Abstract
A systematic review and network-meta analysis (NMA) were performed to estimate significance of the anxiolytic effect of lavender essential oil taken as silexan capsules versus other comparators (i.e., placebo/paroxetine/lorazepam). The outcome of interest was Hamilton Anxiety Scale (HAMA). Weighted mean differences (WMD) were calculated to estimate the treatment effect at the confidence interval of 95%. League tables were generated using treatment effect, for all pairwise comparisons, where WMD < 0 favors the column-defining treatment. Five studies were identified with a total of 524 participants receiving treatment with silexan 80 mg and 121 participants taking silexan 160 mg. The NMA results indicated that consumption of silexan 160 mg resulted in higher decline of HAMA score [WMD −1.14 (−1.10, 3.39)] in comparison to silexan 80 mg, placebo [−2.20 (−4.64, 0.24)] and paroxetine [−1.24 (−5.34, 2.85)]. The effect of silexan 80 mg was observed to be same as that of paroxetine. Overall, silexan 160 mg was noticed to be a more efficient treatment giving significant decline in HAMA score across other comparators. However, no improvements in HAMA score was observed for the group receiving lorazepam 0.5 mg when compared to silexan 160 mg, silexan 80 mg, paroxetine 20 mg, and placebo.
Similar content being viewed by others
Introduction
Among psychiatric disorders, anxiety disorders are more frequent than others1. The global prevalence of anxiety disorders based on the data from 87 studies in 44 countries around the world was estimated to be 7.3%2. The frequency of anxiety in the population differs greatly between countries. The lifetime prevalence of anxiety disorders in United States was reported to be 28.8%3, 14.5% in Europe4 and 20% in Australia5. Anxiety disorders have a huge economic impact on society affecting mainly working-age population. Thus, the prevalence of anxiety disorders in United States for the 18–64 years old population reaches 33.7%6. Anxiety often manifests as a symptom of other psychiatric disorders and also frequently precedes their onset7,8. Anxiety disorders are also more comorbid than other mental disorders9. Their high comorbidity extended to other psychiatric disorders as well as physical illnesses.
Due to chronic nature of anxiety disorders, patients suffer from them for a long time, sometimes decades. However, therapeutic interventions, such as pharmacotherapy and psychotherapy and their combination, are usually beneficial and improve patient’s quality of life often resulting in complete recovery10,11. The meta-analysis estimating efficacy of treatments for anxiety disorders suggested that positive effects of pharmacotherapy exceeded those of psychotherapy1. Therefore, pharmacotherapy is often critically important in managing patients with anxiety disorders. However, the chronic character of anxiety disorders and requirements of the long-term treatment set very high safety and compliance standards for the medications and bring phytotherapy as a treatment option12.
Phytotherapy has been gaining popularity in the treatment of anxiety13,14,15,16,17 with many GABA-modulating medicines of herbal origin undergoing preclinical and clinical investigations18. Particular attention has been paid to anxiolytic-like effects of essential oils, among which a lavender essential oil demonstrated the best pharmacological profile19,20,21.
Lavender (Lavandula angustifolia Miller or Lavandula officinalis Chaix) has a long history of traditional use and its essential oil was found to possess a wide range of biological effects22,23. Evidence of the effectiveness of the lavender essential oil in the pharmacotherapy of mental disorders led to the development of Silexan, which is a standardized essential oil of L. angustifolia flowers prepared by steam distillation24. Silexan is approved in Germany for the treatment of restlessness related to anxiety and marketed as LASEA®25.
Silexan was found to contain 36.8% of linalool and 34.2% of linalyl acetate26. Other components of the lavender essential oil present in substantial quantities include monoterpene alcohol lavandulol, its ester lavandulyl acetate, and bicyclic monoterpenoids borneol, eucalyptol (1,8-cineole) and camphor27,28. Linalool also demonstrated anxiolytic properties in several animal models29,30 and possessed stress-relieving effect in humans under the experimental stress31. Studies on molecular mechanisms of pharmacological effects of lavender essential oil revealed that its effect on CNS could be attributed to the inhibition of voltage dependent calcium channels26. Unlike some other monoterpenes found in the lavender essential oil, linalool and linalyl acetate significantly inhibited voltage dependent calcium channels26. In another study, lavender essential oil was demonstrated to possess affinity to the NMDA receptor and SERT28. Binding to NMDA receptor was also observed for major constituents of lavender essential oil linalyl acetate and linalool, while only linalool demonstrated significant binding to SERT28. Further results of the clinical, randomized, placebo-controlled, double-blind, cross-over study on healthy men using positron emission tomography technology suggested that the anxiolytic effect of lavender essential oil (given as Silexan) could be attributed to the serotonergic system changes, particularly at the 5-HT1A receptor level32.
