Abstract
Reverse transcription quantitative PCR (RT-qPCR) is used to quantify gene expression and require standardization with reference genes. We sought to identify the reference genes best suited for experiments that induce osteogenic differentiation from human induced pluripotent stem cells. They were cultured in an undifferentiated maintenance medium and after confluence, further cultured in an osteogenic differentiation medium for 28 days. RT-qPCR was performed on undifferentiation markers, osteoblast and osteocyte differentiation markers, and reference gene candidates. The expression stability of each reference gene candidate was ranked using four algorithms. General rankings identified TATA box binding protein in the first place, followed by transferrin receptor, ribosomal protein large P0, and finally, beta-2-microglobulin, which was revealed as the least stable. Interestingly, universally used GAPDH and ACTB were found to be unsuitable. Our findings strongly suggest a need to evaluate the expression stability of reference gene candidates for each experiment.
Similar content being viewed by others
Introduction
Quantitative real-time PCR (qPCR) was developed in the 1990s1 and is now widely used in various fields as a tool for the accurate and easy detection as well as quantification of target nucleic acid samples. In particular, reverse transcription quantitative PCR (RT-qPCR) is regarded as an indispensable experimental technique for the analysis of gene expression by quantifying RNA.
RT-qPCR is used to measure the amplification of an exponentially amplified region and quantify its initial template cDNA, that is, mRNA. Accurate assessments require the correction of inter sample variations, particularly of factors such as the total amount and quality of RNA, reverse transcription efficiency, and PCR efficiency. To perform this, internal control genes are selected as reference genes, which are quantified along with the target gene. Standardization is performed by calculating their expression ratios. The “housekeeping genes”, those with constant expression levels in the tissues or cells to be analyzed, are generally used as internal control genes; however, their expression levels may vary with each tissue and each cell. Fluctuations have been reported depending on the conditions such as the stages of development2. The Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE), a set of guidelines compiled to ensure the reproducibility of qPCR, stipulate that reference genes suited for each cell, tissue, and experimental design be selected from numerous candidates so as to ensure reliability of normalisation3. It is evident hence, that the selection of suitable reference genes, along with the provision of data demonstrating their expression stability, is becoming an indispensable part of quantitative analysis of gene expression using RT-qPCR.
To date, research has mainly focused on the osteogenic potential of mesenchymal stem cells (MSCs)4,5. However, since Takahashi and Yamanaka’s6 reports on human induced pluripotent stem (iPS) cells, there have been high expectations for the use of human iPS cells in areas such as regenerative medicine7 and the development of drugs for rare diseases8. Cell culture experiments have focused on hypoxic environments as the original physiological state inside living tissues9,10,11. We reported that when rat MSCs cultured in a hypoxic environment were exposed to a normoxic environment, it enhanced their osteogenic potential12. Based on these results, we expect an examination of the osteogenic differentiation cultures of iPS cells under hypoxic conditions would significantly contribute to osteochondral regenerative medicine and our understanding of the pathology of musculoskeletal diseases such as knee osteoarthritis. Nevertheless, only a few studies have thoroughly verified the expression stability of reference genes used during RT-qPCR for osteogenic differentiation experiments using stem cells, especially iPS cells. Therefore, in the present study we compared multiple internal control genes to identify the most stable reference genes for osteogenic differentiation experiments using RT-qPCR on human iPS cells under various oxygen concentrations.
Results
Tissue staining
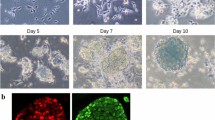
Continuous induction of osteogenic differentiation (Fig. 1a) resulted in areas that revealed calcification (Fig. 1b). Samples cultured on day 28 were stained to verify this. The results presented areas positive for Alizarin Red S throughout the wells, with certain regions exhibiting ALP activity (Fig. 1c).
Osteogenic differentiation in human induced pluripotent stem (iPS) cells. (a) Schema of the osteogenic differentiation protocol. (b) Microscopic images on day − 4, 0, 14 and 28 showed mineralization yield. (c) Mineralization on day 28 with Alizarin red S and alkaline phosphatase (ALP) staining results in six-well culture plates.
Comparison of reference gene candidate stability by RT-qPCR
RT-qPCR was performed to compare the expression stability of internal control genes listed in Table 1 on samples from three wells on days 0, 7, 14, 21, and 28. The resulting threshold cycle (Ct) values (Fig. 2) and PCR efficiency values from the validation data of each primer were used in each method to calculate the results listed in Table 2. None of the samples were excluded as invalid while verifying RNA purity with absorbance measurements, and all Ct values were < 35.
