Abstract
The popular medicinal mushroom Ganoderma lucidum (Fr.) Karst. [Ling Zhi] has been widely used for the general promotion of health and longevity in Asian countries. Continuous cultivation may affect soil microbe and soil properties. However, the effect of G. lucidum cultivation on related wood segments, soil and tree roots microbial communities and soil properties is remain unknown. In our study, the microbial communities of soils, wood segments, and tree roots before and after G. lucidum cultivation were investigated by Illumina Miseq sequencing of both ITS and 16S rDNA, and taxonomic composition of eukaryotic and prokaryotic microorganisms were observed. Indices of microbial richness, diversity and evenness significantly differed between before and after G. lucidum cultivation. Each of the investigated sampling type harbored a distinctive microbial community and differed remarkably before and after G. lucidum cultivation. Ascomycota and Basidiomycota (fungi), Proteobacteria and Actinobacteria (bacteria) showed significant differences after Ling Zhi cultivation. The soil property values also changed after cultivation. The redundancy analysis (RDA) showed that both the fungal and bacterial community structure significantly correlated with soil humus, pH, nitrogen, carbon and trace elements (Fe, Zn, Mn, Cu) contents. The results indicated that G. lucidum cultivation may have significant differed the associated microbial community structures and soil properties. The study will provide useful information for G. lucidum cultivation and under-forest economic development.
Similar content being viewed by others
Introduction
Ganoderma lucidum (Ling Zhi or reishi mushroom,) has been an economically important mushroom, especially in the East Asian countries (i.e. China, Japan, Korea), since 4000 years ago1. The fungi is grown on a widespread commercial scale and is commonly consumed for medicinal and spiritual potency1,2,3. Ling Zhi cultivation under forests has great economic and ecological importance. Problems such as production ability limit, affecting forest resources etc. exist4. Cultivation (continuous monocropping) can lead to disease infection, microbial community composition change, alterations in soil quality and plant growth5,6,7,8. Similarly, Ling Zhi cultivation may play a vital role on microbial communities of soils, wood segments and tree roots as well as soil properties. Meanwhile, related microbial communities could also affect soil quality and Ling Zhi cultivation. However, information on effect of G. lucidum cultivation on these microbial communities and soil properties is still unavailable, making it very difficult to draw any general conclusions.
For microbial community researches, culture-dependent and culture-independent approaches can be adopted9,10. Currently, high-throughput sequencing provides a deep understanding of the biotic and abiotic parameters affecting microbial community composition, having great advantage of generating huge data as well as detecting unculturable species at much lower price, making it easier to extend our knowledge of microbial diversity11,12,13.
Our main study aim is to investigate the differences on the microbial communities of soils, wood segments and tree roots before and after G. lucidum cultivation. Correlation between the microbial community structure and the soil properties is also elucidated. The study will provide a comprehensive view of microbial community and soil properties changes affected by G. lucidum cultivation and useful information for further exploitation and utilization.
Materials and Methods
Ethics
The samples in the study were collected on private land and the owners allowed the full permission to conduct the study on the sites. The experimental materials did not involve in any humans and animals.
Sample collection
The study was carried out in the sampling sites (ca. 100 m × 100 m) of Jiu Long Mountain Forestry Reserve area (39°56′15″N, 116°05′12″E), Mentougou District, Beijing, North China. The samples were collected twice: 1) before G. lucidum cultivation—in October 2015; 2) three years after G. lucidum cultivation—in October 2018. Apart from G. lucidum cultivation, all the other conditions were same. Under forests, Ling Zhi grows on the wood segments, wood segments are half in the soils under forests trees Pinus tabulaeformis Carr. (Supplementary Fig. S1). Each time, three types of samples were collected: soil, wood segments where G. lucidum directly grow and P. tabulaeformis roots in the site. Four biological replicates were chosen for each sample. The sampling sites were evenly divided into four plots (each ca. 50 m \(\times \) 50 m). For soil samples, nine soil cores (2.5 cm diameter) from 0 to 30 cm depth (where directly in touch with wood segments) were randomly collected and mixed for each plot. For wood segments and tree roots, also, nine were randomly collected and mixed for each plot.Non-cultivated negative control samples were set up to make sure the effect of Ling zhi cultivation. Samples were collected into sterile plastic bags and processed within 24 h. Soil physicochemical properties (soil carbon, nitrogen, phosphorus, potassium, sodium, humus, copper, iron, manganese, zinc, pH) were tested at Pony Testing International Group Ltd (Beijing) with standard method in14.