To date, two reviews have been reported in the literature evaluating the efficacy of Silexan in anxiety-disorders33,34. Review by Kasper33 in 2013 was not a systematic review, liable to bias, and the author did not combine results of individual studies using meta-analysis. The systematic review and meta-analysis by Möller et al.34 estimated the efficacy of Silexan in sub-threshold anxiety disorders and reported significant reduction in anxiety as measured by HAMA scale. The present systematic review further builds on our existing knowledge on the efficacy of Silexan in the management of anxiety disorders by comparing the efficacy of different strengths of lavender oil using a network meta-analysis technique. This is a unique technique and is considered superior to meta-analysis when direct multiple comparisons between the intervention and control group do not exist in the literature35. In addition, our systematic review has evaluated safety of lavender oil as well.
Methods
A systematic review was performed to identify potential research papers across 8 databases from inception till December 14, 2017. The NMA was performed, in accordance with the recommendations made by the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses (PRISMA)36, to estimate the overall effectiveness of lavender versus placebo and other medicinal products in anxiety disorders and its adverse effects.
Population intervention comparator and outcomes
-
Population of interest: Patients with anxiety disorder.
-
Intervention: Lavender given orally (capsules).
-
Comparators: Placebo or any other medicinal product.
-
Outcome: Change in Hamilton Anxiety Scale (HAMA) total score of the patients.
Search strategies
Eight electronic databases including Embase via Ovid, MEDLINE via Ovid, Cochrane, PubMed, AMED, PsycINFO, SCOPUS and EBSCOHost using the following search terms: lavender OR lavandula OR alhucema OR aspic OR lavandin OR lavender essential oil OR lavender oil OR lavandin oil OR aspic oil OR lavender extract OR Silexan OR Lasea OR Kalms Lavender One-A-Day OR CalmAid OR MS 1265 OR linalool OR linalyl acetate AND anxiety.
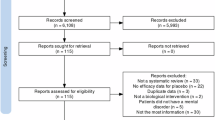
All the relevant papers were identified and imported to an EndNote file to create a combined library. Duplicate records were removed, where appropriate. Title and abstracts were screened by two authors independently and disagreements were resolved through discussions. Full-texts were downloaded and considered for inclusion based on the inclusion/exclusion criteria described below. Also, we manually reviewed the references of the included studies to identify other relevant studies. Additional details about the studies identified are shown in Fig. 1.
Study selection
The following criteria were used to further align the search with the specific outcomes of interest.
Inclusion criteria
-
All English language studies from inception till December 14, 2017 were considered eligible for inclusion in this systematic review.
-
Only human experimental studies, which are clinical trials comparing the effect of lavender (as a tea, powder or capsule) were considered for potential inclusion in order to assess causal assertions. Comparators considered appropriate were: a placebo, reference/control group, or any other active regimen, which was compared with the lavender formulation given orally.
-
The main outcome of interest is the change in Hamilton Anxiety Scale (HAMA) total score of the patients.
Exclusion criteria
-
Systematic reviews, all observational studies, letter to the editors, case reports, case series, personal opinion, qualitative studies, and reviews/communications focusing on individual experience of the use of lavender were excluded.
-
In addition, any data that are not published, reports, and thesis were excluded.
-
Experimental studies on animals and studies using lavender for aromatherapy were excluded.
Outcome of interest
-
Primary outcomes: Change in Hamilton Anxiety Scale (HAMA) total score of the patients.
-
Secondary outcome: Safety and tolerability of lavender preparations as a medicine.
