Abstract
We explored the frequency of germline and somatic mutations in homologous recombination (HR)-associated genes in major histological types of ovarian cancer. We performed targeted sequencing to assess germline and somatic mutations of 16 HR-associated genes and 4 mismatch repair (MMR) genes among 207 ovarian cancer patients (50 high-grade serous carcinomas (HGSC), 99 clear cell carcinomas (CCC), 39 endometrioid carcinomas (EC), 13 mucinous carcinomas (MC), and 6 low-grade serous carcinomas (LGSC)). Germline or somatic mutations of HR-associated genes were detected in 44% of HGSC, 28% of CCC, 23% of EC, 16% of MC, and 17% of LGSC patients. The profile of HR-associated gene mutations was remarkably different among each histological type. Germline BRCA1/2 mutations were frequently detected in HGSC and were rarely observed in CCC, EC, and MC patients. ATM somatic mutation was more frequently detected in CCC (9%) and EC patients (18%) than in HGSC patients (4%). There was a positive correlation between MMR gene mutations and HR-associated gene mutations (p = 0.0072). Our findings might be useful in selection of ovarian cancer patients that should be treated with PARP inhibitors.
Similar content being viewed by others
Introduction
Recently, the prevalence of homologous recombination (HR)-associated gene mutations among many tumor types has been characterized1,2. In particular, HR pathway alterations are most frequently observed in high-grade serous ovarian carcinoma (HGSC) and breast cancer2. It is well-known that around half of HGSC patients exhibit HR deficiency3,4. HR deficiency is also associated with response to platinum-based chemotherapies in patients with ovarian cancer5, and germline BRCA1 and BRCA2 mutations, which are representative alterations causing HR deficiency, are undoubtedly associated with improved prognosis in advanced-stage ovarian cancers6. In a retrospective analysis, Pennington et al.7 have found that the ovarian cancer patients with germline or somatic mutations in 13 HR-associated genes (BRCA1, BRCA2, ATM, BARD1, BRIP1, CHEK1, CHEK2, FAM175A, MRE11A, NBN, PALB2, RAD51C, and RAD51D) had higher platinum sensitivity and prolonged overall survival than those without HR-associated gene mutations.
Clinical use of PARP inhibitors that induces synthetic lethality in HR deficient (HRD) cancer cells has a great impact on treatment strategies for ovarian cancer8. Niraparib maintenance therapy has shown prolonged progression-free survival (PFS) in platinum-sensitive, recurrent ovarian cancer patients with HRD9. Rucaparib maintenance has also improved PFS in platinum-sensitive, recurrent ovarian cancer patients with HRD10. However, the majority of subjects in clinical trials of PARP inhibitors were type II ovarian cancer patients9,10,11,12,13, and thus, the efficacy of PARP inhibitors for type I ovarian cancer, such as clear cell or low-grade endometrioid types, remains unclear. To date, the frequency of germline and somatic HR-associated gene mutations in type I ovarian cancer has yet to be elucidated fully.
In this study, we focused on difference in the distribution of ovarian cancer histologic subtypes between Western countries and Japan14. For example, clear cell carcinoma (CCC) accounts for 25% of ovarian cancer in Japan and less than 10% in the United States15,16. Therefore, we aimed to identify the frequency of germline and somatic HR-associated gene mutations per major histological subtypes of ovarian cancer in Japan, suggesting the therapeutic strategy of PARP inhibitors for ovarian cancers with HR-associated gene mutations.
Results
Clinicopathological characteristics of ovarian cancer patients
Clinicopathological characteristics of 207 patients (197 ovarian, 9 peritoneal, and 1 fallopian tube cancer) are shown in Table 1. Neoadjuvant chemotherapy cases that mainly consisted of HGSC patients were excluded. The median onset age of all patients was 56.0 years and more than half of patients (66%) were diagnosed at stage I. In all patients, the frequency of CCC (48%) was higher than that of HGSC (24%) that was the most common type of epithelial ovarian cancer17. Although more than half of HGSC and low-grade serous carcinoma (LGSC) patients were at stage III, over 60% of CCC, endometrioid carcinoma (EC), and mucinous carcinoma (MC) patients were at stage I.
