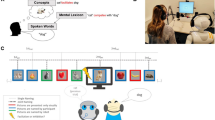
Abstract
The ability to realize the individuals’ impressions during the verbal communication allows social robots to significantly facilitate their social interactions in such areas as child education and elderly care. However, such impressions are highly subjective and internalized and therefore cannot be easily comprehended through behavioural observations. Although brain-machine interface suggests the utility of the brain information in human-robot interaction, previous studies did not consider its potential for estimating the internal impressions during verbal communication. In this article, we introduce a novel approach to estimation of the individuals’ perceived difficulty of stories using the quantified information content of their prefrontal cortex activity. We demonstrate the robustness of our approach by showing its comparable performance in face-to-face, humanoid, speaker, and video-chat settings. Our results contribute to the field of socially assistive robotics by taking a step toward enabling robots determine their human companions’ perceived difficulty of conversations, thereby enabling these media to sustain their communication with humans by adapting to individuals’ pace and interest in response to conversational nuances and complexity.
Similar content being viewed by others
Introduction
Gone are the days when robots were sitting on the factory floors to perform tasks whose instructions were hardcoded in a great detail flawlessly. As the field of robotics matures, human society witnesses a growing integration of these media in individuals’ daily lives and activities. In fact, today’s robotics is less about assisting humans perform their physical tasks and more about facilitating their social interaction1. This observation is evident in growing adaptation of these media in such broad social domains as early child education2,3 and elderly care4,5. Pivotal to these applications is the ability for these agents to engage in social interaction6 and therefore solutions to such hard problems as learning the social norms and dynamics form the foundation for enabling robots understand the intentions of their human companion, thereby allowing them to achieve a sustainable long-term interaction.
However, obtaining these abilities by only observing the human behaviour is not sufficient considering the fact that behavioural cues can often be interpreted in different ways which is even more so during a verbal communication. For example, a frowning face during a conversation can be construed as sign of attentiveness or it may signal the person’s difficulty in following its content. Interestingly, deciphering such cues becomes even harder once we take into account the ability of individuals to disguise their emotions (e.g., smiling in a stressful situation). In the same vein, the fact that such reactions are highly subjective (i.e., they differ from person to person) makes facial analysis approaches7 fall short in decoding the cues associated with verbal communication: shy students that find a lecture difficult can shun the clarifying questions by acting as if they were following the lecture.
On the other hand, the human brain as the source of these subjective and internalized states can provide more reliable information about them. The brain activity cannot be easily suppressed or manipulated and therefore the information that is reflected in such activities has the potential to quantify individuals’ feelings. However, despite substantial progress in utilization of the brain information in such applications as brain machine interface (BMI)8 and human-robot interaction (HRI)9,10, there appears to be a paucity of research (to the best of our knowledge) on the use of brain information for estimating individuals’ assessment of the verbal communication (e.g., its difficulty) during HRI. Robots can particularly benefit from such an ability while interacting with overstressed persons and individuals with selective mutism disorders.
In this article, we propose to estimate the individuals’ perceived difficulty of a verbal communication by quantifying the information content of their prefrontal cortex (PFC) activity. We use the term “perceived difficulty” to refer to the cognitive load that a person’s PFC may endure during such tasks as language processing11, social cognition12, and story comprehension13,14,15. In this respect, functional imaging techniques in conjunction with such tasks as mental arithmetic (MA)16 and n-back17 have enabled researchers to shed light on PFC functioning and change in its activity in response to varying cognitive load16,17,18. Considering these findings, we expect that these tasks can also be useful in estimating the perceived difficulty of a more cognitively demanding task like verbal communication.
We also introduce a novel information-theoretic approach for quantification of such cognitive loads. The choice of information is motivated by the following three observations. First, information is an unbiased measure of association between interacting processes19 (e.g., change in the brain activity in response to varying task’s difficulty) and hence an attractive choice for brain mapping20 as well as the modeling of its inherent complexity21,22,23,24. Second, information allows for a more robust handling of such confounders as residual brain activity prior to the start of the task period (also known as resting state25). This ability plays a central role in preventing an overestimation of the cognitive load due to the prior brain activity that is not induced by the task. Third, brain activation can take place in a shorter time span (i.e., faster) in case of easier than more difficult tasks26 and therefore its differential activities can simply be averaged out27,28,29 if the incurred variability is not accounted for30,31,32. This hinders the ability to differentiate the activation patterns that are induced by tasks with varying level of difficulty. On the other hand, information of a continuous random variable is a function of the variance than the mean33, p. 182] and therefore can preserve the variability of such time series as brain activity34. This, in turn, makes information well-suited for scenarios in which higher variability in individuals’ brain responses is expected (e.g., verbal communication). Therefore, we expect that an information-based quantification of the individuals’ PFC activation to form a reliable biomarker for measuring the cognitive load that is associated with the difficulty of a verbal communication.
In our approach, we first determine a decision boundary that distinguishes between the incurred cognitive loads by one- and two-back auditory tasks on individuals’ PFC (acquired by near-infrared spectroscopy (NIRS)). In this task, the participants are instructed to respond (e.g., through mouse clicks) to the repeated patterns in numerical sequences (e.g., sequential i.e., n = 1 or one-back and every-other repetition i.e., n = 2 or two-back) that are presented auditorily. We use n-back in our study due to its demonstrated ability in inducing differential cognitive load on PFC35 as well as its utility in quantifying the PFC activity in response to individuals’ change in mood18,36. Then in a realtime storytelling scenario, we use this boundary to estimate the individuals’ perceived difficulty of narrated stories, thereby interpreting their perceived difficulty of stories based on induced cognitive load by n-back auditory task.
Our contributions are threefold. First, we introduce a novel information-theoretic approach to quantification of the induced cognitive load on PFC. We present the effectiveness of our approach in quantification of the PFC activity during a WM task. Our results show a substantial improvement on the previous findings17,37. Second, We demonstrate the utility of our approach in estimation of the individuals’ perceived difficulty of the verbally communicated content in a humanoid-mediated storytelling scenario. Third, we provide evidence for robustness of our approach through comparative analysis of its performance in face-to-face, humanoid, speaker, and video-chat system media settings.
In our view, the use of brain information can advance the HRI research on modeling of the human behaviour by providing invaluable information about mechanisms that underlie human behavioural responses. For instance, brain activity can be used as neurophysiological feedbacks about individuals’ mental states6 in multimodal modeling of human behaviour38. This, in turn, can open a new venue for formal analysis of a robotic ToM39 that (in addition to behavioural observations) builds upon critical implications of the humans’ neurological responses during interaction with their synthetic companions.
Methods
Our approach comprises of three steps: (A) information-theoretic formulation of the cognitive load (CL), (B) determination of a decision boundary that identifies the CL quantities that are uniquely associated with differing WM task’s level, (C) realtime estimation of the perceived difficulty of the verbally communicated content. In what follows, we explain each step in details.
Information-theoretic formulation of cognitive load
This step consists of two components: the “quantification of the cognitive load” that estimates the induced cognitive load on the PFC in response to external stimuli per estimation step and the “constrained updating of the induced cognitive load” that dictates an update rule to reduce the effect of the PFC activity’s fluctuations on such a quantification.
Quantification of the cognitive load
Let Xτ represent the time series associated with the task period’s PFC activity at estimation step τ. Let B be the baseline (i.e., resting state) time series that represent the frontal brain activity prior to the start of the task. Furthermore, let H(Xτ) represent the entropy of Xτ (i.e., its average information content). Although H(Xτ) quantifies the PFC’s cognitive load (CL)40 at τ, it is an overestimation of CL if PFC’s residual activity that is carried over from the resting period is not attenuated. It is also crucial to observe that such a residual effect cannot be attenuated by mere subtraction of the expected resting state’s activity (i.e., μB) from Xτ due to the invariant of information to translation [41, Theorem 8.6.3, p. 253]. Therefore, we quantify the PFC’s cognitive load at estimation step τ i.e., CL(Xτ) through conditioning of the PFC activity at τ with respect to its activity prior to the start of the task:
where MI(Xτ; B) represents the mutual information between PFC activity during the task period at estimation step τ and its activation pattern during the resting period.
