Abstract
It remains unclear whether mitochondrial modulators (MMs) are beneficial in the treatment of obsessive–compulsive and related disorders. Thus, in an attempt to answer this clinical question, we performed a systematic review and a random-effects meta-analysis of double-blind, randomized, placebo-controlled trials. The primary outcome was change in overall symptoms as measured using standardized rating scales. Other outcomes were response to treatment; improvement in anxiety-related scales scores, depression-related scale scores, Clinical Global Impression Severity Scale (CGI-S) scores, and Sheehan Disability Scale (SDS) scores; all-cause discontinuation; and individual adverse events. We calculated the standardized mean differences for continuous outcomes and risk ratios for dichotomous outcomes with 95% confidence intervals. We reviewed 17 studies (n = 629, 72.62% female; duration = 2–20 weeks; mean age = 30.47 years) of MMs: eicosapentaenoic acid (K = 1), folic acid (K = 1), lithium (K = 1), N-acetylcysteine (K = 10), inositol (K = 3), and silymarin (K = 1). MMs outperformed placebo in overall improvement in symptoms (p < 0.01) and in improving anxiety-related scale scores (p = 0.05). Subgroup analysis of individual MMs revealed that although overall symptoms were better improved by N-acetylcysteine (p < 0.01) and lithium (p = 0.04), no MMs outperformed placebo in terms of improving anxiety-related scale scores. Neither pooled nor individual MMs outperformed placebo in improving response to treatment, depression-related scale scores, CGI-S scores, SDS scores, or all-cause discontinuation. N-acetylcysteine was no more associated with a higher incidence of individual adverse events including gastrointestinal symptoms, than placebo. In conclusion, N-acetylcysteine was beneficial in the treatment of obsessive–compulsive and related disorders. However, further study with larger samples is necessary to confirm this finding.
Similar content being viewed by others
Introduction
Obsessive–compulsive disorder (OCD), characterized by the presence of obsessions or compulsions, or both [1], is a common chronic mental disorder; the lifetime prevalence of OCD among adults in the United States was 2.3% [2]. Both pharmacotherapy, such as antidepressants, and psychotherapeutic methods, such as exposure and response prevention therapy, are among the primary treatments for OCD [3]. However, not all individuals with OCD respond to these treatments; thus, new therapeutic agents with new pharmacological mechanisms of action that differ from those of conventional psychotropic drugs are needed.
Oxidative stress and free radicals, inflammation, and mitochondrial dysfunction have recently been shown to play key roles in the development of OCD [4, 5]. Mitochondrial dysfunction and oxidative stress are tightly dependent on each other [4, 5]. For example, a positron emission tomographic study demonstrated inflammation in not only the basal ganglia but also in the cortico-striato-thalamo-cortical circuit in individuals with OCD [6]. A cross-sectional study showed that serum levels of 8-hydroxydeoxyguanosine, which is a marker of oxidative DNA damage, were significantly higher in individuals with OCD than in those without OCD and were lower in the patients who received treatment for OCD [7].
To date, 17 double-blind, randomized, placebo-controlled trials of mitochondrial modulators such as N-acetylcysteine and inositol in treating OCD have been conducted (Table 1). Of these, ten focused on N-acetylcysteine, a precursor of endogenous antioxidant glutathione that has antioxidant, anti-inflammatory, and neuroprotective properties [8, 9]. N-acetylcysteine may also have mitochondrial biogenesis effects [10]. Results regarding the efficacy of mitochondrial modulators against OCD were noted to be inconsistent among the double-blind, randomized, placebo-controlled trials (Table 1), and so it remains unclear whether mitochondrial modulators are beneficial in the treatment of OCD. The discrepancy in results may be attributable to the smallness of the samples in these trials. A meta-analysis produces a weighted summary result, in which more weight is given to larger studies. Combining results from more than one study has the advantage of increasing statistical power, which is often inadequate in studies with small sample sizes [11]. Therefore, we conducted a systematic review and meta-analysis of these studies to examine the efficacy, tolerability, and safety of mitochondrial modulators in the treatment of mood disorders in individuals with OCD and OCD-related disorders.
Subjects and methods
The systematic review and meta-analysis were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines (Appendix S1) [12] and were registered with the Open Science Framework (https://osf.io/h4ypt). The literature search, data transfer accuracy, and statistics were each double-checked by at least two of the authors.