First, the ΔCT method revealed that the gene with the lowest mean SD was TBP, and that with the highest value was B2M, which was the only one with a mean SD > 1. Based on BestKeeper threshold of SD = 1, no candidate genes were excluded. GUSB and HMBS had the lowest and highest SDs, respectively. In addition, r value was the highest and lowest for TFRC and B2M, respectively. The general ranking calculated from these two rankings revealed that TFRC had the highest stability, while that of B2M was the lowest. Using NormFinder, all the iPS cell samples in this study that underwent osteogenic differentiation were treated as a single group. Three samples from each day were used to analyze a total of 15 samples. The genes with the lowest stability values were TBP and TFRC, whereas B2M had the highest value. Figure 3 depicts two graphs created using geNorm. The pair with the lowest M value was RPLP0 and TBP with threshold values of less than 0.5, whereas B2M presented the highest value (Fig. 3a). In addition, V2/3–V13/14 were all below 0.15 (Fig. 3b). These results indicate that valid assessments can be performed with the combination of RPLP0 and TBP. While these methods found several candidates with high stability, including TBP and TFRC, all the methods demonstrated the low stability of B2M.
A general ranking was calculated using the geometric means of the rankings in each of the four methods (Table 2). The results indicate that TBP is the gene with the most stable expression and that B2M is the lowest.
Expression of target genes using RT-qPCR
Using TBP as the reference gene based on the results of the aforementioned analysis, RT-qPCR was performed to evaluate the expression of each target gene in the osteogenic differentiation process using the primers listed in Table 3. Like the process with internal control genes, samples from days 0, 7, 14, 21, and 28 were used. To consider the variation in differentiation rates between samples on days 21 and 28, six wells were sampled on these days and three wells were sampled on the other days. For the undifferentiation markers, sampling was performed on days 0, 1, 2, 3, 4, 5, 7, 9, 11, and 14, given that gene expression declines early in the culture period. Due to the variation in differentiation rates between samples, six wells were sampled on days 11 and 14, and three wells were sampled on the other days. Relative expression levels for each gene were calculated using the 2−∆∆Ct method by employing the Ct values obtained by RT-qPCR (Fig. 4). A verification of RNA purity using absorbance measurements revealed that A260/A230 was greater than 1.8 in the following samples: one sample on day 7 and two samples on day 14 in the undifferentiation marker experiment; one sample on day 14 and one sample on day 21 in the experiment conducted for the other markers. These samples were deemed as invalid and excluded from the analysis. As the Ct values of DMP1 exceeded 35 in one sample each on days 7 and 14, these samples were also excluded as they were deemed invalid.
Expression of undifferentiation (a) and osteogenic marker (b) genes standardized by TBP in iPSCs during osteogenic differentiation. The values are represented as the mean ± S.D. from two-six independent samples. One-way analysis of variance (ANOVA) with Bonferroni correction for multiple comparisons was applied. *P < 0.05, **P < 0.01 versus day 0.
Figure 4a depicts the expression of TBP as a reference gene and the changes in the relative expression of undifferentiation markers during osteogenic differentiation up to day 14. TBP expression did not vary markedly over the entire period. The expression of both undifferentiation markers, POU5F1 and NANOG, declined significantly from day 1 until day 14. This suggests that during the osteogenic differentiation process in this study, human iPS cells lost their undifferentiated nature and further differentiated into a certain cell type.
Figure 4b illustrates the expression of TBP as a reference gene and the changes in the relative expression of osteoblast and osteocyte differentiation markers during osteogenic differentiation up to day 28. TBP expression did not vary significantly over the entire period. Expression of the transcription factors, RUNX2 and SP7, and the proteins, COL1A1 and OC, which are osteoblast differentiation markers, exhibited increasing trends starting from day 0. These results indicate that inducing osteogenic differentiation promoted osteoblast differentiation. The protein ALP is not only an osteoblast differentiation marker but also an undifferentiation marker. Its expression level was high on day 0 when the cells were undifferentiated. It then declined over time before it exhibited an increasing trend again on day 28 due to osteoblast differentiation. The protein DMP1 is an osteocyte differentiation marker that is expressed in young osteocytes and the protein SOST is expressed in mature bone cells. Both exhibited a marked increase in expression on day 28. Despite the large variations in DMP1 expression between samples on day 28, osteocyte differentiation is assumed to have progressed at least during the second half of the osteogenic differentiation period.