Data extraction
Data were extracted independently by two review authors (WSY, TMK) and in the case there was a conflict, a third independent assessor AVD assisted in revalidating the data and resolving the conflict. A structured, pilot-tested data collection form was designed to collect data from individual studies. In addition to the data related to the outcomes of interest, data on the number of authors, year of publication, study design, setting and country where the study took place, sample size, patients’ mean age and gender, and inclusion criteria and exclusion criteria were also extracted. The results for the outcome measures included in this review were summarized as mean and SD difference from baseline to endpoint in both the intervention and control groups. When not reported, the mean and SD difference were calculated provided sufficient data were available.
Data analysis
The risk of bias was assessed individually for each RCT included in this review using Cochrane risk of bias assessment tool37. Two authors (MAH, ZJ) assessed risk of bias independently and disagreements were resolved through discussion.
The network meta-analysis (NMA) was performed using STATA version 14®. Random effects model was used and weighted mean differences with 95% confidence interval (CI) were calculated for all continuous outcome measures to compare the effectiveness of the intervention. P-value less than 0.05 was considered statistically significant. Furthermore, in order to generate forest plots for NMA, respective pairwise comparisons for the treatment effect were carried. League tables were created using treatment effect calculated based on weighted mean difference (MD; 95% CI) for all pairwise comparisons. A league table, a square matrix consisting of all pairwise comparison within a meta-analysis, empowers researcher/reader to directly compare the direction and magnitude of treatment effect, encouraging easier interpretation of results35. A negative (−) MD indicate decline in the score for the HAMA and SAS scales.
Results
Included studies
Of the 982 studies screened, five randomized control trials were included in this review (Fig. 1).
Characteristics of the studies and type of interventions
To assess the effect of lavender in treating anxiety, interventions included in the 5 studies were placebo, 80 mg/d silexan, 160 mg/d silexan, 0.5 mg/d lorazepam, and 20 mg/d paroxetine. The study by Woelk & Schalfke38 had a treatment phase of 6 weeks whereas the other 4 studies by Kasper et al.39,40,41,42 had a treatment phase of 10 weeks. All these studies were conducted in Germany. The primary outcome in each study was the change in total score of HAMA. Safety and tolerability of the interventions were assessed by the reporting of adverse events that occurred over the course of the study. Details on study characteristics are available in Table 1.
Although the recruitment criteria for all 5 studies included participants of both genders and of any ethnicity, less than 31% of participants in each study were male and almost all were Caucasian. Woelk & Schlafke38 conducted a multi-center, double-blind, randomised phase III study assessing the comparative effect of 80 mg silexan daily versus 0.5 mg lorazepam daily on a sample size of 78 participants with 23.4% being male. In this study, 40 participants received silexan 80 mg. Kasper et al.39 investigated the effect of 80 mg silexan daily against placebo in a randomized, double-blind, multi-center trial with 20.2% of its 228 participants being male. In this study, 104 participants received treatment with silexan 80 mg.
Following that, Kasper et al.40 conducted a randomized, double-blind, double-dummy, multi-centre trial with four parallel groups receiving 160 mg silexan daily (121 patients), 80 mg silexan daily (135 patients), 20 mg paroxetine daily, and placebo with 26.4% of male in the sample size of 616 participants. Subsequently, Kasper et al. in 201541 investigated the effect of 80 mg silexan daily and used placebo for the reference group. The study design was a randomized, double-blind trial with 28.2% of the study’s 179 participants being male38. In this study, 86 participants were assigned to the silexan 80 mg group. The following year, Kasper et al. published another paper to again investigate the effect of 80 mg silexan daily compared to placebo42. In this double-blind, randomized, parallel-group multi-centre trial, the sample size was 362 participants and 30.8% of them were male. The silexan 80 mg group consisted of 159 participants42. Further details on participant characteristics are shown in Table 2.
Risk of bias
In general, the overall risk of bias was low for most of the domains for the included studies (Figs. 2 and 3). In particular, risk was low for selection bias, performance bias and detection bias for all the studies. One of the studies has unclear risk of bias for allocation concealment (Figs. 2 and 3). In other biases, studies were deemed to be high risk of bias as they were funded by the manufacturer.
Effect on HAMA score: Primary outcome
To estimate the effect of all interventions on HAMA, five studies were included and a NMA was performed to compare and contrast the effect on the HAMA scale. The network plots demonstrate the organization of all available evidences. The width of the lines represents the number of trials and the size of node represents the sample size (Fig. 4).
