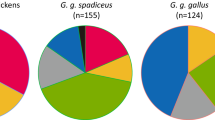Abstract
Trichogramma dendrolimi is one of the most successful biocontrol agents in China. However, an inundative condition is necessary to obtain acceptable parasitism effect. A good solution to this is the application of its thelytokous counterparts which unfortunately are scarce in field. We here report the first case of a natural T. dendrolimi population in China comprising both bisexual wasps and an extremely low proportion of thelytokous wasps. These two forms of T. dendrolimi are phylogenetically related based on the reconstructions of ITS-2 and COI genes. Also, the phylogenetic results suggested a potentially Wolbachia-drived ITS-2 variation. The expression of thelytoky was hardly affected by temperature, which might help control Asian corn borer and Dendrolimus punctatus. Wolbachia are responsible for current thelytoky according to phylogenetic analyses, antibiotic treatment and introgression experiment. We also present the third case of paternal sex ratio chromosome that restrains the expansion of Wolbachia. Moreover, the low frequency of thelytoky may be common in natural populations. Consequently if for biological control it is determined that a thelytokous strain is to be preferred, then large number of field collected females should be set up as isofemale lines, to detect the rare thelytoky.
Similar content being viewed by others
Introduction
Augmentative biological control (ABC) concerns periodical inundative release of natural enemies mass-reared in biofactories to promptly control pests1. For many years, ABC has been an environmentally and economically successful alternative to chemical pest control1,2,3. It has been estimated that in ABC more than 170 natural enemy species are produced and sold globally for controlling more than 100 pest species on 0.16 million km2 (i.e., 0.4% of land under cultivation)4. Among these natural enemies, parasitic wasps Trichogramma are the most commonly used biocontrol agents against lepidopteran pests throughout the world5. Their mass production and inundative release are economically feasible to take a number of most devastating pests under control, such as the Asian corn borer, Ostrinia furnacalis Guenée (Lepidoptera: Crambidae)5, Plutella xylostella (L.) (Lepidoptera: Plutellidae)6, and Tuta absoluta (Lepidoptera: Gelechiidae)7,8.
Different from most other biocontrol agents, these minute egg parasitoids exhibit two modes of reproduction. Typically, they display bisexual reproduction (also referred as arrhenotokous), i.e., the males arise from unfertilized haploid eggs, whereas the females arise from fertilized diploid eggs. But they can also reproduce thelytokously where only diploid females are present. In genus Trichogramma, thelytoky can be under the control of the wasps themselves or their endosymbionts. While genetically determined thelytoky was reported only in Trichogramma cacoeciae9, 15 Trichogramma species have been documented to gain thelytoky after being infected with Wolbachia10,11, the most abundant endosymbionts in arthropods which are famous for their ability to manipulate multiple reproductive phenotypes12,13,14. Though another two microorganisms, Cardinium and Rickettsia, also cause thelytoky of several arthropod species, none of these cases are within Trichogramma11. In general, we can distinguish between genetically determined and endosymbionts-induced thelytoky through revertibility. For the former type, thelytoky should be non-revertible which means the reproductive mode is stable and cannot be changed by antibiotic or heat treatment, whereas the latter will produce male or intersex individuals under such conditions15,16. Additionally, if thelytoky is genetically determined, the bisexual strains will gain thelytokous reproduction function when alleles of thelytokous strains are introgressed into their genomes17. Meanwhile, since endosymbionts are cytoplasmically inherited, the reproductive phenotype induced by them would not change under such introgression.
One important reason we study thelytokous Trichogramma, either genetically determined or induced by Wolbachia, is to utilize them as potent biocontrol agents. Trichogramma wasps are surely preponderant parasitoids of lepidopterans, but an inundative condition is needed to obtain acceptable parasitism effect which makes a high cost18,19,20. Therefore, researchers have been trying to find the best form of Trichogramma that could control the pests in lower amount with higher efficiency21. And the thelytokous Trichogramma may provide us a brilliant future as they offer several advantages: (1) higher population growth rate; (2) less production cost; (3) easier establishment of population; (4) ability to depress host populations to a lower level22,23,24,25,26. Unfortunately, this form is only reported in a few Trichogramma species in field, and is still lacking in species, like T. dendrolimi and T. ostriniae (especially in China), which are predominantly used in biocontrol18. Thelytokous Trichogramma strains or lines can be artificially obtained by transferring Wolbachia inter- or intraspecifically, but in newly infected hosts the expression of induced thelytokous phenotype is generally weak or undetectable, and the infection tends to be lost in most scenarios27,28. On the contrary, Wolbachia infection and the induced phenotype are stably maintained in natural populations29,30. Native strains are also preferred for their better characteristics such as higher tolerance to local environment and higher searching efficiency31. Therefore, native thelytokous Trichogramma may hold greater biocontrol potential.
In China, T. dendrolimi is primarily applied to control Asian corn borer (ACB) which is the most destructive pest of corn occurring in vast majority of Chinese corn-growing areas and cause yield loss ranging from 6 to 9 million tons annually18. During 1980s and 1990s, these parasitoid wasps were released annually across 200,000 to 350,000 ha of corn, which increased to 2.3 million ha annually since 2012 in Jilin province, to keep ACB under control5. T. dendrolimi is also the preponderant parasitoid of Dendrolimus spp. (Lepidoptera: Lasiocampidae), the most important conifer defoliators in China whose outbreak is analogized as the ‘fire without smoke'32. Moreover, in recent studies, T. dendrolimi was identified as the best or promising biological agent of rice stem borer Chilo suppressalis33, oriental fruit moth Grapholita molesta (Lepidoptera: Tortricidae)34, box tree pyralid Cydalima perspectalis (Lepidoptera: Crambidae)35, and oriental armyworm Mythimna separata (Lepidoptera: Noctuidae)36. T. dendrolimi is one of few Trichogramma that could be mass reared on big (e.g., Antheraea pernyi) and artificial eggs18,37. And this wasp species shows a wide distribution in China. It has been found in East (Anhui, Fujian, Jiangsu and Shandong province), South (Guangdong province), North (Beijing municipality and Hebei province), Northeast (Heilongjiang, Jilin and Liaoning province), Northwest (Shaanxi Province) and Southwest (Sichuan province) China (Supplementary Fig. 1). All of these unquestionably make T. dendrolimi one of the most successful biocontrol agents.
