 5 replications of the process from extraction to amplification were conducted to ensure that the final sequencing accuracy is more than 99%. The QIAGEN Multiplex PCR Kit (Qiagen, Hilden, Germany) was used to perform the polymerized chain reaction for the amplification of mitochondrial region 16S39 and IGR40. The PCR cycling parameters used are as follows: initial 95 °C denaturation for 15 min. followed by 35 cycles of 94 °C for 60 s., annealing for 60 s. (LP16SF/LP16SR27: 50 °C; AGAL / DENF: 51 °C), and 72 °C for 60 s., and a final elongation step of 72 °C for 10 min.
The PCR products were checked under UV light after running in 1% agarose gel under 90 V for 45 min. All PCR products were cleaned by incubating with exonuclease I and FastAP Thermosensitive Alkaline Phosphatase (ExoFAP; USB, Cleveland, OH, USA) at 37 °C for 60 min. followed by 85 °C for 15 min. The final products were sequenced in both forward and reverse directions with fluorescently labeled dye terminators according to the manufacturer’s protocols (BigDye, Applied Biosystems Inc., Foster City, CA, USA), and analyzed using an ABI 3130XL Genetic Analyzer (Applied Biosystems). The 16S and IGR sequences were assembled and aligned using the program Geneious R8 (Biomatters Ltd., Auckland, New Zealand) and were uploaded to the GenBank.
Environmental data collection
A hull-mounted Acoustic Doppler Current Profiler (ADCP, 75 kHz Ocean Surveyor) with deep-water profiling capabilities was used to measure the water current and direction at different depths in and around the DWC area. The ADCP was programmed to profile the entire water column from 20 m to near-bottom at an ensemble interval of 5 min with a bin size of 20 m along the track of the ship. The depth range of good velocity data typically extended to 600 m below the vessel, depending on the conditions of the sea. The ADCP profiling was conducted over an area of 5 km × 5 km in the vicinity of the DWC site.
Vertical profiles of temperature, salinity (conductivity), dissolved oxygen (DO), density, and depth, measured at 1 m intervals, were obtained using a Sea-Bird-9 plus CTD system at nine stations (Fig. 1). Water samples obtained during the CTD cast were used for the determination of nutrients. Concentrations of nitrate, nitrite, phosphate, and silicon were photometrically determined using a SKALAR San Plus model Auto-Analyzer using the analytical methods provided by the manufacturer.
References
Roberts, J. M., Wheeler, A. J., Freiwald, A. & Cairns, S. D. Cold-Water Corals: The Biology and Geology of Deep-Sea Coral Habitats (Cambridge University Press, Cambridge, 2009).
Levin, L. A. & Dayton, P. K. Ecological theory and continental margins: where shallow meets deep. Trends Ecol. Evol. 24, 606–617 (2009).
Cairns, S. D. Deep-sea corals: an overview with special reference to diversity and distribution of deep-water scleractinian corals. Bull. Mar. Sci. 81, 311–322 (2007).
Berumen, M. L. et al. The status of coral reef ecology research in the Red Sea. Coral Reefs 32, 737–748. https://doi.org/10.1007/s00338-013-1055-8 (2013).
Roder, C. et al. First biological measurements of deep-sea corals from the Red Sea. Sci. Rep. 3, 2802 (2013).
Qurban, M. A. et al. In-situ observation of deep water corals in the northern Red Sea waters of Saudi Arabia. Deep-Sea Res. PT I(89), 35–43 (2014).
Marenzeller, E. von, 1907. Expedition S.M. Schiff “Pola” in das Rote Meer, nor̈dliche und südliche Hälfte 1895/96–1897/98. Zoologische Ergebnisse 25. Tiefseekor-allen. Denkschriften Der Mathematisch-Naturwissenschaftliche Klasse der Kaiserlichen Akademie der Wissenchaften vol. 80, 13–25.
Taviani, M. et al. Last glacial deep water coral from the Red Sea. Bull. Mar. Sci. 81, 361–370 (2007).
Fricke, H. W. & Schuhmacher, H. The depth limits of Red Sea stony corals: an ecophysiological problem (a deep diving survey by submersible). Mar. Ecol. 4, 163–194 (1983).
