Abstract
Here, we characterized the basic properties of repetitive sequences in essential A and supernumerary B chromosomes of Festuca pratensis Huds. This was performed by comparative analysis of low-pass Illumina sequence reads of B chromosome lacking (−B) and B chromosome containing (+B) individuals of F. pratensis. 61% of the nuclear genome is composed of repetitive sequences. 43.1% of the genome are transposons of which DNA transposons and retrotransposons made up 2.3% and 40.8%, respectively. LTR retrotransposons are the most abundant mobile elements and contribute to 40.7% of the genome and divided into Ty3-gypsy and Ty1-copia super families with 32.97% and 7.78% of the genome, respectively. Eighteen different satellite repeats were identified making up 3.9% of the genome. Five satellite repeats were used as cytological markers for chromosome identification and genome analysis in the genus Festuca. Four satellite repeats were identified on B chromosomes among which Fp-Sat48 and Fp-Sat253 were specific to the B chromosome of F. pratensis.
Similar content being viewed by others
Introduction
The genus Festuca consists of more than 450 species1 and is one of the largest genera in the Poaceae. Festuca sp. and its closely allied genus Lolium sp. belong to the grass family Poaceae, subfamily Pooideae and tribe Poeae2. The genus Festuca is an ancient group and considered as one of the main evolutionary lines in the tribe Poeae3. Festuca species have diverse distribution and are considered as important components of grass ecosystems of the temperate zones4. Ten to 12 species of Festuca grow wildly throughout Iran5,6.
Species inside Festuca vary substantially in DNA C-value and ploidy levels, from diploid (2n = 2x = 14) to dodecaploid (2n = 12x = 84), most of them being allopolyploid7,8,9. There are many cytogenetic studies of different populations of the genus Festuca7,10,11.
Festuca pratensis Huds. (Meadow fescue) is a diploid species in the genus Festuca with 2n = 2x = 14 standard (A) chromosomes (Fp genome). In addition to the essential A chromosomes, one to five supernumerary B chromosomes (Bs) have been reported in F. pratensis, however one and two Bs are common in B-carrying individuals12,13,14. Bs are generally neutral and may have negative effects on the host when they are present in higher numbers. In some plants such as F. pratensis, Secale cereal and Allium schoenoprasum, Bs may confer adaptive advantage under the unfavorable conditions15,16,17. There are reports suggesting that Bs increase allelic variation by altering the frequency and distribution of crossing overs in A chromosome18,19,20 suggesting that Bs are not genetically inert and directly or indirectly affect regulatory mechanisms in the cell.
Transposons and satellites (satDNA) are two major groups of repetitive sequence in eukaryotic genomes and have diverse sequence and distribution patterns21,22,23,24,25. The length of satellite monomers can vary widely but are typically a few hundred nucleotides26,27. Commonly, satellite repeats form tens of kilobases to megabases structural blocks with biased base composition28. The rapid evolution of satellite repeats in plant genomes, has led to their intraspecific homogenization and fixation of species-specific polymorphisms, making satDNA families unique source of molecular and cytogenetic markers to analyze genetic diversity and genome evolution28,29,30,31,32. Repetitive sequences also have accumulated in B chromosomes during the process of B chromosome formation and evolution33.
With the availability of high throughput sequencing technologies and development of assembly-free methods for assessing repetitive sequences34, we were interested to conduct a genome wide repeatome analysis in F. pratensis. Its 1C DNA value is about 3.25 pg (≈3.178 Gbp) as measured for the cultivar ‘Kolumbus’35. Physical mapping of ribosomal DNA sites in some Festuca species revealed that 5S and 45S ribosomal loci in F. pratensis have been localized on the short arms of chromosomes 2 and 3, respectively36. FISH using BAC clones37 and sequencing of chromosome 4F38 have identified some repetitive DNA sequences in the meadow fescue genome. Using five tandem repeats identified from chromosome 4F together with rDNA probes, Křivánková, et al.39 established a molecular karyotype of meadow fescue. Majka, et al.40 identified an A chromosome-specific repetitive sequence in this species.
In order to characterize the repeatome landscape of F. pratensis in plants with and without B chromosomes, we employed low coverage next generation sequencing in combination with the RepeatExplorer software tool and FISH to determine the types, abundance, organization and chromosomal position of repetitive DNA sequences. The first B-specific tandem repeats of F. pratensis decorating entire B chromosomes after FISH were identified.
Results
Identification and classification of transposable elements
In total, 17 million Illumina 150 bp paired end reads were generated from both −B and +2B F. pratensis genotypes corresponding to about 0.4× coverage of the haploid genome. The GC content for the genome showed a value of 46%. Comparative analysis of reads from −B (250410 reads) and +2B (249590) genotypes by the RepeatExplorer pipeline41, showed that highly and moderately repetitive sequences constitute 61% of the nuclear genome. The overall proportions of individual repeat types and monomer frequencies (except for B enriched repeats) was similar in the −B and +2B genotypes (Supplementary Table S1).
