Abstract
Apple bitter rot caused by Colletotrichum species is a growing problem worldwide. Colletotrichum spp. are economically important but taxonomically un-resolved. Identification of Colletotrichum spp. is critical due to potential species-level differences in pathogenicity-related characteristics. A 400-isolate collection from New York apple orchards were morphologically assorted to two groups, C. acutatum species complex (CASC) and C. gloeosporioides species complex (CGSC). A sub-sample of 44 representative isolates, spanning the geographical distribution and apple varieties, were assigned to species based on multi-locus phylogenetic analyses of nrITS, GAPDH and TUB2 for CASC, and ITS, GAPDH, CAL, ACT, TUB2, APN2, ApMat and GS genes for CGSC. The dominant species was C. fioriniae, followed by C. chrysophilum and a novel species, C. noveboracense, described in this study. This study represents the first report of C. chrysophilum and C. noveboracense as pathogens of apple. We assessed the enzyme activity and fungicide sensitivity for isolates identified in New York. All isolates showed amylolytic, cellulolytic and lipolytic, but not proteolytic activity. C. chrysophilum showed the highest cellulase and the lowest lipase activity, while C. noveboracense had the highest amylase activity. Fungicide assays showed that C. fioriniae was sensitive to benzovindiflupyr and thiabendazole, while C. chrysophilum and C. noveboracense were sensitive to fludioxonil, pyraclostrobin and difenoconazole. All species were pathogenic on apple fruit with varying lesion sizes. Our findings of differing pathogenicity-related characteristics among the three species demonstrate the importance of accurate species identification for any downstream investigations of Colletotrichum spp. in major apple growing regions.
Similar content being viewed by others
Introduction
Colletotrichum is a cosmopolitan fungal genus comprised of more than 189 species distributed throughout tropical and temperate regions worldwide1,2,3. Colletotrichum species cause devastating diseases such as anthracnose and fruit rots on a broad range of plant hosts4,5 and affect valuable fruit crops such as banana, strawberry, citrus, avocado, papaya, mango and apple6,7,8,9,10.
Apple (Malus domestica Borkh.), native to central Asia and then introduced to the west and other parts of the world11,12, is a major fruit crop cultivated in temperate regions today. The United States produces about five million metric tons on almost 130,309 hectares, making it the second largest apple producer after China13. In the U.S. in 2018, apples were grown commercially in 20 states with New York ranking as the second largest producer14, with 17 thousand hectares of apples cultivated for over 260 million dollars in value15.
Apple is vulnerable to a wide range of diseases affecting yield and fruit quality. Bitter rot, caused by Colletotrichum spp., is one of the most important fungal diseases of apple causing remarkable economic losses under wet and warm weather conditions in the US and globally6,16,17. Reports of apple fruit losses to bitter rot in New York range from 14–25%18,19 and up to 100% in organic orchards, reach up to 100% in North Carolina20 and are 30% on average in Kentucky, where some orchards were a complete loss21,22. Among the nine major clades of Colletotrichum, the C. gloeosporioides species complex (CGSC) and the C. acutatum species complex (CASC) are the two most common clades that cause bitter rot of apple2,16,17. C. fioriniae, C. nymphaea and C. godetiae23 from CASC and C. fructicola, C. aenigma, C. siamense and C. theobromicola from CGSC3,17,24,25,26, are known so far to cause bitter rot on apple worldwide. Besides causing bitter rot, species such as C. limetticola, C. paranaense, C. melonis in CASC and C. fructicola in CGSC cause Glomerella leaf spot (GLS) of apple27. Although these two diseases are associated with the same fungal genus, differences in pathogenicity, morphology and cultural characteristics of species have been reported28.
Accurate identification of Colletotrichum species causing bitter rot is crucial due to potential species-level variation in pathogenicity-related characteristics. Identifying the causal agent(s) facilitates resistance breeding programs and determines the best control strategies for apple diseases29,30. Identification of Colletotrichum to the species level was traditionally reliant on host, cultural and morphological descriptions, as well as comparison of nuclear rDNA internal transcribed spacer (ITS1-5.8S-ITS2 = ITS) sequences31,32,33,34,35. However, these identification techniques are limited in their effectiveness as growth medium and temperature are known to cause variation in cultural and morphological characteristics, such as size and shape of conidia, colony growth rate and pigmentation of Colletotrichum isolates3,29,34. The ITS regions known as the barcode locus for fungi36,37, is considered insufficient to delimit species in the CGSC37,38,39.
While species delimitation using morphology and ITS-based phylogeny remains insufficient for resolution of Colletotrichum at the species level, multi-locus phylogenetic analyses have proven reliable in addressing challenges in the identification of Colletotrichum species3,10,40,41,42,43. In addition to ITS, loci such as glutamine synthetase (GS), glyceraldehyde-3-phosphate dehydrogenase (GAPDH), calmodulin (CAL), actin (ACT), chitin synthase (CHS–1), β-tubulin (TUB2), DNA lyase (APN2), and the intergenic region between DNA lyase and the mating type (Mat1-2) gene (ApMat) have been used to resolve various species in the CGSC3,26,41,44,45,46,47,48,49,50,51,52,53.
Accurate identification of Colletotrichum species causing bitter rot is a prerequisite to successfully manage this disease in apple production regions because different species of Colletotrichum respond differently to fungicides and vary in traits such as enzyme activity and pathogenicity17,54,55. Accordingly, characterizing fungicide sensitivity, enzyme activity and pathogenicity of species is of extreme importance for future research on control of bitter rot. The ability of Colletotrichum species to produce extracellular enzymes determines their pathogenicity and virulence capacity34. The variable levels of amylolytic, pectolytic, polymethylgalacturonase (PMG) and polygalacturonase (PG) activity was detected in Colletotrichum species associated with different plant diseases55,56,57,58,59.
Several studies showed variable fungicide sensitivity within and between the two main complexes causing bitter rot, CASC and CGSC25,60,61,62, which gives rise to challenges in bitter rot management24,34,63,64. For instance, a response variation among C. acutatum and C. gloeosporioides isolates collected in Kentucky was observed following the in vitro evaluation of fungicides17. Within each complex, The in vitro screening to determine the half maximal effective concentration (EC50) allow tracking the sensitivity of Colletotrichum species to fungicides and manage the risk of fungicide resistance as a rising problem65,66,67, which to some extent stems from applying ineffective concentrations of fungicides68.
In this study we aimed to (1) Identify the Colletotrichum species causing bitter rot of apple in New York; (2) Determine in vitro enzyme activity of Colletotrichum species; (3) Evaluate sensitivity of collected Colletotrichum species to several key fungicides; and (4) Compare the pathogenicity of these species on apple fruit.
Results
Screening of isolates
In 2017 and 2018, we collected a total of 400 Colletotrichum isolates from apple fruit in New York and other states. Isolates were morphologically screened for colony color, growth rate, sporulation capacity and conidial shape on PDA, and organized into two general morphological types. The first morphotype, comprising more than 60% of isolates, included isolates producing a distinct salmon to red colony color, fusiform spores, and slower growth rate on PDA. The second morphotype included two distinct groups: 12.5% isolates producing colonies with mycelia ranging from dominantly white to off-white with slightly light grey centers and rare sporulation; and 25% isolates with light to dark gray mycelium, cylindrical rounded spores and faster growth rate on PDA. The characteristics of isolates in the morphotype 1 and 2 were consistent with the descriptions of CASC and CGSC, respectively. Initially, 100 isolates from morphotypes 1 and 2, representing the morphological variation, geographical and apple cultivar range, were selected for species identification using ITS, TUB2 and GAPDH sequences. Facing challenges in species delimitation within CGSC, we increased the number of partially sequenced genes to eight for isolates belonging to CGSC, adding ACT, ApMat, CAL, GS and APN2, and reduced the number of isolates for identification and downstream analyses to 44 (19 from morphotype 1 and 25 from morphotype 2).
Multiplex PCR assay
An amplicon of approximately 349 bp was recovered using Colletotrichum-specific GAPDH primers GDF1/C-GAPDH-R, confirming that all isolates were Colletotrichum spp. The CAL gene primers specific to species complex confirmed that of the 44 isolates, 19 from morphotype 1 were members of the CASC and 25 from morphotype 2 were members of the CGSC (491 and 649 bp amplicons respectively; Supplementary Fig. S1). Amplified fragments of expected lengths representing the CASC and CGSC support49 that the GAPDH/CAL multiplex PCR approach is satisfactory at differentiating these two species complexes.
Phylogenetic analyses
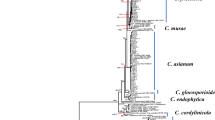
The ITS phylogeny concurred with the multiplex PCR assay in that Colletotrichum isolates collected in this study fell into two Colletotrichum species complexes: C. acutatum (19 isolates) and C. gloeosporioides (25 isolates) with high support (Supplementary Fig. S2). The C. acutatum phylogeny dataset included 85 taxa (including 19 isolates from this study) and 1278 characters consisting of three loci (ITS, TUB2 and GAPDH). Two C. orchidophilum isolates, CBS 119291, and CBS 632.80, were used as an outgroup. All five major C. acutatum clades42 were resolved with high support (BS ≥ 84, PP = 0.99; Fig. 1). Both Bayesian Inference (BI) and Maximum Likelihood (ML) analyses revealed that the 19 isolates collected in this study clustered with C. fioriniae as part of clade 3 of the CASC with full support (BS/PP: 100/1; Fig. 1) and are hereafter designated as C. fioriniae. We found that the majority of isolates in this study included in the CASC phylogentic analysis clustered with C. fioriniae type isolate CBS 128517, with high PP support (0.98), but lacking BS support ≥ 70. The remaining three isolates (ACFK3, ACFK6, ACFK205) fell outside of this group (Fig. 1), though remaining within the highly supported Clade 3 (C. fioriniae). ACFK3 and ACFK6 were placed well-within a different subclade with high BS support (85) but lacking PP support ≥ 0.90.While further analysis is required, we believe this separation of the isolates in this study may be similar to the previous fnding that the C. fioriniae clade is partitioned into two major subclades42.
