Abstract
Current guidelines recommend BRCA1 and BRCA2 genetic testing for individuals with a personal or family history of certain cancers. Three BRCA1/2 founder variants — 185delAG (c.68_69delAG), 5382insC (c.5266dupC), and 6174delT (c.5946delT) — are common in the Ashkenazi Jewish population. We characterized a cohort of more than 2,800 research participants in the 23andMe database who carry one or more of the three Ashkenazi Jewish founder variants, evaluating two characteristics that are typically used to recommend individuals for BRCA testing: self-reported Jewish ancestry and family history of breast, ovarian, prostate, or pancreatic cancer. Of the 1,967 carriers who provided self-reported ancestry information, 21% did not self-report Jewish ancestry; of these individuals, more than half (62%) do have detectable Ashkenazi Jewish genetic ancestry. In addition, of the 343 carriers who provided both ancestry and family history information, 44% did not have a first-degree family history of a BRCA-related cancer and, in the absence of a personal history of cancer, would therefore be unlikely to qualify for clinical genetic testing. These findings may help inform the discussion around broader access to BRCA genetic testing.
Similar content being viewed by others
Introduction
Pathogenic variants in the BRCA1 and BRCA2 genes are linked to an increased risk for female breast and ovarian cancer (including early-onset breast cancer), male breast cancer, prostate cancer, pancreatic cancer, and certain other cancers1. These variants are highly penetrant: Women with a variant have a 45–85% chance of developing breast cancer and up to a 46% chance of developing ovarian cancer by age 702. However, increased surveillance and prophylactic surgery (mastectomy and salpingo-oophorectomy) can greatly reduce the risk of breast and ovarian cancer in women carrying a BRCA1 or BRCA2 mutation3.
The prevalence of pathogenic BRCA1 and BRCA2 variants is estimated to be between 1 in 300 and 1 in 800 in the general population1,2. Among individuals of Ashkenazi Jewish descent, three BRCA1/2 founder variants — 185delAG (c.68_69delAG), 5382insC (c.5266dupC), and 6174delT (c.5946delT) — are present at a frequency of ~1 in 401.
Current U.S. guidelines limit BRCA1/2 genetic testing to individuals with a personal or family history of a relevant cancer, including early-onset breast cancer, multiple primary breast cancers, ovarian cancer, and certain other cancers1,2,4. In addition, Ashkenazi Jewish ancestry is sometimes used to recommend screening for individuals with a personal or family history of a single breast cancer at any age1. However, recent studies have found that about 50% of BRCA carriers have little or no family history of a relevant cancer5,6,7,8. These individuals would likely not qualify for clinical genetic testing unless they developed cancer themselves, representing a missed opportunity for cancer prevention. Because BRCA variants predispose to very high breast and ovarian cancer risks even among carriers without a family history5,9, these findings have spurred calls for broader access to BRCA genetic testing, among Ashkenazi Jews and in the general population5,6,7,10,11.
The 23andMe database provides an ethnically diverse group of genotyped individuals. We sought to characterize a cohort of individuals who carry one or more of the three Ashkenazi Jewish founder variants as related to two characteristics that are typically used to recommend individuals for BRCA testing: self-reported Jewish ancestry and family history of breast, ovarian, prostate, or pancreatic cancer. We focused on these two characteristics because they can enable individuals to learn their BRCA status before developing cancer, thus providing opportunities for cancer prevention and/or early detection.
Results
We identified 2,853 individuals who carry one or more of the three Ashkenazi Jewish BRCA1/2 founder variants (Table 1).
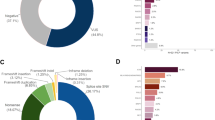
We first characterized the ethnic backgrounds of the carriers. The three variants in this study are most common in people of Ashkenazi Jewish descent; overall, 1 in 46 23andMe research participants with 85–100% Ashkenazi Jewish genetic ancestry had at least one of these variants, consistent with previous reports that the frequency of these variants in the Ashkenazi Jewish population is approximately 1 in 401. However, among the 1,967 carriers who provided self-reported ancestry information, 21% did not report Jewish ancestry (Table 2). Participation in this study was not restricted by ethnicity or country of residence for otherwise eligible participants; additional self-reported ethnicity information is provided in Supplementary Table 1.