We also standardized the results of RT-qPCR using only GAPDH (Supplemental Fig. 1), which is described as one of the most commonly used method in existing studies13,14. When the focus was on the presence of significant differences, results were generally similar. However, we found the relative expression of GAPDH tended to be higher in the second half of the osteogenic differentiation period compared to TBP, and the relative expression of the target genes tended to be relatively low.
These results indicate that human iPS cells used in the present study differentiated into osteoblasts and osteocytes by inducing osteogenic differentiation. They also provide evidence that assessment of the expression of internal control genes, which was the objective of this study, was performed during the induction of osteogenic differentiation in human iPS cells.
Discussion
Presently, we conduct osteogenic differentiation experiments using human iPS cells, and RT-qPCR is used as the primary method to quantify gene expression. This speaks to the importance of the accuracy of RT-qPCR and the need to appropriately evaluate the quality of the extracted RNA and select reagents and reference genes.
In the present study, we initially examined the RNA extraction from calcified samples. First, the absorbance of the RNA samples was measured to calculate A260/A230 and A260/A280 ratios. High RNA purity was observed when A260/A230 > 1.8 and A260/A280 > 2.0. Denatured agarose gel electrophoresis was also performed on the RNA samples to calculate the ribosomal RNA ratio 28S/18S and determine whether it was close to 2:1. Assessing the RNA quality of several extraction reagents revealed that TRIzol had the highest probability of clearing the RNA purity criteria with 28S/18S close to 2:1. This reagent was found to yield maximum RNA from samples of the same size. Based on these results, we determined that TRIzol is the most suitable for extracting RNA in osteogenic differentiation experiments and devised a method of using RNeasy Micro Kit to recover RNA after extraction.
Reference Genes H96 comprises preset control wells for verifying genomic DNA contamination, PCR efficiency, RNA quality, and reverse transcription efficiency. We used this plate in a preliminary experiment for the present study and found a case that did not meet the standard for reverse transcription efficiency during control. This prompted us to compare multiple cDNA synthesis kits using reagents used in control wells for the reverse transcription efficiency of Reference Genes H96. The iScript Advanced cDNA Synthesis Kit had the highest reverse transcription efficiency and was adopted for cDNA synthesis.
RNA concentrations were calculated from A260 measured with a spectrophotometer and cDNA concentrations were calculated from RNA concentration. We recognize that their lack of accuracy may affect the assessment of gene expression levels and is a limitation of this study. However, these concentrations must be standardized by suitable reference genes and hence in the present study, we did not require further accuracy.
After the aforementioned verification process, we verified suitable reference genes, which was the aim of the present study. Standardizing the expression of target genes using reference genes is an essential means of correcting variations between samples in areas such as total RNA, reverse transcription efficiency, and PCR efficiency. The MIQE guidelines state that suitable reference genes should be selected for each tissue, cell, and experimental design3. However, an existing study on osteogenic differentiation using human embryonic stem (ES) cells, MSCs, and iPS cells was found to use customarily used genes such as GAPDH, ACTB, and 18S rRNA13. The use of GAPDH is particularly common, although no basis has been provided for its selection14. Only limited studies have attempted to identify optimal reference genes, and as far as we could find, none were identified for human iPS cells.
To verify the optimal reference genes, the present study used the most common and reliable methods: the ΔCt method, BestKeeper, NormFinder, and geNorm. The ΔCt method is a pairwise comparison and details on the other three algorithms have been cited in the MIQE guidelines3. Reference genes verification results based on these algorithms have been reported in several studies for various cells and differentiation processes15,16,17,18,19,20,21,22,23. For example, Robledo et al.16 and Augustyniak et al.17 performed verifications using the four algorithms we used in the present study. Jacob et al.15 suggested that verifications should be performed with at least three different algorithms. They found that more than 60% of the published studies on RT-qPCR use two algorithms, NormFinder and geNorm, whereas only 2.6% use all four algorithms. This was based on their review of studies published between 2009 and 2012. Although we expect recent studies to apply more algorithms, Jacob et al.’s finding highlighted the fact that only a few studies have conducted thorough reference gene verification.