Maximum-likelihood phylogeny inferred from ITS, TUB2 and GAPDH sequences from the Colletotrichum acutatum species complex. Colletotrichum isolates from this study are denoted with asterisks. The remaining taxa are reference isolates retrieved from NCBI. The phylogeny is rooted with C. orchidophilum (CBS 119291) and C. orchidophilum (CBS 632.80) as the outgroup. Bootstrap support values greater than 70 and posterior probabilities greater than 0.90 are shown on the branches (BS/PP). Type isolates are in bold font. Double hash marks indicate branch lengths shortened at least 2-fold to facilitate visualization. Scale bar represents the estimated number of substitutions per site.
The C. gloeosporioides phylogeny dataset included 201 taxa (including 25 isolates collected in this study and Coll940) and 4890 characters consisting of eight loci (ACT, ApMat, CAL, GAPDH, GS, APN2, ITS and TUB2). The outgroup included one member of the CASC, C. javanense CBS 144963, and two members of the C. boninense complex, C. boninense CBS 123755 and C. hippeastri ICMP17920.
The 26 isolates belonging to CGSC collected in this study from apple in New York, Virginia, and Pennsylvania, were found to group into three distinct clades, two of which represent previously described species within the CGSC. Twelve isolates, AFK17, AFK18, AFK22, AFK26, AFK28, AFK30 and AFK31 from New York, isolates AFK154 and PMAREC-1a from Virginia, and isolates PMKnsl-1, PMCMS-6760 and PMLynd-9a from Pennsylvania, grouped with the ex-type strain of C. chrysophilum with maximum support (BS/PP: 100/1; Fig. 2) and are hereafter designated as C. chrysophilum. To our knowledge, this is the first time that the C. chrysophilum species has been reported to cause bitter rot disease on apple. After C. fioriniae, C. chrysophilum was the second most abundant species causing bitter rot disease in New York.
Maximum-likelihood phylogeny inferred from eight loci (ACT, CAL, GAPDH, GS, ITS, ApMat, APN2 and TUB2) from the Colletotrichum gloeosporioides species complex. Colletotrichum isolates from this study are denoted with asterisks. The remaining taxa are reference isolates retrieved from NCBI. The phylogeny is rooted with C. javanense CBS 144963, C. boninense CBS 123755 and C. hippeastri ICMP17920 as the outgroup. Bootstrap support values greater than 70 and posterior probabilities greater than 0.90 are shown on the branches (BS/PP). Type isolates are in bold font. Double hash marks indicate branch lengths shortened at least 2-fold to facilitate visualization. Scale bar represents the estimated number of substitutions per site.
Two isolates, AFK156 and PMCrwn1 from Virginia, grouped with the ex-type strain of C. fructicola (BS/PP: 91/1; Fig. 2) and are hereafter designated as C. fructicola. Of the two CGSC isolates originally collected from peach in South Carolina49, RR12-1 was found to group with C. fructicola, as previously reported47. The second isolate, RR12-3, previously recognized as C. fructicola using a multi-locus analysis (CAL, GAPDH and TUB249), was found clustered within the fully supported C. chrysophilum clade in our eight-gene multi-locus analysis (Fig. 2). Further, CGSC member GA253, isolated from avocado in Israel69, which was previously identified as C. nupharicola using an ApMat phylogeny as well as a six-gene multi-locus analysis69, was found to cluster within the C. chrysophilum clade (Fig. 2). No isolates belonging to C. fructicola were identified from apple fruit in New York and Pennsylvania.
The remaining 9 isolates, AFKH109, AFK65, AFK220, AFK289, AFK408 and AFK423 from New York and PMEssl-10a, PMCMS-6751 and PMBrms-1 from Pennsylvania, formed a separate, distinct clade with maximum support and independent from any recognized species in the CGSC (BS/PP: 100/1; Fig. 2). This distinct clade included isolate Coll940, which was originally isolated from leaves of black walnut (Juglans nigra) in Oklahoma and had an uncertain placement based on nrITS, TUB2, APN2 and ApMat analyses53. We pursued further analyses to determine if this unique cluster represented a new, undescribed lineage in the CGSC. For phylogenetic models and partitioning schemes see Supplementary Table S1.
Species delimitation
All Colletotrichum isolates from apple were assigned to a lineage containing the ex-type of a previously described species using genealogical concordance phylogenetic species recognition approach (GCPSR) except for AFKH109, AFK65, AFK220, AFK289, AFK408, AFK423, PMEssl-10a, PMCMS-6751, and PMBrms-1. These isolates were strongly supported in the 8-locus concatenated analyses as monophyletic (BS = 100; PP = 1) and sister to C. fructicola, C. nupharicola and C. chrysophilum. Among the independent gene trees, these isolates were strongly supported as monophyletic in the ApMat (BS = 99; PP = 1), APN2 (BS = 100; PP = 1), and GS (BS = 92; PP = 1) phylogenies. These isolates were also inferred to be monophyletic in the ACT phylogeny, although with weak support in both the ML analysis (BS = 54) and BI analysis (PP = 0.85). While they were not monophyletic in the phylogenies inferred from TUB2, ITS, GAPDH and CAL, there was no strongly supported conflict in those trees. Our results are consistent with the criteria of GCPSR for recognizing these isolates as an independent lineage representing a novel species of Colletotrichum, named as C. noveboracense. Phylogenetic models and partitioning schemes used can be found in Supplementary Table S1.
Morphology characterization
We described morphological characteristics including colony color, conidial shape, measurements of colony growth rate and conidial length and width for several isolates of each Colletotrichum species causing apple bitter rot in this study (C. fioriniae, C. chrysophilum, C. fructicola and C. noveboracense). The isolates of C. fioriniae produced salmon to red conidial masses on 7-day-old cultures on PDA in both front and reverse sides and produced fusiform conidia after 10 days on PDA (Fig. 3a–c). Isolates belonging to C. chrysophilum initially presented colonies in white to light gray and progressively turned to dark grey in the center covered with predominantly black acervuli, producing orange conidial masses with longer incubation time. Cylindrical conidia with rounded ends developed after 10 days of incubation on PDA for this species (Fig. 3d–f). C. fructicola formed off-white to slightly gray aerial mycelium and yellowish to grey in reverse, developing cylindrical conidia with rounded ends after 10 days of incubation on PDA (Fig. 3g–i). Comparisons of conidial dimensions and shape, colony growth rates, as well as the description of colony color are presented in Table 1 Detailed morphological description for C. noveboracense is provided in the Taxonomy section.
Morphological characteristics (colony color and conidial shape) of Colletotrichum spp. isolated from bitter rot-infected apple fruit. Colony color of Colletotrichum fioriniae: (a) Front, (b) Reverse, (c) Conidial shape; colony color of Colletotrichum chrysophilum: (d) Front, (e) Reverse, (f) Conidial shape; Colony color of Colletotrichum fructicola (isolate AFK156 from Virginia): (g) Front, (h) Reverse, (i) Conidial shape.
TAXONOMY
Colletotrichum noveboracense F. Khodadadi, P.L. Martin, V.P. Doyle, & J.B. Gonzalez & S.G. Aćimović, sp. nov. MB 836581.
MycoBank MB 833232 (Figs. 4 and 5).
Etymology
The specific epithet is a combination of the long-established Latin name for New York (Noveboracum) state in the United States of America and the Latin -ensis, denoting the origin of the holotype.
Holotype
The United States of America: New York State, Hudson, from fruit lesion of Malus domestica cultivar Idared, July 2017, F. Khodadadi & S.G. Aćimović, BPI 911227.
Ex-holotype culture
CBS 146410; AFKH109.
Description
Growth rate on full strength PDA at 25 °C, 13.1 ± 1.3 mm/d (avg. ± std. dev.) and 11.9 ± 0.9 mm/d on ½ strength PDA. Colonies on PDA white with light gray toward the center, reverse white to pale off-white and slightly grey at the center. Aerial mycelium on PDA white to off-white and cottony. Colony on CMA nearly invisible. Acervuli not observed. Perithecia solitary to clustered on SNA and OMA, dark brown to black, globose to obpyriform; ascospores allantoid, light olive (13.1–)15.9–17.6–19.8(–22.4) × (3.8–)4.4–4.8–5.1(–5.5) μm, length-width ratio (2.7–)3.4–3.7–4.0(–4.6). Conidiophores hyaline, smooth-walled, aseptate, unbranched. Conidiogenous cells hyaline, smooth-walled, cylindrical to ampulliform, monophialidic, often extending percurrently to form new monophialides and conidiogenous loci. Conidia formed from conidiogenous cells, one-celled, smooth-walled, hyaline, and cylindrical, sometimes oblong, contents appearing granular with occasional oil droplets. Conidia on ½ strength PDA (12.19–)12.5–13.03–14.9(–15.6) × (4.6–)4.8-5.3–5.9(–6.4) μm (avg. 13.6 × 5.7 μm, n = 25), length/width ratio (1.9–)2–2.3–2.7(–2.8) (avg. 2.41, n = 25). Appressoria (hyphopodia) in slide cultures, single, or in groups, light to medium brown, smooth-walled, oval, often with undulate margin (4.47–) 5.5–8.6–11.7(–13.5) × (3.7–)4.9–5.5–6.1(–6.2) μm (avg. 8.8 ± 2 × 4.9 ± 0.6 μm, n = 25). Hyphal diameter 1.8–4.5 μm.
Habitat/host
Known from the states of New York and Pennsylvania, causing bitter rot on Malus domestica fruit and a single isolate from Oklahoma as a leaf endophyte on Juglans nigra53.
