Abstract
Mexico has made substantial contributions to marine turtle protection and conservation, especially since 1990. Several conservation projects entail monitoring efforts to recover nesting territories for marine turtles. The Sea Turtle Protection Program of Akumal, in the Mexican state of Quintana Roo, was created in 1993 and was developed by the Akumal Ecological Center. This paper provides the nesting ecology parameters for Caretta caretta over a protection period of 24 years (1995–2018). A well-defined nesting peak was observed in June, with a nesting success rate of 75.2 ± 23.0%. Nesting females showed a mean curved carapace length of 99.0 ± 5.6 cm. The mean clutch size was 108.6 ± 24.6 eggs, with variation among years. The mean incubation period was 57.2 ± 6.2 days. The hatching and emergence success rates were 87.2 ± 16.9% and 78.8 ± 24.4%, respectively. For the 926 tagged females that returned, the remigration interval peaked at 726 days, with a 12-day inter-nesting period. The results show not only the recovery of the nesting population over time but also a decrease in female size; we postulate that this decrease is due to the recruitment of young females, which has been increasingly pronounced since 2010. Hence, the Akumal rookery plays an important role in its corresponding regional management unit (Atlantic Northwest).
Similar content being viewed by others
Introduction
The Quintana Roo beaches of the Yucatán Peninsula belong to the Western Caribbean Marine Ecoregion (the scale unit in the Marine Ecoregions of the World system) in the tropical northwestern Atlantic1,2,3. The beaches in this ecoregion served as nesting4 and foraging grounds5,6 for marine turtles long before humans settled on the American continent7. Human pressures have impeded sea turtle habitats in many ways. Today, tourism and recreational activities in these areas constitute threats5,8,9 to the recovery of small but important rookeries10. However, the conservation of nesting grounds has also flourished, which has successfully enabled marine turtles to nest on protected beaches11.
In the case of loggerheads, the “available long-term series of annual nest counts shows an overall increase over the past three generations for the northwest Atlantic loggerhead subpopulation, which breeds mostly in the southeastern U.S. and the Yucatán Peninsula in Mexico”11. Hence, loggerheads belong to the “least concern” category of the International Union for the Conservation of Nature (IUCN) Red List in the subpopulation/regional management unit (RMU) of the Atlantic Northwest11.
The geographic distribution of subgroups in the area is complex: five subpopulations of loggerheads have been identified; the “Greater Caribbean subpopulation”, which includes Akumal loggerheads, is one of these subpopulations12. This subpopulation is also considered one of the five recovery units within the northwest Atlantic11. In this context, there is evidence of loggerhead metapopulations13, which justifies the designation of a RMU (Atlantic Northwest RMU for loggerheads)3. Trends show that nesting has increased in several rookeries, aided by conservation projects (vide11,14,15).
The Akumal Ecological Center (CEA) Conservation and Research Program was founded in 1995 and has since been protecting marine turtle nesting beaches and foraging grounds (Akumal Bay, Fig. 1). For 24 years, conservation efforts have entailed collecting data, helping restore nesting sites and protecting nesting females and hatchlings of two species: Chelonia mydas and Caretta caretta (Cc). We present data from the CEA monitoring programme from 1995–2018 concerning nesting parameters, hatching success, and temporal distributions of loggerhead (Cc) sea turtle nests. We also debate the implications of the programme and provide recommendations for the future.
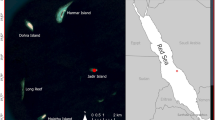
Study beaches at Akumal, Quintana Roo state, Mexico: Half Moon Bay (HMB), Akumal Bay (AB), Jade Bay (or Playa Tortugas (PT)), and South Akumal Bay (SA). The image on the left was designed by Freepik (https://www.freepik.com/free-photos-vectors/travel Travel vector created by Layerace - www.freepik.com) and modified using Adobe Illustrator CC2017; the right image was designed with Adobe Illustrator CC2017 from Landsat images (Landsat image courtesy of the U.S. Geological Survey).
Results
Records
The raw CEA records of the total number of nests per year between 1995 and 2018 are shown in Fig. 2. For the statistical analysis, a total of 3364 (N) records were analysed. Figures 3 and 4 show the distribution of records per monitoring year as percentages. An increasing tendency is shown by the linear tendency lines drawn from the percentages of records of Cc that emerged during the project (Figs. 3 and 4).
Figure 3 shows that the number of records has peaked since 2015, i.e., an increased number of records was observed on Akumal beaches in the last four years of the project.