Search strategy, inclusion criteria, and data extraction
A systematic literature review was performed to characterize patients, interventions, comparisons, and outcome strategies. Patients were adults and children with OCD and OCD-related disorders (body dysmorphic disorder, hoarding disorder, trichotillomania, or excoriation [skin picking] disorder) [1]. Interventions consisted of mitochondrial modulator therapy: N-acetylcysteine, acetyl-l-carnitine, S-adenosylmethionine, coenzyme Q10, alpha-lipoic acid, creatine monohydrate, vitamin C, vitamin D, vitamin E, melatonin, omega-3 polyunsaturated fatty acids, inositol, silymarin, folic acid, resveratrol, sulforaphane, and lithium [4, 13,14,15]. The control conditions involved placebo. Outcomes are listed in the following section.
We identified eligible studies indexed in Embase, PubMed, and the Cochrane Library databases, published in any language, and containing data from the inception of the study to March 14, 2022 (Fig. S1). The search terms in PubMed and the Cochrane Library databases included “placebo”; “random*”; a specific disorder (one of the following: obsessive–compulsive disorder, OCD, body dysmorphic disorder, hoarding disorder, trichotillomania, excoriation, or body-focused repetitive behaviors); and one specific mitochondrial modulator (N-acetylcysteine, acetyl-l-carnitine, S-adenosylmethionine, coenzyme Q10, alpha-lipoic acid, creatine monohydrate, vitamin C, vitamin D, vitamin E, melatonin, omega-3 polyunsaturated fatty acids, docosahexaenoic acid, eicosapentaenoic acid, inositol, silymarin, folic acid, resveratrol, sulforaphane, or lithium). The search terms for Embase included names of the aforementioned disorders, randomized controlled trial and names of the aforementioned individual mitochondrial modulators. The authors also searched the ClinicalTrials.gov (http://clinicaltrials.gov/) and the International Clinical Trials Registry Platform (http://www.who.int/ictrp/en/) to ensure that the search was comprehensive and to minimize publication bias. The reference lists of the retrieved publications were also searched for additional relevant published and unpublished studies, including conference abstracts.
Outcomes and data synthesis
We extracted data from the included articles and entered the data in spreadsheets. We first reviewed the study design and the characteristics of patients and treatments in the studies.
The primary outcome was change in overall symptoms, measured using standardized rating scales such as the Yale–Brown Obsession Compulsion Scale [16], the Massachusetts General Hospital Hair Pulling Scale [17], and the Yale–Brown Obsessive–Compulsive Scale Modified for Neurotic Excoriation [18]. Other outcomes included study-defined response to treatment; scores on anxiety-related scales, such as the Hamilton Anxiety Rating Scale [19] and Beck Anxiety Inventory [20]; scores on depression-related scales, such as the Hamilton Depression Rating Scale [21], the Children’s Depression Inventory [22], the Beck Depression Inventory [23], and the Montgomery–Åsberg Depression Rating Scale [24]; scores on the Clinical Global Impression Severity Scale (CGI-S) [25]; scores on the Sheehan Disability Scale (SDS) [26]; rate of all-cause discontinuation of treatment; and incidence of individual adverse events.
Statistical analysis
To perform the pairwise meta-analysis, we used Review Manager software (version 5.4.1; Copenhagen, The Nordic Cochrane Centre) [27]. Because of the potential for heterogeneity across the included studies, we used a random-effects model [28]. We calculated the standardized mean differences (SMDs) for continuous outcomes and risk ratios for dichotomous outcomes with 95% confidence intervals (CIs). Heterogeneity was tested with the I2 statistic, whereby I2 of ≥50% indicated considerable heterogeneity [11]. As shown in Fig. 1, we found considerable heterogeneity for the primary outcome in our meta-analysis. Only N-acetylcysteine and lithium outperformed placebo in improving overall symptoms. Because only one study focused on lithium, we conducted a sensitivity analysis to include only studies of N-acetylcysteine, using meta-regression to evaluate the following potential confounding factors: (1) mean age (years), (2) diagnosis (OCD or OCD-related disorders), (3) study duration (in weeks), (4) total number of individuals, and (5) percentage of female participants. Because reports of studies of all mitochondrial modulators other than N-acetylcysteine did not include sufficient data for us to perform a meta-analysis regarding safety outcomes, we conducted these meta-analyses for only studies of N-acetylcysteine. We used a funnel plot and Egger’s regression to assess whether small-study effects were attributable to publication bias [29]. For the meta-regression analysis, constructing the funnel plot, and assessing results of Egger’s regression, we used the Comprehensive Meta-Analysis software version V3 (Biostat Inc., Englewood, NJ, USA). We assessed the methodological quality of the included articles according to the Cochrane risk of bias criteria [11].