Diagnosis
Isolates of Colletotrichum noveboracense are strongly supported as monophyletic by the combined analysis of ACT, APN2, GS, CAL, ApMat, GAPDH, ITS and TUB2 and sister to C. nupharicola, C. chrysophilum and C. fructicola. C. noveboracense differs from C. nupharicola by having a faster growth rate on PDA as well as shorter and narrower conidia. C. nupharicola also differs in having an orange colony that turns black with age on PDA versus white to grey colony color for C. noveboracense. C. noveboracense differs from C. fructicola by having shorter conidia and lighter colonies on PDA and differs from C. chrysophilum by having a slower growth rate on PDA. Sequence data from ApMat, APN2, GS and ACT delimit C. noveboracense, but C. noveboracense could not be distinguished by sequences of GAPDH, CAL, TUB2 and ITS.
Additional specimens examined
USA. New York: Ulster County: on fruit of Malus domestica, Jul 2017, F. Khodadadi (AFK220, AFK408, AFK423, and AFK289); USA. New York: Colombia County: on fruit of Malus domestica, Jul 2017, F. Khodadadi (AFK65); USA. Pennsylvania: Adams County: on fruit of Malus domestica, late summer and fall of 2018, P. L. Martin (PMBrms-1); USA. Pennsylvania: Lehigh County: on fruit of Malus domestica, late summer and fall of 2018, P. L. Martin (PMCMS-6751); USA. Pennsylvania: Northumberland County: on fruit of Malus domestica, late summer and fall of 2018, P. L. Martin (PMEssl-10a); USA. Oklahoma: leaf endophyte of Juglans nigra, Cherokee County, 36.028725N, 95.185787W, June 10, 2010, V. Doyle Coll940.
Notes
A very low sporulation rate was observed among the isolates collected from New York. No sporulation was seen on PDA and ½ strength PDA except for few isolates including AFKH109 which sparsely produced conidia on OMA. However, isolates collected from Pennsylvania sporulated on ½ strength PDA and OMA.
Agar plate enzyme activity
All isolates belonging to C. noveboracense, C. fioriniae and C. chrysophilum showed lipolytic, amylolytic and cellulolytic activity after five days of incubation on PDA (Fig. 6). However, none of the isolates showed halos of degradation for the proteolytic activity on skimmed milk. All isolates evaluated in this study showed cellulolytic activity as a yellow halo around the colony in plates including CMC stained with Congo red and secured with NaCl. C. chrysophilum showed a significantly larger mean degradation halo of 8 mm in cellulolytic activity assay when compared to C. noveboracense and C. fioriniae (mean halo zone 6 and 6.5 mm, respectively) (Fig. 6a,b). Colletotrichum isolates of all three species produced hallo around their colonies indicating their ability to produce lipase. C. chrysophilum isolates exhibited significantly the lowest lipid degradation with the mean halo diameter of 17 mm compared to isolates of C. fioriniae and C. noveboracense, with the mean halo diameter of 23 and 27 mm, respectively (Fig. 6c,d). The screening of Colletotrichum isolates on starch agar plates showed that all species produced halo zones reflecting amylase activity after exposure to iodine. The smallest and largest halo sizes for amylase belonged to C. fioriniae and C. noveboracense isolates, with an average of 3 and 8 mm, respectively. However, no significant difference was observed between the mean degradation halo of C. chrysophilum and C. fioriniae (Fig. 6e,f).
Qualitative enzyme activity of Colletotrichum fioriniae, C. noveboracense and C. chrysophilum isolates using the agar plate method at 5 days after incubation. (a) Mean cellulase degradation halo of Colletotrichum isolates; (b) Yellow halo formed after exposure to Congo red and fixation with NaCl; (c) Mean lipid degradation halo of Colletotrichum isolates; (d) White and clear halo representing lipolytic activity; (e) Mean starch degradation halo of Colletotrichum isolates; (f) Clear zone around the colony after exposure to iodine indicating amylolytic activity. C. fioriniae represented with isolates ACFK165, ACFK10, ACFK11, ACFK12, ACFK14, ACFK145, ACFK15, ACFK16, ACFK25, ACFK29, ACFK299, ACFK4, ACFK5, ACFK8, ACFK9, ACFKCM-9, ACFK205, ACFK3 and ACFK6; C. noveboracense represented with isolates AFK220, AFK289, AFK408, AFK423, AFKH109 and AFK65; and C. chrysophilum represented with isolates AFK17, AFK28, AFK154, AFK18, AFK22, AFK26, AFK30 and AFK31. Different letters indicate significant differences between species based on Bonferroni Comparison Posttest (p ≤ 0.05). Error bars represent standard deviation.
Fungicide sensitivity
Isolates belonging to the three Colletotrichum species showed significantly lower sensitivity to natamycin (mean EC50 values ranged from 4 to 5 µg/ml) compared to the other fungicides (mean EC50 values less than 0.5 µg/ml) (Fig. 7). Relative to C. noveboracense and C. chrysophilum, C. fioriniae isolates exhibited greater sensitivity to difenoconazole (EC50 value of 0.09 µg/ml), pyraclostrobin (EC50 value of 0.04 µg/ml) and fludioxonil (EC50 value of 0.12 µg/ml), but had less sensitivity to thiabendazole and benzovindiflupyr with EC50 values of 0.4 and 0.3 µg/ml, respectively. With respect to the relative fungicide sensitivity of individual species within the CGSC, we found that C. noveboracense isolates had significantly higher EC50 values in response to the fungicide difenoconazole and fludioxonil compared to C. chrysophilum. While all members of the CGSC responded similarly to thiabendazole (mean EC50 of 0.2 µg/ml), C. chrysophilum isolates were significantly less sensitive to pyraclostrobin and benzovindiflupyr (mean EC50 values of 0.26 µg/ml) when compared to C. noveboracense with mean EC50 value of 0.17 µg/ml (Fig. 7).
Mean EC50 values (µg/ml) of Colletotrichum fioriniae (isolates ACFK165, ACFK10, ACFK11, ACFK12, ACFK14, ACFK145, ACFK15, ACFK16, ACFK25, ACFK29, ACFK299, ACFK4, ACFK5, ACFK8, ACFK9, ACFKCM-9, ACFK205, ACFK3 and ACFK6); Colletotrichum chrysophilum (isolates AFK17, AFK28, AFK154, AFK18, AFK22, AFK26, AFK30 and AFK31) and Colletotrichum noveboracense (isolates AFK220, AFK289, AFK408, AFK423, AFK65 and AFKH109) for fungicides difenoconazole (Difeno), thiabendazole (Tiaben), pyraclostrobin (Pyraclos), benzovindiflupyr (Benzovin), fludioxonil (Fludiox) and Natamycin (Natam). Colletotrichum species followed by the same letter were not significantly different based on Bonferroni Comparison Posttest (p ≤ 0.05). Error bars represent standard deviation.
Pathogenicity
All isolates caused the typical symptoms of bitter rot as light to dark brown and sunken circular lesions on apple fruit of cultivar ‘Honeycrisp’. To meet the requirements of Koch’s postulates, Colletotrichum isolates were recovered from inoculated apple fruit and re-identified. Symptoms did not develop on the apple fruit inoculated with agar plugs. In the comprehensive pathogenicity test of selected Colletotrichum isolates of each species, the average diameter of lesions varied between the two species complexes and among the species within the CGSC complex (Fig. 8). The average diameter of lesions caused by C. fioriniae isolates on apple fruit of ‘Fuji’ and ‘Gala’ was significantly smaller, 17.7 and 39.6 mm, respectively, compared to that of produced by C. chrysophilum and C. noveboracense (Fig. 8). However, in ‘Red Delicious’, ‘Golden Delicious’ and ‘Honeycrisp’, the average lesion diameter caused by C. fioriniae isolates were the same as that produced by C. chrysophilum isolates (53.2, 76.5 and 44.4 mm) but significantly larger than the lesions developed by C. noveboracense (29, 57, 23.5 mm) (Fig. 8).
Mean lesion diameter (mm) formed on apple fruit of cultivars ‘Fuji’, ‘Gala’, ‘Red Delicious’, ‘Golden Delicious’ and ‘Honeycrisp’ by Colletotrichum species 15 days after inoculation. Colletotrichum fioriniae was represented with isolates ACFK145, ACFK15, ACFK16, ACFK205, ACFK29 and ACFK25; Colletotrichum noveboracense was represented with isolates AFK220, AFK289, AFK408, AFK423, AFKH109 and AFK65; and Colletotrichum chrysophilum was represented with isolates AFK17, AFK28, AFK154, AFK18, AFK31 and AFK22. Colletotrichum species followed by the same letter were not significantly different based on Bonferroni Comparison Posttest (p ≤ 0.05). Error bars represent standard deviation.
Discussion
Effective control of plant diseases caused by Colletotrichum species and determination of host specificity and virulence factors are reliant on precise identification and accurate taxonomical delimitation of species boundaries. The assorting of Colletotrichum isolates recovered from apple fruit in this study to CASC and CGSC using a multiplex-PCR49, confirmed the reliability and affordability of this method to differentiate between these two species complexes. The ITS gene tree placed all the isolates in the CASC and CGSC with strong support, aligning with previous studies confirming the utility of ITS sequencing for classifying Colletotrichum isolates at the species complex level49,52. In addition, the placement of our isolates in this study into the CASC and CGSC supports previous evidence that species from these two species complexes are predominantly involved in causing apple bitter rot16,17,29,43. Although the application of ITS rDNA sequences to identify Colletotrichum species was used in studies in the 1990s70,71,72, ITS data are insufficient for identifying species in the CGSC39,40,73. The multi-locus analyses provided strong resolution and placed the Colletotrichum isolates causing bitter rot of apple in New York orchards in C. fioriniae clade from CASC and C. chrysophilum clade from CGSC. It also contributed to the identification of a new species in this study, C. noveboracense, causing apple bitter rot in New York and Pennsylvania.