The last peak (2016) was different from the peaks in 1999, 2002, 2007/08 and 2012/13; additionally, the records obtained between 1995 and 2013 represented only 50.1% of all records analysed in this study.
Morphometric analysis
For the nesting Cc turtles, the mean curved carapace length (CCL ± SD) was 99.0 ± 5.6 cm (N = 1407 females measured), ranging from 61.1–120.0 cm (Me = 99.3 cm and Mo = 100.0 cm). The histogram in Fig. 5 shows the frequency distribution for the CCL of all samples.
The average Cc curved carapace width (CCW) was 90.5 ± 5.7 cm (N = 1406), ranging from 59.0–111.0 cm (Me = 91.0 cm; Mo = 90.0 cm).
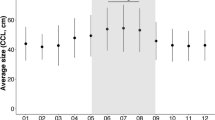
The 24 years of carapace measurements show a Gaussian distribution. The ANOVA for CCL showed significant differences between 1995 and 2015; between 1998 and 2012, 2015, and 2018; between 1999 and 2012, 2015, and 2018; and between 2000 and 2012, 2015 and 2018 (F = 3.795; P = 0.000).
The trio of 1998/1999/2000 showed the highest average CCLs (approximately 101 cm), and the values during these years differed significantly from those in 2012/2015/2018, when the smallest averages were recorded (approximately 97 cm). Similar differences in clutch sizes were also expected to be detected in these years. In fact, the nesting seasons of 2015 and 2018 showed the smallest clutch sizes and contrasted significantly (F = 3.385; P = 0.000) with the years with the largest clutch sizes (1999, 2007, 2008, 2011, and 2017).
Additionally, the variation in morphometrics decreased during the study period (Fig. 6).
Clutch size and nest parameters
The nesting success for Cc was 75.2 ± 23.0%, and the average clutch size was 108.6 ± 24.6 eggs (N = 1710 nests). There were no data regarding the nest contents in 2003 or 2005 (Fig. 7). The year 2018 showed the smallest average clutch size (100.4 ± 23.0 eggs), in contrast with 2014, which had the highest average (132.7 ± 24.9 eggs). The number of clutches in 2014 was atypical since the field effort was compromised (there were no nesting parameter data). The year with the highest average incubation period (IP) value (2010) also had a small clutch size average.
The nesting season is shown in Fig. 8a; it occurs between April and November, but most nesting activity occurred between May and July. June is the peak nesting month for Cc in Akumal. There was no disruption of the nesting season over the 24 years (Fig. 8b). The last years of the programme showed increases in nests.month−1 in June and July (more than 25%; Fig. 8b).
Incubation period (IP)
The overall (1995–2004) average IP was 57.2 ± 6.2 days (Table 1), ranging from 39 to 81 days (Mo = 55.0 days; Me = 57.0 days; N = 1413). The IP showed a slight decreasing tendency over the years (Fig. 9), achieving its lowest peak in 2009 (IP = 54.8 ± 4.8 days) and its highest peak in 2010 (IP = 61.1 ± 6.7 days).
Hatching success (HS) and emergence success (ES)
Generally, HS and ES rates are high in a successful conservation programme. For Cc, the overall HS was 87.2 ± 16.9%. The ES was 78.8 ± 24.4%, with much variability (in fact, the values ranged from 0–100%, meaning that in some of the excavated nests, all the hatchlings (alive or not) were found in the sand). Table 1 summarizes the overall IP, HS and ES averages for Cc, and Table 2 shows the overall averages for each nest parameter.
Inter-nesting period and remigration interval
Tag information revealed that for the Akumal beaches, Cc females returned 12 days after the first emergence (Fig. 10; first chart). The remigration interval was 726.0 days (1.99 years) for 926 unique tags (N). Data dispersion was high (Fig. 10; second chart); the mean of the first cluster of data (Fig. 10b) was approximately 346.1 days, the mean of the second cluster of data was 715.4 days, and the mean of the third cluster of data was 1068.2 days. The averages of the four clusters (Fig. 10b) showed an effective Cc remigration cycle of exactly 345.4 days between the four clusters.
Effects on nesting on each beach
Incubation occurred most frequently in PT and HMB (Fig. 11). However, the clutches were largest in SA, which has the fewest disturbances attributable to tourists and lights from hotels and houses. AB, for example, is very exposed to tourism since two major hotel complexes are present on this beach. HMB also has many houses and tourists. The IP also differed among these beaches. The overall IP average was the longest in SA and the shortest in HMB.
ANOVA by beach suggested that the IP in SA was significantly different from the IPs on the other beaches (P < 0.006; Tukey post hoc test).