Constrained updating of the induced cognitive load
Neuroscientific findings imply that the brain activity occurs in sparse transient42. In other words, observed brain activities are subject to fluctuation. Considering the direct correspondence between information and the variation33,40, such a sparsity can directly affect the calculated CL as formulated in Eq. (1) which, in turn, can result in a false belief about the overall task-induced cognitive load on PFC due to the accumulation of such over/underestimations of CL. In other words, fluctuating patterns in PFC activity can result in rapid changes in signal variability whose discrimination from desirable task-induced changes in PFC activation might not be trivial if one only rely on the computed CL. For instance, an increase/decrease in CL at a given time might solely be explained by a short-lived fluctuation and not the effect of the task per se on PFC. In such a scenario, simply following the computed CL can lead to a false conclusion since a small number of such rapid and short-lived incremental/decremental fluctuations can cancel out and average the actual effect of the task on PFC activity.
Above observations identify the need for additional measures to validate the correspondence between potential differences between two consecutive CLs. More importantly, these measures must take into account the pattern of PFC activity associated with these consecutive CLs to verify whether their observed differences are in fact due to a substantial variation than a mere short-lived fluctuation. In other words, they must allow for constraining the observed differences between two consecutive CLs with the level of change in their respective PFC variability.
Interestingly, these fluctuations can conveniently be accounted for through realization of the MI: a measure of the shared information among interacting processes [41, p. 19 and p. 251]. Specifically, rewriting Eq. (1) as MI(Xτ; B) = H(Xτ)−CL(Xτ) it becomes apparent that an increase/decrease in the cognitive load must, in principle, be accompanied with its corresponding decrease/increase in mutual information between Xτ and B. In fact, if the interacting processes belonged to a well-defined parametric distribution it would have sufficed to solely check for MI(Xτ−1; B) and MI(Xτ; B) to discriminate between potential fluctuations and the legitimate variations in the task-induced cognitive load. However, extent of the brain dynamics that borders with chaotic system43 in conjunction with varying complexity of naturalistic tasks (e.g., conversational nuances and change in difficulty of their contents) do not warrant the utility of such simplifying assumption as imposing a known parametric distribution on observed PFC activity during naturalistic scenarios.
Alternatively, we can verify whether above necessary condition in case of MI is also satisfied at the distribution level of these interacting processes, thereby bypassing any unwarranted assumption on their distributions. This can be achieved by utilization of the Kullback-Leibler divergence (DKL) that reflects the distance between the distribution of interacting processes [41, p. 19 and p. 251]. The utility of DKL is realized by observing that any increase/decrease in MI due to a reduced/increased CL in MI(Xτ; B) = H(Xτ)−CL(Xτ) indeed identifies an increase/decrease in resemblance between their distributions and therefore their reduced/increased divergence.
Therefore, we control for potential fluctuating patterns in PFC during the task performance by evaluating the difference between CL(Xτ−1) and CL(Xτ) through quantification of their respective MI and DKL with respect to B, thereby constraining the updates of computed PFC’s cognitive load. Concretely, we directly use the result from Eq. (1) if the difference between CL(Xτ−1) and CL(Xτ) is warranted by their MI and DKL with respect to B or, alternatively, we compensate for the potential fluctuation by averaging CL(Xτ−1) and CL(Xτ), weighted by their variation of information (VI)44:
and:
Decision boundary determination
Let S1 and S2 denote the CLs that correspond to cognitive loads induced by two-level WM tasks. Computing the decision boundary 𝔻 between S1 and S2 is analogous to determining the midpoint between the CL quantities that uniquely fall within the S1 or S2 intervals i.e., the elements that are not members of their overlapping subset. Algorithm 1 outlines this process. It first sorts S1 and S2 in their descending and ascending orders (steps 1 and 2). Next, it finds the smallest CL in S1 that lies within the S2 interval (step 3) and the largest CL in S2 that is within S1 interval (step 4), thereby marking their overlapping partition. Then, it determines the immediate largest CL in S1 and the immediate smallest CL in S2 that are smaller and larger than S1’s and S2’s respective CLs that mark this overlapping partition. Last, it returns the average of these immediate largest and immediate smallest CLs as the decision boundary 𝔻 that separates the CLs associated with the disjoint S1 and S2 sets.
In this article, we utilize n-back auditory task as the WM task. In this case, S1 and S2 correspond to cognitive loads induced by one- and two-back WM tasks, respectively.
Online estimation of the perceived difficulty of conversation
We utilize the calculated decision boundary 𝔻 for online estimation of the individuals’ perceived difficulty of the verbally communicated content. At every estimation step, our model calculates the CL of the current PFC activity time series. At the end of the verbal communication, it computes the median of these computed CLs and determines whether it is above or below the computed decision boundary 𝔻. Subsequently, it marks the individual’s perceived difficulty of the verbally communicated content as “difficult/easy” if this median is above/below 𝔻.
Ethics statement
This study was carried out in accordance with the recommendations of the ethical committee of the Advanced Telecommunications Research Institute International (ATR) with written informed consent from all subjects in accordance with the Declaration of Helsinki. The protocol was approved by the ATR ethical committee (approval code:16-601-1).
Experiments
We conducted two experiments to evaluate the utility of our model. In the first experiment, we verified whether our proposed measure of cognitive load can distinguish the PFC activation in response to low vs. high cognitive loads in one- and two- back WM task. This allowed us to evaluate the ability of our approach for differential quantification of the induced PFC activation in response to these tasks. It also allowed us to determine the decision boundary 𝔻 between differential cognitive loads on the PFC activity which we used in the second experiment.
The second experiment was for verification of the performance of our approach on estimation of the individuals’ perceived difficulty of the verbal communication in a naturalistic setting. For this purpose, we used storytelling as a first step toward decoding of the conversational communication since stories’ scripts can be kept intact and repeated to different individuals without any change in their contents, thereby allowing for the control of such confounders as subtle differences in conveyed information.
The “perceived difficulty” within the context of the first experimental paradigm then refers to the cognitive psychology notion of cognitive load: measurable change in WM capacity in processing information that is associated with controlled tasks that are specifically designed for WM excitation17,40. In the context of second experiment, on the other hand, it reflects the change in WM capacity at more subjective level (e.g., increase in WM information processing with respect to the change in stories’ difficulty) that is quantified by such fine-grained and well-designed class of WM tasks as n-back.
Experiment 1: Discrimination of differential cognitive load in N-Back WM Task
Purpose
In this experiment, we validated the performance of our approach on quantification of the effect of the WM tasks on PFC activity. Among such tasks, we chose n-back WM task since it forms a better basis for quantification of the verbally communicated contents, considering its effect on PFC35 and its ability in identifying the change in PFC activation in response to individuals’ emotions and change in mood18.
Participants
Thirty three younger adults (fourteen males and nineteen females, M = 30.96 years, SD = 10.84) participated in this experiment. Data from one male and one female were not recorded properly and were discarded. All participants were free of neurological and psychiatric disorders and had no history of hearing impairment. All experiments were carried out with written informed consents from all subjects. We used a job-offering site for university students to recruit our participants.
Paradigm
It included a seventy-second audio (in Japanese) sequences of numerical (1 through 9) one- and two-back WM tasks. Each session consisted of a one- and a two-back WM tasks. We kept the order as well as content of these WM tasks intact for all the participants. We used a speaker to play the audio sequences of numerical one- and two-back WM tasks to the participants. Every participant completed these two tasks. The participants responded to sequential (i.e., one-back) and every-other (in case of two-back) occurrences of these numerical values through mouse-clicks. We used PsychoPy for generating these audio one- and two-back WM tasks.