Studies are listed by names of the first authors and year of publication, with abbreviations for drugs listed in parentheses (A, B, C). A Listing by mitochondrial modulator name. EPA eicosapentaenoic acid, FA folic acid, INO inositol, LIT lithium, NAC N-acetylcysteine, SIL silymarin. B Listing by diagnosis. EXC excoriation disorder, OCD obsessive–compulsive disorder, TRI trichotillomania. C Listing according to adults (A) or children (C).
Results
Results of the literature search
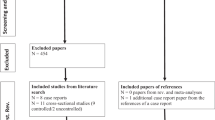
Of the 119 retrieved publications, 37 duplicates and 64 publications based on an abstract or a title review were excluded. In addition, one post hoc study [30] was excluded after a full-text review (Fig. S1). No additional studies were retrieved from the clinical trial registries or in manual searches with reference to the previous systematic reviews [31,32,33,34,35,36,37,38]. Ultimately, we reviewed 17 studies involving a total of 629 patients [39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55].
Study characteristics
Table 1 lists the characteristics of each study and the characteristics of patients included in each study. Eicosapentaenoic acid (1 study) [41], folic acid (1 study) [43], lithium (1 study) [55], N-acetylcysteine (10 studies) [44,45,46,47, 49,50,51,52,53,54], inositol (3 studies) [39, 40, 48], and silymarin (1 study) [42] were the mitochondrial modulators studied. Twelve studies included individuals with OCD [39,40,41, 43,44,45,46,47, 50, 51, 53, 55], four others included individuals with trichotillomania [42, 48, 52, 54], and the remaining study included individuals with excoriation disorder [49]. Thirteen studies included adults [39,40,41, 43, 44, 47,48,49,50,51, 53,54,55], three others included children [45, 46, 52], and one included both adults and children [42]. In all studies except one of inositol [39], the investigators assessed mitochondrial modulator add-on to conventional treatment, such as pharmacotherapy (in most studies, serotonin reuptake inhibitors) or psychotherapy. Patients’ mean age was 30.47 years, 72.62% of participants were female, and study duration ranged from 2 to 20 weeks. In two studies of inositol [39, 40], that is, one of eicosapentaenoic acid [41] and one of silymarin [42], a crossover design was used. For the two inositol studies, data from the first treatment phase could be used to analyze efficacy outcomes, although the data were from a complete analysis. Except for one study of folic acid [43], none of the trials was sponsored by pharmaceutical companies. The methodological quality of most studies was high (Fig. S2).
Overall symptoms
In all 17 studies, the mitochondrial modulators outperformed placebo (n = 632, SMD = −0.61, 95% CI = −0.95 to −0.26, p < 0.01, I2 = 75%; Table 2, Fig. 1). According to subgroup analysis of individual mitochondrial modulators, placebo was outperformed by N-acetylcysteine (in 10 studies involving 453 patients; SMD = −0.77, 95% CI = −1.26 to −0.29, p < 0.01, I2 = 83%) and by lithium (in 1 study involving 20 patients; SMD = −0.98, 95% CI = −1.92 to −0.03, p = 0.04, I2 was not applicable; Fig. 1). When study results were pooled together, the p value of Egger’s regression for the outcome was 0.15, and the funnel plot was visually symmetric (Fig. S3).
A meta-regression analysis demonstrated no associations between effect size and any potential confounding factors (Table 3). Although the funnel plot for only N-acetylcysteine studies was visually symmetric (Fig. S4), the p value of Egger’s regression for the meta-analysis that included only studies of N-acetylcysteine were 0.03.