Although the average clutch size in SA was higher than those on the other three beaches, there were no significant differences between them (Fig. 12).
The HS rates in HMB and PT were significantly different (P < 0.001; Tukey’s post hoc test). Additionally, the ES significantly differed between PT and AB (P = 0.003), HMB (P < 0.001), and SA (P = 0.025) according to Tukey’s post hoc tests.
Discussion
What do the numbers show
The Cc females that were observed in Akumal were increasingly frequent but decreased in size in the last years of the conservation programme. It is probable that many Cc born on Akumal beaches were recruited to nest, showing philopatric behaviour.
The results show that the Cc nesting in Akumal deposited an increasing number of nests, especially since 2010. Although Mexico’s Protected Area Commission (CONAMP)16 indicated a 5% decline in nesting for Cc between 1995 and 2006 based on the analysis of records from index beaches (i.e., beaches that provide data to estimate the trends for the region), the IUCN reported that data from the Greater Caribbean Recovery Units suggested the opposite11. The Akumal Project corroborated IUCN’s results but showed that turtles preferred the beaches in Akumal for other reasons.
The increase in nesting numbers in Akumal warrants explanation. Marine turtle conservation projects have been increasing in the Caribbean, and accordingly, so have the management skills of the monitors16. Mexico fully prohibited the capture of marine turtles in its waters in 199016,17. As more turtles were protected, more turtles were recruited to the waters and nesting grounds as they reached sexual maturity. The marine turtles that were protected in 1995 in Akumal would reach this maturity after 18/20 years (2013/2015; vide the following section), and in fact, 50% of the analysed data were concentrated after 2013 (the last five years of the project). The monitors working for more than 20 years in the area who marked the marine turtles recorded successful nesting by the same turtles on Akumal beaches approximately 18 years later. This view is also corroborated by Labrada-Martagón et al.6.
Morphometry of nesting females
Chaloupka and Limpus18 studied and defined “age classes” for loggerhead turtles. An adult CCL ranges between 85 and 105 cm. The CCL average in Akumal indicated that the nesting females were adults, though a few small-sized females nested on its beaches. The decreasing average CCL value over the years (Fig. 6) was also observed in other scientific studies; “Karen Bjorndal revealed that somatic growth rates for loggerheads (…) throughout the region began to decline in the late 1990s as the result of an ecological regime shift; the decline continues to the present”19. What does this ecological shift entail? Does increased sea surface temperature accelerate growth and sexual maturity? Will relatively small females with a fast growth rate be observed? Or do the numbers mean that the number of young females in the population increased in the last several years? An increased rate of recruitment for first-time breeders may explain the increase in the population registered in recent years.
Nesting parameters
Nesting success in Akumal was higher than that reported in Guanahacabibes Peninsula, Cuba (67%), which belongs to the same RMU. Nesting success values are probably affected by the tourism pressure on three of the main nesting beaches. Light pollution, obstacles in the sand concentrated in specific areas (such as beach furniture), and people on the beach can lead Cc turtles to abandon their nesting attempts. In some areas of the beach, the sand may be too dry or too thick to excavate, with rocks and coral debris, which also leads turtles to abort their nesting intentions.
The remigration interval was 2.0 years, which is in accordance with other publications (vide Hart et al.12), and Cc takes 12 days on average to re-nest/-emerge in the same nesting season. The analysis showed a very predictable population of nesters exhibiting clockwork-like nesting behaviour. For these turtles, nesting seasons occur precisely every 354.4 days on average. Another aspect revealed by the tag analysis was that the first emergence in a nesting season occurred on the exact same beach as it did in the previous nesting season.
The typical average clutch size for Cc is 100–130 eggs20, meaning that the average clutch size in Akumal (108.6 ± 24.6 eggs) exhibits a large variation. In Florida, the average clutch size for Cc is 98.5 ± 1.7 eggs21, which is smaller than that in Akumal, and in the Archie Carr National Wildlife Refuge along the central coast of Florida, the average clutch size is 113.9 ± 1.4 eggs according to Ehrhart et al.14, which is higher than that in Akumal. High clutch sizes were observed in 2014, but there were no significant differences between the average CCL in this year and the average CCLs in other years in the study period. Hence, the difference in clutch size may be due to causes other than female dimensions, even though there were no significant differences between the clutch size in 2014 and the sizes in other years. These turtles are not particularly large (CCL = 100.2 ± 4.9 cm), but they lay a large number of eggs per clutch.