Procedure
Every participant first was seated in an armchair with proper head support in a sound-attenuated testing chamber and gave written informed consent in the experimental room. Then, a male experimenter explained the experiment’s full procedure to the participants. This included the total number of tasks in a session (i.e., a one-back followed by a two-back WM tasks), the duration of each task (i.e., seventy seconds per WM task), instructions on WM tasks procedure (i.e., periodic sequential (i.e., one-back) or every-other (i.e., two-back) occurrences of some of the numerical values), instructions on how to respond if the participants detected such reoccurrences (i.e., through the mouse-click at every detection), instructions about the one-minute rest period prior to the actual session (i.e., sitting still with eyes closed), and the content of the audio sequences (i.e., numerical values 1 through 9). The experimenter also asked the participants to stay focused on listening to the one- and two-back sequences that were played back to the participants through a computer speaker and then began the experimental session. Every one- and two-back WM task started by recording a one-minute rest data which was followed by its seventy seconds WM task period. We recorded the participants’ frontal brain activity time series throughout these tasks’ periods (including their respective one-minute resting).
Once the participants were ready, the experimenter asked them to follow the instructions on the computer screen in front of them (Fig. 1(A)). The instructions on the display informed the participants that they participate in a one- and a two-back WM tasks, that each WM task was seventy seconds long, that the task period was proceeded with a one minute resting period during which they needed to close their eyes and relax as much as possible, and that once this resting period was over a voice (recorded voice of the experimenter through the computer speaker) would announce the start of the WM task after which the task would immediately begin. These instructions also provided the participants with an audio-visual example of the task that was about to begin. For instance, in the case of one-back it displayed a short sequence of numbers which were (in sequential fashion) highlighted by a square around the digit that was being read out and explaining how the value that was just read out was related to the value one-back before (Fig. 1(B)). The participants then started with the resting period of the one-back WM task and immediately engaged in this task once the end of the resting period and the start of the WM task was announced by the voice. For the one-back task, there were ten numerical values that were repeated in one-back fashion (Fig. 1(B)). Once one-back was over, the voice announced the end of the task and instructed the participants to follow the on-screen information for two-back task (similar to the one-back but this time the instruction of the task was about the two- than one-back WM task). The participants then started the two-back WM task which began by its one-minute resting period at the end of which the voice asked the participants to open their eyes and that the task was about to start. Two-back WM task also (similar to one-back) included the repetition of ten numerical values in a two-back fashion (Fig. 1(C)). Once the two-back WM task was over, the experimenter removed the NIRS device from the forehead of the participants and guided them out side the experimental room.
Data acquisition
We used functional near infrared spectroscopy (fNIRS) to collect the frontal brain activity of the participants and acquired their NIRS time series data using a wearable optical topography system called “HOT-1000,” developed by Hitachi High-Technologies Corp. (Fig. 2). Participants wore this device on their forehead to record their frontal brain activity through detection of the total blood flow by emitting a wavelength laser light (810 nm) at a 10.0 Hz sampling rate. Data acquisition was carried out through four channels (L1, L3, R1, and R3, Fig. 2). Postfix numerical values that are assigned to these channels specify their respective source-detector distances. In other words, L1 and R1 have a 1.0 cm source-detector distance and L3 and R3 have a 3.0 cm source-detector distance. Note that whereas a short-detector distance of 1.0 cm is inadequate for the data acquisition of cortical brain activity (e.g., 0.5 cm45, 1.0 cm46, 1.5 cm47, and 2.0 cm48), 3.0 cm is suitable45,47. Therefore, we mainly report the result with the data from L3 in present study and the result with R3 is shown in Supplementary Materials (SM).
Data processing
We first attenuated the effect of systemic physiological artefacts49 (e.g., cardiac pulsations, respiration, etc.) using a one-degree polynomial Butterworth filter with 0.01 and 0.6 Hz for low and high bandpass which was then followed by performing the linear detrending on the data. We then attenuated the effect of the skin blood flow (SBF) using an eigen decomposition technique50. This approach considers the first three principal components of all NIRS recorded channels of the participants’ frontal brain activity during rest period to represent the SBF. Subsequently, it eliminates the SBF effect by removing these three components from participants’ NIRS time series in the task period. Although Sato et al.47 suggested that the use of first principal component than first three components appeared to be sufficient for SBF attenuation, Keshmiri et al.51 demonstrated that the use of first two principal components resulted in both significantly higher SBF attenuation as well as more cortical activity’s information preservation. Therefore, we followed51 and removed the first two principal components of the respective resting period of the participants from the NIRS time series of their frontal brain activity that was recorded during the task period. It is worth emphasizing that we used the same measurement settings (i.e., same equipment, number of measurement channel, and its position) as in Keshmiri et al.51 Similar to our NIRS recording, Zhang et al.50 also used 3.0 cm source-detector distance channels. Cooper et al.52 showed that this filter also attenuates the motion artefact (e.g., head motion).
While quantifying the PFC activation, we used twenty-second NIRS time series segments of participants’ PFC activation with ten-second of overlap between every two consecutive segments to calculate the CLs at every ten-second estimation step. We used our mathematical model in Section 2.1 for CL computation. For the first segment in the task period, we considered its overlap with the last ten seconds of the rest period. We used the last twenty-second of the rest period’s NIRS time series for each participant in Eq. (1). The ten-second estimation step resulted in seven CLs in case of one- and two-back WM tasks (per task).
Analysis
First, we computed the medians (per participant per task) of the CLs for the one- and the two-back WM tasks. Then, we applied Wilcoxon rank sum on these medians to determine the differential significance between these WM tasks’ CLs. Next, we used these medians to determine the discrimination accuracy of our model’s CLs in differentiating between one- and two-back WM tasks. We computed the accuracy of our model using Mdntwo−back > Mdnone−back (Mdn stands for median) criterion per participant. We also computed the Spearman correlation between these medians and the percentage of correct clicks by the participants. We scaled the participants’ number of clicks within [0, … 1] interval. Last, we computed the Spearman correlation between one- and two-back WM tasks’ medians. In order to determine the utility of our model, we also applied these analyses on participants’ average PFC activation (SM Section 1 for left-hemispheric and SM 2.1.2 for right-hemispheric PFC).
To further examine whether the changes in participants’ CLs during two-back WM task were significantly associated with the cognitive load induced by this WM task period than being the residual effect from their one-back WM task period, we performed a one-sample bootstrap test of significance (10,000 simulation runs) at 99.0% confidence interval (CI) on the difference between participants’ CLs during two-back and one-back WM tasks (i.e., CLB2 − CLB1). We then considered the null hypothesis H0: induced change in CLs by two-back WM task after the deduction of one-back’s CLs was non-significant and tested it against the alternative hypothesis H1: two-back WM task’s induced change in CLs after the deduction of one-back’s CLs was significant. Since we considered CLB2 − CLB1, H0 and H1 then represented the situations in which CLB2 − CLB1 ≈ 0 (i.e., zero fell within the 99.0% confidence interval) and CLB2 − CLB1 > 0 (i.e., their 99.0% confidence interval was significantly above zero), respectively. It is worth noting that H1: CLB2 − CLB1 > 0 is equivalent to H1': CLB1 − CLB2 < 0. We reported the mean, standard deviation, and 99.0% confidence interval for left PFC in the main manuscript (for results associated with right PFC, see SM, Section 2 and Fig. 3).
(A) Wilcoxon rank sum between participants’ CLs in one- and two-back WM tasks. Asterisks indicate the significant difference between these CLs. (B) Spearman correlation between participants’ CLs and the number of their correct clicks in one-back WM task. (C) Spearman correlation between participants’ CLs and the number of their correct clicks in response to two-back WM task. (D) Spearman correlation between participants’ CLs in one- and two-back WM tasks. In (B) and (C) correct clicks are normalized within [0, …, 1] interval. In these subplots, “CL” refers to the medians of the CLs for the two tasks.
Next, we computed the Spearman correlations between these CLB2 − CLB1 values and the participants’ correct clicks during the two-back WM task. We chose the participants’ correct clicks during two- than one-back WM tasks since CLB2 − CLB1 values reflected the quantitative changes in participants’ CLs associated with two-back WM task and after the reduction of participants’ CLs during one-back WM task period. We followed this by computing their 95.0% bootstrap (10,000 simulation runs) confidence intervals. For the bootstrap test, we considered the null hypothesis H0: there was no correlation between CLB2 − CLB1 and participants’ correct clicks during two-back WM task and tested it against the alternative hypothesis H1: CLB2 − CLB1 significantly correlated with participants’ correct clicks during two-back WM task. We reported the mean, standard deviation, and 95.0% confidence interval for this test. We also computed the p-value of this test as the fraction of the distribution that was more extreme than the actually observed correlation values. For this purpose, we performed a two-tailed test in which we used the absolute values so that both the positive and the negative correlations were accounted for.