Other efficacy outcomes and all-cause discontinuation of therapy
When study results were pooled together, mitochondrial modulators outperformed placebo in improving anxiety symptoms in 9 studies involving 411 patients (SMD = −0.20, 95% CI = −0.39, −0.00, p = 0.05, I2 = 0%; Table 2, Fig. 2). Subgroup analysis of individual mitochondrial modulators revealed that N-acetylcysteine was marginally superior to placebo in improving anxiety symptoms (p = 0.09), but other individual mitochondrial modulators did not outperform placebo (Fig. 2). Pooled mitochondrial modulators have marginally outperformed placebo in improving, SDS scores (p = 0.05; Table 2, Fig. S5) and response to treatment (p = 0.07; Table 2, Fig. S6). Subgroup analysis of individual mitochondrial modulators revealed that N-acetylcysteine marginally outperformed placebo in improving SDS scores (p = 0.05; Fig. S5) and response to treatment (p = 0.09; Fig. S6). Neither pooled nor individual mitochondrial modulators outperformed placebo in terms of improving CGI-S scores and depression-related scale scores or rates of all-cause discontinuation of treatment (Table 2, Figs. S7, S8 and S9).
Studies are listed by names of the first authors and year of publication, with abbreviations for drugs listed in parentheses (A, B, C). A Listing by mitochondrial modulator name. FA folic acid, INO inositol, LIT lithium, NAC N-acetylcysteine, SIL silymarin. B Listing by diagnosis. EXC excoriation disorder, OCD obsessive–compulsive disorder, TRI trichotillomania. C Listing according to adults (A) or children (C).
Safety outcomes
In comparison with placebo, N-acetylcysteine was not associated with a higher incidence of insomnia, somnolence, fatigue, dizziness, headache, dry mouth, nausea/vomiting, diarrhea, constipation, or rash (Table 4).
Discussion
This is the first systematic review and meta-analysis in which the efficacy, acceptability, and safety of mitochondrial modulators (eicosapentaenoic acid, folic acid, inositol, lithium, N-acetylcysteine, and silymarin) were compared with those of conventional treatment for OCD and OCD-related disorders. Except for one study of inositol [39], all studies involved mitochondrial modulator add-on to conventional treatment, such as serotonin reuptake inhibitors. As a group, mitochondrial modulators have significantly improved overall symptoms of OCD and anxiety. In particular, lithium and N-acetylcysteine significantly improved overall OCD symptoms, and N-acetylcysteine marginally improved anxiety symptoms. As a group, mitochondrial modulators also marginally outperformed placebo in improving SDS scores and response to treatment. In particular, N-acetylcysteine marginally improved SDS scores and response to treatment. Moreover, N-acetylcysteine did not carry any risk of adverse events. Thus, N-acetylcysteine as an antidepressant adjunct might be a novel treatment option for individuals with OCD and OCD-related disorders who received conventional treatment.
Our meta-analysis showed that the overall effect size of N-acetylcysteine was moderate (SMD = −0.77) and comparable with the antidepressant effect sizes of aripiprazole (SMD = −1.35), haloperidol (SMD = −0.82), and risperidone (SMD = −0.59), as indicated by a previous meta-analysis [56]. Lithium was able to improve overall OCD symptoms, but because only one study involving lithium (in 20 patients) was included in our systematic review, further study with larger samples is needed to confirm our findings.
We found considerable heterogeneity in the results of N-acetylcysteine with regard to the primary outcome, but our meta-regression analysis demonstrated no associations between effect size and any confounding factors. The funnel plot was visually symmetric; however, because the p value of Egger’s regression was 0.03, small-study effects might have influenced this result [29]. Another limitation of our study was that the trials included in our meta-analysis were of short duration. Other limitation of our study was that there was only one trial for eicosapentaenoic acid, folic acid, lithium, and silymarin. Therefore, longer studies, as well as larger samples, are necessary to confirm our results.
In conclusion, N-acetylcysteine was beneficial in the treatment of OCD and OCD-related disorders. However, because all studies included in our systematic review had small sample sizes, small-study effects might have influenced our findings. To obtain robust evidence, further studies with larger samples are needed.
Data availability
Data used for the current study were reported in articles as cited in this paper.
References
American Psychiatric Association: Diagnostic and StatisticalManual of Mental Disorders, 5th ed. Washington, DC, American Psychiatric Association, 2013.
Ruscio AM, Stein DJ, Chiu WT, Kessler RC. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15:53–63.
Goodman WK, Storch EA, Sheth SA. Harmonizing the neurobiology and treatment of obsessive-compulsive disorder. Am J Psychiatry. 2021;178:17–29.