The average IP for Cc in Akumal was longer (57.2 ± 6.2 days) than the published value of 50.8 ± 1.222, but it was within the range of other studies (46 to 82 days for Matsuzawa et al.23 study). The range of values was high, probably because there was high seasonal variation in the temperature of the sand.
Since the pivotal IP is approximately 52.6 days in the Mediterranean24, one can hypothesize that a balanced ratio of males and females per nest is produced in Akumal. Additionally, the IP depends on temperature fluctuations25. The pivotal temperature for Cc incubation is 28.74 °C26, although Mrosovsky et al.24 determined this value to be 29.3 °C. Another recent experimental study emphasized that the optimal range for Cc incubation was 28.5–31 °C25. Temperatures above 31 °C may impact the hatchling survivorship rate25, which probably explains why some ES values were so low. Humidity, air temperature, and precipitation are probably the main climatic drivers of hatchling production; sea surface temperature and wind speed, though important, do not have significant influences27. It would be very important to determine, for example, the temperature fluctuations in Akumal sand/nests to understand how they affect incubation conditions (are they female-biased with an increasing trend?).
The Cc HS values were similar or even increased when compared to those in other studies (e.g., similar to 87.3 ± 17.8%27; higher than 68% ± 4%21, 55.1 ± 4.0%14), although the ES was decreased and varied most likely due to the difficulties faced by hatchling when leaving the nest. Abiotic factors in Akumal vary due to strong precipitation or flooding due to storms and hurricanes and cause pre-emergent mortality23. In a Florida study, the HS rates for loggerheads decreased from 1985–200321. In a study by Ehrhart et al.14, the HS and ES values were very low due egg washing caused by beach erosion (55.1 ± 4.0%; 53.3 ± 3.7%). On Japanese beaches, the HS determined by Matsuzawa et al.23 was relatively low, which was possibly due to the following conditions: compacted/desiccated sand, hatchlings trapped in the nest due to heat (inhibition of movement), or oxygen deficiency inside the nest due to accelerated metabolic rates23. It is possible that in Akumal, the ES is compromised by one of these constraints; this possibility needs to be further considered.
Conservation implications
Akumal, where snorkelling and observation of nesting females are possible activities for tourists, is certainly important in many aspects8. These opportunities have provided alternative livelihoods for villagers that settled in the region, a pattern observed in other southeastern Mexican coastal locations (vide the Kanzul beach case9) and in other Caribbean locations5.
On the beaches and in the foraging grounds off the southeastern Mexican coast, efforts have been made to enhance the protection of juveniles, females and nests. Activities related to tourism are more efficiently controlled by local environmental authorities28. Additionally, it is very important to improve citizens’ awareness of the recovery and protection of nesting, development and foraging territories. For example, touristic developments should focus on offering information and responsible activities to tourists. The respect of sea turtle habitats and niches by people is crucial. Cc is still vulnerable even with all the apparent recovery suggested by the numbers and indicators and the protection provided by the conservation teams.
To guarantee the success of the Inter-American Convention for the Protection and Conservation of Sea Turtles agenda in the future, other measures need to be taken. Cases such as those in the Cayman Islands, where migratory green and loggerhead nesting populations are critically reduced13, must be addressed. The protection of all territories and the interconnections among them inside the RMU will provide additional opportunities for population recovery. Additional evidence, such as the results obtained by Blumenthal et al.29, who emphasized that “oceanic juveniles from some rookeries appear to be dispersed among multiple foraging grounds, while those from other rookeries appear to be more locally constrained”, must also be considered to maximize protection.
Efforts are needed to identify the role of the rookeries in Akumal and other Quintana Roo regions in supporting the migratory subpopulations/cohorts of the RMU (e.g., for the Florida Atlantic coast, the Gulf of Mexico, the Cuban and Bahamian waters, and even the eastern Atlantic waters10,30,31,32, among other destinations). Field biologists are collecting data and filling gaps to enhance the knowledge of these long-lived species19. The information provided here provides indicators for the Yucatán Peninsula and can be used to compare nesting parameters with other rookeries inside the RMU. Genetic30 or telemetric studies and cross-tagging information analyses are mandatory. These approaches can help reveal the potential connections, genetic drift of genes, molecular diversity and the metapopulation29 structure within the wider Caribbean region. The State of the World’s Sea Turtles (SWOT) has emphasized the need for cooperation among teams.