DNA extraction, amplification of ITS and 16 rDNA region and Illumina sequencing
TIANamp Soil DNA Kit (TIANGEN Biotech Co., Ltd., Beijing, China) was used to extract DNA from homogenized soil samples. Genomic DNA of the wood segments and tree roots were extracted with a standard cetyl–trimethyl ammonium bromide (CTAB) method with modifications15. The concentrations of the DNA were measured with NanoDrop ND-2000 spectrophotometer (Thermo Fisher Scientific, USA).
Fungal primers ITS1F (CTTGGTCATTTAGAGGAAGTAA) and ITS2R (GCTGCGTTCTTCATC GATGC) were used to amplify ITS1 region of rDNA16. Bacterial 16S rDNA was amplified with primers 338 F (ACTCCTACGGGAGGCAGCA) and 806 R (GGACTACHVGGGTWTCTAAT)17. The PCR products were purified and sequenced with Illumina MiSeq. platform at Shanghai Majorbio Science and Technology Ltd. All sequences were deposited at the Sequence Read Archive (SRA) of the National Center for Biotechnology Information (NCBI) under project accession number PRJNA538936.
Pre-processing and analysis of ITS rDNA sequences
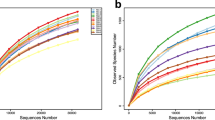
Raw reads quality was pre-processed with FLASH18 and Trimmomatic19. Mothur standard operation pipeline (SOP, v.1.37.6)20 was used to analyze the data and classify sequences into OTUs (Operational Taxonomic Units) at 97% similarity against UNITE Database v. 7.221 (fungi) and Silva Database release 12822 (bacteria). Sequence reads were subsampled for each sample with the minimum number (43410 fungi; 41074 bacteria) of reads among all samples before comparative analysis. The indices richness (Sobs), diversity (Invsimpson) and evenness (Simpsoneven)23 were calculated in Mothur. Data for rarefaction curves were also generated in Mothur. One-way ANOVA (analysis of variance) was used to identify differences in community richness, diversity and evenness, and microbial abundance. R language platform24 was used for analysis and visualization of the data sets of the microbial diversity and abundances in different samples (Rarefaction curves, Venn, bar chart, PCoA, RDA, PERMANOVA).
Results
MiSeq sequencing data
In the study, after sequence quality evaluation, a total of 1,305,273 fungal and 1,217,144 bacterial high quality sequences were recovered. The samples were with an average of 54,386 ± 2952 fungal and 50,714 ± 1348 bacterial sequences (mean ± SD). The average sequence length was 272 bp and 438 bp for fungi and bacteria, respectively.
Richness, diversity and evenness of microbial communities
Quality-filtered sequences were clustered into 1,831 fungal OTUs and 3,549 bacterial OTUs (excluding singletons). The soil samples had the highest richness, diversity and evenness, and the wood segments had the lowest richness, diversity and evenness in all the microbial communities (Table 1). The fungi showed higher richness, diversity and evenness in soil and wood segments samples after G. lucidum cultivation. The bacteria indexes are higher in wood segments after cultivation, while, showed an opposite trend in soil and tree root samples (Table 1). Microbial richness, diversity and evenness indexes of samples showing statistically significant differences were shown in Supplementary Fig. S2 labeled with asterisk (*). Rarefaction curve showed that the OTUs abundance were saturated in all samples (Supplementary Fig. S3A,B). [LA– Soil after G. lucidum cultivation; LB– Soil before G. lucidum cultivation; MA–Wood segments after G. lucidum cultivation; MB–Wood segments before G. lucidum cultivation; GA– P. tabulaeformis roots of the sites after G. lucidum cultivation; GB– P. tabulaeformis roots of the sites before G. lucidum cultivation].