Next, we assessed mutation status of five genes (TP53, ARID1A, PIK3CA, KRAS, and PTEN), which were frequently mutated in ovarian cancer18, for each histologic type (Table 1). Most of HGSC (82%) and MC (62%) patients harbored TP53 somatic mutations. CCC patients were characterized by high frequency of ARID1A (70%) and PIK3CA (64%) somatic mutations. EC patients harbored KRAS (46%) and PTEN (39%) somatic mutations in addition to ARID1A (46%) and PIK3CA (41%) somatic mutations.
Landscape of HR-associated gene mutations in ovarian cancer
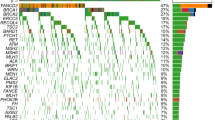
We investigated germline and somatic mutations of 16 HR-associated genes in 207 ovarian cancer samples. The average sequencing depth and the percentage of the target lesion that covered at least 20 reads were on average 98.6 and 98.9% in all samples, respectively. All the somatic mutations in HR-associated genes are listed in Supplementary Table S2. Missense mutation was the most frequent type of mutation (64%), followed by stopgain mutation (21%), frameshift insertion and deletion (11%), and splicing mutation (3%). Among 207 samples, 42 samples (20%) harbored at least one HR-associated gene mutation. The frequencies of germline and somatic HR-associated mutation in each histological subtype are shown in Fig. 1. Germline or somatic HR-associated gene mutations were detected in 44% of HGSC, 28% of CCC, 23% of EC, and 16% of MC, and 17% of LGSC patients. We investigated the correlation between stage and HR-associated gene mutations in each histological subtype (HGSC, CCC and EC). All ECs harboring HR-associated gene mutations or germline BRCA mutations were diagnosed as stage I. On the other hand, there was no obvious difference of HR-associated mutation frequency per stage in HGSC and CCC (Supplementary Fig. S1).
Frequency of HR-associated gene mutation based on histology. The frequency of HR-associated gene mutation based on histology is shown in each pie chart. The mutation data were classified into seven categories – germline BRCA mutation (gBRCA m), somatic BRCA mutation (sBRCA m), both germline and somatic BRCA mutation (bBRCA m), other germline gene mutation (gOther m), other somatic gene mutation (sOther m), other germline and somatic gene mutation (bOther m), and no mutation.
All germline mutations identified in our dataset are shown in Table 2. We detected 24 germline mutations in 22 ovarian cancer patients. Among the 24 germline HR-associated gene mutations, 12 (50%) were detected in HGSC patients, and almost all of them were either BRCA1 or BRCA2. Intriguingly, BRCA2 mutation was identified in one patient each of CCC, EC, and MC. When we focused on ovarian or breast cancer history in first- or second-degree relatives, 6 of 22 patients (27%) with a germline HR-associated gene mutation had family history with ovarian cancer or breast cancer. Of them, five patients were diagnosed with HGSC harboring germline BRCA1/2 mutations.
Somatic HR-associated gene mutations per histological subtype
Next, we compared the frequency of HR-associated gene alterations among three major histological subtypes (HGSC, CCC, and EC) of ovarian cancer (Fig. 2). BRCA1/2 somatic mutations were detected more frequently in HGSC patients (12%) than in CCC (5%) or EC patients (5%). However, ATM somatic mutations were detected more frequently in CCC (9%) and EC patients (18%) than in HGSC patients (4%). Most of the other HR-associated gene mutations were detected in a small population of each histological subtype.
Clinical significance of HR-associated gene mutations
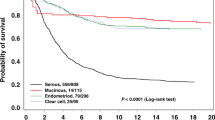
We divided HGSC, CCC, and EC into two subgroups based on the status of HR-associated gene mutation and compared clinical characteristics between two subgroups in each histological subtype (Supplementary Table S3). EC patients with HR-associated gene mutation had younger age of onset than those without HR-associated gene mutations. No significant prognostic difference was observed between patients with and without HR-associated gene mutation irrespective of histology (Fig. 3). When we focused on only BRCA1/2 mutations, there were no significant differences in progression-free or overall survival between patients with and without BRCA1/2 mutations (Supplementary Fig. S2).