Last, to ensure that the observed changes in the participants’ CLs during two-back WM tasks were due to the PFC activity during this WM task than artefacts (e.g., noise or an affine transformation of one-back’s CLs as a result of the underlying linear property of the hemodynamic responses53,54), we applied a one-sample bootstrap test of significance (10,000 simulation runs) at 99.0% confidence interval on the Kullback-Leibler divergence DKL of participants’ CLs during two- and one-back WM tasks (i.e., DKL(CLB2, CLB1). we considered the null hypothesis H0: difference in the distribution of CLs in two- and one-back WM tasks were non-significant (hence one-back’s CLs can be used to explain the observed CLs during two-back WM task) and tested it against the alternative hypothesis H1: distribution of CLs during two-back WM task was significantly different from one-back’s CLs. We reported the mean, standard deviation, and 99.0% confidence interval for this test. It is worthy of note that whereas H0 in this test was satisfied if zero fell within the computed DKL’s 99.0% confidence interval, H1’s satisfaction was associated with the case in which 99.0% confidence interval was significantly above zero (or equivalently significantly below zero in the case of DKL(CLB1, CLB2).
The earlier studies on n-back35 WM tasks and the language processing11 reported on a higher activation in left- than right-hemispheric PFC. On the other hand, the recent findings on the role of PFC in n-back17 and story comprehension13,14,15 indicate that such a distinction is not necessarily warranted. Therefore, we considered both left as well as right PFC in our study. However, we focused on the left PFC in the main manuscript since the activity in left PFC formed the common themes among these previous findings and provided the results pertinent to the right PFC in SM.
Results
Wilcoxon rank sum (Fig. 3(A)) identified a significant difference between the participants’ CLs in one- and two-back WM tasks (p < 0.001, W(60) = 3.59, r = 0.46, Mone−back = 1.83, SDone−back = 0.59, Mtwo−back = 2.48, SDtwo−back = 0.64). Our model achieved an 87.10% prediction accuracy for classification of these tasks. Table 1 summarizes these results.
We found a significant correlation between participants’ CLs and their number of correct clicks in response to one-back (Fig. 3(B)) WM task (r = 0.52, p < 0.01, MClicks = 0.88, SDClicks = 0.05). Similarly, this correlation was significant in two-back (Fig. 3(C)) WM task (r = 0.46, p < 0.01, MClicks = 0.69, SDClicks = 0.15). Last, we observed (Fig. 3(D)) a significant correlation between participants’ CLs in one- and two-back WM tasks (r = 0.41, p < 0.03).
One-sample bootstrap test of significance (10,000 simulation runs) at 99.0% confidence interval (CI) on the difference between participants’ CLs during two-back and one-back WM tasks (i.e., CLB2 − CLB1) verified that (Fig. 4(A)) the changes in participants’ CLs during two-back WM task were significantly associated with the cognitive load associated with this WM task period (i.e., CLB2 − CLB1 > 0.0) than being the residual effect from their one-back WM task period (MCLB2−CLB1 = 0.96, SDCLB2−CLB1 = 0.71, CICLB2−CLB1 = [0.74 1.18] where M and SD refer to the mean difference and the standard deviation of such a difference between the two compared states and CI shows the 99.0% confidence interval of their difference).
(A) One-sample bootstrap test of significance (10,000 simulation runs) at 99.0% confidence interval on the difference between participants’ CLs during one- and two-back WM tasks. In this subplot, the x-axis shows CLB2 − CLB1. The blue line marks the null hypothesis H0 i.e., non-significant change in CLs during two-back and with respect to one-back WM task. The red lines are the boundaries of the 99.0% confidence interval. The yellow line shows the location of the average CLB2 − CLB1 value for 10,000 simulation runs, the red-lines mark the 99.0% confidence interval, and the blue is H0. (B) Spearman correlation between participants’ number of correct clicks in response to two-back WM task and the difference between their CLs in one- and two-back WM tasks (i.e. CLB2 − CLB1). (C) Bootstrap correlation test (10,000 simulation runs) at 95.0% confidence interval in which the observed correlation between CLB2 − CLB1 and participant’s correct click during two-back WM task was verified. (D) One-sample bootstrap test of significance (10,000 simulation runs) at 99.0% confidence interval on Kullback?Leibler divergence DKL between participants’ CLs distribution in two- versus one-back WM tasks. In this subplot, the axis shows the calculated DKL between CLs’ distributions in two- (B2) and one-back (B1) (i.e., DKL(CLB2, CLB1)). The yellow line shows the location of the average CLB2 − CLB1 value for 10,000 simulation runs, the red-lines mark the 99.0% confidence interval, and the blue is H0 i.e., the non-divergence between the two distributions.
We also observed a significant correlation between participants’ CLB2 − CLB1 values and their correct click during their two-back WM task period (Fig. 4(B), r = 0.39, p = 0.03) which was further supported by their corresponding bootstrap tests (10,000 simulation runs) at 95.0% confidence interval (Fig. 4(C), CI95.0% = [0.05 0.67]).
Finally, the bootstrap test of significance (10,000 simulation runs) at 99.0% confidence interval on the Kullback-Leibler divergence (Fig. 4(D)) between participants’ CLs distribution in two- versus (i.e., DKL(CLB2, CLB1)) identified a significant difference in the distribution of the participants’ CLs in two-back (i.e., B2 in this subplot) and their corresponding CLs during one-back (i.e., B1 in this subplot) WM tasks (MDKL(B2, B1) = 3.44, SDDKL(B2, B1) = 0.87, CIDKL(B2, B1) = [1.48 5.93]). This test ruled out that the observed changes in the participants’ CLs during the two-back were primarily due to the proceeding one-back task (e.g., effect of noise, linear scaling, or affine transformation).
Experiment 2: Estimation of the perceived difficulty during naturalistic storytelling
The first experiment showed that our proposal for quantification of the cognitive load can significantly discriminate the differential load of the WM tasks on PFC activity. In the second experiment, we verified the ability of our approach in estimation of the individuals’ perceived difficulty of the verbally communicated content in a naturalistic storytelling. We used our CL’s formulations and the decision boundary computed with the data used in the second experiment. We also investigated the performance of our approach in four media settings: face-to-face, humanoid, speaker, and video-chat. These media settings allowed us to validate the robustness of our approach. Precisely, the face-to-face setting laid down a reliable basis for verification of our model’s performance: throughout the history, stories have been made and narrated by the people for the people. On the other hand, the speaker and the video-chat verified the utility of our approach in capturing the PFC activation in response to the content of the story than such potential factors as embodiment and novelty effect (in case of the humanoid). Taken together, comparable performance of our model on these media settings in conjunction with its accuracy in case of the humanoid demonstrated its generalizability and therefore effectiveness of the brain-based quantification of the perceived difficulty of the communicated content using PFC pattern of activation.
Contents of Sections 3.2.2 through 3.2.4 are also appeared in Keshmiri et al.55. For the sake of clarity, we provide their outline in this article as well.
Participants
Our participants consisted of twenty three younger adults (fifteen females and eight males, M = 22.39, SD = 2.82). Data from three females were not recorded properly and were discarded. All participants in the first and second experiments were free of neurological and psychiatric disorders and had no history of hearing impairment. All experiments were carried out with written informed consents from all subjects. We used a job-offering site for university students to recruit our participants.
Media
They included a humanoid robotic medium, an audio speaker, a video-chat system, and a human. We chose a minimalist teleoperated humanoid called, Telenoid R4TM (Telenoid hereafter)56. Telenoid is approximately 50.0 cm long and weighs about 3.0 kg. It comes with nine degrees-of-freedom (3 for its eyes, 1 for its mouth, 3 for its neck, and 2 for its arms) and is equipped with an audio speaker on its chest. It is primarily designed to investigate the basic and essential elements of embodiment for the efficient representation and transfer of a humanlike presence. Therefore, its design follows a minimalist anthropomorphic principle to convey a gender-and-age-neutral look-and-feel. In present study, we chose a minimalist anthropomorphic embodiment to eliminate the projection of such physical traits as gender and age onto our robotic medium.