Due to the weighting of SD and r values, Bestkeeper may be the least user-friendly out of the four algorithms. As no clear ranking method is provided, some studies have chosen not to use this method18. In the present study, after excluding items with SD > 1 as invalid, we calculated a general ranking by measuring the geometric mean ranking of SD and r value. A web-based tool called RefFinder (https://www.leonxie.com/referencegene.php) automatically executes BestKeeper, NormFinder, and geNorm algorithms when Ct values are entered, and calculates the stability rankings based on each algorithm, as well as a general ranking. Although many studies have used the verification results of RefFinder15,19,20,21, we did not adopt it in the present study because; (1) it does not take into account PCR efficiency, which can affect PCR results21; (2) its BestKeeper results only include SD but not r; and (3) the method used to determine the general ranking is not stipulated.
Of the 14 preset candidate genes in Reference Genes H96, we found B2M to be the most unsuitable reference gene. This finding was consistent across all the algorithms and our results indicate that B2M has high fluctuations compared to the other candidates. Consistent with Jacob et al.15 and Vandesompele et al.’s24 recommendation to evaluate combinations of three reference genes, our results found TBP, TFRC, and RPLP0 to be suitable reference genes. The details of our findings are presented in Table 2.
However, in the present study we obtained these results only on the osteogenic differentiation of human iPS cells under normoxic conditions. We plan to perform further experiments under hypoxic conditions and with the exclusion of candidate genes that fluctuate with changes in oxygen concentration. GAPDH is a typical example of such genes. Among the three recommended genes, TFRC is involved in the hypoxic response, and as it is presumed to fluctuate depending on the oxygen concentration, its exclusion is highly recommended25. Hence, if only one reference gene was to be used, it should be TBP. If a combination of genes was to be used, geNorm results indicate that the standard should be TBP and RPLP0. The present study found TBP to have the highest stability, a finding which is consistent with that of Rauh et al.22 They found TBP to be the most stable reference gene in NormFinder and geNorm in three-dimensional osteogenic differentiation culture experiments of human bone marrow derived MSC. Consistent with their results, we also found TFRC to be the second most stable reference gene. However, instead of TBP or TFRC, their investigation of reference genes in a two-dimensional culture experiment identified completely different genes as the most suitable. Li et al.23 found that TBP to be one of the least stable genes and B2M as one of the most stable in osteogenic differentiation and adipose differentiation culture experiments of human bone marrow-derived and fetal tissue-derived MSCs. Their results appear to be contradictory to our findings. However, this issue of reversed stability ranking of genes has been raised by Rauh et al.22 and suggests that the selection of reference genes suited to each cell, tissue, and experimental design requires an investigation of the expression stability indicated by reference gene candidates in each experiment.
We would like to highlight that the commonly used GAPDH was not highly ranked; its position was 5 out of 14, whereas ACTB was positioned at 11. Our results indicate that commonly used reference genes such as GAPDH and ACTB were unsuitable in the current experiment and that using them may have led to incorrect interpretations of gene expression levels. This is consistent with De Jonge et al.’s26 analysis of data from public samples of 13,629 microarrays performed in various tissues and conditions; they found that of the 13,037 genes, none of the commonly used reference genes—ACTB, GAPDH, HPRT1, B2M—were ranked in the top 50 for stability.
Our study standardized the results of RT-qPCR on target genes in the osteogenic differentiation process using only TBP, which was used to evaluate the expression level of each gene. In this study, POU5F1 and NANOG were used as undifferentiation markers and RUNX2, SP7, ALP, COL1A1, and OC were used as osteoblast differentiation markers. These genes were adopted because they were commonly used in studies27,28,29,30 reporting similar osteogenic differentiation experiments on human iPS cells. We considered osteocyte differentiation markers to be necessary as well. We used DMP1, which is expressed in relatively young osteocytes, and SOST, which is a marker of mature osteocytes. We found that the levels of undifferentiation markers declined markedly within 2 weeks, indicating a loss of undifferentiation, whereas those of the osteoblast and osteocyte differentiation markers increased within 4 weeks. This indicates that iPS cells differentiate into osteoblasts and osteocytes. This observation was also supported by results from two types of staining. We were able to achieve the main purpose of this study, that is to verify the reference genes in an osteogenic differentiation experiment on human iPS cells. When the reference gene was changed from TBP to GAPDH, there was a slight difference in the trend of relative expression of the target genes. Although it did not appear as a clear difference in this study, there is a risk that even a slight difference in the trend of relative expression could lead to errors in the assessment of target gene expression levels.