Overall analysis
Outcome of interventions versus placebo at HAMA scale
Multivariate analysis was performed to estimate the effect of all the active treatments silexan 160 mg, silexan 80 mg, lorazepam 0.5 mg, and paroxetine 20 mg versus placebo as the reference arm. Results have revealed that effect of placebo was not superior in comparison to silexan. Overall, silexan 160 mg −4.963 [−7.167–−2.759], p ≤ 0.001, I2 = 0.00%, Tau2 = 0), and silexan 80 mg −3.820 [−5.261–−2.380], p ≤ 0.001, I2 = 0.00%, Tau2 = 0), were noticed to have a significantly higher decline in HAMA score in comparison to placebo. Detailed comparison of all treatment with reference arm is shown in Table 3.
Furthermore, a pairwise analysis was carried out to estimate the comparative effect of all the treatments versus placebo and other interventions. Using the pairwise comparison, a league table was generated to present all possible pairwise comparisons between any two of the five treatments (Fig. 5). Treatment effect, that is, the weighted mean difference of each pairwise comparison was calculated and reported along with the 95% CI. It was revealed that lorazepam 0.5 mg had no positive effect and did not cause a decline in HAMA score. The treatment with silexan 160 mg was found to be effective in comparison to placebo, paroxetine 20 mg, and lorazepam 0.5 mg. Overall, the effect of silexan 160 mg [−1.14 (−1.10, 3.39)], assessed on the basis of declining HAMA score, was superior in comparison to silexan 80 mg. In addition, the effect of silexan 80 mg [−1.06 (−3.32,1.21)] was observed to be slightly better than that of placebo. The decline in HAMA score was higher for silexan 160 mg in comparison to placebo and paroxetine 20 mg. Overall, all interventions including placebo were observed to be more effective in declining HAMA score compared to lorazepam 0.5 mg. Details are described in Table 4.
Safety of silexan
In the analyzed 5 studies, adverse events, attributable to silexan use, consisted mainly of gastrointestinal problems such as nausea, eructation or breath odour, and diarrhea. A number of patients also reported having headaches in the 2015 study by Kasper et al.41 Nevertheless, the number of patients that experienced these mild adverse events comprised of a small percentage of the sample size. No serious adverse event was found to be linked to the use of silexan.
Fatigue is a known side effect of lorazepam. In the study conducted by Woelk & Schlafke38, 16.2% of patients receiving treatment with lorazepam had fatigue whereas none in the silexan group experienced fatigue. However, 10.0% in the silexan group had nausea whereas only 2.7% had nausea in the lorazepam group.
The actual number and type of total adverse events experienced by the participants in the study conducted by Kasper et al.36,37 were not reported in details. Nonetheless, it was noted by Kasper et al.37 that the participants receiving treatment with silexan experienced a 3% increase in risk of gastrointestinal problems compared to those in the placebo group. Further details on adverse events reported in the studies can be found in Table 5.
Discussion
To date, perhaps it is the first NMA estimating the effect of oral dosage form of lavender i.e. silexan in comparison to placebo, paroxetine and lorazepam on the anxiety score at the HAMA scale. Systematic reviews and meta-analysis have proven to be an effective tool to estimate the clinical efficacy and safety of herbs, and further comparative analysis using network meta-analysis will provide researcher an opportunity to estimate the clinical efficacy and safety of a herb versus various treatments or comparators43. Therefore, the current systematic review and NMA will serve as a useful tool in analyzing the use of silexan in clinical settings in an effective manner. The overall analysis revealed that administration of silexan 160 mg produced a higher decline in HAMA score [WMD −1.14 (−1.10, 3.39)] in comparison to silexan 80 mg. The effect of silexan 80 mg was observed to be similar to paroxetine 20 mg. Overall, the treatment with silexan 160 mg resulted in more significant decline in the HAMA score across all the comparators and therefore can be considered as the most effective anxiolytic intervention for the included studies. However, it should be noted that the dose of lorazepam used in the included RCT, and subsequently in this NMA, was only 0.5 mg which is the lowest daily dose recommended. Comparative efficacy of silexan with higher dose of lorazepam is not known and needs further research.