Telenoid conveyed the vocal information of its teleoperator through its speaker. Its motion was generated based on the operator’s voice, using an online speech-driven head motion system57. However, its eyes and arms were motionless in this study. We placed Telenoid on a stand approximately 1.40 meters from the participant’s chair to prevent any confounding effect due to tactile interaction (e.g., holding, hugging, etc.). We adjusted this stand to resemble an eye-contact setting between Telenoid and the participant. We maintained the same distance in the case of the other media as well as for the face-to-face setting. In face-to-face condition, we adjusted the storyteller’s seat to maintain eye-contact with the participant. For the video-chat, we adjusted its placeholder in such a way that the storyteller’s appearance on the screen resembled an eye-contact setting. In the speaker setting, we placed the video-chat screen in front of the participant (like in the video-chat condition) and placed the speaker behind its screen.
We used the same audio device in the speaker and video-chat settings to prevent any confounding effect due to audio quality. We used the same recorded voice of a woman, who was naive to the purpose of this study, in speaker, video-chat, and Telenoid. These recordings took place in a single session in which we recorded her voice and video while telling stories. In Telenoid setting, we played back the same prerecorded voice for the speaker through the audio speaker on its chest. In face-to-face setting, the same woman read the stories to the participants.
We asked our female storyteller to stay as neutral as possible while reading these stories. However, we are unable to confirm the absence of any difference in emotional impact of the stories’ content on her during the face-to-face or the voice/video recordings.
Paradigm
It consisted of three-minute storytelling sessions in which a woman narrated three-minute stories from Greek mythology through three kinds of communication media: an audio speaker, a video-chat system, and Telenoid. As a control setting, we included the face-to-face scenario in which we told these stories to the participants in-person. Every individual participated in all four storytelling sessions. We also controlled the field of view of the participants within the same spatial limit by placing their seat in a cubicle in all these sessions (height = 130.0 cm, width = 173.0 cm, depth = 210.0 cm) which further enabled us to prevent the potential confounding effect of visual distraction.
Procedure
It started with collecting the written informed consents from the participants which was then followed by a male experimenter explaining the experimental procedure to them. In this step, the experimenter explained to the participants that each storytelling session was approximately three minutes in length and that each session would start with a one-minute resting period during which they were required to sit still with their eyes closed. He then briefed them about the content of stories and instructed them to stay focused on these stories’ content as much as possible. After leading the participants to the experimental room, the experimenter helped them be seated and ensured the proper adjustment of the medium (or helped the storyteller get in her proper position during the face-to-face setting) and began the experimental session. In every session, after the experimenter acquired the one-minute resting data, he asked the participants to open their eyes and get ready for the start of the story which was then immediately followed by the story being told. During the face-to-face setting, we asked the storyteller to maintain as much eye-contacts with the participants as possible. We also used the Tobii Eye Tracker 4C controller which has a sampling rate of 90.0 Hz to acquire the participants’ eye-movement data. Tobii was placed at approximately 80.0 cm from the participant’s seat and 30.0 cm above the ground. To avoid any interference between the eye-tracker and the fNIRS devices, we placed it between the stand of the media and the chair of participants.
Once a storytelling session was over, the participants filled in a questionnaire that asked them how difficult they thought the story content was. We used an 8-point scale questionnaire in which “1” meant “not difficult at all” and “8” denoted “very difficult” story content. We gave our participants a one-minute rest period prior to the commencement of each of the storytelling sessions and asked them to keep their eyes closed. In this period, we prepared the setting for the next storytelling session. We video-recorded all the activities throughout the experiment.
Every individual participated in each of the four storytelling sessions. We kept the content of the stories and their orders in these sessions intact (i.e., the first story was always the same for all participants and regardless of the medium) while randomizing the order of the media. Each experiment took about 90 minutes for each participant.
Analysis
We used the decision boundary 𝔻 that was determined based on one- and two-back WM tasks’ CLs in the second experiment during the realtime storytelling experiment.
We followed the same procedure as in the first experiment for data acquisition and processing. At every estimation step in current implementation, our model calculated the CL of the current PFC activity time series (per participant per medium). At the end of the storytelling session, our model computed the median of these computed CLs and determined whether it was above or below the decision boundary 𝔻. Subsequently, it marked the individual’s perception of the story content as “difficult” if this median was above the decision boundary. Otherwise, it marked it as “easy.”
We also used these medians per participant along with their self-assessed responses to the difficulty of the story’s content per medium to determine our model’s true positive (tp), true negative (tn), false positive (fp), and false negative (fn). Concretely, we considered self-assessed responses 1 through 4 and 5 though 8 to represent “easy” and “difficult” content, respectively. We then evaluated our model’s estimate as a “Hit” if MdnCLparticipant was above the decision boundary 𝔻 and the self-assessed response ≥5 or if MdnCLparticipant was below the decision boundary 𝔻 and the self-assessed response ≤4 (i.e., if they matched). Otherwise, we evaluated its estimate as a “Miss.” Every “Hit” by our model contributed to a “tp” (i.e., MdnCLparticipant was above the decision boundary 𝔻 and the self-assessed response ≥5) or a “tn” (i.e., MdnCLparticipant was below the decision boundary 𝔻 and the self-assessed response ≤4). On other hand, we considered the model’s estimate as a “fp” if MdnCLparticipant was above the decision boundary 𝔻 and the participant’s self-assessed response ≤4. Similarly, we considered it as a “fn” if MdnCLparticipant was below the decision boundary 𝔻 and the participant’s self-assessed response ≥5. We used these tp, tn, fp, and fn values to calculate the confusion matrix, the accuracy, the precision, the recall, and the F1-score of our model in each of the media settings.
Last, we computed the Spearman correlation between participants’ self-assessed responses to difficulty of story content and their CLs. In case of all results, we reported the results for the left-hemispheric PFC in the manuscript. We provided the results for the right-hemispheric PFC in SM 2.2.
Results
Figure 5(A) shows the distribution of one- (red) and two-back (blue) CLs prior to the application of Algorithm 1. Figure 5(B) plots their resulting non-overlapping CLs after the application of this algorithm (nine CLs per task discarded in total). Wilcoxon rank sum indicated that (Fig. 5(C)) the significant difference between one- and two-back CLs after the refinement step was preserved (p < 0.001, W(42) = 5.67, r = 0.85, Mone−back = 1.65, SDone−back = 0.43, Mtwo−back = 3.07, SDtwo−back = 0.37).
(A) One- (red) and two-back (blue) CLs prior to application of Algorithm 1. (B) Non-overlapping CLs of one- (red) and two-back (blue) WM tasks. (C) Wilcoxon rank sum between refined CLs of one- and two-back WM tasks. Asterisks mark their differential significance. (D) Refined one- and two-back CLs along with the medians of the CLs of the participants’ PFC activation during storytelling experiment (as computed by our proposed model) in speaker (S), video-chat (V), Telenoid (T), and face-to-face (F) media settings. The decision boundary 𝔻 at CL ≈ 2.30 is shown in this subplot. The CLs associated with one- and two-back WM tasks are also presented in this subplot to better visualize the correspondence between the decision boundary 𝔻 and these CLs distributions.
Figure 5(D) shows the median of the participants’ CLs during the realtime storytelling experiment in speaker (S), video-chat (V), Telenoid (T), and face-to-face (F) media settings. These CLs are presented next to one- and two-back CLs for better visualization of their distribution with respect to these WM tasks’ CLs. The decision boundary 𝔻 by Algorithm 1 𝔻 at CL ≈ 2.30 is shown in this subplot.
Our model was able to predict the participants’ perceived difficulty of the story content with 80.0% prediction accuracy in the speaker setting. In addition, its accuracy was 85.0% in case of the video-chat and Telenoid media. Last, it predicted their perceived difficulty of the story content with 90.0% accuracy during the face-to-face setting. Table 2 summarizes the performance statistics of our model in these media settings during the realtime storytelling experiment.