The stage of differentiation that the human iPS cells were at each time in the culture period of our experiments was not revealed. This is another limitation of this experiment. However, given that the purpose of this study was to validate the reference genes which can be used appropriately throughout the entire culture period, we decided that it was not necessary to verify the differentiation stage.
As we continue to develop our osteogenic differentiation experiments using iPS cells, it will be necessary to verify the transition of the differentiation stage by quantification of target proteins and other factors. We believe that this will allow us to verify suitable reference genes at each differentiation stage. In addition, the verification of more genes that have not been included in the reference gene candidates so far, will allow room for the identification of more suitable reference genes. We hope that these verification data will allow to perform more accurate RT-qPCR.
Materials and methods
Human iPS cell line and induction of osteogenic differentiation
We used the commercially available Cellartis Human iPS Cell Line 12 (Takara Bio Inc., Kusatsu, Japan) as the human iPS Cell line. This was seeded on wells coated with the Matrigel basement membrane matrix for feeder-free culturing (Corning Inc., Corning, NY, USA) and then cultured in the undifferentiated maintenance medium Essential 8 (E8; Thermo Fisher Scientific Inc., Waltham, MA, USA). The medium was changed daily and after reaching confluence (about 80%), the cells were detached using Versene (Thermo Fisher Scientific Inc.) for 12 min in 37 °C and replated. This passage procedure was repeated.
Cells obtained by cloning in this manner were used in the osteogenic differentiation experiment (Fig. 1a). Confluent cells (defined as Day 0) were switched to Dulbecco’s modified Eagle’s medium (DMEM; Nacalai Tesque Inc.) containing fetal bovine serum (FBS, 15%; GE Healthcare Life Sciences, Logan, UT, USA), 1% nonessential amino acids (Sigma-Aldrich Inc., St. Louis, MO, USA), 1% antibiotic–antimycotic mixed stock solution (Nacalai Tesque Inc., Kyoto, Japan), and 100 μM β-mercaptoethanol (Nacalai Tesque Inc.). This medium was changed daily. From day 5,10 mM β-glycerophosphate (Sigma-Aldrich), 50 μg/ml l-ascorbic acid (Wako Pure Chemical Ind., Osaka, Japan), 10 nM dexamethasone (Sigma-Aldrich), and 50 nM 1,25-(OH)2 vitamin D3 (Wako Pure Chemical Ind.) were added to the above-mentioned medium to induce osteogenic differentiation and the culture was changed once every two days.
Tissue staining
For the staining assessments, the human iPS cells that underwent osteogenic differentiation were fixed with 95% ethanol. To evaluate Ca deposition, they were stained with Alizarin Red S (Nacalai Tesque Inc.). To assess ALP activity, they were stained with Naphthol AS-MX phosphate disodium salt (Sigma-Aldrich) and Fast Red Violet LB Salt (Nacalai Tesque Inc.) in 56 mM 2-amino-2-methyl-1,3-propanediol buffer (Wako Pure Chemical Ind.) adjusted to pH 9.9.
Gene expression analysis
For total RNA extraction, TRIzol (Invitrogen Life Technologies, Carlsbad, CA, USA) was added and left to stand at room temperature for 10 min. The resulting solution was transferred to a QIA shredder spin column (Qiagen Inc., Valencia, CA, USA) and centrifuged at 15,000 rpm for 2 min at room temperature. The resulting filtrate was transferred to a new 1.5 ml microtube, 200 μl of chloroform was added, mixed well, and allowed to stand at room temperature for 5 min. This was centrifuged at 15,000 rpm for 15 min at 4 °C before the upper aqueous layer was transferred to a new 1.5 ml microtube, and the same amount of 70% ethanol was added and mixed well. This sample was then transferred to RNeasy Micro Kit spin column (Qiagen Inc.) and total RNA was recovered according to the procedure described in the product’s instruction manual. Next, DNase treatment was performed to prevent genomic DNA contamination. The resulting solution was diluted 20-fold with Tris–EDTA buffer (pH 8.0) (Nippon Gene, Toyama, Japan), and then analyzed with a Ultrospec 3,000 spectrophotometer (Biochrom Ltd., Cambridge, England) at 230, 260, and 280 nm to measure the absorbance, based on which the purity of RNA was confirmed and its concentration was calculated. RNA template (1 μg) was prepared and reverse transcription reaction was performed with iScript Advanced cDNA Synthesis Kit (Bio-Rad Laboratories, Hercules, CA, USA) according to the procedure described in the product’s instruction manual. The StepOnePlus Real-Time PCR System (Applied Biosystems, Foster City, CA, USA) was used for RT-qPCR.