43.13% of the repeats are composed of transposons with majority (40.81%) of which being retrotransposons. On the other hand, class II transposons contributed to only 2.32% of the repeats. LTR retrotransposons are the most abundant mobile elements and composed 40.75% of the genome. LTRs divided to Ty3-gypsy and Ty1-copia super families with 32.97% and 7.78% of genome, respectively. More details about the types and proportions of the identified repeats are presented in Fig. 1.
Types of highly and moderately repeated DNA sequences in F. pratensis genome. The proportion of each repeat type or family is shown inside parenthesis. Repeat proportions were calculated by annotating clusters representing at least 0.01% of the genome in RepeatExplorer. For mobile elements, members of each repeat family is indicated in outer layers.
Satellite repeat identification
Using the tandem repeat analyzer (TAREAN) pipeline42, eighteen different satellite repeats representing 3.93% of the genome in the F. pratensis were in silico identified (Table 1). Five clusters were identified as high confident putative satellites and thirteen clusters as low confident putative satellites. The proportion of each of these tandem repeats in the genome and other details including consensus length and satellite probability are shown in Table 1. All these repeats exhibited graphics with a globular or circular layout and showed C (Connected component index) and P (Pair completeness index) indices close to 1. Both C and P are close to 1 for putative satellite repeats. Although the parameter C facilitates the identification of clusters representing tandemly repeated genomic sequences by being close to 1, it does not efficiently discriminate clusters derived from satellite DNA from those representing other types of tandem repeats. In fact, the proportions of broken pairs are much higher in tandem repeats scattered in the genome in a high number of short arrays42. Fraction of complete pairs in cluster is assessed by the pair completeness index P42.
Physical mapping of satellite repeats
The consensus monomers of five tandem repeats were selected for PCR and FISH analysis based on their frequency in −B and +B genotypes and satellite probability (Table 2, Supplementary Table S1).
Labelled Fp-Sat2, Fp-Sat84 (Fig. 2) and Fp-Sat37 (Fig. 3) repeats produced hybridization signals on the A chromosomes. Only repeat Fp-Sat2 was not detected on the B chromosomes (Fig. 2C). Fp-Sat84 and Fp-Sat37 repeats also produced signals on Bs (Figs. 2F and 3). Fp-Sat37 produced stronger signals on the Bs compare with the A chromosomes (Fig. 3). Fp-Sat84 gave rather dispersed signals on all F. pratensis chromosomes although some signal clusters were detected (Fig. 2F). In order to test whether this repeat is species-specific, we performed FISH with the chromosomes of the allopolyploid species F. arundinacea Schreb. (2n = 6x = 42, FpFgFg’ genome); a species were F. pratensis contributed a subgenome36,43,44. In total, a subset of 14 of the 42 chromosomes showed Fp-Sat84-specific signals (Figs. 2H,I and 3) although the signals were not evenly distributed and some cross-hybridization occurred on a few other chromosomal regions possibly due to probe patterns itself or inter-subgenomic translocations (Fig. 2I).
Localization of Fp-Sat2 and Fp-Sat84 tandem repeats on the Festuca chromosomes. (A–C) DAPI stained chromosomes and banding patterns of Fp-Sat2 (green signals) on F. pratensis chromosomes. (D–F) DAPI stained chromosomes and Fp-Sat84 localization showing overall distribution on the F. pratensis chromosomes (red signals). Arrows point to the B chromosomes. (G–I) DAPI stained chromosomes of F. arundinacea (genome FpFgFg’), distribution patterns of Fp-Sat84 repeat on its chromosomes and the resulting merged image. Scale bar = 5 µm.
FISH with the FP-Sat2 repeat resulted in chromosome-specific banding patterns in F. pratensis and proved to be a suitable cytological marker for chromosomes identification (Fig. 2C). BLAST analysis revealed that FP- Sat2 has 82% similarity with a middle repetitive sequence of L. perenne (accession AF063226.1) and 78% similarity with a cDNA from gliadin genes in Triticum aestivum (accession MG560141.1) (Table 1). Furthermore, Fp-Sat84 and Fp-Sat162 showed 98% and 96% similarity with satellite TR7 (accession JX624133.1) and TR15 (accession JX624136.1) respectively, which previously were identified from sequencing data of chromosome 4F38. No similarity was found for Fp-Sat131, FP-Sat37, Fp-Sat71, Fp-Sat75, Fp-Sat130, Fp-Sat159, Fp-Sat175, Fp-Sat236, and the B chromosome enriched repeats FP-Sat48 and FP-Sat253.