Figure 6 shows the confusion matrices of our model during the storytelling experiment in speaker (S), video-chat (V), Telenoid (T), and face-to-face (F) media settings. We observed that our model was stronger in differentiating the participants’ PFC activation in response to perceived difficulty of the story content in case of the Telenoid (Fig. 6T, True Positive block). Similarly, it differentiated best the content that was perceived “easy” by the participants in case of the face-to-face setting (Fig. 6F, True Negative block). On the other hand, it did slightly worse for estimation of the perceived difficulty of the story content in case of speaker (Fig. 6S, False Negative). Whereas it misidentified two “easy” cases as “difficult” content in case of the Telenoid (Fig. 6T, False Positive block), such a misestimation was one in case of the speaker (Fig. 6S, False Positive block) and the video-chat (Fig. 6V, False Positive block).
Confusion matrices of our model during the storytelling experiment for speaker (S), video-chat (V), Telenoid (T), and face-to-face (F) media. In this figure, “Difficult” and “Easy” represent the number of participants whose self-assessed responses indicated “difficult” or “easy” story content. Results of the participants’ self-assessment responses to difficulty of the story content identified that fifteen and five participants considered the story content difficult and easy in the speaker and the face-to-face. These numbers were sixteen and four in the video-chat and the Telenoid settings.
These observations were further supported by the precision, recall, and F1-score of the model’s performance. Table 2 indicates that whereas our model’s performance obtained its highest precision in the face-to-face setting that was followed by the video-chat and speaker, its recall was highest in the case of Telenoid. Interestingly, this table also reveals a direct correspondence between the physical embodiment of the medium and the performance of our model. Specifically, we observed that face-to-face and Telenoid were associated with the first and the second highest F1-score values among the four media settings. We also observed that our model achieved its highest accuracy in the face-to-face setting which was followed by its higher accuracy in the case of Telenoid and the video-chat while the speaker setting was associated with lowest model accuracy among the four media settings.
Last, we observed significant correlations (Fig. 7) between the participants’ self-assessed responses to difficulty of the story content and their CLs in speaker (r = 0.50, p < 0.03, uncorrected), video-chat (r = 0.43, p < 0.05, uncorrected), Telenoid (r = 0.48, p < 0.05, uncorrected), and face-to-face (r = 0.47, p < 0.05, uncorrected) settings.
Discussion
In this article, we presented a novel information-theoretic approach to quantification of the cognitive loads associated with the human subjects’ PFC activity. Our choice of information theory41 for modeling of the PFC pattern of activity was motivated by the neuroscientific findings that have provided compelling empirical30,31,32 and theoretical58,59 evidence that emphasize the crucial role of signal variability in the quantification of the brain activity. This, in turn, has resulted in emerging viewpoints that identify the role of entropy in brain functioning60. For instance, our quantification of the induced cognitive load on PFC in terms of the interplay between mutual information (i.e., convergence) and Kullback-Leibler divergence is motivated by a new thesis on the entropic nature of the brain61 that itself is based on findings that identify the functioning of the brain near criticality62,63.
We showed that the use of this approach (in conjunction with a decision boundary that corresponded to the refined cognitive loads induced by the one- and two-back auditory tasks) was successful in predicting the individuals’ perceived difficulty of communicated content in a storytelling scenario. Our results indicated that our model maintained a significantly above average (50.0% chance level in case of binary classification) prediction accuracy with a comparable performance in face-to-face, humanoid, speaker, and video-chat media settings. This verified that the quantified participants’ PFC activation in the form of CL was primarily due to the effect of communicated content than such confounders as choice of the medium. This observation was also supported by the comparative analysis of the participants’ self-assessment responses in different media settings (SM Section 3). More importantly, our model was able to estimate the effect of stories’ difficulty on individuals’ PFC activity bilaterally which was in line with the neuroscientific findings on bilateral effect of stories on human subjects’ PFC13,14,15.
Previous findings have shown that certain patterns of brain activity can provide means to uncover the subjective contents that might at least partly be shared among individuals64,65,66. Our results extended these findings by demonstrating that such potential shared spaces can be utilized for decoding of the individuals’ PFC activity to predict their perceived difficulty of the narrated stories. From a broader perspective, our approach was related to research that focuses on the application of machine learning67 and other computational paradigms68 for decoding of the brain activity in response to stimuli.
An interesting observation during the storytelling experiment was the incremental improvement of the prediction accuracy of our model from speaker (i.e., total absence of storyteller) to face-to-face settings. In addition, we observed that our model achieved its highest correctly predicted easy (i.e., true negative) and difficult (i.e., true positive) contents in the face-to-face and the humanoid settings, respectively. These results may suggest the potential benefit of the physical embodiment on quantification of the human subjects’ PFC activation in response to verbally communicated contents. However, further investigation of the extent of such an effect is necessary prior to drawing any conclusion on this possibility.
Our results contribute to such socially assistive robotics1,69 scenarios as child education and elderly care. For instance, our model can enable these media to determine whether their level of interaction (e.g., socialization2, reading and comprehension3) is exceeding the comfort level of children, thereby allowing for modulation of their communicated contents and/or behavioural interaction. Our model can also further enhance the use of these media in robot-assistive cognitive training of the older people70. For example, robots equipped with our model can be used during cognitive training of older people in elderly care facilities to monitor these individuals’ brain activity during their training, thereby allowing the cognitive trainers to determine the older people’s level of comfort in continuing their training session4,5.
Our model’s performance during the storytelling experiment that was in line with the neuroscientific findings on the effect of story on human subjects’ PFC activation13,14,15 indicated a promising first step toward the use of brain information for quantification of one of the basic component of the human mental state: perceived difficulty of verbally communicated contents. These results benefit such HRI paradigms as interactive learning38 by allowing these algorithms to utilize the individuals’ patterns of brain activity as real-time neurological feedbacks to refine their interaction strategy. In a broader perspective, our findings can benefit the research on PFC activation during social cognition12,13 and its involvement in story-based ToM analyses71. More importantly, the ability of the robots to estimate their human companions’ perceived difficulty of their verbal communication can contribute to formal analysis of a robotic ToM39 through critical investigation of its implications in humans’ neurological responses while interacting with these agents.
Limitations and future direction
Although our model indicated a promising first step toward quantification of the human subjects’ PFC activation in response to difficulty of verbally communicated content, a larger human sample is necessary for an informed conclusion on its utility. Moreover, our participants were limited to the younger adults. Therefore, it is necessary to investigate the performance of our model in other age groups (e.g., kids, adolescents, older people) to verify that its performance is unaffected by this factor.
Our analyses indicated that our model was able to quantify the PFC activity in response to one- and two-back auditory tasks differentially, thereby allowing for their classification with a high accuracy. An important issue in this regard that demands further investigation is the order by which the participants performed these WM tasks. In particular, in our study every individual first performed the one-back which was then followed by the two-back task. Although our further analyses identified that the CLs associated with two-back WM task was significantly due to the effect of this task than a residual effect of one-back task. it is crucial for the future studies to investigate whether counter-balancing the order of these task may impose any impact on the performance of the proposed approach.
We also observed that participants’ self-assessed difficulty of stories correlated with change in their PFC activity. This highlighted the potential presence of a direct correspondence between individuals’ subjective evaluation of the communicated contents (e.g., stories) and the effect of such contents on their PFC information processing. On the other hand, whereas we observed that such correlations were bilateral in the case of speaker, Telenoid, and face-to-face settings, it was only present in left-hemispheric PFC in the case of video-chat. This differed from our prediction results in which we observed that our model was able to significantly predict the difficulty of story content in all media settings, that such significantly above chance predictions were present bilaterally in the case of all media, and that the increase of such predictions were incremental in the level of embodiment. Therefore, the future research is necessary to further investigate the source of such a difference between correlation and prediction analyses.
Moreover, our experimental setting was limited to a storytelling scenario in which participants listened to a verbally communicated content without any requirement for their response. Therefore, it is crucial to examine the performance of our model in such verbal communication scenarios as conversation to ensure its performance is unaffected by any potential PFC activation induced by such bidirectional verbal communications.