Correlation between mismatch repair gene mutations and HR-associated gene mutations
We investigated germline and somatic mutations of four mismatch repair (MMR) genes (MLH1, MSH2, MSH6, and PMS2) in 207 ovarian cancer cases (Table 3). Ten patients (5%) harbored deleterious germline or somatic mutations in MMR genes. The frequency of MMR gene mutation was 2% (HGSC), 3% (CCC), and 10% (EC) and there was no significant difference in the frequency of MMR gene mutation among histological subtypes. Subsequently, we investigated the correlation between MMR and HR-associated gene mutations. More than half of HR-associated gene mutation and MMR gene mutation were somatic mutations (63.3% and 70%) (Supplementary Fig. S3A). Patients with germline or somatic MMR gene mutation exhibited significantly higher frequency of germline or somatic HR-associated gene mutations (p = 0.0072) (Supplementary Fig. S3B).
Discussion
In this study, we demonstrated the frequency of germline and somatic HR-associated gene mutations in Japanese patients with ovarian cancer. In Japan, three studies19,20,21 have clarified the frequency of germline BRCA1/2 mutation in ovarian cancer but not somatic BRCA1/2 mutation. Pennington et al.7 have assessed the frequency of germline and somatic HR-associated gene mutations in 390 ovarian cancer dataset largely composed of HGSC. The Cancer Genome Atlas (TCGA) network4 has performed exome sequencing and clarified the frequency of germline and somatic HR-associated gene mutations in 316 HGSC cases. These studies have demonstrated that HR-associated gene mutations might correlate with better prognosis of HGSC patients. On the other hand, there was no report to assess the frequency and clinical significance of germline and somatic HR-associated gene mutations in a large scale of non-HGSC cases. Therefore, we focused on difference in the frequency of germline and somatic HR-associated gene mutations among major histological types of ovarian cancer.
Major histological subtypes of ovarian cancer are characterized by several cancer-associated gene mutations22. For example, TP53 mutation was detected in 97% of HGSC patients4 and more than 50% of MC patients23,24. KRAS mutation was found in 50% of MC and 18% of LGSC25 but not in HSGC. Similarly, ARID1A and PIK3CA were frequently mutated in CCC and EC patients but not in HGSC26,27,28. Our result was almost consistent with those of previous studies. Although the distribution of ovarian cancer histological subtypes is different between Japan and Western countries, representative mutation profiles per histological subtype are similar beyond ethnicity.
Corresponding to previous studies4,7, our findings showed that germline and somatic BRCA1/2 mutations were frequently identified in HGSC patients. Somatic BRCA1/2 mutations were also identified in a part of CCC (5%) and EC (5%). Interestingly, germline BRCA2 mutation was identified in one sample each of CCC, EC, and MC. HR-associated gene mutations were also detected in HGSC, CCC, and EC samples24,29. Surprisingly, our data indicated that a small part of the MC patients harbored HR-associated gene mutations and thereby, PARP inhibitors might be a potent therapeutic alternative for these cancers. However, the sample size of MC patients in this study was small (n = 13). Further analysis will be needed to elucidate the clinical significance of HR-associated gene mutations in MC, which is usually refractory to conventional platinum-taxane chemotherapy30.
The frequency of somatic ATM mutation was higher in CCC and EC than in other histological subtypes. The ATM protein kinase plays an important role in DNA damage response31. Genetic alterations of ATM were found in many cancers32, such as colorectal cancer (10%), prostate cancer (8.8%), lung cancer (7.3%), and ovarian cancer (4.5%). The frequency of ATM mutation in CCC was previously reported to be 7% (3/48)27. ATM-deficient tumor cells, such as mantle cell lymphoma33 and colorectal cancer32 exhibited high sensitivity to the PARP inhibitor, olaparib in vitro and in vivo34. In a phase 2 clinical trial to assess the effectiveness of olaparib for metastatic prostate cancer and gastric cancer, ATM mutated cases had better prognosis than ATM wildtype cases35,36. Therefore, PARP inhibitors might provide a survival advantage to ATM-mutated ovarian cancer, especially for CCC and EC types and tumor sequencing might be important not to miss these somatic mutations,.