Fungal and bacterial community composition among soils, wood segments and tree roots
The sequences assigned to Fungi kingdom were classified into 7 fungal phyla. The relative abundances of phyla exceeding 1% in samples were shown in Fig. 1a. Totally, Ascomycota was the most abundant group (59.93%) followed by Basidiomycota (26.68%) and Zygomycota (6.83%). Others such as Glomeromycota weremuch less (<0.1%). Dominant fungi phyla changed after cultivation. For example, Ascomycota predominated in wood segments before cultivation, while, Basidiomycota took the place after cultivation. Abundance of Ascomycota and Basidiomycota showed significant differences between samples before and after G. lucidum cultivation (Supplementary Fig. S4A). Seven classes had a relative abundance of more than 1% (Fig. 1b), which include Agaricomycetes, Leotiomycetes, Sordariomycetes, Eurotiomycetes, Dothideomycetes, Pezizomycetes and Tremellomycetes. Agaricomycetes and Leotiomycetes showed significant differences between samples before and after cultivation (Supplementary Fig. S4B). Regarding the bacterial population, overall, Proteobacteria was most abundant, followed by Actinobacteria and Bacteroidetes. Phyla abundance were shown in Fig. 1c. Class Alphaproteobacteria, Actinobacteria and Gammaproteobacteria were most abundant (Fig. 1d). Abundance of 14 bacterial phyla including three phyla mentioned above and 14 classes showed significant differences before and after cultivation (Supplementary Fig. S3E,F).
At family level, relative abundance of 17 fungal and 18 bacterial groups exceeded 2% (Fig. 2a,c), such as eukarytic Mortierellacea (LA and LB), Ganodermatceae (MA and MB), Hydnaceae(GA and GB) and prokaryotic Streptomycetaceae (LA and LB; GA and GB), Sphingobacteriaceae (MA and MB) [Parentheses indicate great differences were shown between sampls].
As to genera, their relative abundance changed a lot after cultivation (Fig. 2b,d), such as eukarytic Mortierella (soil LA and LB), Peniophorella (MA and MB, GA and GB) and prokaryotic Mucilaginibacter (LA and LB), Sphingomonas (MA and MB, GA and GB). Many sequences can be just classified to genus or higher level. Relative abundance of 9 families and 11 genera (fungal), and 13 families and 7 genera (bacterial) including taxa mentioned above showed significant differences after cultivation (Supplementary Fig. S3C,D,G,H).
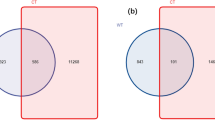
The common and unique OTUs of samples were illustrated in Fig. 3a,b. Fungi of LA had the most unique OTUs, while, Bacteria of LB had the most unique OTUs.
The relationship between microbial community structure and G. lucidum cultivation and soil properties
Principal Coordinate Analysis (PCoA) showed that soil microbial communities formed individual cluster before and after G. lucidum cultivation as well as in wood segments and tree roots (Fig. 4a,b). Subsequent PERMANOVA test confirmed the significant differences in community structures of all samples betwween two time stage (all p < 0.05).
Soil property values are enclosed in Table 2. The values can be divided into three groups according to their change trend after cultivation: 1) reduced—humus; 2) almost stable—P, K, Na; 3) increased—pH, C, N, Fe, Zn, Mn, Cu. The RDA tests showed that the both fungal and bacterial community structure was significantly correlated with soil humus, pH, N, C, Fe, Cu, Zn and Mn (0.76 ≤ r2 ≤ 0.88; 0.001 ≤ p ≤ 0.002) (Fig. 5a,b). Trace elements (Fe, Zn, Mn, Cu) clustered closely, indicating similar effect degree, and showed same trend with C, N, pH (Fig. 5a,b). The tests results were somehow consistent with soil properties change trend.
Discussion
In our study, the microbial communities of soils, wood segments and tree roots before and after G. lucidum cultivation were mainly studied by Next-generation sequencing. 7 phyla, 28 classes, 95 orders, 206 families, 405 genera and 1,831 OTUs were identified in Fungi kingdom, 42 phyla, 93 classes, 198 orders, 401 families, 818 genera and 3,549 OTUs were determined in Bacteria, supplying a whole view for the uninvestigated microbial diversity and microbial succession during Ling Zhi cultivation. The study will also provide helpful information and potential use for G. lucidum cultivation and other edible and medicinal mushrooms cultivations to develop under-forest economic development.
Microbial richness, diversity and evenness indexes showing great differences after G. lucidum cultivation. The fungi cultivation can be taken as one kind of land use. Land use was considered as a main driving factor of bacteria for multiple land uses (arable, forest, grasslands)25. Previous studies also showed that microbial indexes could be influenced by land-use in many scale26,27,28. Some opposite change trends of fungi and bacteria in our research may indicate their interaction during Ling Zhi cultivation.