We used the participants’ self-assessed responses to difficulty of stories to investigate the accuracy of our model. This constrained our validation procedure in that we were forced to wait until the storytelling ended and therefore were not able to examine its performance finer time scales such as minute-prediction-cycle or shorter. This constraint was imposed by the fact any finer-scale prediction cycle inevitably needs us to interrupt of the individuals’s listening to the stories to inquire about their subjective feeling of the story’s difficulty, thereby disrupting their thoughts as well as the course of verbal communication. Therefore, future research to look into alternative strategies to acquire individuals’ self-assessment during the communication is necessary to draw a more informed conclusion on performance of our model.
We observed an incremental prediction accuracy from the speaker to face-to-face media settings. We also observed that our model achieved its highest correctly predicted easy (i.e., true negative) and difficult (i.e., true positive) contents in the face-to-face and the humanoid settings, respectively. This suggested a potential positive effect of embodiment on quantification of the PFC activation during verbal communication. However, present study did not include other types of physically embodied media (e.g., mechanical looking robots, pet robots, etc.). Therefore, it is necessary to determine the correspondence between the media embodiment and the observed incremental accuracy of our model. It is also important to verify whether different embodiments can induce differential impact on this incremental pattern of accuracy.
Our approach is based on information theory. Integral to this formalism is the Data Processing Inequality (DPI) [41, p. 34, Theorem 2.8.1] which states that the more the data manipulation (e.g., data pre/processing steps) the more the loss of information. Therefore, it was crucial for us to apply the pre/processing steps as efficiently as possible. In this regard, Cooper et al.52 showed that PCA-based motion correction algorithm yielded a significant reduction in the mean-squared error (MSE) and a significant increase in contrast-to-noise ratio (CNR) in comparison with no correction and/or the process of rejecting the motion-contaminated trials. Considering the fNIRS more immunity to body movement as compared with other neuroimaging techniques72, the minimal physical activity in our experimental settings (e.g., clicking mouse in n-back), and the recent findings that indicated that the measured PFC activity during listening to stories was not affected by such behavioural responses as eye-movement55, we found the application of PCA sufficient for addressing both SBF attenuation and potential minimal motion artefacts correction, thereby adhering with DPI principle as much as possible. However, the future research requires to take into consideration the effectiveness of our methodology in the use of PCA in scenarios in which substantially more physical activity and/or embodied interaction is expected, thereby examining the utility of other approaches for motion artefacts correction52.
References
Matarić, M. J. Socially assistive robotics: Human augmentation versus automation. Science Robotics 2, eaam5410 (2017).
Tanaka, F., Cicourel, A. & Movellan, J. R. Socialization between toddlers and robots at an early childhood education center. Proceedings of the National Academy of Sciences 104, 17954–17958 (2007).
Michaelis, J. E. & Mutlu, B. Reading socially: Transforming the in-home reading experience with a learning-companion robot. Science Robotics 3, eaat5999 (2018).
Robinson, H., MacDonald, B., Kerse, N. & Broadbent, E. The psychosocial effects of a companion robot: a randomized controlled trial. Journal of the American Medical Directors Association (JAMA) 14, 661–667 (2013).
Valentí Soler, M. et al. Social robots in advanced dementia. Frontiers in Aging Neuroscience 7, 133 (2015).
Yang, G.-Z. et al. The grand challenges of Science Robotics. Science Robotics 3, eaar7650 (2018).
Corneanu, C. A., Simón, M. O., Cohn, J. F. & Guerrero, S. E. Survey on RGB, 3D, thermal, and multimodal approaches for facial expression recognition: History, trends, and affect-related applications. IEEE Transactions on Pattern Analysis and Machine Intelligence 38, 1548–1568 (2016).
Donoghue, J. P. Connecting cortex to machines: recent advances in brain interfaces. Nature Neuroscience 5, 1085 (2002).
Canning, C. & Scheutz, M. Functional near-infrared spectroscopy in human-robot interaction. Journal of Human-Robot Interaction 2, 62–84 (2013).
Esfahani, E. T. & Sundararajan, V. Using brain? Computer interfaces to detect human satisfaction in human-robot interaction. International Journal of Humanoid Robotics 8, 87–101 (2011).
Gabrieli, J. D., Poldrack, R. A. & Desmond, J. E. The role of left prefrontal cortex in language and memory. Proceedings of the National Academy of Sciences 95, 906–913 (1998).
Forbes, C. E. & Grafman, J. The role of the human prefrontal cortex in social cognition and moral judgment. Annual Review of Neuroscience 33, 299–324 (2010).
Mar, R. A. The neural bases of social cognition and story comprehension. Annual Review of Psychology 62, 103–134 (2011).
Mar, R. A. The neuropsychology of narrative: Story comprehension, story production and their interrelation. Neuropsy chologia 42, 1414–1434 (2004).
Lerner, Y., Honey, C. J., Silbert, L. J. & Hasson, U. Topographic mapping of a hierarchy of temporal receptive windows using a narrated story. Journal of Neuroscience 31, 2906–2915 (2011).
Verner, M., Herrmann, M. J., Troche, S. J., Roebers, C. M. & Rammsayer, T. H. Cortical oxygen consumption in mental arithmetic as a function of task difficulty: a near-infrared spectroscopy approach. Frontiers in Human Neuroscience 7, 217 (2013).
Owen, A. M., McMillan, K. M., Laird, A. R. & Bullmore, E. N-back working memory paradigm: A meta-analysis of normative functional neuroimaging studies. Human Brain Mapping 25, 46–59 (2005).
Ozawa, S., Matsuda, G. & Hiraki, K. Negative emotion modulates prefrontal cortex activity during a working memory task: a NIRS study. Frontiers in Human Neuroscience 8, 46 (2014).
Kinney, J. B. & Atwal, G. S. Equitability, mutual information, and the maximal information coefficient, Proceedings of the National Academy of Sciences, 201309933 (2014).
Kriegeskorte, N., Goebel, R. & Bandettini, P. Information-based functional brain mapping. Proceedings of the National Academy of Sciences 103, 3863–3868 (2006).
Zhang, Y.-C. Complexity and 1/f noise. A phase space approach. Journal de Physique I 7, 971–977 (1991).
Borst, A. & Theunissen, F. E. Information theory and neural coding. Nature Neuroscience 2, 947 (1999).
Quiroga, R. Q. & Panzeri, S. Extracting information from neuronal populations: information theory and decoding approaches. Nature Reviews Neuroscience 10, 173 (2009).
Sengupta, B., Stemmler, M. B. & Friston, K. J. Information and efficiency in the nervous system - a synthesis. PLoS Computational Biology 9, e1003157 (2013).
Gusnard, D. A. & Raichle, M. E. Searching for baseline: functional imaging and the resting human brain, Nature Review. Neuroscience 2, 685–694 (2001).
Heekeren, H. R., Marrett, S. & Ungerleider, L. G. The neural systems that mediate human perceptual decision making. Nature Reviews Neuroscience 9, 467–479 (2008).
Ben-Yakov, A., Honey, C. J., Lerner, Y. & Hasson, U. Loss of reliable temporal structure in event-related averaging of naturalistic stimuli. NeuroImage 63, 501–506 (2012).
Haynes, J. D. & Rees, G. Decoding mental states from brain activity in humans. Nature Reviews Neuroscience 7, 523–534 (2007).
Spiers, H. J. & Maguire, E. A. Decoding human brain activity during real-world experiences. Trends in Cognitive Sciences 11, 356–365 (2007).
Garrett, D. D., Kovacevic, N., McIntosh, A. R. & Grady, C. L. The importance of being variable. Journal of Neuroscience 31, 4496–4503 (2011).
Heisz, J. J., Shedden, J. M. & McIntosh, A. R. Relating brain signal variability to knowledge representation. Neuroimage 63, 1384–1392 (2012).
Garrett, D. D. et al. Moment-to moment brain signal variability: a next frontier in human brain mapping? Neuroscience & Biobehavioural Reviews 37, 610–624 (2013).
Applebaum, D. Probability and information: An integrated approach, Cambridge University Press, Cambridge (1996).