Identification of B chromosome-specific repeats in F. pratensis
We identified plants harbouring 0B, 1B, 2B and 3B chromosomes. A similar number of Bs were present in various cells of both roots and leaves of each B chromosome containing plant (Fig. 4) so Bs might be retained in all the different plant tissues. The Bs can easily be distinguished in F. pratensis by the morphology, being roughly one third to one fourth the length of the A chromosomes and with a subterminal centromere (Fig. 4A). Comparative analysis in RepeatExplorer recognized the tandem repeats Fp-Sat37, Fp-Sat48, Fp-Sat84 and Fp-Sat253 with monomers of 347, 667, 343 and 372 bp, respectively as potentially B-enriched sequences (Fig. 5; Table 2). The full-length consensus monomer of the B-specific Fp-Sat253 (372 bp) is shown in Fig. 6, rebuilt from the most frequent k-mers (11-,15-, 19-, 23- and 27-mers) by TAREAN. The B-specific candidate repeats were amplified from the +B genotype by PCR, although all of these repeats could also be amplified by using the −B genotype DNA as template. Fp-Sat253 primers produced a rather clear distinct band using the genomic DNA of −B genotype but a smear product (with occasionally a faint band) using the genomic DNA of +2B genotype after electrophoresis of PCR products in agarose gel (Supplementary Figs. S1 and S2).
B chromosome painting in F. pratensis. (A) A DAPI-stained metaphase cell of a F. pratensis plant with two B chromosomes. Enlarged Bs are also shown in top-right corner of the picture; (B,C) B-chromosome labelling with the B-specific probes Fp-Sat253 and Fp-Sat48, respectively. Arrow heads indicate B chromosomes; (D) Combined picture resulting from merging A, B and C subfigures showing painted B chromosomes. (E) FISH using Fp-Sat48 (green signals) probe generated strong signals on Bs and faint signals on some of the A chromosomes. FISH also resulted in B-specific localization of Fp-Sat253 (red signals), although week telomeric associated signals on one of the A chromosome pairs (arrows) were detectable after an extended exposure time. Arrowheads point to the B chromosomes; (F) FISH on leaf interphase nuclei detecting the number of B chromosomes in F. pratensis plants. This picture shows the result of FISH using B specific probes Fp-Sat48 (green) and Fp-Sat253 (red) on nuclei from a plant having 2 Bs. Scale bar = 5 µm.
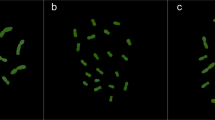
(A) In silico identification of consensus sequences of satellite DNA enriched in F. pratensis B chromosomes. Potential B enriched repeats (red dots) were identified by comparison of genomic proportions of individual repeat families in −B and +B genotypes. Dots on the plot represent individual repeat clusters. The vertical axis is the relative abundance in −B and +2B genomes indicated by the percentage cluster size in the +B genotype (harboring 2 B chromosomes) divided by its size in the −B (lacking B chromosome) genotype, resulting in a value of 50 for sequences with the same sized in both genotypes. (B) Graphical layouts (generated by TAREAN) of repeats potentially enriched in Bs.
Sequence logos for the full-length consensus monomer of the B specific Fp-Sat253 (372 bp) satellite DNA in F. pratensis. This sequence was rebuilt from the most frequent k-mers (11-,15-, 19-, 23- and 27-mers). K-mer frequencies for each satellite is calculated from Illumina reads and is used to reconstruct their most conserved fragments. These fragments are then merged to create full-length consensus monomers shown here for Fp-Sat253. The height of the letters reflects the frequencies bases in the corresponding k-mers. The arrows indicate forward and reverse primer sequences used for the amplification of Fp-Sat253 satellites by PCR.
FISH showed a B-specific localization of Fp-Sat253 along both chromosome arms (Fig. 4B). Additional A-located signals were found from Fp-Sat253 repeat after an extension of the exposure time (Fig. 4E). Probe Fp-Sat48 generated strong signals on Bs and a few faint signals on some of the A chromosomes at normal exposure ranges (Fig. 4C; Supplementary Fig. S3). Fp-Sat253 repeat seems to be localized at the telomeric or subtelomeric regions while Fp-Sat48 concentrated at the pericentromeric regions of the B chromosomes (Fig. 4D). Fp-Sat37 satellite sequence produced strong signals on Bs, although it was available on the essential A chromosomes as well (Fig. 3). Among the 5 studies satellite repeats, FP-Sat2 was the only repeat that did not produce signals on the B chromosomes. Because of the difficulty of chromosome preparation due to the small size of F. pratensis roots, we were interested to test FISH on leaf nuclei to identify +B genotypes. FISH using both B-specific probes on nuclei isolated from leaf proved to be a fast and efficient way of screening the +B plants (Fig. 4F).