Materials and Methods
Monitoring programme and field effort
In Mexico, this species is protected under Federal laws and specific Norms such as NOM-059-SEMARNAT-201033, which protects threatened species of flora and fauna, and NOM-162-SEMARNAT-201234, which specifies the protection, recovery and management of the sea turtle populations and their nesting habitats. In Akumal, the nesting season is from May to November/December4,34,35. Four main beaches are protected by the Akumal Ecological Center (Fig. 1) and monitored daily during the nesting season. Every night during the nesting season, the beaches were patrolled from 9 pm until 4 am and every morning (6 am) when eggs began to hatch by instructed monitors. During these patrols, several variables were observed/collected on paper forms, and detected nests were identified with a tag (e.g., BA Cc 0001). The nest variables were also collected. After the IP and according to NOM-162-SEMARNAT-201234 protocols, each nest was excavated, and the shells, dead hatchlings, and live hatchlings were counted.
Nesting sea turtles were tagged following the Eckert and Beggs36 methodology with Monel tags, which were generally applied between the 2nd and 3rd plates on one of the front flippers. Prior to tagging, marking scars and other tags were checked on both the flippers and paddles. Nests were carefully monitored after the first evidence of hatching; they were not disturbed until there was evidence of the emergence of hatchlings. Many emergence events were observed in situ since the beaches are on small bays. Three to five days after the eggs hatched, the nests were excavated to perform counts.
Collected/measured variables
For each patrol time, the date (and sometimes the GPS coordinates) of the emergence/nesting events, curved carapace length (CCL, notch to tip, n-t) and width (CCW) were recorded according to Bolton’s method37, and the track width, presence of tags and respective codes, and presence of epibionts on the carapace were among the variables collected during the night patrols. Whenever a nest was constructed, an identification code was attributed; if oviposition was observed, variables such as nest depth and temperatures inside the nest and of the sand were collected. Additionally, whenever possible, the number of eggs was counted. After incubation, the following nest content variables were recorded: emerged hatchlings, shells, live and dead hatchlings, undeveloped eggs, unhatched and unhatched term (UHT) (“unhatched apparently full-term embryo in eggshell or pipped, i.e., with a small amount of external yolk material”38) eggs, and predated eggs. Nests were also monitored during incubation, and relevant events were recorded (flooding, predation events). All nests at risk after oviposition were moved to the hatchery in Jade Bay (Fig. 1). The computed variables were the IP, clutch size, hatching success (HS) and emergence success (ES), inter-nesting interval and remigration interval.
Study site/beaches
The CEA patrols and protects four main beaches: Half Moon Bay (HMB), Akumal Bay (AB), Jade Bay (or Playa Tortugas (PT)), and South Akumal Bay (SA) (Fig. 1). The project has a hatchery situated in PT. The four beaches are composed of biogenic sand, and PT hosts the largest number of nests.
Each monitored beach has geographical, biological and physical differences, as well as different lengths and widths. HMB is 700 m long and 18 m wide at the widest part and 5 m wide at the narrowest part. The beachfront contains approximately 80% human development, such as condo buildings and rental houses. AB is approximately 1200 m long; however, only 900 m serves as a nesting area due to sea turtle preference. Approximately 90% of the beachfront is occupied by hotels and restaurants. The width varies from 5 to 20 m. PT is 500 m long, with 60% human occupancy by one low-density hotel, one beach club and houses. Due to these structures, the minimum width is 2 m at some sites and 10 m at the widest part. SA has the smallest nesting area, at approximately 400 m long; however, the total length of the beach is 700 m. Due to the beach’s shape, the central area is 20 m wide and has the highest density, and the ends are only 2 m wide. This beach has the least human disturbance, hosting only private houses39.
Statistical analysis
The statistical analysis was performed with PASW Statistics 18, from SPSS an IBM company, and Microsoft Office Excel 2007 software. Significance was estimated at the 95% confidence level. Variables such as IP, clutch size, HS and ES were compared using t-tests and one-way ANOVA and Games-Howell, Tukey’s or Scheffe’s post hoc tests when statistically significant differences were detected (p < 0.05). For all the variables, the overall data were estimated, and then averages for each nesting season (which in this case coincided with the annual data) were determined; the annual values were used for ANOVAs.
To determine the inter-nesting period, the remigration interval tag numbers of the females that emerged were used. The inter-nesting interval was considered the number of days between a successful nesting event and the first subsequent attempt by the same female in a particular nesting season; the remigration interval was considered the period between the observations of a female in different but subsequent nesting seasons20. The analysis was complex due to the number of tags and records. Therefore, an automation process in Microsoft Office Excel 2007 was required to run the data and generate the two variables. The automation formulas consisted of identifying the unique tag numbers in the overall sample (all years), the number of times a unique tag was repeated, and the date of each repetition; building a matrix for the tags; and finally calculating the difference (number of days) between the dates.