Mismatch repair (MMR) is a repair system of base mismatch pairing caused in DNA replication, and MMR genes (MLH1, MSH2, MSH6, and PMS2) often react to errors of a DNA single strand break. MMR gene mutations lead to microsatellite instability (MSI), and loss of function in germline MMR gene alterations cause Lynch syndrome37. PD-1 antibody contributes to a favorable benefit for patients with MSI-high cancer38. Our results showed positive correlation between mutation status of HR-associated genes and mismatch repair genes. It is inconclusive that defective MMR might contribute to platinum sensitivity for HGSC39. However, Fleury et al. have reported that downregulation of both HR pathway and MMR pathway increases sensitivity of PARP inhibitor40. Furthermore, the phase 2 study for advanced or metastatic triple-negative breast cancer has revealed that combination therapy of Niraparib with Pembrolizumab confers higher response rates in patients with tumor BRCA mutation41. Therefore, clinical trials evaluating the efficacy of PARP inhibitor and PD-1 antibody combination should be conducted for ovarian cancer patients.
Our findings indicated the possibility of HRD in major histological types of ovarian cancer. When we divided each histological group into two subgroups on the basis of HR-associated gene status in this study, there were no significant difference in clinical outcome between two subgroups. This might be due to several reasons. First, most of patients in this study were stage 1. Second, each HR-associated gene mutation was not evaluated at protein level for its pathogenicity. At least, we might need to evaluate an association of HR-associated gene mutation with other HRD assessment such as HRD score42 and HRDetect43. Third, the sample size of each histological subtype was still small, and we could not exclude any influence of other prognostic factors such as stage, residual disease, and chemotherapy.
In conclusion, we elucidated the mutation profile of HR-associated genes in major histological types of ovarian cancer. PARP inhibitors might provide survival advantage to ovarian cancer patients with HR-associated gene mutations beyond histological subtypes.
Methods
Clinical specimens
This study was performed in conformity with the Declaration of Helsinki and approved by the institutional ethics review board at Niigata University (G2016-0006). The subjects were 207 epithelial ovarian cancer patients who had undergone initial surgery between July 2006 and September 2017 at Niigata University Medical and Dental Hospital44. During the study period, we had enrolled 376 epithelial ovarian cancer patients. At first, 41 cases receiving neoadjuvant chemotherapy were excluded from this study. Then, 128 cases that could not provide the required DNA quality for blood and/or tumor samples were excluded. Finally, 207 epithelial ovarian cancer samples were collected as a cohort. All patients provided written informed consent for sample collection and subsequent analysis. Fresh-frozen tissue samples were obtained from primary tumor tissues. Staging of ovarian cancer was done following the criteria of the International Federation of Gynecology and Obstetrics (FIGO)45. Histological diagnosis was performed by two gynecological pathologists belonging to the Japanese Society of Pathology and assessed on formalin-fixed and paraffin-embedded hematoxylin and eosin sections. Histological subtypes were diagnosed according to WHO classification of ovarian tumors46. Tumor DNA was extracted from frozen tissues containing more than 70% of tumor cells, revealed by histological evaluation. PFS time was evaluated for the interval from primary surgery of a disease to disease progression or recurrence. Disease progression was defined as at least a 20% growth in the size of the tumor or the longest diameters of lesions or as the appearance of one or more new lesions and/or unequivocal progression existing non-target lesions since primary surgery following standard Response Evaluation Criteria In Solid Tumors (RECIST) guidelines47. Overall survival time was evaluated for the interval from primary surgery to the death by ovarian cancer.
DNA extraction
Tumor DNA extraction was performed with Tissue Genomic DNA Extraction Mini Kit (FAVORGEN), according to the manufacturer’s instructions. Blood DNA was extracted with the QIAamp DNA Blood Maxi kit (Qiagen) following the manufacturer’s instructions. Genomic DNA was quantified using a Qubit dsDNA HS Assay Kit (Thermo Fisher Scientific).