Vallee A, Vallee JN, Lecarpentier Y. Lithium: a potential therapeutic strategy in obsessive-compulsive disorder by targeting the canonical WNT/beta pathway. Transl Psychiatry. 2021;11:204.
Vallee A, Lecarpentier Y, Vallee JN. Possible actions of cannabidiol in obsessive-compulsive disorder by targeting the WNT/beta-catenin pathway. Mol Psychiatry. 2022;27:230–48.
Attwells S, Setiawan E, Wilson AA, Rusjan PM, Mizrahi R, Miler L, et al. Inflammation in the Neurocircuitry of Obsessive-Compulsive Disorder. JAMA Psychiatry. 2017;74:833–40.
Alici D, Bulbul F, Virit O, Unal A, Altindag A, Alpak G, et al. Evaluation of oxidative metabolism and oxidative DNA damage in patients with obsessive-compulsive disorder. Psychiatry Clin Neurosci. 2016;70:109–15.
Berk M, Malhi GS, Gray LJ, Dean OM. The promise of N-acetylcysteine in neuropsychiatry. Trends Pharmacol Sci. 2013;34:167–77.
Kato T. Current understanding of bipolar disorder: Toward integration of biological basis and treatment strategies. Psychiatry Clin Neurosci. 2019;73:526–40.
Berk M, Turner A, Malhi GS, Ng CH, Cotton SM, Dodd S, et al. A randomised controlled trial of a mitochondrial therapeutic target for bipolar depression: mitochondrial agents, N-acetylcysteine, and placebo. BMC Med 2019;17:18.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA. Cochrane Handbook for Systematic Reviews of Interventions version 6.2. Cochrane, 2021. Available from www.training.cochrane.org/handbook.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
Nierenberg AA, Kansky C, Brennan BP, Shelton RC, Perlis R, Iosifescu DV. Mitochondrial modulators for bipolar disorder: a pathophysiologically informed paradigm for new drug development. Aust N Z J Psychiatry. 2013;47:26–42.
Liang L, Chen J, Xiao L, Wang Q, Wang G. Mitochondrial modulators in the treatment of bipolar depression: a systematic review and meta-analysis. Transl Psychiatry. 2022;12:4.
Liu Y, Yang Z, Du Y, Shi S, Cheng Y Antioxidant interventions in autism spectrum disorders: a meta-analysis. Prog Neuropsychopharmacol Biol Psychiatry. 2022. https://pubmed.ncbi.nlm.nih.gov/34793863/. in press.
Goodman WK, Price LH, Rasmussen SA, Mazure C, Fleischmann RL, Hill CL, et al. The Yale-Brown Obsessive Compulsive Scale. I. Development, use, and reliability. Arch Gen Psychiatry. 1989;46:1006–11.
Keuthen NJ, O’Sullivan RL, Ricciardi JN, Shera D, Savage CR, Borgmann AS, et al. The Massachusetts General Hospital (MGH) Hairpulling Scale: 1. development and factor analyses. Psychother Psychosom 1995;64:141–5.
Arnold LM, Mutasim DF, Dwight MM, Lamerson CL, Morris EM, McElroy SL. An open clinical trial of fluvoxamine treatment of psychogenic excoriation. J Clin Psychopharmacol 1999;19:15–8.
Hamilton M. The assessment of anxiety states by rating. Br J Med Psychol 1959;32:50–5.
Beck AT, Epstein N, Brown G, Steer RA. An inventory for measuring clinical anxiety: psychometric properties. J Consult Clin Psychol 1988;56:893–7.
Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62.
Kovacs M. The Children’s Depression, Inventory (CDI). Psychopharmacol Bull. 1985;21:995–8.
Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J. An inventory for measuring depression. Arch Gen Psychiatry. 1961;4:561–71.
Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry. 1979;134:382–9.
Guy W, Bonato RR. Manual for the ECDEU assessment battery, 2nd ed. Chevy Chase, Md: National Institute of Mental Health; 1970.
Sheehan D. The anxiety disease. New York, NY: Scribner; 1983.
The Cochrane Collaboration. Review Manager (RevMan 5.4.1.). The Nordic Cochrane Centre, Copenhagen, 2020.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Controlled Clin Trials. 1986;7:177–88.