Reference Genes H96 (Bio-Rad Laboratories) was used in RT-qPCR to compare the expression levels of several internal control genes that were candidate reference genes. Reference Genes H96 is a predesigned plate with wells containing primers for 14 types of validated human internal control genes (Table 1), which can be used as control assays for evaluating genomic DNA contamination, PCR efficiency, RNA quality, and reverse transcription efficiency. A reaction solution of 20 µl comprising 10 μl 2 × Sso Advanced universal SYBR Green Supermix (Bio-Rad), 1 μl cDNA template (25 ng), and 9 μl sterile distilled water was added per sample in each well. For the reaction conditions, initial activation was performed at 95 °C for 2 min, followed by 40 cycles of thermal denaturation at 95 °C for 5 s and then annealing and elongation at 60 °C for 30 s, and eventually the melting curve was analyzed.
The internal control genes selected based on the data analysis described in the following manner were used as the target genes in RT-qPCR to analyze the expression of undifferentiation, osteoblast differentiation, and osteocyte differentiation markers (Table 3). PrimePCR SYBR Green Assay (Bio-Rad) or Taqman Gene Expression Assay (Applied Biosystems) was used as the primer. For the former, 5 μl of 2 × SsoAdvanced universal SYBR Green Supermix, 0.5 μl of 20 × SYBR Green Assay, 1 μl (25 ng) of cDNA template, and 3.5 μl of sterile distilled water were used as the reaction solution, for a total of 10 μl per sample dispensed in each well. The reaction conditions were the same for Reference Genes H96. For the latter, 10 μl of 2 × Taqman Fast Universal PCR Master Mix, 1 μl of 20 × Gene Expression Assay, 2 μl (50 ng) of cDNA template, and 7 μl of sterile distilled water were used as the reaction solution, for a total of 20 µl per sample dispensed in each well. For the reaction conditions, initial activation was performed at 95 °C for 20 s, followed by 40 cycles of thermal denaturation at 95 °C for 1 s, and annealing and elongation at 60 °C for 20 s.
Data analysis
To select suitable reference genes for osteogenic differentiation experiments in human iPS cells, Ct values, PCR efficiency, and relative expression values based on the internal control genes obtained using RT-qPCR were used in the following methods for analysis.
ΔCt method
Silver et al.31 reported the ΔCt method for comparing the stability of candidate genes based on pairwise comparison. A pair is created with candidate gene A and another candidate gene B. Thereafter, the difference in their Ct values in the same sample is calculated to obtain ΔCt. The standard deviation (SD) of ΔCt in all samples for the pair (gene A and B) is then calculated (A vs. B). This calculation is also performed for all the pairs containing gene A such as A vs. C and A vs. D. The arithmetic means of the all SD (mean SD) for gene A represents the variation in the expression of gene A with respect to all the candidate genes, allowing it to be compared with other genes. Accordingly, the gene with the lowest value is the most stable reference gene. A characteristic of this method is that assessments can be performed by merely repeating a relatively simple calculation.
BestKeeper
The BestKeeper algorithm of Pfaffl et al.32 is characterized by directly inputting the crossing point (CP) value, which is equal to Ct, and PCR efficiency for all the samples of each candidate reference gene to automatically calculate two indicators. The first indicator is the SD of the CP values of each gene, with the lowest value being the most stable reference gene. The developers recommend removing any candidate gene with SD > 1. Next, the geometric mean of the CP values of all genes in each sample is calculated as the BestKeeper index, and Pearson's correlation coefficient (r) between this and the CP value of each gene is measured as the second indicator. The highest value represents the most stable reference gene. Since BestKeeper requires an evaluation of both SD and r, we considered the geometric mean of the ranking for both indicators, which was used to create a general ranking for BestKeeper.