Microbial composition is influenced by land-use25. Featured fungal taxon was significantly related with distinct systems of land use25. Ascomycota predominated in wood segments and tree roots before Ling Zhi cultivation, while, Basidiomycota took the place after cultivation. Ascomycota showed an opposite trend in soils. Ascomycota and Basidiomycota showed significant differences between samples before and after G. lucidum cultivation. Changes of fungal community of soil were also reported especially distinctive among trophic groups29. Bacterial Proteobacteria and Actinobacteria were most abundant and also differed greatly in our study. The most represented phyla Proteobacteria and Actinobacteria were also found in different land use soils along European transect25. The dominance of the two phyla is in agreement with other studies of soill30,31. Class Agaricomycetes and Leotiomycetes showed significant differences between samples before and after cultivation. Agaricomycetes has about 21,000 recorded mushroom-forming species as pathogens, decayers and mutualists of terrestrial and aquatic habitats32. Their abundance was gradually declined with longer length with grazing exclusion in soil layers33. Leotiomycetes is one of the largest nonlichen-forming ascomycetous groups, include varieties of ecology of saprobes, plant endophytes, plant pathogens, fungi-parasites, and insect and other animal associates34. Actinobacteria is one of the largest groups of Bacteria, very significant in the biodegradation and recycling of organic matter35. Abundance changes exhibit the effects of Ling Zhi cultivation.
Abundance of dominant taxa including Mortierella, Ganoderma, Hydnaceae etc. changed after cultivation. Mortierella of Mortierellacea have been reported to produce arachidonic acid36 and single cell oil (SCO)37. Ganoderma of Ganodermatceae showed significant difference of MA and MB. This is not difficult to understand since Ling Zhi grew directly on wood segments. Ling Zhi cultivation obviouslyincrease Ganoderma abundance. Hydnaceae includes many wood−decay fungi, and is vital in carbon and nutrient recycling in forest ecosystems38. Abundance of some prokaryotic taxa also altered remarkably. Streptomycetaceae belongs to Actinobacteria, members of it can produce a wide array of clinically important bioactivemolecules39, and have been found in plants roots, and affect tree growth40. Its abundance showed great changes after cultivation, especially in soils and tree roots. Sphingobacteriaceae, were found existed in all of soil samples of different days after paraquat use41, among various strains of Tyrophagus putrescentiae42. Mucilaginibacter members exist in many soil samples43,44. The genus was hypothesized to play an important role in processing carbon44. Sphingomonas has been reported having abilities of degradation of polyphenols and biodegradation of tributyl phosphate (TBP)45,46. The ecological roles and functions of the taxa in our study merits further research.
Structure of microbial communities differed significantly before and after G. lucidum cultivation. Each of the sample (soils, wood segments and tree roots) in our study harbored a different microbial assemblage. Continuous monoculture of watermelon for twenty years significantly change the soil microbes, adding to diversity of bacteria, while, reducing fungal diversity8. Microbes are critical in farmland ecosystems, particularly in improving soil fertility. Significant changes were also found in microbial communities caused by mudflat reclamation under rice cultivation47. The fungal community composition of soil showed notable differences among the first, the secondand the third rotation plantation during consecutive monoculture in Casuarina equisetifolia plantations48. The results in our study show consistency with these findings. However, wood preservatives with copper can delay wood breaking down and altered fungi but not bacteria composition of soil49. More studies are needed in the future.