In our study, C. fioriniae was the dominant species causing bitter rot on apple which is consistent with previous work in Kentucky where C. fioriniae was also the most abundant species17. C. fioriniae causes bitter rot on apple in the US, Korea and Croatia17,43,74,75. Seventy percent of isolates recovered from apple orchards in Arkansas, North Carolina and Virginia were identified as C. acutatum76, the taxon assigned to all Colletotrichum strains with acute conidia which later were assigned to CASC of over a dozen species42.
C. fructicola was reported as a causal agent of bitter rot of apple in the USA, Brazil, Korea and Uruguay24,43. However, in our work, C. fructicola was recovered only from symptomatic apple fruit received from Virginia, not from apple orchards in New York and Pennsylvania. C. fructicola was reported to represent the most biological and geographical diversity in the CGSC3. Its host range and distribution were reported from coffee berries in Thailand, peach in USA, avocado in Australia and apple in USA, Brazil and Korea, to name a few examples of the geographic and host diversity from which this species has been isolated3.
First, consisting of two strongly supported monophyletic subclades, C. ignotum was described as an endophyte of Genipa americana, Tetragastris panamensis and Theobroma cacao52. This species was later synonymized with C. fructicola3, with the ex-type of C. ignotum and C. fructicola nested within the same subclade. Later, it was determined that the second subclade within C. fructicola represented an independent evolutionary lineage and was described as C. chrysophilum51. We detected C. chrysophilum for the first time as pathogen on apple in New York and Pennsylvania. It ranks as the second most common species identified in apple orchards in New York, after C. fioriniae. In classification of Colletotrichum isolates causing anthracnose of peach, using CAL, GAPDH and TUB2, isolate RR12-3 clustered with C. fructicola reference strain ICMP 18645 with bootstrap value 9449. By adding five more partial gene sequences, RR12-3 was re-identified as C. chrysophilum in our phylogenetic analyses. In addition, we re-identified isolate GA253 as C. chrysophilum, that was previously identified as C. nupharicola69. These findings expand the known host range and geographic distribution of C. chrysophilum, which has been identified on cacao and genipa (Genipa americana; Panama52), fern (Terpsichore taxifolia; Puerto Rico53), avocado (Israel69), peach (South Carolina49) and banana (Brazil51).
Colletotrichum noveboracense was identified as a new species in Colletotrichum genus causing apple bitter rot disease in New York and Pennsylvania. The nine C. noveboracense isolates from New York and Pennsylvania, as well as a single endophytic isolate from Juglans nigra in Oklahoma, formed a distinct clade with high support. In our initial phylogenetic analyses using Bayesian inference, the isolates later attributed to C. noveboracense formed a distinct clade in a three-gene multi-locus analysis (ITS, TUB2, ApMat) with full support (BI PP 1.0). Additionally, in our initial Bayesian analysis of seven loci (ACT, TUB2, CAL, GAPDH, GS, ITS and ApMat) and other different combinations of loci, C. noveboracense was sister to C. nupharicola (PP = 0.95). C. nupharicola is easily distinguished within the CGSC in terms of morphology. This host-specific species has very slow growth on PDA and both the length and width of the conidia are much greater than other species in CGSC53,77. The morphological differences between C. noveboracense and C. nupharicola prompted us to expand the analysis to include a much larger dataset, include an additional locus (APN2) known to provide better resolution in CGSC53, and evaluate the new clade under GCPSR criteria78. This led us to identify these isolates as a strongly supported clade, distinct from other taxa in CGSC.
Fungi have developed a plethora of adaptive mechanisms, including extracellular enzyme secretion79. In our study, using the skimmed milk agar plates to detect proteolytic activity, it was impossible to observe visible halos of degradation for the assessed isolates. Several possibilities might contribute to the lack of visualization of proteolytic activity in Colletotrichum species. First, the ability and the level of protease gene expression in fungi could differ based on the nutrient source used in agar medium. Aspergillus isolates showed ability to produce proteases in agar medium supplemented with gelatin and casein as two different sources of protein80. Second, the difference in range of pH in culture medium also affects the proteolytic activities80. Finally, although the degradation halo indicating the protease activity in C. fructicola isolates was detected easily on skimmed milk agar plate55, sometimes the detection of the degradation zones is not possible unless a developing agent like bromocresol green dye is used81. The three Colletotrichum species in our study showed different level of amylolytic, cellulolytic and lipolytic activities. Prior to the present work, only a few studies investigated the enzyme activity of Colletotrichum isolates. C. fructicola isolates causing bitter rot and leaf spot on apple in Brazil were compared for their ability to produce amylolytic, pectolytic, lipolytic and proteolytic activity, and showed higher amylolytic and pectolytic activity compared to the isolates causing leaf spot, while they were the same in lipolytic and proteolytic activity55. Our results show species variation in enzymatic activity, which might be related to variable ability of different Colletotrichum species to effectively penetrate and spread in host plant tissues and the higher level of virulence55,82. This hypothesis must be further evaluated by investigating the contribution of these enzymes in pathogenicity.
To control bitter rot disease, applications of different fungicides are recommended. We observed statistically different fungicide sensitivity between and within the complexes in our study, which is supported by the previous studies on apple where the CASC was more tolerant to thiophanate-methyl, myclobutanil, trifloxystrobin, captan and demethylation inhibitor (DMI) fungicides in comparison to the CGSC17,83,84,85. In addition, within the CASC, isolates from apple orchards in Brazil showed different levels of sensitivity (25–83%) to mancozeb, thiophanate-methyl and azoxystrobin62. All the fungicides in our study showed high mycelial growth inhibition against all the species. Several studies support our findings. Benzovindiflupyr was highly active against mycelial growth of C. gloeosporioides, C. acutatum, C. cereale and C. orbiculare with EC50 values lower than 0.1 μg/ml86. Similar efficiency of this fungicide was seen in germination of conidia and germ tube growth of isolates with EC50 values 0.1 and 1 μg/ml86. Few isolates belonging to C. fioriniae, C. fructicola and C. siamense were sensitive to fludioxonil, and benzovindiflupyr with EC50 values < 0.1 μg/ml and <0.1 to 0.33 μg/ml, respectively87. In our study, in vitro toxicity of natamycin with an EC50 of 5 μg/ml against Colletotrichum species was significantly lower than that of the other fungicides with EC50 values ranging from 0.04 to 0.4 μg/ml. This is consistent with the previous work in which the toxicity of natamycin against mycelial growth of C. acutatum ranged from 0.5 to 1.9 μg/ml in EC50 values and was considerably lower compared to fludioxonil, azoxystrobin and cyprodinil85. Our data show strong in vitro activity of fungicides used in this study and likely would provide effective control of bitter rot in orchards or storages. Although the susceptibility profiles of Colletotrichum species against fungicides in apple orchards across the United States are limited, the efficacy of benzovindiflupyr and pyraclostrobin against bitter rot and GLS was reported in two recent trials in North Carolina88,89. Future studies should continue to validate the effectiveness of these and other fungicides against apple bitter rot.
In conclusion, three Colletotrichum species, C. fioriniae, C. chrysophilum and a novel species C. noveboracense, were identified as the causal agent of apple bitter rot in New York. Also, our study for the first time describes C. chrysophilum as the causal agents of bitter rot on apple in Virginia and Pennsylvania and C. noveboracense in Pennsylvania. We determined that the three species varied in pathogenicity, enzyme activity and fungicide sensitivity, which are important characteristics for bitter rot management. Our results highlight the significance of accurate identification of Colletotrichum species causing bitter rot in apple production regions in order to manage this economically important disease and secure the profitability of apple industry.
Methods
Sample collection and fungal isolation
In 2017 and 2018, apple fruit with typical symptoms of bitter rot disease were collected from a variety of apple cultivars in commercial and private apple orchards in the Hudson Valley area, New York (Table 2). Around 400 Colletotrichum isolates were obtained from apple fruit disinfected with 5% bleach for 2 min and rinsed with sterile distilled water. After removing the peel around the lesion, three small pieces of fruit pulp cut from the margin of each lesion were placed onto potato dextrose agar (PDA, Difco Laboratories, Detroit, MI, US). Plates were stored at 25 °C in the dark and colonies were purified by hyphal tip method.
Selection of isolates for molecular analysis
Besides sample collection from New York, we also received bitter-rot infected apple fruit from commercial orchards in Pickerel and Cana, Virginia and Thurmond, North Carolina in 2017 (provided by Virginia Tech Research Station, Winchester, VA) and isolates from Pennsylvania State University’s Fruit Research and Extension Center in Biglerville, PA, for identification and comparison. Moreover, two isolates (Cg)RR12-1 and (Cg)RR12-3 identified as C. fructicola recovered from peach fruit49 were received from School of Agricultural, Forest and Environmental Sciences, Clemson University, SC, for re-identification and comparison. All isolates collected in this study were placed into two morphological typesbased on growth rate, colony texture and color, sporulation and conidial shape on PDA. In total, 44 isolates (31 from New York and 13 from other states) from the two morphologically distinct typeswere selected based on geographical distribution and apple cultivar for identification to the species complex using ITS sequencing and multiplex PCR assay, and subsequently to the species level using multi-locus phylogenetic analyses (Table 2). Isolates collected from New York were used for enzyme activity assay, fungicide sensitivity and pathogenicity test.