Ethical approval and informed consent
Fieldwork in Akumal, Mexico, was conducted by the staff from the non-governmental organization Centro Ecológico de Akumal (CEA) in accordance with the guidelines and permits (non-extractive use of sea turtle permits, with the following references SGPA/DGVS/04368/17, SGPA/DGVS/006855/18) provided by SEMARNAT, Secretaria de Medio Ambiente y Recursos Naturales, and through the Dirección General de Vida Silvestre (DGVS). All methods were carried out according to Mexican regulations (vide NOM-059-SEMARNAT-201033, NOM-162-SEMARNAT-201234) imposed by CEA and SEMARNAT. All experimental protocols were approved by CEA and SEMARNAT.
References
Dow, W., Eckert, K., Palmer, M. & Kramer, P. An Atlas of Sea Turtle Nesting Habitat for the Wider Caribbean Region. The Wider Caribbean Sea Turtle Conservation Network and The Nature Conservancy. WIDECAST Technical Report No. 6. Beaufort. North Carolina. 267 p. plus electronic Appendices (2007).
Spalding, M. D. et al. Marine Ecoregions of the World: A Bioregionalization of Coastal and Shelf Areas. BioScience 57(7), 573–583, https://doi.org/10.1641/B570707 (2007).
Wallace, B. P. et al. Regional Management Units for Marine Turtles: A Novel Framework for Prioritizing Conservation and Research across Multiple Scales. PLoS One 5, e15465, https://doi.org/10.1371/journal.pone.0015465 (2010).
Anastácio, R., Gonzalez, J. M., Kathy, S. & Pereira, M. J. Software for Field Survey of Nesting Marine Turtles. Scientific Reports 7, 10796, https://doi.org/10.1038/s41598-017-11245-6 (2017).
Gil, M. A., Renfro, B., Figueroa-Zavala, B., Penié, I. & Dunton, K. H. Rapid tourism growth and declining coral reefs in Akumal. Mexico. Mar Biol. 162(11), 2225–2233, https://doi.org/10.1007/s00227-015-2748-z (2015).
Labrada-Martagón, V., Tenería, F. A. M., Herrera-Pavón, R. & Negrete-Philippe, A. Somatic growth rates of immature green turtles Chelonia mydas inhabiting the foraging ground Akumal Bay in the Mexican Caribbean Sea. Journal of Experimental Marine Biology and Ecology 487, 68–78, https://doi.org/10.1016/j.jembe.2016.11.015 (2017).
Lagueux, C. J. Status & Distribution of the Green Turtle. Chelonia mydas. in the Wider Caribbean Region. In: Eckert. K. L. & F. A. Abreu Grobois (eds.) 2001. Proceedings of the Regional Meeting: “Marine Turtle Conservation in the Wider Caribbean Region: A Dialogue for Effective Regional Management.” Santo Domingo. 16–18 November 1999. WIDECAST. IUCN-MTSG. WWF. and UNEP-CEP. xx+154 pp. (2001).
Penié, I., Lozano, M. & Slater, K. Ancient Mayan “Place of the Turtles” Copes with Modern-Day Tourism. SWOT Report, pp. 28–29 (2010).
Oliver de la Esperanza, A., Arenas Martínez, A., Tzeek Tuz, M. & Pérez-Collazos, E. Are anthropogenic factors affecting nesting habitat of sea turtles? The case of Kanzul beach, Riviera Maya-Tulum (Mexico). Journal of Coastal Conservation 21, 85–93, https://doi.org/10.1007/s11852-016-0473-5 (2017).
Gavilán, F. M. Status & Distribution of the Loggerhead Turtle. Caretta caretta. in the Wider Caribbean Region In: Eckert. K. L. & Abreu Grobois F.A. (eds.) 2001. Proceedings of the Regional Meeting: “Marine Turtle Conservation in the Wider Caribbean Region: A Dialogue for Effective Regional Management.” Santo Domingo. 16–18 November 1999. WIDECAST. IUCN-MTSG. WWF. and UNEP-CEP. xx+154 pp. (2001).
Ceriani, S. A. & Meylan, A. B. Caretta caretta (North West Atlantic subpopulation)(amended version of 2015 assessment). The IUCN Red List of Threatened Species 2017: e.T84131194A119339029. https://doi.org/10.2305/IUCN.UK.2017-2.RLTS.T84131194A119339029.en (2017).
Hart, K. M., Sartain, A. R. & Fujisaki, I. Bahamas connection: residence areas selected by breeding female loggerheads tagged in Dry Tortugas National Park, USA Animal. Biotelemetry 3(3), 1–17, https://doi.org/10.1186/s40317-014-0019-2 (2015).