One possible explanation for the sizable fraction of carriers who did not report Jewish ancestry is that they were unaware of their Ashkenazi Jewish ancestry. To test this hypothesis, we explored the relationship between self-reported ancestry and genetic ancestry. As expected, individuals with a greater proportion of estimated Ashkenazi Jewish genetic ancestry were more likely to report Jewish ancestry (Table 3 and Fig. 1); fewer than half of individuals with less than 20% Ashkenazi Jewish genetic ancestry (roughly equivalent to one grandparent or great-grandparent who was Ashkenazi Jewish) reported Jewish ancestry. Furthermore, most (62%, 258 of 415) of the BRCA carriers who did not report Jewish ancestry did have at least 1% Ashkenazi Jewish genetic ancestry. However, a lack of knowledge of Ashkenazi Jewish ancestry could not fully account for the 21% of carriers who did not report Jewish ancestry, as 8.4% (166 of 1,967) of individuals carrying an Ashkenazi Jewish founder variant had no detectable Ashkenazi Jewish genetic ancestry. These data (Table 2, Table 3 and Supplementary Table 1) are consistent with previous reports that BRCA1 185delAG and 5382insC are also found in people of other ethnicities12. Nine individuals self-reported Jewish ancestry but had no detectable Ashkenazi Jewish genetic ancestry.
The primary criterion for BRCA genetic testing is a personal or family history of breast, ovarian, or certain other cancers (including prostate and pancreatic cancer)1. We therefore assessed whether the carriers in our cohort had a family history of cancer. 393 carriers provided family history information.
Among participants who reported Jewish ancestry, 41% reported no first-degree family history of a BRCA-related cancer (Table 4). Similarly, 54% of participants who did not report Jewish ancestry reported no first-degree family history of cancer. Although our family history data differ substantially from guidelines used to determine genetic testing eligibility (which often include age of diagnosis and more than one cancer of certain types), these data are consistent with previous reports that about 50% of BRCA carriers would not be eligible for genetic testing based on family history alone5,6,7,8.
Discussion
In this study, we describe a cohort of approximately 2,800 individuals identified through direct-to-consumer genetic testing who carry one or more of the three Ashkenazi Jewish BRCA1/2 founder variants. Among eligible participants, this cohort was not restricted by genetic ancestry, self-reported ethnicity, or country of residence. In characterizing the ancestry and family cancer histories of these individuals, we made two key observations.
First, we found that a sizable proportion (21%) of carriers do not self-report Jewish ancestry. Of these individuals, more than half (62%) do have detectable Ashkenazi Jewish genetic ancestry, although frequently in very low percentages (Table 3 and Fig. 1). Interestingly, 8% of carriers have no detectable Ashkenazi Jewish genetic ancestry, consistent with reports that BRCA1 185delAG and 5382insC are found in women of other ethnicities who are referred for clinical genetic testing12. One potential caveat is that the check-all-that-apply survey question format used to ascertain self-reported Jewish ancestry (“Do any of the following cultural group labels describe your ancestry? Please check all that apply.”) may mis-classify some individuals relative to a forced-choice format (e.g., “Do you have any Jewish ancestry?”)13. Thus, it is possible that we may be overestimating the fraction of carriers who do not know they have Jewish ancestry.
Second, we observed that nearly half of individuals carrying an Ashkenazi Jewish BRCA variant have no first-degree family history of a BRCA-related cancer and, in the absence of a personal cancer history, would therefore be unlikely to qualify for clinical genetic testing. This percentage is consistent with published reports that about 50% of BRCA carriers lack a strong family history of cancer5,6,7,8. Based on these findings, many individuals identified in our study likely would not have learned their BRCA status through traditional clinical testing; indeed, a recent study suggests that more than 80% of individuals with a pathogenic BRCA variant do not know they have one8.
One strength of this study is that we were able to analyze data from a large population not restricted by genetic ancestry, self-reported ethnicity, or prior personal or family history of cancer. However, there are likely to be differences between the 23andMe customers who consented to participate in this research and the general U.S. population that could impact the generalizability of our results, including education level, income, ethnicity, and knowledge of/interest in genetics. In addition, while we have no reason to believe that people with a family history of cancer are more likely to become 23andMe research participants, we cannot exclude this possibility.