Multiplex PCR assay
DNA from mycelia of 7-day-old Colletotrichum cultures was extracted using the DNeasy Plant Mini Kit (Qiagen Inc., Valencia, CA, USA) according to the manufacturer’s instructions. A multiplex PCR assay was performed to differentiate isolates of the CGSC and CASC by partial amplification of the GAPDH and CAL genes using primer pairs GDF1/C-GAPDH-R, CALF1/Cg-R, and CALF1/Ca-R149. PCR amplifications were carried out in 25 μL volumes containing 10X PCR buffer (includes 20 mM MgCl2) (Dream Taq, Thermo Fisher Scientific, Waltham, MA, US), 200 ng of gDNA, 2 mM dNTP, 1 u/μL of Taq DNA polymerase (Dream Taq, Thermo Fisher Scientific, Waltham, MA, US) and 10 μM of each primer using Applied Biosystems 2720 Thermo Cycler (Thermo Fisher Scientific, Waltham, MA, US). Cycling conditions were as follows: initial denaturation of 4 min at 94 °C, followed by 35 cycles of denaturation at 94 °C for 40 s, annealing at 56 °C for 40 s and extension at 72 °C for 1 min, with a final extension at 72 °C for 5 min. PCR products were visualized in 1% (w/v) agarose gels in 1xTAE buffer electrophoresed at 94.1 V for 45 min.
DNA extraction, PCR amplification and sequencing
DNA was extracted from mycelia of 7-day-old cultures of 44 isolates using the DNeasy Plant Mini Kit (Qiagen Inc, Valencia, CA, US). The partial nucleotide sequences were amplified from eight loci (ITS, CAL, TUB2, GAPDH, GS, ACT, ApMat and APN2), and from three loci (ITS, TUB2 and GAPDH) for isolates belonging to the CGSC and CASC, respectively (primer pairs described in Table 3). PCR reactions were performed in 30-μL volumes, including 200 ng of genomic DNA, 10X Dream Taq Green PCR buffer (includes 20 mM MgCl2) (Dream Taq, Thermo Fisher Scientific, Waltham, MA, US), 2 mM dNTP, 1 u/μL Taq DNA polymerase (Dream Taq, Thermo Fisher Scientific, Waltham, MA, US) and 10 μM of each primer. Cycling conditions were as follows3,52: initial denaturation of 4 min at 95 °C, followed by 35 cycles of denaturation at 95 °C for 30 s, 30 s annealing at 52 (ITS), 59 (CAL and GS), 55 (TUB2), 60 (GAPDH), 58 (ACT and ApMat) and 56 °C (APN2), extension of 45 s at 72 °C, final extension at 72 °C for 10 min. PCR products were examined in 1% (w/v) agarose gels in 1x TAE buffer electrophoresed at 94.1 V for 45 min. PCR product purification and Sanger sequencing were performed by Eurofins Genomics, Louisville, KY, USA.
Phylogenetic analyses
Consensus sequences were obtained by assembling forward and reverse reads using Geneious Pro v. 11.1.490. In order to confirm the placement of the isolates within species complexes, ITS sequences collected from 44 isolates and references from representatives of each of the nine major clades10 were used to construct the ITS phylogeny. To evaluate the placement of isolates at the species level, the C. acutatum phylogeny was constructed using three loci (ITS, TUB2 and GAPDH), whereas the C. gloeosporioides phylogeny was constructed using eight loci (ACT, CAL, GAPDH, GS, ITS, ApMat, APN2 and TUB2).
All three phylogenies were constructed using Bayesian inference (BI) and maximum likelihood (ML) approaches. Reference sequences (Supplementary Table S2) were downloaded from GenBank and aligned using MAFFT v7 on-line91,92, specifying the G-INS-i iterative refinement strategy. The alignments were trimmed using Gblocks v0.91b93 specifying the less stringent criteria. Model selection was conducted using PartitionFinder 294, specifying the Greedy algorithm95, “MrBayes” models for BI using PhyML96, or “all” models for ML analysis using RAxML97 and the AICc metric. Bayesian inference was conducted using MrBayes v3.2.698 implementing the BEAGLE library99. For the ITS and C. acutuatum phylogenies posterior probabilities were estimated using two runs of 2,000,000 generations with 25% burn-in. For the C. gloeosporioides phylogeny, 10,000,000 generations were used. ML analysis was conducted using RAxML v8.2.1297 specifying 1k bootstrap replications. Clade support was determined by mapping the bootstrap replicates onto the ML best trees using the DendroPy v4.4.0 program SumTrees v4.4.0100. BI and ML clade support values were mapped onto the ML best trees using the DendroPy v4.4.0 program SumLabels v2.0.0. Model selection and Bayesian and ML analyses were conducted using the CIPRES Science Gateway101. Data handling/formatting was facilitated using AliView v1.26102, Mesquite v3.51103, SequenceMatrix v1.8104 and Geneious v11.1.3 (https://www.geneious.com). Phylogenetic trees were plotted in R v3.4.3105 using the Ape package106 in the RStudio v1.1.383 environment107 and finished using Adobe Illustrator 2020. Sequences generated in this study were deposited in GenBank (Table 2) and the taxonomic novelty in MycoBank. The alignment files and trees were deposited in TreeBase (http://purl.org/phylo/treebase/phylows/study/TB2:S25647).
Species delimitation
Initial phylogenetic analyses revealed that several CGSC isolates collected in this study, in addition to isolate Coll94053, clustered together with high support and were distinct from any clade containing the ex-type of any previously described species, suggesting that these isolates may represent a novel lineage. In order to determine whether this new clade formed a distinct phylogenetic lineage, we applied GCPSR78. In this approach, a clade is determined to represent an independent evolutionary lineage if the clade satisfies one of two criteria: genealogical concordance or nondiscordance. The genealogical concordance criterion is satisfied if the clade is found well-supported (e.g. both ML and BI analysis ≥70% and ≥0.95, respectively) in most individual gene trees. The nondiscordance criterion is satisfied if the clade is found well-supported in at least one gene tree and members not found strongly supported in contradictory placement (e.g. clustering with the type isolate of another species) in any other individual gene trees.
To apply the GCPSR approach, individual gene trees were constructed for each of the eight genes used in the multi-locus C. gloeosporioides phylogeny. Evolutionary model selection and gene tree constructed were as described above, except that 5,000,000 generations were used to infer posterior probabilities for the Bayesian approach. Placement of clade members in each Bayesian and ML tree were evaluated for each individual gene tree.
Morphological characterization
Colony color, growth rate, conidial shape, length and width of Colletotrichum spp. in this study was evaluated by transferring 4-mm diameter plugs from the periphery of 5-day-old cultures, grown at 25 °C in dark, onto PDA and ½ strength PDA. Colony color was described after 7 days of incubation on PDA at 25 °C in dark. Colony growth rate was determined by measuring the colony diameter of each isolate grown on PDA daily over the course of 7 days at 25 °C in dark. To study the morphology of isolates belonging to the novel species, slide culture technique108 was used to induce the isolates to produce appressoria. Synthetischer nahrstoffarmer agar (SNA i.e. synthetic nutrient-poor medium)109 and oatmeal agar (OMA)110 were used to induce sporulation.
Microscopic observations, with 25 measurements per each structure, were viewed with an Olympus BX51 microscope (Olympus Corporation of the Americas, Center Valley, PA, US) using the differential interference contrast (DIC) setting. Statistical analysis was conducted by one-way analyses of variance (ANOVA) using Graph Pad Prism software v5 (San Diego, CA, U.S.A).
Agar-plate enzyme activity
To perform the qualitative enzyme activity, isolates were grown on PDA at 25 °C for 7 days in the dark. For lipolytic and proteolytic activities, we transferred a mycelial plug from the growing part of each colony onto peptone agar medium (10 g peptone, 5 g NaCl, 0.1 g CaCl2 2H2O, 15 g agar, pH 6.0) supplemented with 1% Tween 20111 and onto PDA containing 1% soluble skim milk112, respectively. After five days of incubation at 25 °C in dark, the size of the clear zone indicating lipolytic and proteolytic activity around each colony was measured in millimeters (mm) using a caliper. For amylolytic activity, isolates were transferred to starch hydrolysis agar medium (pH 7) and kept at 25 °C for 5 days in dark111. After flooding with 1 ml of Gram Iodine solution, the clear halo around each colony was measured. Isolates were cultured on PDA supplemented with 0.5% carboxy-methylcellulose (CMC) for 5 days at 25 °C in dark for cellulolytic activity. The plates were treated with 1% Congo red solution and shaken for 15 min. After removing Congo red, cultures were treated with 1 M NaCl and shaken for 15 min. Subsequently, clear zones indicating cellulolytic activity were measured113. Three replicates per each isolate was used. Data were analyzed by one-way ANOVA with Graph Pad Prism software v5 (GraphPad Software, San Diego, CA, US).
Fungicide sensitivity
We evaluated sensitivity of Colletotrichum isolates to the technical grade of fungicides pyraclostrobin (Merivon, Pristine, BASF Corporation), difenoconazole (Inspire Super, Syngenta Crop Protection), benzovindiflupyr (Aprovia, Syngenta Crop Protection), thiabendazole (Mertect 340-F, Syngenta Crop Protection), fludioxonil (Scholar, Syngenta Crop Protection) and bio-fungicide natamycin, by using colony growth inhibition assays. We selected these active ingredients as they are registered in the US by the Environmental Protection Agency (EPA) for application in apple orchards or storages. Each isolate was sub-cultured on PDA and grown at 25 °C for 5 days in the dark. Three-mm mycelial disks cut from actively growing parts of each colony were transferred to PDA plates supplemented with pyraclostrobin and thiabendazole at 0, 0.0001, 0.001, 0.01, 0.1, 0.2, 0.5, 1 and 10 μg/ml; difenoconazole at 0, 0.0001, 0.01, 0.02, 0.05, 0.1, 0.2, 0.5, 1 and 10 μg/ml; benzovindiflupyr at 0, 0.0001, 0.001, 0.01, 0.1, 0.2, 0.5, 1, 10 and 20 μg/ml, and fludioxonil at 0, 0.0001, 0.01, 0.03, 0.1, 0.2, 1, 10 and 20 μg/ml. All fungicides were dissolved in acetone. Natamycin was dissolved in methanol and used at 0, 0.2, 1, 2.5, 5, 7.5, 10, 20, 40 μg/ml. Each concentration for each fungicide was replicated five times and the experiment was performed twice. To calculate the EC50 values, mean colony diameter and growth rate of each isolate were measured after 5 days incubation at 25 °C in dark114. The data were fit to a sigmoidal dose-response curve and EC50 values were determined by nonlinear regression using Graph Pad Prism software v5 for Windows OS (GraphPad Software, San Diego, CA, US). Mean EC50 of each fungicide for all isolates was compared using two-way ANOVA with Bonferroni Comparison Posttest using GraphPad Prism v5.