The lavender in different forms has been used in cosmetic and therapeutic applications for centuries. These applications were driven by unique scent of lavender essential oil. This essential oil became popular in aromatherapy and its therapeutic effectiveness in this form has been assessed clinically in a number of trials. Particularly, aromatherapy with lavender essential oil was effective in managing preoperative stress and anxiety in various settings significantly decreasing anxiety level compared to placebo44,45,46. However, similar interventions applied after surgery revealed no difference with control group on anxiety level and mental stress47,48.
The aromatherapy with lavender essential oil improved quality of sleep and reduced level of anxiety in patients with coronary artery disease49, significantly reduced anxiety in older patients with acute coronary syndrome50 and myocardial infarction51. Lower indexes for perceived stress and objective stress were observed in the intensive care unit patients receiving aromatherapy with lavender essential oil52. The inhaled lavender essential oil helped to alleviate anxiety in burn patients53 and postpartum women54.
However, no significant long-term improvements were observed in control of anxiety for patients suffering from cancer receiving aromatherapy treatment with lavender essential oil55. No significant difference in the anxiety level was also observed between cancer patients receiving inhalation with lavender essential oil during radiotherapy and the placebo group56. Overall, the aromatherapy with lavender as an anxiolytic agent has not been supported by sufficient evidence of therapeutic efficacy57. However, our NMA was based on the systemic administration of the lavender essential oil (Silexan) and demonstrated positive evidences for its anxiolytic effect. More detailed studies are required for the conclusion regarding other methods for the administration of lavender essential oil, particularly topically or via inhalation.
Strengths and limitations
The exclusion of non-English articles can be one of the limitations of this NMA. This was mainly due to a lack of resources to translate articles written in other languages. There is a slight possibility that some relevant data from these non-English literature, if combined with the current analysis, might change the significance of lavender effects versus other comparators. In addition, all the studies investigating the effect of lavender used its essential oil in form of Silexan capsules. These studies were from Germany and the outcome might be potentially affected by the patients’ genetics. To get more comprehensive analysis and conclusions on the effect of lavender and particularly Silexan capsules, similar clinical studies using ethnically more diverse populations are required. The lavender preparations like Silexan capsules might have different results on HAMA scale in other races from different genetic makeup. Moreover, the current analysis estimated only the overall effect of the treatments on HAMA score, the current study was unable to compare the multiple treatments based on the decline in symptoms, severity and relapse of anxiety, which was mainly due to variable reporting of the results across the selected studied for the NMA.
Overall, the other bias was observed across all the five included studies. However, one of the main strength of this NMA is heterogeneity among the one-on-one comparison and pairwise comparison was 0% which reflects no inconsistencies among the studies. A number of factors including similarities between the study populations across RCTs included in this NMA as all studies were conducted in Germany, similarities in intervention (all in the form of Silexan capsules) and duration of follow-up and use of same tool (HAMA) to assess anxiety in all included studies could explain low heterogeneity observed in NMA.
Conclusion
Results (through pairwise comparisons) revealed statistically significant effect of silexan 160 mg versus placebo, silexan 80 mg and paroxetine 20 mg. In addition, the effect of paroxetine 20 mg was also observed superior than placebo and silexan 80 mg. However, silexan consumption has shown some gastrointestinal side effect such as nausea, eructation or breath odor and diarrhea, which were tolerated by the patients recruited in the silexan arm. There is a need of more methodological strong studies to further investigate the effect of silexan among the patients from other regions to get a comprehensive picture about its clinical efficacy and safety.
Data availability
All materials, data and associated protocols are available to readers without restrictions.
References
Bandelow, B. & Michaelis, S. Epidemiology of anxiety disorders in the 21st century. Dialogues in clinical neuroscience 17, 327 (2015).
Baxter, A., Scott, K., Vos, T. & Whiteford, H. Global prevalence of anxiety disorders: a systematic review and meta-regression. Psychological medicine 43, 897–910 (2013).
Kessler, R. C. & Wang, P. S. The descriptive epidemiology of commonly occurring mental disorders in the United States. Annu. Rev. Public Health 29, 115–129 (2008).