Fricke, H. W. & Hottinger, L. Coral bioherms below the euphotic zone in the Red Sea. Mar. Ecol. Prog. Ser. 11, 113–117 (1983).
Fricke, H. W. & Knauer, B. Diversity and spatial pattern of coral communities in the Red Sea upper twilight zone. Oecologia 71, 29–37 (1986).
Alcock, A. Diagnoses and descriptions of new species of corals from the Sibogaexpedition. Tijdschr. Ned. Dierkd. Ver. 7(2), 89–115 (1902).
Zibrowius, H. Les Scleractiniares de la Méditerranée et de l’Atlantique nord-oriental. Mem. Inst. Oceanogr. (Monaco) 11, 1–284 (1980).
Cairns, S. D. A revision of the shallow-water azooxanthellate Scleractinia of the western Atlantic. Stud. Nat. Hist. Caribb. Reg. 75, 1–321 (2000).
Tempera, F. et al. Eguchipsammia (Dendrophylliidae) topping on the cone. Mar. Biodiv. 45, 3–4. https://doi.org/10.1007/s12526-014-0220-9 (2015).
Cairns, S. D. The marine fauna of New Zealand: Scleractinia (Cnidaria: Anthozoa). NZ Oceanogr. Inst. Mem. 103, 210 (1995).
Evenhuis, N. L. & Eldredge, L. G. Records of the Hawaii Biological Survey for 2004–2005. Bishop Mus. Occas. Pap. 87, 66 (2006).
Gordon, D. New Zealand Inventory of Biodiversity. Volume One: Kingdom Animalia: Radiata, Lophotrochozoa, Deuterostomia (ed. Gordon, D. P.) 566 (Christchurch (New Zealand): Canterbury University Press, 2009).
Vine, P. Red Sea Invertebrates. IMMEL Publishing: London. ISBN 0–907151–11–6. 224 (1986).
Roik, A., Röthig, T., Roder, C., Müller, P. J. & Voolstra, C. R. Captive rearing of the deep-sea coral Eguchipsammia fistula from the Red Sea demonstrates remarkable physiological plasticity. PeerJ. 3, e734 (2015).
Yum, L. K. et al. Transcriptomes and expression profiling of deep-sea corals from the Red Sea provide insight into the biology of azooxanthellate corals. Sci. Rep. 7, 6442. https://doi.org/10.1038/s41598-017-05572-x (2017).
Hoegh-Guldberg, O., Poloczanska, E. S., Skirving, W. & Dove, S. Coral reef ecosystems under climate change and ocean acidification. Front. Mar. Sci. 4, 158 (2017).
Wienberg, C. et al. The giant mauritanian cold-water coral mound province: oxygen control on coral mound formation. Quat. Sci. Rev. 185, 135–152. https://doi.org/10.1016/j.quascirev.2018.02.012 (2018).
Raitsos, D. E. et al. Abrupt warming of the Red Sea. Geophys. Res. Lett. 38(1–5), L14601. https://doi.org/10.1029/2011GL047984 (2011).
Hoegh-Guldberg, O. et al. The Ocean. In Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part B: Regional Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds. Barros, V. R., Field, C. B., Dokken, D. J., Mastrandrea, M. D., Mach, K. J., Bilir, T. E., Chatterjee, M., Ebi, K. L., Estrada, Y. O., Genova, R. C., Girma, B., Kissel, E. S., Levy, A. N., MacCracken, S., Mastrandrea, P. R. & White, L. L.) 1655–1731 (Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, 2014).
Cantin, N. E., Cohen, A. L., Karnauskas, K. B., Tarrant, A. M. & McCorkle, D. C. Ocean warming slows coral growth in the central Red Sea. Science 329, 322–325. https://doi.org/10.1126/science.1190182 (2010).
Roberts, J. M., Wheeler, A. J. & Freiwald, A. Reefs of the deep: the biology and geology of cold-water coral ecosystems. Science 312, 543–547 (2006).