Targeted sequencing
Targeted sequencing of 16 HR-associated genes, 5 ovarian cancer-associated genes, and 4 mismatch repair genes (Supplementary Table S1) was conducted with the pre-capture pooling method described in our previous study48. In summary, 20 ng of DNA was simultaneously fragmented and adapter-ligated with a SureSelect QXT Library Prep Kit (Agilent Technologies). The fragmented libraries with distinct indexed adapters were preserved at equimolar amounts. Subsequently, target enrichment was performed using the SeqCap EZ Choice System (Roche Diagnostics). A DNA probe set complementary to the target region was selected by NimbleDesign (https://design.nimblegen.com). The libraries were sequenced on a MiSeq platform (Illumina). The paired-end read data were aligned to a human reference genome (hg19) using BWA49 and SAMtools50. The aligned reads were processed for removal of PCR duplicates using Picard tools (v.1.111; broadinstitute.github.io/picard). Local realignments and base-quality recalibrations were conducted using GATK (v.3.2.2)51,52. The averages of depth and the coverages were calculated by the DepthOfCoverage and CallableLoci tools in GATK. Somatic single nucleotide variants (SNVs) and short insertions and deletions (indels) were called in each pair of tumor and matched normal blood samples using Strelka (v.1.0.14) workflow software53. Sites with a depth greater than or equal to 20 in both tumor and matching normal blood samples, were subjected to somatic variant calling. We set the quality-score threshold to greater than or equal to 30 for SNVs and 50 for indels. Functional annotation of the identified somatic variants was implemented by ANNOVAR54. The prevalence of somatic mutations indicated in previous genome-wide screenings in various cancer types were collected from the COSMIC database (v.79)55. The detected variants in germline BRCA1/2 were interpreted using BRCA Exchange56 and confirmed that there was no pathogenic germline missense BRCA1/2 mutation. In germline mutation analysis, stopgain, frameshift, and splicing mutations were used. In addition to these mutation types, non-frameshift indel and missense mutations were used in somatic mutation analysis.
Statistical analysis
All computations were conducted using R. Standard statistical tests were used as appropriate, including unpaired t-test, Welch’s exact test, and logrank test.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Alexandrov, L. B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421, https://doi.org/10.1038/nature12477 (2013).
Knijnenburg, T. A. et al. Genomic and Molecular Landscape of DNA Damage Repair Deficiency across The Cancer Genome Atlas. Cell Rep 23, 239–254 e236, https://doi.org/10.1016/j.celrep.2018.03.076 (2018).
Konstantinopoulos, P. A., Ceccaldi, R., Shapiro, G. I. & D’Andrea, A. D. Homologous Recombination Deficiency: Exploiting the Fundamental Vulnerability of Ovarian Cancer. Cancer Discov 5, 1137–1154, https://doi.org/10.1158/2159-8290.CD-15-0714 (2015).
Cancer Genome Atlas Research, N. Integrated genomic analyses of ovarian carcinoma. Nature 474, 609–615, https://doi.org/10.1038/nature10166 (2011).
Tumiati, M. et al. A Functional Homologous Recombination Assay Predicts Primary Chemotherapy Response and Long-Term Survival in Ovarian Cancer Patients. Clin. Cancer Res. 24, 4482–4493, https://doi.org/10.1158/1078-0432.CCR-17-3770 (2018).
Bolton, K. L. et al. Association between BRCA1 and BRCA2 mutations and survival in women with invasive epithelial ovarian cancer. JAMA 307, 382–390, https://doi.org/10.1001/jama.2012.20 (2012).
Pennington, K. P. et al. Germline and somatic mutations in homologous recombination genes predict platinum response and survival in ovarian, fallopian tube, and peritoneal carcinomas. Clin Cancer Res 20, 764–775, https://doi.org/10.1158/1078-0432.CCR-13-2287 (2014).
Lord, C. J. & Ashworth, A. PARP inhibitors: Synthetic lethality in the clinic. Science 355, 1152–1158, https://doi.org/10.1126/science.aam7344 (2017).
Mirza, M. R. et al. Niraparib Maintenance Therapy in Platinum-Sensitive, Recurrent Ovarian Cancer. N Engl J Med 375, 2154–2164, https://doi.org/10.1056/NEJMoa1611310 (2016).