NormFinder
The NormFinder algorithm of Andersen et al.33 is characterized by evaluating stability through the amount of change in expression within and between groups. A “stability value” is obtained by inputting the relative expression value, with the lowest value representing the most stable reference gene. Each group needs at least eight samples for appropriate assessments and a sample of five to ten candidate reference genes is recommended.
geNorm
The geNorm algorithm of Vandesompele et al.24 is based on pairwise comparisons. Inputting the relative expression values yields two indicators. The “M value” indicates the stability of expression, with the lowest value representing the most stable reference gene. In the developer’s experimental data, genes with stable expression had a mean M value < 0.534. When evaluating the stability of two or more reference gene combinations, a pairwise variation (Vn/n+1) is calculated to determine the appropriate number of reference genes to be combined, and Vn/n+1 < 0.15 is considered as an appropriate cutoff value.
Statistical analysis
The expression of each gene is presented as mean ± standard deviation (SD). When a significant difference was observed in a one-way analysis of variance (ANOVA) with day as the factor, multiple comparisons with the Bonferroni method were performed. P < 0.05 was considered as statistically significant. SPSS 22.0 J for Windows (SPSS Inc., Chicago, IL, USA) was the statistical software used for this analysis.
References
Higuchi, R., Fockler, C., Dollinger, G. & Watson, R. Kinetic PCR analysis: real-time monitoring of DNA amplification reactions. Biotechnology (NY) 11, 1026–1030 (1993).
Bustin, S. A. Quantification of mRNA using real-time reverse transcription PCR (RT-PCR): trends and problems. J. Mol. Endocrinol. 29, 23–39 (2002).
Bustin, S. A. et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 55, 611–622 (2009).
Akahane, M. et al. Osteogenic matrix sheet-cell transplantation using osteoblastic cell sheet resulted in bone formation without scaffold at an ectopic site. J. Tissue Eng. Regen. Med. 2, 196–201 (2008).
Tohma, Y. et al. Bone marrow-derived mesenchymal cells can rescue osteogenic capacity of devitalized autologous bone. J. Tissue Eng. Regen. Med. 2, 61–68 (2008).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Tsumaki, N., Okada, M. & Yamashita, A. iPS cell technologies and cartilage regeneration. Bone 70, 48–54 (2015).
Hino, K. et al. Activin-A enhances mTOR signaling to promote aberrant chondrogenesis in fibrodysplasia ossificans progressiva. J. Clin. Investig. 127, 3339–3352 (2017).
Grayson, W. L., Zhao, F., Bunnell, B. & Ma, T. Hypoxia enhances proliferation and tissue formation of human mesenchymal stem cells. Biochem. Biophys. Res. Commun. 358, 948–953 (2007).
Hu, X. et al. Transplantation of hypoxia-preconditioned mesenchymal stem cells improves infarcted heart function via enhanced survival of implanted cells and angiogenesis. J. Thorac. Cardiovasc. Surg. 135, 799–808 (2008).
Ren, H. et al. Proliferation and differentiation of bone marrow stromal cells under hypoxic conditions. Biochem. Biophys. Res. Commun. 347, 12–21 (2006).
Inagaki, Y. et al. Modifying oxygen tension affects bone marrow stromal cell osteogenesis for regenerative medicine. World J. Stem Cells 9, 98–106 (2017).
Yang, X. et al. Bone to pick: the importance of evaluating reference genes for RT-qPCR quantification of gene expression in craniosynostosis and bone-related tissues and cells. BMC Res. Notes 5, 222 (2012).
Bustin, S. A., & Nolan, T. Data analysis and interpretation in A-Z of Quantitative PCR (ed. Bustin, S. A.) 441–492 (International University Line, 2004).
Jacob, F. et al. Careful selection of reference genes is required for reliable performance of RT-qPCR in human normal and cancer cell lines. PLoS ONE 8, e59180 (2013).
Robledo, D. et al. Analysis of qPCR reference gene stability determination methods and a practical approach for efficiency calculation on a turbot (Scophthalmus maximus) gonad dataset. BMC Genom. 15, 648 (2014).
Augustyniak, J., Lenart, J., Lipka, G., Stepien, P. P. & Buzanska, L. Reference gene validation via RT-qPCR for human iPSC-derived neural stem cells and neural progenitors. Mol. Neurobiol. 56, 6820–6832 (2019).
Piehler, A. P., Grimholt, R. M., Ovstebø, R. & Berg, J. P. Gene expression results in lipopolysaccharide-stimulated monocytes depend significantly on the choice of reference genes. BMC Immunol. 11, 21 (2010).