Soil property values also changed much after cultivation, humus reduced, pH, C, N, and trace elements (Fe, Zn, Mn, Cu) increased, indicating cultivation put profound effect on soil properties. Microbial community structure was also obiviously connected with soil properties, exhibiting soil may affect the associated microbial community. Importance of plants and soil on belowground microbial diversity has been investigated in the last decades50. Studies at local to continental-level showed that soil properties are major driver effectors of microbial communities of soil, and also profound with plant communities and land use25. Close mutual relationship was made between plants and soils. Plants can call for their rhizosphere microbes from the soil pool51. The microbial communities structure of pear trees also obviously correlated with properties of the growth soil microbial communities of root and rhizosphere are much more similar52. In our study, soil, G. lucidum cultivation, and forest tree roots may have a very complicated interaction. Wood segments, soils and tree roots can provide nutrients and affect Ling Zhi growth, and Ling Zhi growth can produce many beneficial molecular and compounds1,2,3, influencing wood segments, soils, and tree roots and tree growth. A recent study showed that planting with peanut monocropping and crop rotation greatly affected the rhizosphere microbial assembly of the peanut53. It also has reported that G. lucidum can produce a lot of chemicals like Ganoderma lucidum Polysaccharide, Ganodenic acid etc., having the ability of anti-tumor, anti-aging and detoxification1,2,3. Recent studies have found that G. lucidum polysaccharides produce anti-inflammatory, anti-obesity and anti-diabetes effects by modulating gut microbiota composition in mice54,55. These may help explain soil and microbial differences after Ling Zhi cultivation in some degree. The microbial group in the study might be isolated and used to improve soil quality in the future. The results could be drawn for other mushrooms cultivation under forests. The study will also provide information for sustainable management of soil and make better understandings of the influences of cultivations on microbes.
Conclusions
Microbial compositions of the samples (soils, wood segments and tree roots) in our study were not same. Structure of microbial communities differed significantly before and after G. lucidum cultivation. Soil property values also changed much after cultivation. Both composition of fungal and bacterial community had a remarkble correlation with soil humus, pH, N, C, Fe, Cu, Zn and Mn contents. Our results indicate that G. lucidum cultivation might have important effects on the associated composition of microbial community, soil properties and tree growth. The microbial roles of ecology and functions will merit be studied in the future.
References
Wasser, S. P. Reishi or ling zhi (Ganoderma lucidum). Encyclopedia of dietary supplements 1, 603–622 (2005).
Boh, B., Berovic, M., Zhang, J. & Zhi-Bin, L. Ganoderma lucidum and its pharmaceutically active compounds. Biotechnol. Annu. Rev. 13, 265–301 (2007).
Qin, L. H., Wang, C., Qin, L. W., Liang, Y. F. & Wang, G. H. Spore powder of Ganoderma lucidum for Alzheimer’s disease: A protocol for systematic review. Medicine 98, e14382 (2019).
Hua, M. Current situation and development countermeasures of Ganoderma lucidum industry in China. Northern Economy Trade 10, 82–83 (2010). (in Chinese).
Buckley, D. H. & Schmidt, T. M. The structure of microbial communities in soil and the lasting impact of cultivation. Microb. Ecol. 42, 11–21 (2001).
Wasof, S. et al. Linkages between aboveground and belowground community compositions in grasslands along a historical land-use intensity gradient. Plant Soil 434, 289–304 (2019).
Clermont-Dauphin, C., Cabidoche, Y. M. & Meynard, J. M. Effects of intensive monocropping of bananas on properties of volcanic soils in the uplands of the French West Indies. Soil Use Manage 20, 105–113 (2010).
Wang, T. et al. Characterizing differences in microbial community composition and function between Fusarium wilt diseased and healthy soils under watermelon cultivation. Plant Soil 438, 1–13 (2019).
Fierer, N., Jackson, J. A., Vilgalys, R. & Jackson, R. B. Assessment of soil microbial community structure by use of taxon-specific quantitative PCR assays. Appl. Environ. Microbiol. 71, 4117–4120 (2005).
He, J. Z., Zheng, Y., Chen, C. R., He, Y. Q. & Zhang, L. M. Microbial composition and diversity of an upland red soil under long-term fertilization treatments as revealed by culture-dependent and culture-independent approaches. J. Soil Sediment 8, 349–358 (2008).
Muller, D. B., Vogel, C., Bai, Y. & Vorholt, J. A. The plant microbiota: systems-level insights and perspectives. Annu. Rev. Genet. 50, 211–234 (2016).
Hong, C., Si, Y., Xing, Y. & Li, Y. Illumina MiSeq sequencing investigation on the contrasting soil bacterial community structures in different iron mining areas. Environ. Sci. Pollut. Res. 22, 10788–10799 (2015).
Frindte, K., Pape, R., Werner, K., Löffler, J. & Knief, C. Temperature and soil moisture control microbial community composition in an arctic–alpine ecosystem along elevational and micro-topographic gradients. ISME J. 13, 2031–2043 (2019).
Black, C. A., Evans, D. D. & Dinauer, R. C. Methods of soil analysis. American Society of Agronomy: Madison, USA, 1–1178 (1965).