Discussion
Low-pass Illumina sequencing of the genomic −B/+B DNA followed by the repeat characterization from the sequence reads using RepeatExplorer41 and TAREAN42 pipelines provided a comprehensive view about the types, abundance and organization of repetitive DNA sequences in F. pratensis. A low coverage sequence of 0.4x was more than enough to elucidate the repetitive sequence landscape. The chromosomal distribution of five different repeats was determined by FISH. The analysis revealed that 61% of the genome of F. pratensis is composed of repetitive sequences. Such a high proportion of repeats is commonly found in plants which also show a wide range of genome size45.
Transposons (with the majority being LTR retrotransposons), were the most predominant repeats. The abundance of retrotransposons could be related to their active mechanisms of self-proliferation throughout the plant genome via RNA mediates46,47,48. Among LTR retrotransposons, the frequency of Ty3-gypsy retrotransposons (32.97%) was three times as high as that of Ty1-copia (7.78%) in the genome. In Avena sativa and Triticum aestivum, Ty3-gypsy elements occupy 40% and 46% of the genomes, respectively, approximately three times more abundant than Ty1-copia elements in both species49,50. A high proportion of the Ty3-gypsy superfamily has also been reported for chromosome 4F of F. pratensis38. Based on FISH data, Křivánková, et al.39 reported Athila as a highly frequent retroelement in F. pratensis genome. Our data showed that Retand51, with the frequency of 17.28% of the genome, is at least three times abundant than Athila retroelement (Fig. 1).
Satellites are believed to be amplified via uneven crossover and slippage replication52,53. These highly repeated tandem arrays provide useful markers for the analysis of chromosome evolution. According to our data, a relatively high number of satellite repeats represent 4% of the F. pratensis genome (Table 1). Satellite sequences compose 1% of Capsicum annuum L (3.1 pg/1 C)54, 3% of the genome in Glycine max L. (1.1 pg/1C)55, 2% of the genome in Avena sativa (12.85 pg/1C)49 and less than 0.1% of the genome in Passiflora edulis (1.26 pg/1C)56.
Křivánková, et al.39 established a FISH -based karyotype of F. pratensis based on different tandem repeats and rDNA probes. We also identified a tandem repeat (Fp-Sat2) that produce distinct banding patterns on all the A chromosome pairs although it was the only repeat not available on the B chromosomes. Repeat Fp-Sat84 showed a rather dispersed distribution along F. pratensis chromosomes and labelled the corresponding subgenome in F. arundinacea 6x. This evidence confirms previous conclusions that F. pratensis is one of the parental species of F. arundinacea43,44. Although Fp-Sat84 didn’t show sequence similarity to LTR retrotransposons, dispersed patterns of this repeat suggests that Fp-Sat84 may has evolved from retrotransposons or retrotransposition may has played a role in its evolution and distribution throughout the genome57,58.
Fp-Sat48 and Fp-Sat253 were identified as the first B chromosome-specific repeats of F. pratensis (Fig. 4). Fp-Sat37 also showed to be a B-enriched satellite sequence, but it was also abundant on some of the A chromosomes mainly as pericentromeric bands (Fig. 3). The identification of weak A chromosome-located signals and the presence of PCR amplicons in −B samples of four of the five studied repeats suggests that the Bs of F. pratensis resulted from the A chromosome complement of the same species. A similar evolutionary link between A and B chromosomes was found for a number of different species33. As an example, Bs in Plantago lagopus are composed of mainly 5S rDNA-derived sequences and various types of repetitive elements59,60. We did FISH using 5S and 45S rDNA probes on a +B genotype of F. pratensis but no trace of the corresponding signals was observed on B chromosomes (data not shown). In rye and Ae. speltoides, the Bs are enriched in organelle DNA61,62. However, based on RepeatExplorer analysis, abundance of organelle DNA sequence were not different between −B and +B DNA genomes of F. pratensis in the repsent study.
Conclusions
We analyzed the repetitive DNA content of F. pratensis by low-pass Illumina sequencing of the genomic −B/+B DNA followed by the repeat characterization from the sequence reads using RepeatExplorer41 and TAREAN42 pipelines. 61% of the genome is composed of repetitive sequences and transposable elements are the major component of the genome. 4% of the F. pratensis genome is composed of satellite repeats which were partly harnessed as cytological markers for chromosome identification and genome analysis in the genus. Furthermore, the identified B-specific tandem repeats can be used as a marker to trace the dynamics of supernumerary chromosomes to provide direct insight into the cellular basis of the B chromosome drive mechanism in F. pratensis in future.