Other limitations of this study include a potential ascertainment bias related to family cancer history within the 23andMe database itself, as individuals with such histories may be more likely to answer questions about family cancer history. In addition, due to the limited depth of our family history survey, we defined family history as having a first-degree relative with breast, ovarian, prostate, or pancreatic cancer; clinical testing criteria are typically stricter, requiring an early age of diagnosis and/or more than one affected family member. Together, these two points suggest that our estimate of the fraction of individuals who would be ineligible for testing based on family history alone under existing screening guidelines is likely lower than the true fraction. Finally, because not all individuals in this study provided ancestry and family history information, the number of individuals included in some analyses is fairly small.
Our data suggest that a sizable fraction of individuals with detectable Ashkenazi Jewish genetic ancestry are unaware of that ancestry. This phenomenon is likely not unique to Ashkenazi Jewish ancestry. In addition to BRCA-related cancers, many other conditions are more common in specific ancestral groups, including Tay-Sachs disease, Canavan disease, and Gaucher disease type 1 in Ashkenazi Jews; sickle cell anemia in individuals with African ancestry; and beta-thalassemia in individuals with Mediterranean and certain other ancestries. For individuals who are unaware of their genetic ancestry, perceived risk for diseases could thus differ substantially from actual risk, which could lead to missed opportunities for genetic screening, prevention, and early intervention.
In recent years, several groups have called for broader access to BRCA genetic testing among Ashkenazi Jews and among women in the general population, which could enable women and men with a BRCA variant to learn their status, take steps to reduce their cancer risk, and encourage cascade testing of close family members5,6,7,10,11. Among Ashkenazi Jews, where testing for the three founder variants can identify most BRCA carriers, population-wide screening is cost-effective or even cost-saving14; in other ethnicities, more comprehensive genetic testing would be required to identify most individuals carrying a BRCA variant, but depending on the source of testing, this may also be cost-effective15. Our data may help inform the discussion around this growing call for expanded BRCA testing.
Methods
Participants were drawn from the customer base of 23andMe. All participants provided informed consent and answered surveys online according to a research protocol approved by Ethical and Independent Review Services, an external AAHRPP-accredited institutional review board. Data on ancestry and family cancer history were collected by self-report via online surveys; see Supplementary Information for survey questions. All consented 23andMe research participants 18 years or older and genotyped on one of the two arrays described below were eligible for the study, regardless of self-reported or genetic ancestry. Analyses were run on phenotypic data collected before October 10, 2017. All research was performed in accordance with relevant guidelines and regulations.
DNA extraction and genotyping were performed on saliva samples by CLIA-certified and CAP-accredited clinical laboratories of Laboratory Corporation of America. Samples were genotyped on one of two custom Illumina genotyping arrays16: the OmniExpress+ Bead chip (V3) or a fully custom array (V4). The three variants included in this study (185delAG, 5382insC, and 6174delT) are returned to 23andMe customers as part of 23andMe’s Health + Ancestry Service and have been analytically validated on the most recent versions of our genotyping chip.
Proportions of Ashkenazi Jewish genetic ancestry were estimated via an analysis of local genetic ancestry as described previously17. To account for imprecision in genetic ancestry estimates, we characterized estimates of Ashkenazi Jewish genetic ancestry <1% as “not detectable”.
Data availability
All data generated or analyzed during this study are included in this published article.
References
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Genetic/Familial High-Risk Assessment: Breast, Ovarian, and Pancreatic. Version 1.2020, https://www.nccn.org/professionals/physician_gls/pdf/genetics_bop.pdf.
Committee on Practice Bulletins–Gynecology. Practice Bulletin No 182: Hereditary Breast and Ovarian Cancer Syndrome. Obstet Gynecol 130, e110–e126 (2017).
Rebbeck, T. R., Kauff, N. D. & Domchek, S. M. Meta-analysis of Risk Reduction Estimates Associated With Risk-Reducing Salpingo-oophorectomy in BRCA1 or BRCA2 Mutation Carriers. JNCI J. Natl. Cancer Inst. 101, 80–87 (2009).
Owens, D. K. et al. Risk Assessment, Genetic Counseling, and Genetic Testing for BRCA-Related Cancer: US Preventive Services Task Force Recommendation Statement. JAMA 322, 652–665 (2019).