Taviani, M. et al. The “Sardinian cold-water coral province” in the context of the Mediterranean coral ecosystems. Deep Sea Res. II(145), 61–78. https://doi.org/10.1016/j.dsr2.2015.12.008 (2017).
Castellan, G., Angeletti, L., Taviani, M. & Montagna, P. The Yellow Coral Dendrophyllia cornigera in a Warming Ocean. Front. Mar. Sci. 6, 692; 10.3389/fmars.2019.00692 (2019).
van der Land, J. UNESCO-IOC Register of Marine Organisms (URMO, 2008).
Addamo, A. M. et al. Merging scleractinian genera: the overwhelming genetic similarity between solitary Desmophyllum and colonial Lophelia. BMC Evol Biol 16, 108. https://doi.org/10.1186/s12862-016-0654-8 (2016).
Orejas, C. et al. The effect of flow speed and food size on the capture efficiency and feeding behaviour of the cold-water coral Lophelia pertusa. J. Exp. Mar. Biol. Ecol. 481, 34–40 (2016).
Qurban, M., Wafar, M. & Heinle, M. Phytoplankton and primary production in the Red Sea. In Oceanographic and Biological Aspects of the Red Sea (eds Rasul, N. & Stewart, I.) 491–506 (Springer, Berlin, 2019).
Carlier, A. et al. Trophic relationships in a deep Mediterranean cold-water coral bank (Santa Maria di Leuca, Ionian Sea). Mar. Ecol. Progr. Ser. 397, 125–137 (2009).
Elias-Piera, F., Rossi, S., Gili, J. M. & Orejas, C. Trophic ecology of seven Antarctic gorgonian species. Mar. Ecol. Progr. Ser. 477, 93–106 (2013).
Wafar, M. et al. Patterns of distribution of inorganic nutrients in Red Sea and their implications to primary production. J. Mar. Syst. 156, 86–98 (2016).
Qurban, M. A., Balala, A. C., Kumar, S., Bhavya, P. S. & Wafar, M. Primary production in the northern Red Sea. J. Mar. Syst. 132, 75–82 (2014).
Roberts, J. M. & Cairns, S. D. Cold-water corals in a changing ocean. Curr. Opin. Environ. Sustain. 7, 118–126. https://doi.org/10.1016/j.cosust.2014.01.004 (2014).
Le Goff-Vitry, M. C., Pybus, O. & Rogers, A. Genetic structure of the deep-sea coral Lophelia pertusa in the northeast Atlantic revealed by microsatellites and internal transcribed spacer sequences. Mol. Ecol. 13, 537–549 (2004).
Arrigoni, R. et al. A phylogeny reconstruction of the Dendrophylliidae (Cnidaria, Scleractinia) based on molecular and micromorphological criteria, and its ecological implications. Zool. Scr. 43, 661–688 (2014).
Acknowledgements
We thank the Center for Environment & Water, Research Institute, King Fahd University of Petroleum & Minerals, Dhahran, Saudi Arabia, for providing research facilities. The authors thank the ROV Max Rover operation team, scientists, technicians, and crew of the Research Vessel RV Aegaeo, belonging to the Hellenic Centre for Marine Research (HCMR), Greece, for their skilful support during the cruise. Sequences have been deposited in NCBI GenBank under accession numbers [Submission ID: 2328258 16S and 2328249 IGR] (Eguchipsammia fistula).
Author information
Authors and Affiliations
Contributions
M.A.Q., K.P.M, T.T.M., and T.V.J. conceived the research cruise, collected samples, and carried out on board measurements. M.A.Q., P.K.K., and S.D.C. designed the manuscript. G.S. and D.T. analysed images. S.D.C. examined and photographed the specimens, and confirmed the identification of the species. S.H. conducted the DNA analysis. All authors contributed to data analyses and manuscript preparation. P.K.K. consolidated the sections prepared by other authors and completed the draft. All authors reviewed the draft and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Qurban, M.A., Krishnakumar, P.K., Joydas, T.V. et al. Discovery of deep-water coral frameworks in the northern Red Sea waters of Saudi Arabia. Sci Rep 10, 15356 (2020). https://doi.org/10.1038/s41598-020-72344-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-72344-5
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.