Lin L, Shi L, Chu H, Murad MH. The magnitude of small-study effects in the Cochrane Database of Systematic Reviews: an empirical study of nearly 30 000 meta-analyses. BMJ Evid Based Med. 2020;25:27–32.
Schumer MC, Panza KE, Mulqueen JM, Jakubovski E, Bloch MH. Long-term outcome in pediatric trichotillomania. Depress Anxiety. 2015;32:737–43.
Hoffman J, Williams T, Rothbart R, Ipser JC, Fineberg N, Chamberlain SR, et al. Pharmacotherapy for trichotillomania. Cochrane Database Syst Rev. 2021;9:CD007662.
Meyer J. Inflammation, obsessive-compulsive disorder, and related disorders. Curr Top Behav Neurosci. 2021;49:31–53.
Farhat LC, Olfson E, Nasir M, Levine JLS, Li F, Miguel EC, et al. Pharmacological and behavioral treatment for trichotillomania: an updated systematic review with meta-analysis. Depress Anxiety. 2020;37:715–27.
Couto JP, Moreira R. Oral N-acetylcysteine in the treatment of obsessive-compulsive disorder: a systematic review of the clinical evidence. Prog Neuropsychopharmacol Biol Psychiatry. 2018;86:245–54.
Smith L, Tracy DK, Giaroli G. What future role might N-acetyl-cysteine have in the treatment of obsessive compulsive and grooming disorders?: a systematic review. J Clin Psychopharmacol. 2016;36:57–62.
Mukai T, Kishi T, Matsuda Y, Iwata N. A meta-analysis of inositol for depression and anxiety disorders. Hum Psychopharmacol. 2014;29:55–63.
Rothbart R, Amos T, Siegfried N, Ipser JC, Fineberg N, Chamberlain SR, et al. Pharmacotherapy for trichotillomania. Cochrane Database Syst Rev. 2013;CD007662. https://pubmed.ncbi.nlm.nih.gov/24214100/.
Camfield DA, Sarris J, Berk M. Nutraceuticals in the treatment of obsessive compulsive disorder (OCD): a review of mechanistic and clinical evidence. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35:887–95.
Fux M, Levine J, Aviv A, Belmaker RH. Inositol treatment of obsessive-compulsive disorder. Am J Psychiatry. 1996;153:1219–21.
Fux M, Benjamin J, Belmaker RH. Inositol versus placebo augmentation of serotonin reuptake inhibitors in the treatment of obsessive-compulsive disorder: a double-blind cross-over study. Int J Neuropsychopharmacol. 1999;2:193–5.
Fux M, Benjamin J, Nemets B. A placebo-controlled cross-over trial of adjunctive EPA in OCD. J Psychiatr Res. 2004;38:323–5.
Grant JE, Redden SA, Chamberlain SR. Milk thistle treatment for children and adults with trichotillomania: a double-blind, placebo-controlled, crossover negative study. J Clin Psychopharmacol. 2019;39:129–34.
Tural U, Corapcioglu A, Bosgelmez S, Koroglu G, Unver H, Duman C, et al. Double blind controlled study of adding folic acid to fluoxetine in the treatment of OCD. Psychiatr Danub. 2019;31:69–77.
Sarris J, Byrne G, Castle D, Bousman C, Oliver G, Cribb L, et al. N-acetyl cysteine (NAC) augmentation in the treatment of obsessive-compulsive disorder: a phase III, 20-week, double-blind, randomized, placebo-controlled trial. Prog Neuropsychopharmacol Biol Psychiatry. 2022. https://pubmed.ncbi.nlm.nih.gov/35304155/. in press.
Li F, Welling MC, Johnson JA, Coughlin C, Mulqueen J, Jakubovski E, et al. N-acetylcysteine for pediatric obsessive-compulsive disorder: a small pilot study. J Child Adolesc Psychopharmacol. 2020;30:32–7.
Ghanizadeh A, Mohammadi MR, Bahraini S, Keshavarzi Z, Firoozabadi A, Alavi, et al. Efficacy of N-acetylcysteine augmentation on obsessive compulsive disorder: a multicenter randomized double blind placebo controlled clinical trial. Iran J Psychiatry. 2017;12:134–41.
Costa DLC, Diniz JB, Requena G, Joaquim MA, Pittenger C, Bloch MH, et al. Randomized, double-blind, placebo-controlled trial of N-acetylcysteine augmentation for treatment-resistant obsessive-compulsive disorder. J Clin Psychiatry. 2017;78:e766–e73.