Coleman, R. L. et al. Rucaparib maintenance treatment for recurrent ovarian carcinoma after response to platinum therapy (ARIEL3): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 390, 1949–1961, https://doi.org/10.1016/S0140-6736(17)32440-6 (2017).
Kurman, R. J. & Shih, I. M. The origin and pathogenesis of epithelial ovarian cancer: a proposed unifying theory. Am. J. Surg. Pathol. 34, 433–443, https://doi.org/10.1097/PAS.0b013e3181cf3d79 (2010).
Pujade-Lauraine, E. et al. Olaparib tablets as maintenance therapy in patients with platinum-sensitive, relapsed ovarian cancer and a BRCA1/2 mutation (SOLO2/ENGOT-Ov21): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol 18, 1274–1284, https://doi.org/10.1016/S1470-2045(17)30469-2 (2017).
Moore, K. et al. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N Engl J Med 379, 2495–2505, https://doi.org/10.1056/NEJMoa1810858 (2018).
Matz, M. et al. Worldwide comparison of ovarian cancer survival: Histological group and stage at diagnosis (CONCORD-2). Gynecol Oncol 144, 396–404, https://doi.org/10.1016/j.ygyno.2016.11.019 (2017).
Sugiyama, T. et al. Clinical characteristics of clear cell carcinoma of the ovary: a distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer 88, 2584–2589 (2000).
Konstantinopoulos, P. A. et al. Phase II study of single-agent cabozantinib in patients with recurrent clear cell ovarian, primary peritoneal or fallopian tube cancer (NRG-GY001). Gynecol Oncol 150, 9–13, https://doi.org/10.1016/j.ygyno.2018.04.572 (2018).
Torre, L. A. et al. Ovarian cancer statistics, 2018. CA Cancer J Clin 68, 284–296, https://doi.org/10.3322/caac.21456 (2018).
Rojas, V., Hirshfield, K. M., Ganesan, S. & Rodriguez-Rodriguez, L. Molecular Characterization of Epithelial Ovarian Cancer: Implications for Diagnosis and Treatment. Int J Mol Sci 17, https://doi.org/10.3390/ijms17122113 (2016).
Sakamoto, I. et al. BRCA1 and BRCA2 mutations in Japanese patients with ovarian, fallopian tube, and primary peritoneal cancer. Cancer 122, 84–90, https://doi.org/10.1002/cncr.29707 (2016).
Hirasawa, A. et al. Prevalence of pathogenic germline variants detected by multigene sequencing in unselected Japanese patients with ovarian cancer. Oncotarget 8, 112258–112267, https://doi.org/10.18632/oncotarget.22733 (2017).
Enomoto, T. et al. The first Japanese nationwide multicenter study of BRCA mutation testing in ovarian cancer: CHARacterizing the cross-sectionaL approach to Ovarian cancer geneTic TEsting of BRCA (CHARLOTTE). Int J Gynecol Cancer 29, 1043–1049, https://doi.org/10.1136/ijgc-2019-000384 (2019).
Ramirez, O., Vaughan, C., Herrera, G. & Guries, R. Temporal and spatial resource use by female three-toed sloths and their young in an agricultural landscape in Costa Rica. Rev Biol Trop 59, 1743–1755 (2011).
Ryland, G. L. et al. Mutational landscape of mucinous ovarian carcinoma and its neoplastic precursors. Genome Med 7, 87, https://doi.org/10.1186/s13073-015-0210-y (2015).
Mueller, J. J. et al. Massively parallel sequencing analysis of mucinous ovarian carcinomas: genomic profiling and differential diagnoses. Gynecol Oncol 150, 127–135, https://doi.org/10.1016/j.ygyno.2018.05.008 (2018).
Jones, S. et al. Low-grade serous carcinomas of the ovary contain very few point mutations. J Pathol 226, 413–420, https://doi.org/10.1002/path.3967 (2012).
Itamochi, H. et al. Whole-genome sequencing revealed novel prognostic biomarkers and promising targets for therapy of ovarian clear cell carcinoma. Br. J. Cancer 117, 717–724, https://doi.org/10.1038/bjc.2017.228 (2017).