Methods
Plant material
An ecotype of F. pratensis (2n = 2x = 14) originally collected from Iran and received from the Research Institute of Forests and Rangelands of Iran, showed to carry B chromosomes. Single seeds of F. pratensis were inspected by chromosome counting using a dropping method as described in Abdolmalaki, et al.63 for the presence or absence of B chromosomes. For this, one or two primary root tips of each single seed were analyzed and the corresponding seed was transferred to a pod to grow. We could identify plants harbouring 0B, 1B, 2B and 3B chromosomes. DNA samples were extracted from the leaves of F. pratensis individual plants possessing 0B (−B) or 2B chromosomes (+2B) and sequenced using the Illumina HiSeq 2500 system at low coverage.
Sequence analysis and identification of A- and B-specific repeats
Identification of F. pratensis repeats specific to the essential A and supernumerary B chromosomes was performed by similarity-based clustering of Illumina reads using the RepeatExplorer pipeline41,55. The CG content of the reads was estimated from the F. pratensis genomes using the Illumina R1 and R2 output files with the FastQC tool. All sequences were filtered by quality with 95% of bases equal to or above the quality cut of value of 10. Paired reads were joint into a single FASTA file using FASTA interlacer. The clustering was performed using the default setting of 90% similarity over 55% of the read length. Comparative clustering analysis for the identification of B chromosome specific repeats were performed after assigning 0B and 2B prefix codes to the respective reads of −B and +2B genotypes. Each set of data was down-sampled to 500000 reads to represent 0.1 coverage of each genome followed by their concatenation into a single data set. The analysis allowed for the identification of repeats enriched on B chromosomes. Monomer lengths and consensus sequences of the identified satDNA families were determined using TAREAN42. WebLogo64 was used to generate de Brujin graph of the reconstructed consensus sequences of satellites predicted by TAREAN. Consensus monomers of satellites were used to design PCR primers for amplification of satellites including Fp-Sat2, Fp-Sat37, Fp-Sat48, Fp-Sat84 and Fp-Sat253 (Table 2).
Preparation of FISH probes
Genomic DNA of −B/+2B genotypes were used as template in polymerase chain reaction (PCR). The PCR mixture contained 25 ng template DNA, 5 pmol of each primer, 2.5 mM of each dNTP, 2.5 mM MgCl2 and 0.5 U Taq polymerase. Amplification was for 5 min at 94 °C, followed by 30 cycles of 45 s at 94 °C, 45 s at 58–61 °C (depending on primers), 90 s at 72 °C and a final step of 7 min at 72 °C. PCR products were purified by ethanol precipitation. PCR products were labelled with Atto-488-11-dUTP or Atto-550-11-dUTP using a nick translation kit (Jena Bioscience, Jena, Germany), recovered by ethanol precipitation and used as a probe in FISH.
Slide preparation for FISH
Seeds were germinated on moist filter paper in Petri dishes for 3–6 days at room temperature. In order to stop cell division at metaphase, root tips of about 1–1.5 cm length were cut and placed in microtubes punched at the top. Roots were subjected to nitrous oxide (N2O) gas at 10 bar pressure for 2 hours. Treated roots were fixed in ice-cold 90% acetic acid for 10 minutes, and then transferred to 75% ethanol and stored at −20 °C until use. Roots were washed in ice-cold water, followed by 0.01 M citrate buffer each for 10 minutes. Meristematic part of the roots (about 1 to 1.5 mm of the root tips) were cut and placed in a microtube containing 30 µl enzyme mixture (0.7% cellulase (CalBiochem 219466), 0.7% cellulase R10 (Duchefa C8001), 1% cytohelicase (Sigma C8274) and 1% pectolyase (Sigma P3026) prepared in 0.01 M citrate buffer, (0.01 M citric acid and 0.01 M sodium citrate, pH 4.8). Root tips were digested at 37 °C for 60 to 90 minutes and slides from digested root tips were prepared using dropping method according to Abdolmalaki, et al.63. The slides were fixed in 4% paraformaldehyde in 1× PBS (3 mM NaH2PO4, 7 mM Na2HPO4, 0.13 M NaCl, pH 7.4) for 10 minutes at room temperature, followed by washing in 2× SSC (0.3 M sodium chloride, 0.03 M sodium citrate, pH 7.0) and dehydrating in 96% ethanol.
For the preparation of nuclei, a small leaf of approximately 2 cm2 in total, from each F. pratensis plant was chopped with a sharp razor blade in 0.5 ml 45% acetic acid. The nuclei suspension was filtered through a Partec (Partec, Münster, Germany) 30 µm nylon mesh filter. 10 µl of the nuclei suspension was dropped on a microscopic slide and briefly heated to dry for subsequent FISH.