Diesel, L. F. et al. Stability of reference genes during tri-lineage differentiation of human adipose-derived stromal cells. J. Stem Cells 10, 225–242 (2015).
Taïhi, I. et al. Validation of housekeeping genes to study human gingival stem cells and their in vitro osteogenic differentiation using real-time RT-qPCR. Stem Cells Int. 2016, 6261490 (2016).
De Spiegelaere, W. et al. Reference gene validation for RT-qPCR, a note on different available software packages. PLoS ONE 10, e0122515 (2015).
Rauh, J., Jacobi, A. & Stiehler, M. Identification of stable reference genes for gene expression analysis of three-dimensional cultivated human bone marrow-derived mesenchymal stromal cells for bone tissue engineering. Tissue Eng. Part C Methods 21, 192–206 (2015).
Li, X. et al. Identification of optimal reference genes for quantitative PCR studies on human mesenchymal stem cells. Mol. Med. Rep. 11, 1304–1311 (2015).
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3, research0034 (2002).
Bianchi, L., Tacchini, L. & Cairo, G. HIF-1-mediated activation of transferrin receptor gene transcription by iron chelation. Nucleic Acids Res. 27, 4223–4227 (1999).
De Jonge, H. J. et al. Evidence based selection of housekeeping genes. PLoS ONE 2, e898 (2007).
Kato, H. et al. Promoting effect of 1,25(OH)2 vitamin D3 in osteogenic differentiation from induced pluripotent stem cells to osteocyte-like cells. Open Biol. 5, 140201 (2015).
Sparks, N. R. L., Martinez, I. K. C., Soto, C. H. & Zur Nieden, N. I. Low osteogenic yield in human pluripotent stem cells associates with differential neural crest promoter methylation. Stem Cells 36, 349–362 (2018).
Ardeshirylajimi, A., Hosseinkhani, S., Parivar, K., Yaghmaie, P. & Soleimani, M. Nanofiber-based polyethersulfone scaffold and efficient differentiation of human induced pluripotent stem cells into osteoblastic lineage. Mol. Biol. Rep. 40, 4287–4294 (2013).
Kanke, K., Masaki, H., Saito, T., Komiyama, Y. & Hojo, H. Stepwise differentiation of pluripotent stem cells into osteoblasts using four small molecules under serum-free and feeder-free conditions. Stem Cell Rep. 2, 751–760 (2014).
Silver, N., Best, S., Jiang, J. & Thein, S. L. Selection of housekeeping genes for gene expression studies in human reticulocytes using real-time PCR. BMC Mol. Biol. 7, 33 (2006).
Pfaffl, M. W., Tichopad, A., Prgomet, C. & Neuvians, T. P. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper—excel-based tool using pair-wise correlations. Biotechnol. Lett. 26, 509–515 (2004).
Andersen, C. L., Jensen, J. L. & Ørntoft, T. F. Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res. 64, 5245–5250 (2004).
Hellemans, J., Mortier, G., De, P. A., Speleman, F. & Vandesompele, J. qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol. 8, R19 (2007).
Acknowledgements
This work was supported by Grants from JSPS KAKENHI [JP18K1663 and 20K09508 to Y.I., JP20H03199 to E.M.], AMED Brain/MINDS Beyond [JP20dm0307032 to E.M.], Takeda Science Foundation to E.M., Uehara Memorial Foundation to E.M., Naito Foundation to E.M., MSD Life Science Foundation to E.M., and the unrestricted funds provided to E.M. from Dr. Taichi Noda (KTX Corp., Aichi, Japan) and Dr. Yasuhiro Horii (Koseikan, Nara, Japan). The authors would like to thank Ms. Fumika Kunda for her technical assistance in the laboratory and Ms. Keren-Happuch E Fan Fen for her critical reading of the manuscript.
Author information
Authors and Affiliations
Contributions
K.O. designed and conducted the experiments, analyzed the data and drafted the manuscript. Y.I. and E.M. designed the study and wrote and revised the manuscript. T.K.M., M.M. and T.K. conducted the experiments. T.K.M. and M.M. supervised the experiments and analysis of data. O.M., E.M. and Y.T. supervised this project. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Okamura, K., Inagaki, Y., Matsui, T.K. et al. RT-qPCR analyses on the osteogenic differentiation from human iPS cells: an investigation of reference genes. Sci Rep 10, 11748 (2020). https://doi.org/10.1038/s41598-020-68752-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-68752-2
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.