Pathogenicity assay
Pathogenicity of all isolates was first tested on apple fruit of cultivar ‘Honeycrisp’ to reproduce bitter rot symptoms. Later, six Colletotrichum isolates from each species were inoculated on the apple fruit of cultivars ‘Golden Delicious’, ‘Honeycrisp’, ‘Red Delicious’, ‘Fuji’ and ‘Gala’ obtained from a grocery store and washed with detergent and water to ensure that no fungicide residues remain on the surface. Three fruit per each cultivar were disinfected for 2 min in 5% bleach, rinsed twice with sterile distilled water and then wounded with a 3-mm corkborer43. Two opposite sides of each fruit were inoculated with 3-mm mycelial plugs of each isolate with aerial mycelia facing the flesh. Control apple fruit received uninoculated agar plugs. Plastic boxes containing inoculated apple fruit placed on moist paper towels were incubated at 25 °C in the dark. Lesion diameter was measured 15 days after inoculation . Data were analyzed by two-way ANOVA with Bonferroni Posttest using Graph Pad Prism software v5 (GraphPad Software, San Diego, CA, US). P-values ≤ 0.05 were considered significant. To fulfill Koch’s postulates, strains were re-isolated and morphologically re-identified.
Data availability
Alignments and tree files generated during the current study are available in the TreeBase (Access: http://purl.org/phylo/treebase/phylows/study/TB2:S25647). All sequence data are available in NCBI GenBank following the accession numbers in the manuscript.
Change history
24 November 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Freeman, S. & Rodriguez, R. Differentiation of Colletotrichum species responsible for anthracnose of strawberry by arbitrarily primed PCR. Mycol. Res. 99, 501–4 (1995).
Jayawardena, R. S. et al. Notes on currently accepted species of Colletotrichum. Mycosphere. 7, 1192–1260 (2016).
Weir, B. S., Johnston, P. R. & Damm, U. The Colletotrichum gloeosporioides species complex. Stud. Mycol. 73, 115–180 (2012).
Bailey, J. A., Jeger, M. J. & British Society for Plant Pathology. Colletotrichum. Biology, pathology and control. (British Society for Plant Pathology, London, 1992).
Dean, R. et al. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 13, 414–430 (2012).
Sutton, T. B. Compendium of apple and pear diseases. 15–16 (The American Phytopathological Society, 1990).
Freeman, S. Occurrence and identification of Colletotrichum acutatum responsible for strawberry anthracnose in Israel. (Abstr.). Phytoparasitica. 24, 137 (1996).
Damm, U., Woudenberg, J. H. C., Cannon, P. F. & Crous, P. W. Colletotrichum species with curved conidia from herbaceous hosts. Fungal Divers. 39, 45–87 (2009).
Kim, W. G., Hong, S. K., Choi, H. W. & Lee, Y. K. Occurrence of anthracnose on highbush blueberry caused by Colletotrichum species in Korea. Mycobiol. 37, 310–2 (2009).
Cannon, P. F., Damm, U., Johnston, P. R. & Weir, B. S. Colletotrichum – current status and future directions. Stud. Mycol. 73, 181–213 (2012).
Dzhangaliev, A. D. The wild apple tree of Kazakhstan. Hort. Rev. 29, 63–371 (2003).
Hancock, J. F., Luby, J. J., Brown, S. K. & Lobos, G. A. Apples. (ed. Hancock, J. F.) Temperate fruit crop breeding: germplasm to genomics. 1–37 (Springer Science and Business Media, Berlin, Germany, 2008).
Apples|Agricultural Marketing Resource Center – AgMRC, https://www.agmrc.org/commodities-products/fruits/apples (2019).
Noncitrus Fruits and Nuts 2018 Summary (June 2019) USDA, National Agricultural Statistics Service, https://www.nass.usda.gov/Publications/Todays_Reports/reports/ncit0619.pdf (2018).
USDA/NASS 2018 State Agriculture Overview for New York, https://www.nass.usda.gov/Quick_Stats/Ag_Overview/stateOverview.php?state=NEW%20YORK (2018).
Peres, N. A., Timmer, L. W., Adaskaveg, J. E. & Correll, J. C. Lifestyles of Colletotrichum acutatum. Plant Dis. 89, 784–796 (2005).
Munir, M., Amsden, B., Dixon, E., Vaillancourt, L. & Gauthier, N. A. W. Characterization of Colletotrichum species causing bitter rot of apple in Kentucky orchards. Plant Dis. 100, 2194–2203 (2016).
Iungerman, K. Prior high heat stress opened pathways for bitter pit entry. Tree Fruit News, https://rvpadmin.cce.cornell.edu/pdf/enych_newsletter/pdf44_pdf.pdf (2013).
Aćimović, S. G. et al. Summary of research and extension activities Hudson Valley Research Laboratory 2016–2017, https://cpb-use1.wpmucdn.com/blogs.cornell.edu/dist/f/3191/files/2018/02/2016-2017-Biannual-HVRL-Report-Draft.review.Jentsch.Final-11uliox.pdf (2018).
Mertz, L. Bitter rot spotting in Northeast and Midwest. Good Fruit Grower, https://www.goodfruit.com/bitter-rot-spotting/ (2019).
Gauthier, N. W. et al. A profile of commercial apple production in Kentucky 2017, https://ipmdata.ipmcenters.org/documents/cropprofiles/KY_Apple_CropProfile.pdf, Google Scholar (2017).
McCulloch, M. J., Gauthier, N. W. & Vaillancourt, L. J. First report of bitter rot of apple caused by a Colletotrichum sp. in the C. kahawae clade in Kentucky. Plant Dis. 104, 289 (2019).
Sato, T. & Moriwaki, J. Re-classification of the Colletotrichum acutatum group A2 and A4 and re-identification of strains belonging to the groups in Japan. Plant Protec. 67, 367–372 (2013).
Velho, A. C., Alaniz, S., Casanova, L., Mondino, P. & Stadnik, M. L. New insight into the characterization of Colletotrichum species associated with apple diseases in southern Brazil and Uruguay. Fungal. Biol. 30, 1–16 (2015).
Wang, B., Li, B. H., Dong, X. L., Wang, C. X. & Zhang, Z. F. Effects of temperature, wetness duration, and moisture on the conidial germination, infection, and disease incubation period of Glomerella cingulata. Plant Dis. 99, 249–256 (2015).
Yokosawa, S., Eguchi, N., Ken-ichi, K. K. I. & Sato, T. Phylogenetic relationship and fungicide sensitivity of members of the Colletotrichum gloeosporioides species complex from apple. J. Gen. Plant Pathol. 83, 291–298 (2017).
Moreira, R. R., Peres, N. A. & May De Mio, L. L. Colletotrichum acutatum and C. gloeosporioides species complexes associated with apple in Brazil. Plant Dis. 103, 268–275 (2019).
Taylor, J. A. necrotic leaf blotch and fruit rot of apple caused by strain of Glomerella cingulata. Phytopathol. 61, 221–224 (1971).
González, E., Sutton, T. B. & Correll, J. C. Clarification of the etiology of Glomerella leaf spot and bitter rot of apple caused by Colletotrichum spp. based on morphology and genetic, molecular, and pathogenicity tests. Phytopathol. 96, 982–92 (2006).
Silva, D. D. D. et al. Identification, prevalence and pathogenicity of Colletotrichum species causing anthracnose of Capsicum annuum in Asia. IMA Fungus. 10, 8 (2019).
Sutton, B. C. The coelomycetes. Fungi imperfecti with pycnidia, acervuli and stromata. (Commonwealth Mycological Institute, Kew, England, 1980).
Freeman, S., Katan, T. & Shabi, E. Characterization of Colletotrichum species responsible for anthracnose diseases of various fruit. Plant Dis. 82, 596–605 (1998).
Copes, W. E. & Thomson, J. L. Survival analysis to determine the length of the incubation period of Camellia twig blight caused by Colletotrichum gloeosporioides. Plant Dis. 92, 1177–1182 (2008).
Cai, L. et al. A polyphasic approach for studying. Colletotrichum. Fungal Divers. 39, 183–204 (2009).
Guo, M., Pan, Y. M., Dai, Y. L. & Gao, Z. M. First report of brown blight disease caused by Colletotrichum gloeosporioides on Camellia sinensis in Anhui Province, China. Plant Dis. 98, 284 (2014).
Schoch, C. L. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc. Natl. Acad. Sci. USA 109, 6241–6246 (2012).
Hyde, K. D. et al. Incorporating molecular data in fungal systematics: a guide for aspiring researchers. Curr. Res. Environ. Appl. Mycol. 3, 1–32 (2013).
Sutton, B. C. The genus Glomerella and its anamorph Colletotrichum. (ed. Bailey, J. A. & Jeger, M. J.) Colletotrichum: biology, pathology and control. 1–26 (CAB International: Wallingford, 1992).
Gazis, R., Rehner, S. & Chaverri, P. Species delimitation in fungal endophyte diversity studies and its implications in ecological and biogeographic inferences. Mol. Ecol. 20, 3001–3013 (2011).
Crouch, J. A., Clarke, B. B. & Hillman, B. I. What is the value of ITS sequence data in Colletotrichum systematics and species diagnosis? A case study using the falcate-spored graminicolous Colletotrichum group. Mycologia. 101, 648–56 (2009).