Shibuya, Y. et al. Identification of somatic genetic alterations in ovarian clear cell carcinoma with next generation sequencing. Genes Chromosomes Cancer 57, 51–60, https://doi.org/10.1002/gcc.22507 (2018).
Wiegand, K. C. et al. ARID1A mutations in endometriosis-associated ovarian carcinomas. N Engl J Med 363, 1532–1543, https://doi.org/10.1056/NEJMoa1008433 (2010).
Hanley, G. E. et al. A population-based analysis of germline BRCA1 and BRCA2 testing among ovarian cancer patients in an era of histotype-specific approaches to ovarian cancer prevention. BMC Cancer 18, 254, https://doi.org/10.1186/s12885-018-4153-8 (2018).
Ricci, F., Affatato, R., Carrassa, L. & Damia, G. Recent Insights into Mucinous Ovarian Carcinoma. Int J Mol Sci 19, https://doi.org/10.3390/ijms19061569 (2018).
Lieber, M. R. The mechanism of double-strand DNA break repair by the nonhomologous DNA end-joining pathway. Annu Rev Biochem 79, 181–211, https://doi.org/10.1146/annurev.biochem.052308.093131 (2010).
Wang, C., Jette, N., Moussienko, D., Bebb, D. G. & Lees-Miller, S. P. ATM-Deficient Colorectal Cancer Cells Are Sensitive to the PARP Inhibitor Olaparib. Transl Oncol 10, 190–196, https://doi.org/10.1016/j.tranon.2017.01.007 (2017).
Weston, V. J. et al. The PARP inhibitor olaparib induces significant killing of ATM-deficient lymphoid tumor cells in vitro and in vivo. Blood 116, 4578–4587, https://doi.org/10.1182/blood-2010-01-265769 (2010).
Murai, J. et al. Trapping of PARP1 and PARP2 by Clinical PARP Inhibitors. Cancer Res 72, 5588–5599, https://doi.org/10.1158/0008-5472.CAN-12-2753 (2012).
Mateo, J. et al. DNA-Repair Defects and Olaparib in Metastatic Prostate Cancer. N Engl J Med 373, 1697–1708, https://doi.org/10.1056/NEJMoa1506859 (2015).
Bang, Y. J. et al. Randomized, Double-Blind Phase II Trial With Prospective Classification by ATM Protein Level to Evaluate the Efficacy and Tolerability of Olaparib Plus Paclitaxel in Patients With Recurrent or Metastatic Gastric Cancer. J Clin Oncol 33, 3858–3865, https://doi.org/10.1200/JCO.2014.60.0320 (2015).
Zhao, H. et al. Mismatch repair deficiency endows tumors with a unique mutation signature and sensitivity to DNA double-strand breaks. Elife 3, e02725, https://doi.org/10.7554/eLife.02725 (2014).
Yi, M. et al. Biomarkers for predicting efficacy of PD-1/PD-L1 inhibitors. Mol Cancer 17, 129, https://doi.org/10.1186/s12943-018-0864-3 (2018).
Helleman, J. et al. Mismatch repair and treatment resistance in ovarian cancer. BMC Cancer 6, 201, https://doi.org/10.1186/1471-2407-6-201 (2006).
Fleury, H. et al. Cumulative defects in DNA repair pathways drive the PARP inhibitor response in high-grade serous epithelial ovarian cancer cell lines. Oncotarget 8, 40152–40168, https://doi.org/10.18632/oncotarget.10308 (2017).
Vinayak, S. et al. Open-Label Clinical Trial of Niraparib Combined With Pembrolizumab for Treatment of Advanced or Metastatic Triple-Negative Breast Cancer. JAMA Oncol, https://doi.org/10.1001/jamaoncol.2019.1029 (2019).
Hodgson, D. R. et al. Candidate biomarkers of PARP inhibitor sensitivity in ovarian cancer beyond the BRCA genes. Br J Cancer 119, 1401–1409, https://doi.org/10.1038/s41416-018-0274-8 (2018).
Davies, H. et al. HRDetect is a predictor of BRCA1 and BRCA2 deficiency based on mutational signatures. Nat Med 23, 517–525, https://doi.org/10.1038/nm.4292 (2017).