Fluorescence in situ hybridization (FISH)
Twenty µl of hybridization mixture was placed on each slide and covered with a plastic coverslip. Slides were then denatured at 80 °C for 2 minutes on a hot plate. Hybridization mixture contained 2× SSC, 50% formamide, 20% dextran sulfate, 1 µg sheared salmon testes DNA and 20–30 ng of each labeled probes. For hybridization, slides were incubated in a humidified plastic container at 37 °C. Coverslips were removed and slides were washed in 2× SSC for 20 minutes in a water bath at 56 °C. Slides were dehydrated in 96% ethanol and dried at room temperature. A drop of Vectashield mounting medium (Vector Laboratories) containing 1 µg/ml DAPI (4′, 6-diamidino-2-phenylindole) was added to each slide as counterstain and a glass coverslip was applied. Slides were inspected with a fluorescence Olympus BX51 microscope (Olympus, Japan) and images were captured, using a DP72 digital camera (Olympus, Japan).
References
Clayton, W. D. & Renovize, S. A. Genera Gramium. Grasses of the world. KewBull Addit Ser 13, 389 (1986).
Soreng, R. J. & Davis, J. I. Phylogenetic and character evolution in the grass family (Poaceae): simultaneous analysis of morphology and chloroplast DNA restriction site character sets. Bot. Rev. 64, 1–85 (1998).
Tzvelev, N. N. Grasses of the Soviet Union. Zlaki SSSR, Naukova Dumka, Leningrad. (1976).
Ospina-Gonzalez, J. C. New lectotypifications and new synonyms in Festuca (Poaceae, Pooideae, Loliinae) from the central Andes. Phytotaxa 247, 247–258 (2016).
Parsa, A. Flore del Iran. Vol. 5 736–745 (Publication du Ministere de L’education Museum d’Histoire Naturelle de Tehran, 1950).
Bor, N. L. In Flora Iranica (ed. Reschinger, K. H.) Ch. 105–141, (Akademische Druck, (1970).
Loureiro, J., Kopecký, D., Castro, S., Santos, C. & Silveira, P. Flow cytometric and cytogenetic analyses of Iberian Peninsula Festuca spp. Plant Syst. Evol. 269, 89–105 (2007).
Smarda, P., Bures, P., Horova, L., Foggi, B. & Rossi, G. Genome size and GC content evolution of Festuca: ancestral expansion and subsequent reduction. Ann. Bot. 101, 421–433 (2008).
Hand, M. L., Cogan, N. O., Stewart, A. V. & Forster, J. W. Evolutionary history of tall fescue morphotypes inferred from molecular phylogenetics of the Lolium-Festuca species complex. BMC Evol. Biol. 10, 303 (2010).
Abyaneh, E. B., Hejazi, S. M. H., Gholami, S. & Khaniki, G. B. Study on the cytotaxonomy of some species of the genus Festuca (Poaceae). Iran. J. Sci. Technol. 42, 1689–1696 (2018).
Sheidai, M. & Shabestarei, E. B. Cytotaxonomy of some Festuca species and populations in Iran. Acta Bot. Croat. 66, 143–151 (2007).
Bosemark, N. O. On accessory chromosomes in Festuca pratensis. I. Cytological inrestigations. Hereditas, 346–376 (1954).
Bosemark, N. O. Accessory chromosomes in Festuca pratensis. Hereditas 36, 366–368 (1950).
Jauhar, P. P. In Monographs on Theoretical and Applied Genetics Vol. 18 (eds. Frankel, R. et al.) (Springer Science & Business Media, (1993).
Tsitlenok, S. I. & Vaishlya, O. B. B chromosomes in Festuea pratensis. Botanicheskii Zhurnal 73, 1116–1119 (1988).
Pereira, H. S. et al. Rye (Secale cereale) supernumerary (B) chromosomes associated with heat tolerance during early stages of male sporogenesis. Ann. Bot. 119, 325–337 (2017).
Holmes, D. S. & Bougourd, S. M. B-chromosome selection in Allium schoenoprasum L. natural populations. Heredity 63, 83–87 (1989).
Harper, J. et al. B chromosomes are associated with redistribution of genetic recombination towards lower recombination chromosomal regions in perennial ryegrass. J. Exp. Bot. 69, 1861–1871 (2018).
Jones, R. N. & Rees, H. B Chromosomes. (Academic Press, 1982).
Jauhar, P. P. & Crane, C. F. Meiotic behavior and effects of B chromosomes in tall fescue. J. Eur. Econ. Assoc. 81, 156–159 (1990).
Maumus, F. & Quesneville, H. Impact and insights from ancient repetitive elements in plant genomes. Curr. Opin. Plant Biol. 30, 41–46 (2016).