Prihastuti, H., Cai, L., Chen, H., McKenzie, E. H. C. & Hyde, K. D. Characterization of Colletotrichum species associated with coffee berries in northern Thailand. Fungal Divers. 39, 89–109 (2009).
Damm, U., Cannon, P. F., Woudenberg, J. H. & Crous, P. W. The Colletotrichum acutatum species complex. Stud. mycol. 73, 37–113 (2012).
Parka, M. S., Kimb, B. R., Parkb, I. H. & Hahmb, S. S. First report of two Colletotrichum species associated with bitter rot on apple fruit in Korea – C. fructicola and C. siamense. Mycobiol. 46, 154–158 (2018).
Wikee, S. et al. Colletotrichum species from Jasmine (Jasminum sambac). Fungal Divers. 46, 171–182 (2011).
Fu, D. D. et al. Colletotrichum fructicola, first record of bitter rot of apple in China. Mycotaxon. 126, 23–30 (2013).
Huang, F. et al. Colletotrichum species associated with cultivated citrus in China. Fungal Divers. 61, 61–74 (2013).
Lima, N. B. et al. Five Colletotrichum species are responsible for mango anthracnose in northeastern Brazil. Fungal Divers. 61, 75–88 (2013).
Sharma, G., Kumar, N., Weir, B. S., Hyde, K. D. & Shenoy, B. D. The ApMat marker can resolve Colletotrichum species: a case study with Mangifera indica. Fungal Divers. 61, 117–38 (2013).
Hu, M. J., Grabke, A. & Schnabel, G. Investigation of the Colletotrichum gloeosporioides species complex causing anthracnose fruit rot of peach in South Carolina. Plant Dis. 99, 797–805 (2015).
Liu, F. et al. Unravelling Colletotrichum species associated with Camellia: employing ApMat and GS loci to resolve species in the C. gloeosporioides complex. Persoonia. 35, 63–86 (2015).
Vieira, W. A. S. et al. The impact of phenotypic and molecular data on the inference of Colletotrichum diversity associated with Musa. Mycologia. 109, 912–934 (2017).
Rojas, E. I. et al. Colletotrichum gloeosporioides sl associated with Theobroma cacao and other plants in Panama: multilocus phylogenies distinguish hostassociated pathogens from asymptomatic endophytes. Mycologia. 102, 1318–1338 (2010).
Doyle, V. P., Oudemans, P. V., Rehner, S. A. & Litt, A. Habitat and host indicate lineage identity in Colletotrichum gloeosporioides s. l. from wild and agricultural landscapes in North America. Plos One. 8, 62394 (2013).
Lima, N. B., Lima, W. G., Tovar-Pedraza, J. M., Michereff, S. J. & Câmara, M. P. S. Comparative epidemiology of Colletotrichum species from mango in northeastern Brazil. Europ. J. Plant Pathol. 141, 697–688 (2014).
Velho, A. C., Mondinob, P. & Stadnika, M. J. Extracellular enzymes of Colletotrichum fructicola isolates associated to apple bitter rot and Glomerella leaf spot. Mycology. 2, 145–154 (2018).
Yakoby, N. et al. Colletotrichum gloeosporioides pelB is an important virulence factor in avocado fruit-fungus interaction. Mol. Plant Microb. In. 14, 988–995 (2001).
Wattad, C., Freeman, S., Dinoor, A. & Prusky, D. A nonpathogenic mutant of Colletotrichum magna is deficient in extracellular secretion of pectate lyase. Mol. Plant Microb. In. 8, 621–626 (1995).
Raynaud, C., Etienne, G., Peyron, P., Lanéelle, M. A. & Daffé, M. Extracellular enzyme activities potentially involved in the pathogenicity of Mycobacterium tuberculosis. Microbiol. 144, 577–87 (1998).
Ramos, A. M., Gally, M., García, M. C. & Levin, L. Pectinolytic enzyme production by Colletotrichum truncatum, causal agent of soybean anthracnose. Rev. Iberoam. Micol. 27, 186–190 (2010).
Mondal, S., Bhatia, A., Shilts, T. & Timmer, L. Baseline sensitivities of fungal pathogens of fruit and foliage of citrus to azoxystrobin, pyraclostrobin and fenbuconazole. Plant Dis. 89, 1186–94 (2005).
Kenny, M., Galea, V. & Price, T. Effect of fungicides in vitro and on detached berries on control of coffee berry anthracnose caused by Colletotrichum acutatum and C. gloeosporioides. Plant Prot. Q. 27, 59–63 (2012).
Moreira, R. R., Hamada, N. A., Peres, N. A. & May De Mio, L. L. Sensitivity of the Colletotrichum acutatum species complex from apple trees in Brazil to Dithiocarbamates, Methyl Benzimidazole Carbamates, and Quinone outside inhibitor fungicides. Plant Dis. 103, 2569–2576 (2019).
Bernstein, B., Zehr, E., Dean, R. & Shabi, E. Characteristics of Colletotrichum from peach, apple, pecan, and other hosts. Plant Dis. 79, 478–82 (1995).
Maymon, M., Zveibil, A., Pivonia, S., Minz, D. & Freeman, S. Identification and characterization of benomyl-resistant and-sensitive populations of Colletotrichum gloeosporioides from Statice (Limonium spp.). Phytopathol. 96, 542–8 (2006).
Freeman, S., Nizani, Y., Dotan, S., Even, S. & Sando, T. Control of Colletotrichum acutatum in strawberry under laboratory, greenhouse and field conditions. Plant Dis. 81, 749–52 (1997).
Vega, B. & Dewdney, M. Distribution of QoI resistance in populations of tangerine infecting Alternaria alternata in Florida. Plant Dis. 98, 67–76 (2013).
Oliver, C., Chair, N. M., Baudoin, A., Bush, E. & Yoder, K. Investigation of wine grape cultivar and cluster developmental stage susceptibility to grape ripe rot caused by two fungal species complexes, Colletotrichum gloeosporioides, and C. acutatum, and the evaluation of potential controls. PhD thesis, Virginia Polytechnic Institute and State University, VA (2016).
Beckerman, J., Sundin, G. & Rosenberger, D. Do some IPM concepts contribute to the development of fungicide resistance? Lessons learned from the apple scab pathosystem in the United States. Pest Manag. Sci. 71, 331–42 (2015).
Sharma, G., Maymon, M. & Freeman, S. Epidemiology, pathology and identification of Colletotrichum including a novel species associated with avocado (Persea americana) anthracnose in Israel. Sci. Rep. 7, 15839 (2017).
Mills, R. R., Sreenivasaprasad, S. & Brown, A. E. Detection and differentiation of Colletotrichum gloeosporioides isolates using PCR RAPD bind patterns of Colletotrichum isolates from banana from Australia. FEMS Microbiol. Lett. 98, 137–144 (1992).
Sherriff, C. et al. Ribosomal DNA sequence analysis reveals new species groupings in the genus Colletotrichum. Experiment. Mycol. 18, 121–138 (1994).
Sreenivasaprasad, S., Mills, P. R., Meehan, B. & Brown, A. E. Phylogeny and systematics of 18 Colletotrichum species based on ribosomal DNA sequences. Genome. 39, 499–512 (1996).
Du, M., Schardl, C. L., Nuckles, E. M. & Vaillancourt, L. J. Using mating-type gene sequences for improved phylogenetic resolution of Colletotrichum species complexes. Mycologia. 97, 641–658 (2005).
Ivic, D., Voncina, D., Sever, Z., Simon, S. & Pejic, I. Identification of Colletotrichum species causing bitter rot of apple and pear in Croatia. J. Phytopathol. 161, 284–286 (2013).
Kou, L. P., Shaanxi, Y., Gaskins, V., Luo, Y. G. & Jurick, W. M. First report of Colletotrichum fioriniae causing postharvest decay on ‘Nittany’ apple fruit in the United States. Plant Dis. 98, 993.2–993.2 (2014).
Shi, Y., Correll, J. C. & Guerber, J. C. Frequency of Colletotrichum species causing bitter rot of apples in the southeastern United States. Plant Dis. 80, 692–696 (1996).
Johnson, D. A., Carris, L. M. & Rogers, J. D. Morphological and molecular characterization of Colletotrichum nymphaeae and C. nupharicola sp. nov. on water-lilies (Nymphaea and Nuphar). Mycol. Res. 101, 641–649 (1997).
Dettman, J. R., Jacobson, D. J. & Taylor, J. W. A multilocus genealogical approach to phylogenetic species recognition in the model eukaryote Neurospora. Evolution. 57, 2703–2720 (2003).
Monod, M. & Borg-on Zepelin, M. Secreted proteinases and other virulence mechanisms of Candida albicans. Chem. Immunol. 81, 114–128 (2002).
Díaz, G. V., Zapata, P. D., Villalba, L. L. & Fonseca, M. I. Evaluation of new xylanolytic-producing isolates of Aspergillus from Misiones subtropical rainforest using sugarcane bagasse. Arab. J. Basic Appl. Sci. 26, 292–301 (2019).
Vijayaraghavan, P., Gnana, S. & Vincent, P. A simple method for the detection of protease activity on agar plates using bromocresol green dye. J. Biochem. Technol. 4, 628–630 (2013).
Cox, G. M. et al. Extracellular phospholipase activity is a virulence factor for Cryptococcus neoformans. Mol. Microbiol. 39, 166–175 (2001).
Peres, N., Souza, N., Zitko, S. & Timmer, L. Activity of benomyl for control of postbloom fruit drop of citrus caused by Colletotrichum acutatum. Plant Dis. 86, 620–4 (2002).
Chen, S. N., Luo, C. X., Hu, M. J. & Schnabel, G. Sensitivity of Colletotrichum species, including C. fioriniae and C. nymphaeae, from peach to demethylation inhibitor fungicides. Plant Dis. 100, 2434–2441 (2016).