Yoshihara, K. et al. High-risk ovarian cancer based on 126-gene expression signature is uniquely characterized by downregulation of antigen presentation pathway. Clin Cancer Res 18, 1374–1385, https://doi.org/10.1158/1078-0432.CCR-11-2725 (2012).
Mutch, D. G. & Prat, J. 2014 FIGO staging for ovarian, fallopian tube and peritoneal cancer. Gynecol Oncol 133, 401–404, https://doi.org/10.1016/j.ygyno.2014.04.013 (2014).
Meinhold-Heerlein, I. et al. Erratum to: The new WHO classification of ovarian, fallopian tube, and primary peritoneal cancer and its clinical implications. Arch Gynecol Obstet 293, 1367, https://doi.org/10.1007/s00404-016-4073-2 (2016).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45, 228–247, https://doi.org/10.1016/j.ejca.2008.10.026 (2009).
Ahmadloo, S. et al. Rapid and cost-effective high-throughput sequencing for identification of germline mutations of BRCA1 and BRCA2. J Hum Genet 62, 561–567, https://doi.org/10.1038/jhg.2017.5 (2017).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25, 1754–1760, https://doi.org/10.1093/bioinformatics/btp324 (2009).
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079, https://doi.org/10.1093/bioinformatics/btp352 (2009).
DePristo, M. A. et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet 43, 491–498, https://doi.org/10.1038/ng.806 (2011).
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20, 1297–1303, https://doi.org/10.1101/gr.107524.110 (2010).
Saunders, C. T. et al. Strelka: accurate somatic small-variant calling from sequenced tumor-normal sample pairs. Bioinformatics 28, 1811–1817, https://doi.org/10.1093/bioinformatics/bts271 (2012).
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38, e164, https://doi.org/10.1093/nar/gkq603 (2010).
Forbes, S. A. et al. COSMIC: exploring the world’s knowledge of somatic mutations in human cancer. Nucleic Acids Res 43, D805–811, https://doi.org/10.1093/nar/gku1075 (2015).
Cline, M. S. et al. BRCA Challenge: BRCA Exchange as a global resource for variants in BRCA1 and BRCA2. PLoS Genet 14, e1007752, doi:10.1371/journal.pgen.1007752 (2018).
Acknowledgements
We thank the tissue donors and our supporting medical staff for making this study possible. We are grateful to Anna Ishida, Junko Kajiwara, Junko Kitayama and Yumiko Sato for technical assistance. This work was supported in part by Japan Society for the Promotion of Science (JSPS) KAKENHI grant number JP16H06267 (Grant-in-Aid for Young Scientists A for K. Yoshihara), JP15K10707 (Grant-in-Aid for Scientific Research C for M.Yamaguchi.), and JP16K11132 (Grant-in-Aid for Scientific Research C for K. Kashima.).
Author information
Authors and Affiliations
Contributions
K.Y. and T.E. designed the research plan. K.S., R.T., N.Y., M.Y., Y.M., K.Y., K.S., T.I., S.A. and K.Y. collected tissue samples. H.N. and I.I. performed sequencing experiments. K.S. and R.T. conducted all analyses. K.S. and R.T. wrote the main manuscript and prepared all figures and tables. K.S., R.T., H.N., I.I., K.Y. and T.E. discussed the results. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sugino, K., Tamura, R., Nakaoka, H. et al. Germline and somatic mutations of homologous recombination-associated genes in Japanese ovarian cancer patients. Sci Rep 9, 17808 (2019). https://doi.org/10.1038/s41598-019-54116-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-54116-y
This article is cited by
-
Proteomic landscape of epithelial ovarian cancer
Nature Communications (2024)
-
Integrated genomic/epigenomic analysis stratifies subtypes of clear cell ovarian carcinoma, highlighting their cellular origin
Scientific Reports (2024)
-
A comprehensive molecular analysis of 113 primary ovarian clear cell carcinomas reveals common therapeutically significant aberrations
Diagnostic Pathology (2023)
-
Genomic landscapes of ovarian clear cell carcinoma from latin countries reveal aberrations linked to survival and progression
BMC Cancer (2023)
-
Maintenance therapy for newly diagnosed epithelial ovarian cancer– a review
Journal of Ovarian Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.