Mehrotra, S. & Goyal, V. Repetitive sequences in plant nuclear DNA: Types, distribution, evolution and function. Genom. Proteom. Bioinf. 12, 164–171 (2014).
Bao, W., Kojima, K. K. & Kohany, O. Repbase Update, a database of repetitive elements in eukaryotic genomes. Mobile. DNA 6, 11 (2015).
Piégu, B., Bire, S., Arensburger, P. & Bigot, Y. & evolution. A survey of transposable element classification systems–a call for a fundamental update to meet the challenge of their diversity and complexity. Mol. Phylogenet. Evol. 86, 90–109 (2015).
Wicker, T. et al. A unified classification system for eukaryotic transposable elements. Nat. Rev. Genet. 8, 973 (2007).
Flynn, J. M., Long, M., Wing, R. A. & Clark, A. G. Evolutionary dynamics of abundant 7 bp satellites in the genome of Drosophila virilis. BioRxiv, 693077 (2019).
Gong, Z. et al. Repeatless and repeat-based centromeres in potato: implications for centromere evolution. Plant Cell 24, 3559–3574 (2012).
Garrido-Ramos, M. Satellite DNA: An evolving topic. Genes 8, 230 (2017).
Belyayev, A. et al. Natural history of a satellite DNA family: From the ancestral genome component to species-specific sequences, concerted and non-concerted evolution. Int. J. Mol. Sci. 20, 1201 (2019).
Elder, J. F. & Turner, B. J. Concerted evolution of repetitive DNA sequences in eukaryotes. Q. Rev. Biol. 70, 297–320 (1995).
Ugarkovic, D. & Plohl, M. Variation in satellite DNA profiles-causes and effects. EMBO 2, 5955–5959 (2002).
Samoluk, S. S., Robledo, G., Bertioli, D. & Seijo, J. G. Evolutionary dynamics of an at-rich satellite DNA and its contribution to karyotype differentiation in wild diploid Arachis species. Mol. Genet. Genomics 292, 283–296 (2017).
Marques, A., Klemme, S. & Houben, A. Evolution of plant B chromosome enriched sequences. Genes 9, 515 (2018).
Lower, S. S., McGurk, M. P., Clark, A. G. & Barbash, D. A. Satellite DNA evolution: old ideas, new approaches. Curr. Opin. Genet. Dev. 49, 70–78 (2018).
Kopecký, D. et al. Physical distribution of homoeologous recombination in individual chromosomes of Festuca pratensis in Lolium multiflorum. Cytogenet. Genome Res. 129, 162–172 (2010).
Thomas, H. M., Harper, J. A., Meredith, M. R., Morgan, W. G. & King, I. P. Physical mapping of ribosomal DNA sites in Festuca arundinacea and related species by in situ hybridization. Genome 40, 406–410 (1997).
Kopecký, D., Lukaszewski, A. & Doležel, J. Meiotic behaviour of individual chromosomes of Festuca pratensis in tetraploid Lolium multiflorum. Chromosome Res. 16, 987 (2008).
Kopecký, D. et al. Flow sorting and sequencing meadow fescue chromosome 4F. Plant Physiol. 163, 1323–1337 (2013).
Křivánková, A., Kopecký, D., Stočes, Š., Doležel, J. & Hřibová, E. Repetitive DNA: A versatile tool for karyotyping in Festuca pratensis Huds. Cytogenet. Genome Res. 151, 96–105 (2017).
Majka, J., Książczyk, T., Kiełbowicz-Matuk, A., Kopecký, D. & Kosmala, A. Exploiting repetitive sequences and BAC clones in Festuca pratensis karyotyping. PloS One 12, e0179043 (2017).
Novák, P., Neumann, P., Pech, J., Steinhaisl, J. & Macas, J. RepeatExplorer: a Galaxy-based web server for genome-wide characterization of eukaryotic repetitive elements from next-generation sequence reads. Bioinformatics 29, 792–793 (2013).
Novák, P. et al. TAREAN: a computational tool for identification and characterization of satellite DNA from unassembled short reads. Nucleic Acids Res. 45, e111–e111 (2017).
Humphreys, M. et al. Discriminating the ancestral progenitors of hexaploid Festuca arundinacea using genomic in situ hybridization. Heredity 75, 171 (1995).
Chandrasekharan, P. & Thomas, H. Studies in Festuca. 5. Cytogenetic relationships between species of Bovinae and Scariosae. Z Pflanzenzucht 65, 345–354 (1971).
Macas, J. et al. In depth characterization of repetitive DNA in 23 plant genomes reveals sources of genome size variation in the legume tribe Fabeae. PLoS One 10, e0143424 (2015).