Haack, S. E., Ivors, K. L., Holmes, G. J., Förster, H. & Adaskaveg, J. E. Natamycin, a new biofungicide for managing crown rot of strawberry caused by QoI-resistant Colletotrichum acutatum. Plant Dis. 102, 1687–1695 (2018).
Zhen, I. F., Hu, M., Li, X. & Schnabel, G. Efficacy of SDHI fungicides, including benzovindiflupyr, against Colletotrichum species Hideo. SCI., https://doi.org/10.1002/ps.4216. (wileyonlinelibrary.com) (2016).
Chechi, A., Stahlecker, J., Dowling, M. E. & Schnabel, G. Diversity in species composition and fungicide resistance profiles in Colletotrichum isolates from apples. Pestic. Biochem. Physiol. 158, 18–24 (2019).
Villani, S. M., Johnson, K. A., Kreis, R. A. & Justus, C. Evaluation of fungicides for the management of Glomerella leaf spot and bitter rot on ‘Gala’ apple in NC, 2017. PDMR. 12, PF025 (2018).
Villani, S. M., Johnson, K. A., Kreis, R. A. & Justus, C. Evaluation of fungicides for the management of Glomerella leaf spot and bitter rot on ‘Gala’ apple in NC, 2018. PDMR. 12, PF025 (2019).
Kearse, M. et al. Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics. 28, 1647–1649 (2012).
Katoh, K., Rozewicki, J. & Yamada, K. D. MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinformatics. 20, 1160–1166 (2019).
Kuraku, S., Zmasek, C. M., Nishimura, O. & Katoh, K. aLeaves facilitates on-demand exploration of metazoan gene family trees on MAFFT sequence alignment server with enhanced interactivity. Nucleic Acids. Res. 41, W22–W28 (2013).
Castresana, J. Selection of conserved blocks from multiple alignments for their use in phylogenetic analysis. Mol. Biol. Evol. 17, 540–552 (2000).
Lanfear, R., Frandsen, P. B., Wright, A. M., Senfeld, T. & Calcott, B. PartitionFinder 2: new methods for selecting partitioned models of evolution for molecular and morphological phylogenetic analyses. Mol. Biol. Evol. 34, 772–773 (2016).
Lanfear, R., Calcott, B., Ho, S. Y. & Guindon, S. PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Mol. Biol. Evol. 29, 1695–1701 (2012).
Guindon, S. et al. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst. Biol. 59, 307–321 (2010).
Stamatakis RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 30, 1312–1313 (2014).
Ronquist, F. et al. MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 61, 539–542 (2012).
Ayres, D. L. et al. BEAGLE: an application programming interface and high-performance computing library for statistical phylogenetics. Syst. Biol. 61, 170–173 (2012).
Sukumaran, J. & Holder, M. T. DendroPy: a Python library for phylogenetic computing. Bioinformatics. 26, 1569–1571 (2010).
Miller, M. A., Pfeiffer, W. & Schwartz, T. Creating the CIPRES Science Gateway for inference of large phylogenetic trees. Gateway Computing Environments Workshop (GCE) (2010).
Larsson, A. AliView: a fast and lightweight alignment viewer and editor for large datasets. Bioinformatics. 30, 3276–3278 (2014).
Madison, W. P. & Maddison, D. R. Mesquite: a modular system for evolutionary analysis. Version 3.51, http://mesquiteproject.org (2018).
Vaidya, G., Lohman, D. J. & Meier, R. SequenceMatrix: concatenation software for the fast assembly of multi‐gene datasets with character set and codon information. Cladistics. 27, 171–180 (2011).
R Core Team R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, http://www.R-project.org/ (2017).
Paradis, E., Claude, J. & Strimmer, K. APE: analyses of phylogenetics and evolution in R language. Bioinformatics. 20, 289–290 (2004).
RStudio Team RStudio: Integrated Development for R. RStudio, Inc., Boston, MA, http://www.rstudio.com/ (2016).
Johnston, P. R. & Jones, D. Relationships among Colletotrichum isolates from fruit-rots assessed using rDNA sequences. Mycologia. 89, 420–430 (1997).
Nirenberg, H. I. Untersuchungen über die morphologische und biologischeDifferenzierung in der Fusarium-Sektion Liseola. Mitteilungen aus der Biologischen Bundesanstalt für Land- und Forstwirtschaft Berlin-Dahlem. 169, 1–117 (1976).
Crous, P. W., Verkleij, G. J. M., Groenewald, J. Z. & Samson, R. A. Fungal Biodiversity. In: CBS Laboratory manual series no. 1. Centraalbureau voor Schimmelcultures, Utrecht (2009).
Hankin, L. & Anagnostakis, S. L. The use of solid media for detection of enzyme production by fungi. Mycologia. 67, 597–607 (1975).
Liao, C. Y. et al. Characterization of three Colletotrichum acutatum isolates from Capsicum spp. Europ. J. Plant Pathol. 133, 599–608 (2012).
Sazci, A., Erenler, K. & Radford, A. Detection of cellulolytic fungi by using Congo red as an indicator: a comparative study with the dinitrosalicyclic acid reagent method. J. App. Bacteriol. 61, 559–562 (1986).
Wong, F. P. & Wilcox, W. F. Sensitivity to azoxystrobin among isolates of Uncinula necator. Baseline distribution and relationship to myclobutanil sensitivity. Plant Dis. 86, 394–404 (2002).
Stephenson, S. A. et al. Cloning and characterization of glutamine synthetase from Colletotrichum gloeosporioides and demonstration of elevated expression during pathogenesis on Stylosanthes guianensis. Curr. Genet. 5, 447–454 (1997).
Templeton, M. D., Rikkerink, E. H. A., Solon, S. L. & Crowhurst, R. N. Cloning and molecular characterization of the glyceraldehyde-3- phosphate dehydrogenase-encoding gene and cDNA from the plant pathogenic fungus Glomerella cingulata. Gene. 122, 225–230 (1992).
Gardes, M. & Bruns, T. D. ITS primers with enhanced specificity for basidiomycetes‐application to the identification of mycorrhizae and rusts. Mol. Ecol. 2, 113–118 (1993).
White, T. J. et al. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. (eds. Innis, M. A. et al.), PCR protocols: a guide to methods and applications. 315–322 (Academic Press, San Diego, California, USA 1990).
O’Donnell, K. & Cigelnik, E. Two divergent intragenomic rDNA ITS2 types within a monophyletic lineage of the fungus Fusarium are nonorthologous. Mol. Phylogenet. Evol. 7, 103–116 (1997).
Carbone, I. & Kohn, L. M. A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia. 91, 553–556 (1999).
Acknowledgements
This material is based upon the work supported by the New York Apple Research and Development Program grant numbers 86698 and 83268 to SGA. This work is supported in part by the New York State Specialty Crop Block Grant Program grant number 89379-P001/SCBG 19 006 to SGA and the SGA’s discretionary program funds. We thank David Lewis for sharing the resources of Vassar College microscopy facility at Biology Department in support of this work and Gillian Turgeon and Kathie Hodge at Cornell University for occasional advising. We acknowledge Keith Yoder at Virginia Polytechnic Institute and State University for providing the diseased apple fruit samples from Virginia. At last, we thank the staff and faculty of Cornell University’s HVRL, Christopher Meredith, Lucas Canino, Ricardo Delgado Santander, David Rosenberger and Peter Jentsch for help collecting and mapping apple samples or supporting this research at its initial stages. We acknowledge all apple growers for supporting this work directly or indirectly by allowing access to their farms, contributing HVRL membership and donations, funding facility upgrades and maintenance of HVRL laboratories and experiment plots.
Author information
Authors and Affiliations
Contributions
FK drafted the manuscript, collected isolates, performed all assays, prepared and interpreted the sequence data, contributed to phylogenetic analyses, performed microscopy examination and data analysis. JBG conducted phylogenetic analyses, wrote phylogeny-related sections. PLM collected and sequenced fungal isolates from Pennsylvania, contributed to microscopic examination of fungal material, advised on the interpretation of the taxonomy. EG annotated the sequences and submitted to NCBI, assisted in providing sequences for several genes for the holotype of new species. GJB contributed fungal isolate sequences, edited the manuscript and provided project funding for EG. KAP reviewed the manuscript, provided project funding for PLM and helped with sampling and fungal isolation in Pennsylvania. VPD provided guidance in phylogenetic approaches, performed GCPSR and was the crucial person to confirm the presence of a new species, contributed to microscopic examination of the teleomorph phase of the new species, assisted in writing the taxonomy and species delimitation section for the new species. SGA originated the study ideas, secured the funding and equipment, collected the samples from New York orchards and organized their delivery from other states, provided the facilities and contributed in editing and improving the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Khodadadi, F., González, J.B., Martin, P.L. et al. Identification and characterization of Colletotrichum species causing apple bitter rot in New York and description of C. noveboracense sp. nov.. Sci Rep 10, 11043 (2020). https://doi.org/10.1038/s41598-020-66761-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-66761-9
This article is cited by
-
Comprehensive antifungal investigation of green synthesized silver nanoformulation against four agriculturally significant fungi and its cytotoxic applications
Scientific Reports (2024)
-
The LasmiR858-MYB111 module enhances the antioxidant capacity of green leaf lettuce by promoting flavonoid biosynthesis
Plant Growth Regulation (2024)
-
Identification of mycoparasitism-related genes in Trichoderma harzianum T4 that are active against Colletotrichum musae
Archives of Microbiology (2024)
-
Draft genome sequence of the apple pathogen Colletotrichum chrysophilum strain M932
Journal of Plant Pathology (2023)
-
Identification, prevalence and pathogenicity of Colletotrichum species associated with chilli anthracnose in India
Journal of Plant Pathology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.