Chang, S., Puryear, J. & Cairney, J. A simple and efficient method for isolating RNA from pine trees. Plant. Mol. Biol. Rep. 11, 113–116 (1993).
White, T. J., Bruns, T., Lee, S. & Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR protocols: A guide to methods and applications; Innis, M. A., Gelfand, D. H., Sninsky, J. J., White, T. J. Eds.; Academic Press: New York, USA, 315–322 (1990).
Ai, C. et al. Distinct responses of soil bacterial and fungal communities to changes in fertilization regime and crop rotation. Geoderma 319, 156–166 (2018).
Magoč, T. & Salzberg, S. L. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27, 2957–2963 (2011).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Schloss, P. D., Gevers, D. & Westcott, S. L. Reducing the effects of PCR amplification and sequencing artifacts on 16S rRNA–based studies. PloS One 6, e27310 (2011).
UNITE Community. UNITE mothur release. Version 01.12.2017. UNITE Community, https://doi.org/10.15156/BIO/587478 (2017).
Quast, C., et al The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 41 (Database), D590–D596 (2013).
Mothur Calculators. Available online: https://www.mothur.org/wiki/Calculators (04-26-2019).
R Core Team. R: A language and environment for statistical computing. R foundation for Statistical Computing: Vienna, Austria (2013).
Plassart, P. et al. Soil parameters, land use, and geographical distance drive soil bacterial communities along a European transect. Sci. Rep. 9, e605 (2019).
Lienhard, P. et al. Pyrosequencing evidences the impact of cropping on soil bacterial and fungal diversity in Laos tropical grassland. Agron. Sustain. Dev. 34, 525–533 (2014).
Constancias, F. et al. Mapping and determinism of soil microbial community distribution across an agricultural landscape. MicrobiologyOpen 4, 505–517 (2015).
Terrat, S. et al. Mapping and predictive variations of soil bacterial richness across France. PloS One 12, e0190128 (2017).
Brinkmann, N. et al. Intensive tropical land use massively shifts soil fungal communities. Sci. Rep. 9, e3403 (2019).
Lauber, C. L., Ramirez, K. S., Aanderud, Z., Lennon, J. & Fierer, N. Temporal variability in soil microbial communities across land-use types. ISME J. 7, 1641–1650 (2013).
Delgado-Baquerizo, M. et al. A global atlas of the dominant bacteria found in soil. Science 359, 320–325 (2018).
Hibbett, D. S. et al. 14 Agaricomycetes, In Systematics and Evolution. The Mycota (A Comprehensive Treatise on Fungi as Experimental Systems for Basic and Applied Research), vol. 7A; McLaughlin D., Spatafora J. Eds.; Springer: Berlin, Germany, 373–429 (2014).
Wang, Z. et al. Impact of long-term grazing exclusion on soil microbial community composition and nutrient availability. Biol. Fert. Soils 55, 121–134 (2019).
Zhang, N. & Wang. Z. 3 Pezizomycotina: Sordariomycetes and Leotiomycetes. In Systematics and Evolution. The Mycota (A Comprehensive Treatise on Fungi as Experimental Systems for Basic and Applied Research), vol 7B; McLaughlin D., Spatafora J. Eds.; Springer: Berlin, Germany, 57–88 (2015).
Trujillo, M. E. Actinobacteria. eLS, 1–16, https://doi.org/10.1002/9780470015902.a0020366.pub2 (2001).
Shinmen, Y., Shimizu, S., Akimoto, K., Kawashima, H. & Yamada, H. Production of arachidonic acid by Mortierella fungi. Appl. Biochem. Biotech. 31, 11–16 (1989).
Papanikolaou, S., Komaitis, M. & Aggelis, G. Single cell oil (SCO) production by Mortierella isabellina grown on high-sugar content media. Bioresource Technol. 95, 287–291 (2004).
Bari, E. et al. Ecology of wood‐inhabiting fungi in northern forests of Iran. Forest Pathol. 49, e12501 (2019).
Cho, M. A. et al. Streptomyces Cytochrome P450 Enzymes and Their Roles in the Biosynthesis of Macrolide Therapeutic Agents. Biomol. Ther. 27, 127 (2019).
Prudence, S. et al. Root-associated archaea: investigating the niche occupied by ammonia oxidising archaea within the wheat root microbiome. Access Microbiol. 1A, https://doi.org/10.1099/acmi.ac2019.po0112 (2019).