Alonso, J. & Lépine, J.-P. Overview of key data from the European Study of the Epidemiology of Mental Disorders (ESEMeD). The Journal of clinical psychiatry (2007).
McEvoy, P. M., Grove, R. & Slade, T. Epidemiology of anxiety disorders in the Australian general population: findings of the 2007 Australian National Survey of Mental Health and Wellbeing. Australian and New Zealand Journal of Psychiatry 45, 957–967 (2011).
Kessler, R. C., Petukhova, M., Sampson, N. A., Zaslavsky, A. M. & Wittchen, H. U. Twelve‐month and lifetime prevalence and lifetime morbid risk of anxiety and mood disorders in the United States. International journal of methods in psychiatric research 21, 169–184 (2012).
Kessler, R. C., Ruscio, A. M., Shear, K. & Wittchen, H.-U. In Behavioral neurobiology of anxiety and its treatment 21–35 (Springer, 2009).
Stein, D. J., Scott, K. M., de Jonge, P. & Kessler, R. C. Epidemiology of anxiety disorders: from surveys to nosology and back. Dialogues in clinical neuroscience 19, 127 (2017).
Toft, T. et al. Mental disorders in primary care: prevalence and co-morbidity among disorders. Results from the functional illness in primary care (FIP) study. Psychological medicine 35, 1175–1184 (2005).
Baldwin, D. S. et al. Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. Journal of Psychopharmacology 28, 403–439 (2014).
Bandelow, B., Michaelis, S. & Wedekind, D. Treatment of anxiety disorders. Dialogues in clinical neuroscience 19, 93 (2017).
Sarris, J., Panossian, A., Schweitzer, I., Stough, C. & Scholey, A. Herbal medicine for depression, anxiety and insomnia: a review of psychopharmacology and clinical evidence. European neuropsychopharmacology 21, 841–860 (2011).
Ernst, E. Herbal remedies for anxiety–a systematic review of controlled clinical trials. Phytomedicine 13, 205–208 (2006).
Sarris, J. Herbal medicines in the treatment of psychiatric disorders: a systematic review. Phytotherapy Research 21, 703–716 (2007).
Sarris, J., McIntyre, E. & Camfield, D. A. Plant-based medicines for anxiety disorders, part 1. CNS drugs 27, 207–219 (2013).
Sarris, J., McIntyre, E. & Camfield, D. A. Plant-based medicines for anxiety disorders, part 2: a review of clinical studies with supporting preclinical evidence. CNS drugs 27, 301–319 (2013).
Yeung, K. S., Hernandez, M., Mao, J. J., Haviland, I. & Gubili, J. Herbal medicine for depression and anxiety: A systematic review with assessment of potential psycho‐oncologic relevance. Phytotherapy Research 32, 865–891 (2018).
Savage, K., Firth, J., Stough, C. & Sarris, J. GABA‐modulating phytomedicines for anxiety: A systematic review of preclinical and clinical evidence. Phytotherapy Research 32, 3–18 (2018).
Edris, A. E. Pharmaceutical and therapeutic potentials of essential oils and their individual volatile constituents: a review. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives 21, 308–323 (2007).
Dobetsberger, C. & Buchbauer, G. Actions of essential oils on the central nervous system: An updated review. Flavour and Fragrance Journal 26, 300–316 (2011).
de Sousa, D., Hocayen, P., Andrade, L. & Andreatini, R. A systematic review of the anxiolytic-like effects of essential oils in animal models. Molecules 20, 18620–18660 (2015).
Lis‐Balchin, M. & Hart, S. Studies on the mode of action of the essential oil of LavenderLavandula angustifolia P. Miller. Phytotherapy Research: An International Journal Devoted to Pharmacological and Toxicological Evaluation of Natural Product Derivatives 13, 540–542 (1999).
Cavanagh, H. & Wilkinson, J. Biological activities of lavender essential oil. Phytotherapy Research 16, 301–308 (2002).
Kasper, S. et al. Silexan in anxiety disorders: Clinical data and pharmacological background. The world journal of biological psychiatry 19, 412–420 (2018).
Lasea. Silexan, https://everything-lavender.com/lasea-lavender-oil-silexan-capsule.html (2018).