Vitte, C. & Panaud, O. LTR retrotransposons and flowering plant genome size: emergence of the increase/decrease model. Cytogenet. Genome Res. 110, 91–107 (2005).
Kumar, A. & Bennetzen, J. L. Plant retrotransposons. Annu. Rev. Genet. 33, 479–532 (1999).
Vicient, C. M. & Casacuberta, J. M. Impact of transposable elements on polyploid plant genomes. Ann. Bot. 120, 195–207 (2017).
Liu, Q. et al. The repetitive DNA landscape in Avena (Poaceae): chromosome and genome evolution defined by major repeat classes in whole-genome sequence reads. BMC Plant Biol. 19, 226 (2019).
Wicker, T. et al. Impact of transposable elements on genome structure and evolution in bread wheat. Genome Biol. 19, 103 (2018).
Kejnovsky, E. et al. Retand: a novel family of gypsy-like retrotransposons harboring an amplified tandem repeat. Mol. Genet. Genomics 276, 254–263 (2006).
Biscotti, M. A., Olmo, E. & Heslop-Harrison, J. P. Repetitive DNA in eukaryotic genomes. Chromosom Res. 23, 415–420 (2015).
Charlesworth, B., Sniegowski, P. & Stephan, W. The evolutionary dynamics of repetitive DNA in eukaryotes. Nature 371, 215–220 (1994).
Zhou, H. C., Waminal, N. E. & Kim, H. H. In silico mining and FISH mapping of a chromosome-specific satellite DNA in Capsicum annuum L. Genes Genomics 41, 1001–1006 (2019).
Novák, P., Neumann, P. & Macas, J. Graph-based clustering and characterization of repetitive sequences in next-generation sequencing data. BMC Bioinformatics 11, p.378 (2010).
Pamponét, V. C. C. et al. Low coverage sequencing for repetitive DNA analysis in Passiflora edulis Sims: citogenomic characterization of transposable elements and satellite DNA. BMC Genomics 20, 262 (2019).
Macas, J., Koblížková, A., Navrátilová, A. & Neumann, P. Hypervariable 3′ UTR region of plant LTR-retrotransposons as a source of novel satellite repeats. Genes 448, 198–206 (2009).
Vondrak, T. et al. Characterization of repeat arrays in ultra-long nanopore reads reveals frequent origin of satellite DNA from retrotransposon‐derived tandem repeats. Plant J., https://doi.org/10.1111/tpj.14546 (2019).
Dhar, M. K., Kour, J. & Kaul, S. Origin, behaviour, and transmission of B chromosome with special reference to Plantago lagopus. Genes 10, 152 (2019).
Kumke, K. et al. Plantago lagopus B chromosome is enriched in 5S rDNA-derived satellite DNA. Cytogenet. Genome Res. 148, 68–73 (2016).
Ruban, A. et al. B chromosomes of Aegilops speltoides are enriched in organelle genome-derived sequences. PLos One 9, e90214 (2014).
Martis, M. M. et al. Selfish supernumerary chromosome reveals its origin as a mosaic of host genome and organellar sequences. Proc. Natl. Acad. Sci. USA 109, 13343–13346 (2012).
Abdolmalaki, Z., Mirzaghaderi, G., Mason, A. S. & Badaeva, E. D. Molecular cytogenetic analysis reveals evolutionary relationships between polyploid Aegilops species. Plant Syst. Evol. 305, 459–475 (2019).
Crooks, G. E., Hon, G., Chandonia, J. M. & Brenner, S. E. WebLogo: A sequence logo generator. Genome Res. 14, 1188–1190 (2004).
Qi, X. et al. A different function for a member of an ancient and highly conserved cytochrome P450 family: from essential sterols to plant defense. Proc. Natl. Acad. Sci. USA 103, 18848–18853 (2006).
Acknowledgements
This work was supported by the University of Kurdistan, Iran.
Author information
Authors and Affiliations
Contributions
G.M. conceived and designed research and wrote the manuscript. R.E. conducted experiments. A.H. assisted in genomic DNA sequencing and contributed to critical discussions and manuscript revisions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ebrahimzadegan, R., Houben, A. & Mirzaghaderi, G. Repetitive DNA landscape in essential A and supernumerary B chromosomes of Festuca pratensis Huds. Sci Rep 9, 19989 (2019). https://doi.org/10.1038/s41598-019-56383-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-56383-1
This article is cited by
-
Meiotic segregation and post-meiotic drive of the Festuca pratensis B chromosome
Chromosome Research (2023)
-
The non-Mendelian behavior of plant B chromosomes
Chromosome Research (2022)
-
Out of patterns, the euchromatic B chromosome of the grasshopper Abracris flavolineata is not enriched in high-copy repeats
Heredity (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.