He, W. et al. Effect of paraquat on soil bacteria diversity in a young eucalypt plantation. Acta Scientiae Circumstantiae 32, 2857–2864 (2012).
Lee, J. et al. Comparative microbiome analysis of Dermatophagoides farinae, Dermatophagoides pteronyssinus, and Tyrophagus putrescentiae. J. Allergy Clin. Immun 143, 1620–1623 (2019).
Huq, M. A., Akter, S. & Lee, S. Y. Mucilaginibacter formosus sp. nov., a bacterium isolated from road-side soil. Antonie van Leeuwenhoek 112, 513–521 (2019).
Gadkari, P., Haggblom, M., Barkay, T., Kerkhof, L. & Mannisto, M. Ecology and physiological potential of tundra soil bacteria. Detail 2020, 01–31 (2019).
Feng, Y. et al. Effect of Sphingomonas sp. strain on degradation of polyphenols in redried tobacco leaves. Acta Tabacaria Sinica 25, 19–24 (2019).
Liu, J., Lin, H., Dong, Y. & Li, B. Elucidating the biodegradation mechanism of tributyl phosphate (TBP) by Sphingomonas sp. isolated from TBP-contaminated mine tailings. Environmental Pollution 250, 284–291 (2019).
Zhang, Y., Li, Q., Chen, Y., Dai, Q. & Hu, J. Mudflat reclamation causes changes of gene abundance in nitrogen cycle under long‐term rice cultivation. J. basic microbiol, https://doi.org/10.1002/jobm.201800660 (2019).
Liuting, Z. et al. Variation in soil fungal community structure during successive rotations of Casuarina equisetifolia plantations as determined by high-throughput sequencing analysis. Plant Growth Regulation 87, 445–453 (2019).
Lasota, S., Stephan, I., Horn, M. A., Otto, W. & Noll, M. Copper in wood preservatives delayed wood decomposition and shifted soil fungal but not bacterial community composition. Appl. Environ. Microbiol. 85, e02391–18 (2019).
Fischer, C. et al. Plant species richness and functional groups have different effects on soil water content in a decade‐long grassland experiment. J. Ecol. 107, 127–141 (2019).
Colin, Y. et al. Taxonomic and functional shifts in the beech rhizosphere microbiome across a natural soil toposequence. Sci. Rep. 7, e9604 (2017).
Ren, F., Dong, W., Sun, H. & Yan, D. H. Endophytic Mycobiota of Jingbai Pear Trees in North China. Forests 10((3)), 260 (2019).
Li, X. et al. Legacy of land use history determines reprogramming of plant physiology by soil microbiome. ISME J. 13((3)), 738 (2019).
Khan, I. et al. Mushroom polysaccharides from Ganoderma lucidum and Poria cocos reveal prebiotic functions. J. Funct. Food. 41, 191–201 (2018).
Chang, C. J. et al. Ganoderma lucidum reduces obesity in mice by modulating the composition of the gut microbiota. Nature Commun. 6, 7489 (2015).
Acknowledgements
This research was funded by the Fundamental Research Funds for the Central Non-profit Research Institution of Chinese Academy of Forestry (1project CAFYBB2017ZC006 and 2project CAFYBB2017MA019). Ms. Qing Hua Ma is acknowledged for kindly provide Ling Zhi cultivation photo.
Author information
Authors and Affiliations
Contributions
F.R., Y.G.Z., Y.A.Z. design of the work; F.R. and H.Y. collected and make acquisition of the samples; F.R. analysis, or interpretation of data; F.R. have drafted the of the manuscript; all authors have read and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ren, F., Zhang, Y., Yu, H. et al. Ganoderma lucidum cultivation affect microbial community structure of soil, wood segments and tree roots. Sci Rep 10, 3435 (2020). https://doi.org/10.1038/s41598-020-60362-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-60362-2
This article is cited by
-
The Changes of Microbial Diversity and Isolation of Microorganism in Soil for Alleviating the Production Decreasing After Continuous Cultivation of Ganoderma lucidum
Current Microbiology (2024)
-
Bacterial Community Shifts in Casing Soil Before and After the Cultivation of Oudemansiella raphanipes
Journal of Soil Science and Plant Nutrition (2022)
-
Different pioneer plant species have similar rhizosphere microbial communities
Plant and Soil (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.