Blumenthal, J. M. et al. Ecology of Hawksbill Turtles. Eretmochelys imbricata. on a Western Caribbean Foraging Ground. Chelonian Conservation and Biology 8(1), 1–10, https://doi.org/10.2744/CCB-0758.1 (2009).
Ehrhart, L., Redfoot, W., Bagley, D. & Mansfield, K. Long-Term Trends in Loggerhead (Caretta caretta) Nesting and Reproductive Success at an Important Western Atlantic Rookery. Chelonian Conservation and Biology 13(2), 173–181, https://doi.org/10.2744/CCB-1100.1 (2014).
Azanza-Ricardo, J. et al. Possible Effect of Global Climate Change on Caretta caretta (Testudines, Cheloniidae) Nesting Ecology at Guanahacabibes Peninsula, Cuba. Chelonian Conservation and Biology 16(1), 12–19, https://doi.org/10.2744/CCB-1241.1 (2017).
Pineda, O. G. & Rocha, A. R. B. (Coord.) Las tortugas marinas en México: Logros y perspectivas para su conservación. Comisión Nacional de Áreas Naturales Protegidas (CONANP), Ciudad de Mexico. ISBN 978-607-97436-0-4 (2016).
CONANP (Comision Nacional de Areas Naturales Protegidas) Programa de Acción para la Conservación de la Especie: Tortuga Caguama, Caretta caretta. Dirección General de Operación Regional, Dirección de Especies Prioritarias para la Consevación, Programa Nacional de Conservación de Tortugas Marinas, Ciudad de Mexico (2011).
Chaloupka, M. & Limpus, C. Trends in the abundance of sea turtles resident in Southern Great Barrier Reef waters. Biological Conservation 102, 235–249 (2001).
Witherington, B. et al. Atlantic loggerheads, Why Isn’t the Best Understood Sea Turtle Recovering? State of the World’s Sea Turtles. (SWOT) Report XIV, 12–19 (2019).
Eckert, K. L., Bjorndal, K. A., Abreu-Grobois, F. A. & Donnelly, M. Eds. Research and Management Techniques for the Conservation of Sea Turtles. No. 4. IUCN/ Species Survival Commission Marine Turtles Specialist Group Publication. Washington D.C. (1999).
Antworth, R. L., Pike, D. A. & Stiner, J. C. Nesting Ecology. Current Status. and Conservation of Sea Turtles on an Uninhabited Beach in Florida. USA. Biological Conservation 130, 10–15 (2006).
Kawana, L. T., Burney, C. M. & Fisher, L. The Effect of Air Temperature on the Incubation Period and Hatching Success of In Situ Loggerhead Sea Turtle (Caretta caretta) Clutches in Broward County, Florida. Oceanography Faculty Proceedings. Presentations. Speeches. Lectures 305. http://nsuworks.nova.edu/occ_facpresentations/305 (2011).
Matsuzawa, Y., Sato, K., Sakamoto, W. & Bjorndal, K. A. Seasonal fluctuations in sand temperature: effects on the incubation period and mortality of loggerhead sea turtle (Caretta caretta) pre-emergent hatchlings in Minabe. Japan Marine Biology 140, 639–646, https://doi.org/10.1007/s00227-001-0724-2 (2002).
Mrosovsky, N., Kamel, S., Rees, A. F. & Margaritoulis, D. Pivotal temperature for loggerhead turtles (Caretta caretta) from Kyparissia Bay. Greece. Can. J. Zool. 80, 2118–2124, https://doi.org/10.1139/Z02-204 (2002).
Fisher, L. R., Godfrey, M. H. & Owens, D. W. Incubation Temperature Effects on Hatchling Performance in the Loggerhead Sea Turtle (Caretta caretta). PLoS One 9(12), e114880, https://doi.org/10.1371/journal.pone.0114880 (2014).
Davenport, J. Temperature and the life-history strategies of sea turtles. J Therm Biol 22(6), 479–488, https://doi.org/10.1016/S0306-4565(97)00066-1 (1997).
Montero, N., Ceriani, S. A., Graham, K. & Fuentes, M. M. P. B. Influences of the Local Climate on Loggerhead Hatchling Production in North Florida: Implications From Climate Change. Front. Mar. Sci. 5, 262, https://doi.org/10.3389/fmars.2018.00262 (2018).