Keshmiri, S., Sumioka, H., Yamazaki, R. & Ishiguro, H., Differential entropy preserves variational information of near-infrared spectroscopy time series associated with working memory, Frontiers in Neuroinformatics, 12 (2018).
Li, T., Luo, Q. & Gong, H. Gender-specific hemodynamics in prefrontal cortex during a verbal working memory task by near-infrared spectroscopy. Behavioural Brain Research 209, 148–153 (2010).
Sato, H., Dresler, T., Haeussinger, F. B., Fallgatter, A. J. & Ehlis, A. C. Replication of the correlation between natural mood states and working memory-related prefrontal activity measured by near-infrared spectroscopy in a German sample. Frontiers in Human Neuroscience 8, 37 (2014).
Fishbum, F., Norr, M., Medvedev, A. & Vaidya, C. Sensitivity of fNIRS to cognitive state and load, Frontiers in Human Neuroscience 8 (2014).
Clabaugh, C. & Matarić, M. J. Robots for the people, by the people: Personalizing human-machine interaction. Science Robotics 3, eaat7451 (2018).
Scassellati, B. Theory of mind for a humanoid robot. Autonomous Robots 12, 13–24 (2002).
Miller, G. A. The magical number seven, plus or minus two: Some limits on our capacity for processing information. Psychological Review 63, 81 (1956).
Cover, T. M. & Thomas, J. A. Elements of information theory Second Edition, John Wiley & Sons, Inc., NY (2006).
Lundqvist, M., Herman, P. & Miller, E. K. Working memory: delay activity, yes! persistent activity? maybe not. Journal of Neuroscience 38, 7013–7019 (2018).
Stam, C. J. Nonlinear dynamical analysis of EEG and MEG: review of an emerging field. Clinical Neurophysiology 116, 2266–2301 (2005).
Meilă, M. Comparing clusterings - an information based distance. Journal of Multivariate Analysis 98, 873–895 (2007).
Takahashi, T. et al. Influence of skin blood flow on near-infrared spectroscopy signals measured on the forehead during a verbal fluency task. NeuroImage 57, 991–1002 (2011).
Gagnon, L. et al. Improved recovery of the hemo dynamic response in diffuse optical imaging using short optode separations and state-space modeling. NeuroImage 56, 1362–1371 (2011).
Sato, T. et al. Reduction of global interference of scalp-hemodynamic in functional near-infrared spectroscopy using short distance probes. NeuroImage 141, 120–132 (2016).
Yamada, T., Umeyama, S. & Matsuda, K. Multidistance probe arrangement to eliminate artifacts in functional near-infrared spectroscopy. Journal of Biomedical Optics 14, 120–132 (2009).
Tak, S. & Ye, J. C. Statistical analysis of fNIRS data: a comprehensive review. NeuroImage 85, 72–91 (2014).
Zhang, Y., Brooks, D. H., Franceschini, M. A. & Boas, D. A. Eigenvector-based spatial filtering for reduction of physiological interference in diffuse optical imaging. Journal of Biomedical Optics 10, 011014 (2005).
Keshmiri, S., Sumioka, H., Okubo, M. & Ishiguro, H. An Information-Theoretic Approach to Quantitative Analysis of the Correspondence Between Skin Blood Flow and Functional Near-Infrared Spectroscopy Measurement in Prefrontal Cortex Activity. Frontiers in Neuroscience 13, 79 (2019).
Cooper, R. et al. A systematic comparison of motion artifact correction techniques for functional near-infrared spectroscopy. Frontiers in Neuroscience 6, 147 (2012).
Friston, K. et al. Statistical parametric maps in functional imaging: A general linear approach. Human Brain Mapping 2, ? (1994).
Cardoso, M. M., Sirotin, Y. B., Lima, B., Glushenkova, E. & Das, A. The neuroimaging signal is a linear sum of neutrally distinct stimulus-and task-related components. Nature Neuroscience 15, 1298–1306 (2012).
Keshmiri, S., Sumioka, H., Yamazaki, R. & Ishiguro, H. Differential Effect of the Physical Embodiment on the Prefrontal Cortex Activity as Quantified by Its Entropy. Entropy 21, 875 (2019).
Sumioka, H., Nishio, S., Minato, T., Yamazaki, R. & Ishiguro, H. Minimal human design approach for sonzai-kan media: investigation of a feeling of human presence. Cognitive Computation 6, 760–774 (2014).
Sakai, K., Minato, T., Ishi, C. T. & Ishiguro, H., Novel Speech Motion Generation by Modelling Dynamics of Human Speech Production. Frontiers in Robotics and AI, 4 (2017).
Pouget, A., Drugowitsch, J. & Kepecs, A. Confidence and certainty: distinct probabilistic quantities for different goals. Nature Neuroscience 19, 366–374 (2016).
Friston, K. The free-energy principle: a unified brain theory? Nature Reviews Neuroscience 11, 127–138 (2010).
Tononi, G., Boly, M., Massimini, M. & Koch, C. Integrated information theory: from consciousness to its physical substrate. Nature Reviews Neuroscience 17, 450–461 (2016).
Carhart-Harris, R. L. The entropic brain-revisited. Neuropharmacology 142, 167–178 (2018).
Shew, W. L. & Plenz, D. The functional benefits of criticality in the cortex. The Neuroscientist 19, 88–100 (2013).
Fagerholm, E. D. et al. Cascades and cognitive state: focused attention incurs subcritical dynamics. Journal of Neuroscience 35, 4626–4634 (2015).
Akbari, H., Khalighinejad, B., Herrero, J. L., Mehta, A. D. & Mesgarani, N. Towards reconstructing intelligible speech from the human auditory cortex. Scientific Reports 9, 874 (2019).
Mitchell, T. M. et al. Predicting human brain activity associated with the meanings of nouns. Science 320, 1191–1195 (2008).
Liu, Y. & Ayaz, H. Speech recognition via fNIRS based brain signals. Frontiers in Neuroscience 12, 695 (2018).
Mitchell, T. M. et al. Learning to decode cognitive states from brain images. Machine Learning 57, 145–175 (2004).
Haxby, J. V., Connolly, A. C. & Guntupalli, J. S. Decoding neural representational spaces using multivariate pattern analysis. Annual Review of Neuroscience 37, 435–456 (2014).
Matarić, M.J. & Scassellati, B. 2017. Socially assistive robotics, Springer Handbook of Robotics, Springer International Publishing,Siciliano, B. & O. Khatib, O., Eds, 1973–1994 (2016).
Kim, G. H. et al. Structural brain changes after traditional and robot-assisted multi-domain cognitive training in community-dwelling healthy elderly. PloS One 10, e0123251 (2015).
Molenberghs, P., Johnson, H., Henry, J. D. & Mattingley, J. B. Understanding the minds of others: A neuroimaging meta-analysis. Neuroscience & Biobehavioural Reviews 65, 276–291 (2016).
Dieler, A. C., Tupak, S. V. & Fallgatter, A. J. Functional near-infrared spectroscopy for the assessment of speech related tasks. Brain and Language 12, 90–109 (2012).
Acknowledgements
This research was supported by JSPS KAKENHI Grant Number JP19K20746, JST CREST Grant Number JPMJCR18A1, JST ImPACT Grant Number 2014-PM11-07-01, and JSPS KAKENHI Grant Number 16K16480.
Author information
Authors and Affiliations
Contributions
S.K. proposed the model and carried out the analyses. H.S. designed the experiments and supervised their progress. R.Y. conducted the experiments and performed the data collection. As the head of Hiroshi Ishiguro Laboratories (HIL), H.I. oversees the entire activity of all research teams and themes, ensuring the soundness of all proposals, quality of results, and their validity. S.K., H.S. and M.S. contributed equally in preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that the research was conducted in the absence of any commercial, financial, and/or non-financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Keshmiri, S., Sumioka, H., Yamazaki, R. et al. Information Content of Prefrontal Cortex Activity Quantifies the Difficulty of Narrated Stories. Sci Rep 9, 17959 (2019). https://doi.org/10.1038/s41598-019-54280-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-54280-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.