Leppink EW, Redden SA, Grant JE. A double-blind, placebo-controlled study of inositol in trichotillomania. Int Clin Psychopharmacol. 2017;32:107–14.
Grant JE, Chamberlain SR, Redden SA, Leppink EW, Odlaug BL, Kim SW. N-acetylcysteine in the treatment of excoriation disorder: a randomized clinical trial. JAMA Psychiatry. 2016;73:490–6.
Paydary K, Akamaloo A, Ahmadipour A, Pishgar F, Emamzadehfard S, Akhondzadeh S. N-acetylcysteine augmentation therapy for moderate-to-severe obsessive-compulsive disorder: randomized, double-blind, placebo-controlled trial. J Clin Pharm Ther. 2016;41:214–9.
Sarris J, Oliver G, Camfield DA, Dean OM, Dowling N, Smith DJ, et al. N-acetyl cysteine (NAC) in the treatment of obsessive-compulsive disorder: a 16-week, double-blind, randomised, placebo-controlled study. CNS Drugs. 2015;29:801–9.
Bloch MH, Panza KE, Grant JE, Pittenger C, Leckman JF. N-Acetylcysteine in the treatment of pediatric trichotillomania: a randomized, double-blind, placebo-controlled add-on trial. J Am Acad Child Adolesc Psychiatry. 2013;52:231–40.
Afshar H, Roohafza H, Mohammad-Beigi H, Haghighi M, Jahangard L, Shokouh P, et al. N-acetylcysteine add-on treatment in refractory obsessive-compulsive disorder: a randomized, double-blind, placebo-controlled trial. J Clin Psychopharmacol. 2012;32:797–803.
Grant JE, Odlaug BL, Kim SW. N-acetylcysteine, a glutamate modulator, in the treatment of trichotillomania: a double-blind, placebo-controlled study. Arch Gen Psychiatry. 2009;66:756–63.
McDougle CJ, Price LH, Goodman WK, Charney DS, Heninger GR. A controlled trial of lithium augmentation in fluvoxamine-refractory obsessive-compulsive disorder: lack of efficacy. J Clin Psychopharmacol. 1991;11:175–84.
Dold M, Aigner M, Lanzenberger R, Kasper S. Antipsychotic augmentation of serotonin reuptake inhibitors in treatment-resistant obsessive-compulsive disorder: an update meta-analysis of double-blind, randomized, placebo-controlled trials. Int J Neuropsychopharmacol. 2015;18:pyv047. https://pubmed.ncbi.nlm.nih.gov/25939614/.
Acknowledgements
We thank all participants in the 17 studies and all researchers who conducted these studies.
Author information
Authors and Affiliations
Contributions
TK had complete access to all study data and is responsible for data integrity and analysis accuracy. TK assisted in the conceptualization and design of the study, as well as the statistical analysis. All the authors handled the acquisition and interpretation of data and wrote the manuscript. NI supervised the review.
Corresponding author
Ethics declarations
Competing interests
The authors declare that there are no conflicts of interest in relation to this study. We declare the following interests within the past 3 years: TK, KS, and NI. TK has received speaker’s honoraria from Sumitomo, Eisai, Janssen, Otsuka, Meiji, MSD, Yoshitomi, and Takeda; a Health Labor Sciences Research Grant, Japan Agency for Medical Research and Development grant, Grant-in-Aid for Scientific Research (C) grant, and a Fujita Health University School of Medicine research grant. KS has received speaker’s honoraria from Eisai, Kissei, Meiji, Otsuka, and Torii; a Fujita Health University School of Medicine research grant; Japan Agency for Medical Research and Development grant, and a Grant-in-Aid for Young Scientists (B) grant. NI has received speaker’s honoraria from Astellas, Sumitomo, Eli Lilly, GlaxoSmithKline, Janssen, Yoshitomi, Otsuka, Meiji, Shionogi, Novartis, and Pfizer and research grants from Daiichi Sankyo, Sumitomo, Meiji, and Otsuka.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kishi, T., Sakuma, K. & Iwata, N. Mitochondrial modulators for obsessive–compulsive and related disorders: a systematic review and meta-analysis. Transl Psychiatry 12, 263 (2022). https://doi.org/10.1038/s41398-022-02026-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41398-022-02026-5