DOF (Diario Oficial de la Federación) ACUERDO por el que se establece con el nombre de Bahía de Akumal el área de refugio para la protección de las especies que se indican, la porción marina que se señala en el Estado de Quintana Roo. https://dx.doi.org/07/03/2016, www.dof.gob.mx/nota_detalle.php?codigo=5428829&fecha=07/03/2016&print=true (2016).
Blumenthal, J. M. et al. Turtle groups or turtle soup: dispersal patterns of hawksbill turtles in the Caribbean. Molecular Ecology 18, 4841–4853, https://doi.org/10.1111/j.1365-294X.2009.04403.x (2009).
Dow Piniak, W. E. & Eckert, K. L. Sea turtle nesting habitat in the Wider Caribbean Region. Endang Species Res. 15, 129–141, https://doi.org/10.3354/esr00375 (2011).
Putman, N. F., Verley, P., Shay, T. J. & Lohmann, K. J. Simulating transoceanic migrations of young loggerhead sea turtles: merging magnetic navigation behavior with an ocean circulation model. The Journal of Experimental Biology 215, 1863–1870, https://doi.org/10.1242/jeb.067587 (2012).
Musick, J. A. & Limpus, C. Habitat utilizations and migration in juvenile sea turtles. p. 137–163. In: Lutz. P. L. & Musick. J. A. (Eds.) The Biology of Sea Turtles. CRC Press, New York (1997).
DOF (Diario Oficial de la Federacion) Norma Oficial Mexicana NOM-059-SEMARNAT-2010, Protección ambiental-Especies nativas de México de flora y fauna silvestres-Categorías de riesgo y especificaciones para su inclusión, exclusión o cambio-Lista de especies en riesgo. Secretaria de Medio Ambiente y Recursos Naturales (2010).
DOF (Diario Oficial de la Federacion) Que establece las especificaciones para la protección, recuperación y manejo de las poblaciones de las tortugas marinas en su hábitat de anidación (NOM-162-SEMARNAT-2012, NORMA Oficial Mexicana NOM-162-SEMARNAT-2012 (2013).
Slater, K. Sea Turtle Nesting Site Preferences. 5p (CEA-OPERATION WALLACEA, Akumal http://opwall.com/wp-content/uploads/YA291-Turtle-monitoring-project-overview.pdf (2015).
Eckert, K. L. & Beggs, J. Marine Turtle Tagging: A Manual of Recommended Practices. WIDECAST (Wider Caribbean Sea Turtle Conservation Network) Technical Report No 2 (Revised Edition) 40 pp, Beaufort, North Carolina (2006).
Bolten, A. B. Techniques for Measuring Sea Turtles. In: Eckert, K. L., Bjorndal, K. A., Abreu-Grobois, F. A. & Donnelly, M. (Eds.). Research and Management Techniques for the Conservation of Sea Turtles. IUCN/Species Survival Commission Marine Turtles Specialist Group. Washington, D.C. Pp. 110–114 (1999).
Miller, J. D. Determining Clutch Size and Hatching Success. In: Eckert, K. L., Bjorndal, K. A., Abreu-Grobois, F. A. & Donnelly, M. (Eds.) Research and Management Techniques for the Conservation of Sea Turtles. IUCN/Species Survival Commission Marine Turtles Specialist Group. Washington, D.C. Pp. 124–129 (1999).
Cubedo, H. A. L., López, J. M. G. & Miranda G. J. L. Informe Técnico de Resultados del Programa de Protección, Conservación y Observación de Tortuga Marina del Centro Ukana I Akumal A.C. Temporada 2018. Tulum, Quintana Roo, México. 48 p. (2018).
Acknowledgements
We would like to thank all the people who contributed to collecting data over these 24 years, who are difficult to name since there was a considerable number of contributors. However, we would like to especially acknowledge all tortugueros and volunteers who monitored the Akumal beaches. Additionally, we would like to thank all the CEA staff who, even through hard times, kept the conservation and education programmes going. Finally, we would like to acknowledge all the CEA supporters, donors and people dedicated to our conservation cause. The publishing of this manuscript was supported by the FCT/Ministério da Ciência, Tecnologia e Ensino Superior national funding to CESAM, Universidade de Aveiro.
Author information
Authors and Affiliations
Contributions
J.M.G. and H.A.L.C. led the field research and coordinated the data collection field work by the CEA team; J.M.G., R.A. and M.J.P. performed the analyses and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
González, J.M., Anastácio, R., Lizárraga-Cubedo, H.A. et al. Caretta caretta nesting activity on Akumal Beaches, Mexico. Sci Rep 10, 3020 (2020). https://doi.org/10.1038/s41598-020-60018-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-60018-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.