Schuwald, A. M. et al. Lavender oil-potent anxiolytic properties via modulating voltage dependent calcium channels. PloS one 8, e59998 (2013).
Da Porto, C., Decorti, D. & Kikic, I. Flavour compounds of Lavandula angustifolia L. to use in food manufacturing: Comparison of three different extraction methods. Food Chemistry 112, 1072–1078 (2009).
López, V., Nielsen, B., Solas, M., Ramírez, M. J. & Jäger, A. K. Exploring pharmacological mechanisms of lavender (Lavandula angustifolia) essential oil on central nervous system targets. Frontiers in pharmacology 8, 280 (2017).
Cline, M. et al. Investigation of the anxiolytic effects of linalool, a lavender extract, in the male Sprague-Dawley rat. AANA journal 76 (2008).
Linck, Vd. M. et al. Effects of inhaled Linalool in anxiety, social interaction and aggressive behavior in mice. Phytomedicine 17, 679–683 (2010).
Höferl, M., Krist, S. & Buchbauer, G. Chirality influences the effects of linalool on physiological parameters of stress. Planta medica 72, 1188–1192 (2006).
Baldinger, P. et al. Effects of Silexan on the serotonin-1A receptor and microstructure of the human brain: a randomized, placebo-controlled, double-blind, cross-over study with molecular and structural neuroimaging. International Journal of Neuropsychopharmacology 18 (2015).
Kasper, S. An orally administered lavandula oil preparation (Silexan) for anxiety disorder and related conditions: an evidence based review. International Journal of Psychiatry in Clinical Practice 17(sup1), 15–22 (2013).
Möller, H. J., Volz, H. P., Dienel, A., Schläfke, S. & Kasper, S. Efficacy of Silexan in subthreshold anxiety: meta-analysis of randomised, placebo-controlled trials. European Archives of Psychiatry and Clinical Neuroscience 269(2), 183–193 (2019).
Rouse, B., Chaimani, A. & Li, T. Network meta-analysis: an introduction for clinicians. Internal and Emergency Medicine 12(1), 103–111 (2017).
Moher, D., Liberati, A., Tetzlaff, J. & Altman, D. G. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Annals of internal medicine 151, 264–269 (2009).
Lundh, A. & Gøtzsche, P. C. Recommendations by Cochrane Review Groups for assessment of the risk of bias in studies. BMC medical research methodology 8, 22 (2008).
Woelk, H. & Schläfke, S. A multi-center, double-blind, randomised study of the Lavender oil preparation Silexan in comparison to Lorazepam for generalized anxiety disorder. Phytomedicine 17, 94–99 (2010).
Kasper, S. et al. Silexan, an orally administered Lavandula oil preparation, is effective in the treatment of ‘subsyndromal’anxiety disorder: a randomized, double-blind, placebo controlled trial. International clinical psychopharmacology 25, 277–287 (2010).
Kasper, S. et al. Lavender oil preparation Silexan is effective in generalized anxiety disorder–a randomized, double-blind comparison to placebo and paroxetine. International Journal of Neuropsychopharmacology 17, 859–869 (2014).
Kasper, S., Anghelescu, I. & Dienel, A. Efficacy of orally administered Silexan in patients with anxiety-related restlessness and disturbed sleep–A randomized, placebo-controlled trial. European neuropsychopharmacology 25, 1960–1967 (2015).
Kasper, S., Volz, H.-P., Dienel, A. & Schläfke, S. Efficacy of Silexan in mixed anxiety–depression–A randomized, placebo-controlled trial. European neuropsychopharmacology 26, 331–340 (2016).
Izzo, A. A., Hoon‐Kim, S., Radhakrishnan, R. & Williamson, E. M. A critical approach to evaluating clinical efficacy, adverse events and drug interactions of herbal remedies. Phytotherapy Research 30, 691–700 (2016).
Fayazi, S., Babashahi, M. & Rezaei, M. The effect of inhalation aromatherapy on anxiety level of the patients in preoperative period. Iranian journal of nursing and midwifery research 16, 278 (2011).
Hosseini, S., Heydari, A., Vakili, M., Moghadam, S. & Tazyky, S. Effect of lavender essence inhalation on the level of anxiety and blood cortisol in candidates for open-heart surgery. Iranian journal of nursing and midwifery research 21, 397 (2016).