Gabai-Kapara, E. et al. Population-based screening for breast and ovarian cancer risk due to BRCA1 and BRCA2. Proc. Natl. Acad. Sci. U.S.A. (2014).
Manchanda, R. et al. Population Testing for Cancer Predisposing BRCA1/BRCA2 Mutations in the Ashkenazi-Jewish Community: A Randomized Controlled Trial. JNCI J. Natl. Cancer Inst. 107 (2014).
Metcalfe, K. A. et al. A comparison of the detection of BRCA mutation carriers through the provision of Jewish population-based genetic testing compared with clinic-based genetic testing. Br. J. Cancer 109, 777–779 (2013).
Manickam, K. et al. Exome Sequencing–Based Screening for BRCA1/2 Expected Pathogenic Variants Among Adult Biobank Participants. JAMA Netw. Open. 1 e182140 (2018).
Kuchenbaecker, K. B. et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 317, 2402–2416 (2017).
King, M.-C., Levy-Lahad, E. & Lahad, A. Population-based screening for BRCA1 and BRCA2: 2014 Lasker Award. JAMA 312, 1091–1092 (2014).
Akbari, M. R., Gojska, N. & Narod, S. A. Coming of age in Canada: a study of population-based genetic testing for breast and ovarian cancer. Curr. Oncol 24, 282–283 (2017).
Hall, M. et al. BRCA1 and BRCA2 mutations in women of different ethnicities undergoing testing for hereditary breast-ovarian cancer. Cancer 115, 2222–33 (2009).
Callegaro, M., Murakami, M. H., Tepman, Z. & Henderson, V. Yes–no Answers versus Check-all in Self-Administered Modes: A Systematic Review and Analyses. Int. J. Mark. Res 57, 203–224 (2015).
Manchanda, R. et al. Cost-effectiveness of population based BRCA testing with varying Ashkenazi Jewish ancestry. Am. J. Obstet. Gynecol., https://doi.org/10.1016/j.ajog.2017.06.038 (2017).
Long, E. F. & Ganz, P. A. Cost-effectiveness of Universal BRCA1/2 Screening: Evidence-Based Decision Making. JAMA Oncol 1, 1217–1218 (2015).
Tian, C. et al. Genome-wide association and HLA region fine-mapping studies identify susceptibility loci for multiple common infections. Nat. Commun. 8, 599 (2017).
Durand, E. Y., Do, C. B., Mountain, J. L. & Macpherson, J. M. Ancestry Composition: A Novel, Efficient Pipeline for Ancestry Deconvolution. bioRxiv 010512, https://doi.org/10.1101/010512 (2014).
Acknowledgements
We thank the research participants and employees of 23andMe for making this work possible. We also thank Uta Francke, Jennifer C. McCreight, and Elizabeth S. Noblin for critical review of the manuscript; David A. Hinds for scientific input; and the 23andMe Health Team (Robert K. Bell, Katarzyna Bryc, Alison L. Chubb, Stacey B. Detweiler, Anne E. Greb, Esther Kim, Michaela Johnson, Joanna L. Mountain, Jamaica R. Perry, Jeffrey D. Pollard, Catherine Wilson, Shirley Wu) for support and infrastructure to enable the research presented here.
Author information
Authors and Affiliations
Contributions
Study concept and design: J.Y.T. and R.I.T. Acquisition and analysis of data: J.Y.T., S.B.L., B.L.K. and M.H.M. Interpretation of data: R.I.T., S.B.L., B.L.K., M.H.M., and J.Y.T. Drafting of the manuscript: R.I.T. Critical revision of the manuscript for important intellectual content: R.I.T., S.B.L., B.L.K., M.H.M. and J.Y.T.
Corresponding author
Ethics declarations
Competing interests
R.I.T., S.B.L., B.L.K, M.H.M. and J.Y.T. are employees of and have stock, stock options, or both, in 23andMe, Inc. The authors declare no other financial or non-financial competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tennen, R.I., Laskey, S.B., Koelsch, B.L. et al. Identifying Ashkenazi Jewish BRCA1/2 founder variants in individuals who do not self-report Jewish ancestry. Sci Rep 10, 7669 (2020). https://doi.org/10.1038/s41598-020-63466-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-63466-x
This article is cited by
-
Genomic health data generation in the UK: a 360 view
European Journal of Human Genetics (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.