Wotman, M. et al. The efficacy of lavender aromatherapy in reducing preoperative anxiety in ambulatory surgery patients undergoing procedures in general otolaryngology. Laryngoscope investigative otolaryngology 2, 437–441 (2017).
Seifi, Z. et al. The effect of lavender essential oil on anxiety level in patients undergoing coronary artery bypass graft surgery: A double-blinded randomized clinical trial. Iranian journal of nursing and midwifery research 19, 574 (2014).
Bikmoradi, A. et al. Effect of inhalation aromatherapy with lavender essential oil on stress and vital signs in patients undergoing coronary artery bypass surgery: A single-blinded randomized clinical trial. Complementary therapies in medicine 23, 331–338 (2015).
Karadag, E., Samancioglu, S., Ozden, D. & Bakir, E. Effects of aromatherapy on sleep quality and anxiety of patients. Nursing in critical care 22, 105–112 (2017).
Bahrami, T. et al. Effect of aromatherapy massage on anxiety, depression, and physiologic parameters in older patients with the acute coronary syndrome: A randomized clinical trial. International journal of nursing practice 23, e12601 (2017).
Najafi, Z., Taghadosi, M., Sharifi, K., Farrokhian, A. & Tagharrobi, Z. The effects of inhalation aromatherapy on anxiety in patients with myocardial infarction: a randomized clinical trial. Iranian Red Crescent Medical Journal 16 (2014).
Cho, E. H., Lee, M.-Y. & Hur, M.-H. The effects of aromatherapy on intensive care unit patients’ stress and sleep quality: A nonrandomised controlled trial. Evidence-Based Complementary and Alternative Medicine 2017 (2017).
Seyyed-Rasooli, A. et al. Comparing the effects of aromatherapy massage and inhalation aromatherapy on anxiety and pain in burn patients: A single-blind randomized clinical trial. Burns 42, 1774–1780 (2016).
Kianpour, M., Mansouri, A., Mehrabi, T. & Asghari, G. Effect of lavender scent inhalation on prevention of stress, anxiety and depression in the postpartum period. Iranian journal of nursing and midwifery research 21, 197 (2016).
Soden, K., Vincent, K., Craske, S., Lucas, C. & Ashley, S. A randomized controlled trial of aromatherapy massage in a hospice setting. Palliative medicine 18, 87–92 (2004).
Graham, P., Browne, L., Cox, H. & Graham, J. Inhalation aromatherapy during radiotherapy: results of a placebo-controlled double-blind randomized trial. Journal of Clinical Oncology 21, 2372–2376 (2003).
Perry, R., Terry, R., Watson, L. & Ernst, E. Is lavender an anxiolytic drug? A systematic review of randomised clinical trials. Phytomedicine 19, 825–835 (2012).
Author information
Authors and Affiliations
Contributions
All authors contributed to the preparation of the manuscript for publication as shown below. W.S.Y.- drafted the work, conception, data acquisition, interpretation of data. A.V.D.- data analysis, re-validation of data, Z.J.- revised manuscript drafts substantively. M.A.H.-revised manuscript drafts substantively. T.M.K.-conception, data acquisition, interpretation of data, data analysis, revised manuscript drafts substantively.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yap, W.S., Dolzhenko, A.V., Jalal, Z. et al. Efficacy and safety of lavender essential oil (Silexan) capsules among patients suffering from anxiety disorders: A network meta-analysis. Sci Rep 9, 18042 (2019). https://doi.org/10.1038/s41598-019-54529-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-54529-9
This article is cited by
-
Lavender oil preparation Silexan is effective in mild-to-moderate major depression: a randomized, placebo- and reference-controlled trial
European Archives of Psychiatry and Clinical Neuroscience (2024)
-
Beneficial effects of Silexan on co-occurring depressive symptoms in patients with subthreshold anxiety and anxiety disorders: randomized, placebo-controlled trials revisited
European Archives of Psychiatry and Clinical Neuroscience (2023)
-
Efficacy of Silexan in patients with anxiety disorders: a meta-analysis of randomized, placebo-controlled trials
European Archives of Psychiatry and Clinical Neuroscience (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.