Previously Grenier et al.27 reported a successful case of transferring thelytoky-inducing Wolbachia into uninfected T. dendrolimi. But only partial induction of thelytoky was observed in their newly infected wasps. A stable thelytokous T. dendrolimi line was obtained for the first time in our laboratory by artificially transferring Wolbachia from native host T. embryophagum10. However, this line suffers from several drawbacks, such as the longer parasitization cycle, the poor capacity to evaluate the nutritional quality, the higher superparasitism rate and the fewer host eggs oviposited into20. We here report, to our knowledge, the first case in China of a natural T. dendrolimi population with extremely low proportion of thelytokous individuals. In this study, experiments were conducted to focus primarily on the following questions: (1) Are these thelytokous and bisexual wasps phylogenetically related? (2) Is the thelytoky stable and thus could be applied to biocontrol practice? (3) What is responsible for the thelytoky of T. dendrolimi? 4) What restrains the expansion of thelytoky in this population? In addition, suggestions of how to detect rare thelytoky in field collected populations were provided. Our finding may offer a powerful option for biocontrol of lepidopteran pests.
Methods
Insects
The eggs of Dendrolimus punctatus Walker (Lepidoptera: Lasiocampidae) parasitized by T. dendrolimi were collected on April 16, 2017 from the forest of Chinese red pine in Huanren County, Benxi City of Liaoning Province in China (41°10′N, 125°25′E). Hereafter, the wasps were reared at 25 ± 1 °C, 70 ± 5% relative humidity (RH) with a 16: 8 h light: dark photoperiod on eggs of Corcyra cephalonica (Lepidoptera: Pyralidae). And the C. cephalonica were reared at 26 ± 1 °C, 70 ± 5% RH with a 16: 8 h light: dark photoperiod on maize flour and wheat bran38. All the wasps emerged from D. punctatus eggs were stored in 70% ethanol after they finished parasitization on C. cephalonica.
At first glance, the wasps appeared to be bisexual for their seemingly normal sex ratio. Certain females were then found to produce only daughters during isofemale line establishment. These females were separated and kept individually in a glass tube which allowed them to lay unfertilized eggs. More than five generations of continuous reproduction without sex and all female offspring confirmed the existence of thelytoky. In the following contents, the bisexual and thelytokous wasps were designated as Td-HR and TdT-HR strain respectively.
Genetic backgrounds of Td-HR and TdT-HR strains
The DNA (of 5 individuals randomly selected from all cohorts for each strain) was extracted using Chelex method39: briefly, wasps were placed into a 1.5 mL eppendorf tube with 50 μL 5% Chelex-100 and 2.5 μL Proteinase K (20 mg ml−1), then incubated for 1 h at 55 °C, and finally followed by 10 min at 99 °C. Nuclear ribosomal DNA (nrDNA) internal transcribed spacer 2 (ITS-2) and two mitochondrial genes cytochrome oxidase I (COI) and cytochrome b (Cytb) were amplified through PCR. These genes were chosen as they appear to represent different modes of evolution. Mitochondrial DNA (mtDNA) was used due to its maternal inheritance, very high mutation rate and lack of recombination40, while ITS-2 is a noncoding and therefore rapidly evolving region41. Primer pairs used for amplification were: forward 5′-TGTGAACTGCAGGACACATG-3′ and reverse 5′-GTCTTGCCTGCTCTGAG-3′ for ITS-2, forward C1-J-1718 (5′-GGAGGATTTGGAAATTGATTAGTTCC-3′) and reverse C1-N-2191 (5′-CCCGGTAAAATTAAAATATAAACTTC-3′) for COI, and forward CB-J-10933 (5′-TATGTACTACCATGAGGACAAATATC-3′) and reverse CB-N-11367 (5′- ATTACACCTCCTAATTTATTAGGAAT-3′) for Cytb42,43. The amplification was performed in a total volume of 50 µL, containing 25 µL 2 × Es Taq Mastermix (CWBIO, Beijing, China), 1 µL each of forward and reverse primers (10 μM), 2 µL DNA template and 21 µL ddH2O. Amplification condition for ITS-2 was 3 min at 94 °C, 33 cycles of 40 s at 94 °C, 45 s at 53 °C, and 45 s at 72 °C, followed by 10 min at 72 °C44, while for COI and Cytb was 3.5 min at 94 °C, 33 cycles of 35 s at 94 °C, 35 s at 49 °C, and 45 s at 72 °C, followed by 10 min at 72 °C. Resulting PCR products from ITS-2 primers were run on 1% agarose gel and clear target bands were recycled, purified and cloned to pGEM- Teasy vectors (Promega Co., Madison, Wisconsin, USA). Positive ITS-2 clones, COI and Cytb PCR products were sequenced by Invitrogen Trading Co., LTd. (Shanghai, China). The consequent ITS-2 sequences of both Td-HR and TdT-HR strains were directly submitted to the NCBI GenBank database and were assigned accession numbers MG890332 and MG890333 respectively. Only one trimmed COI or Cytb sequence was deposited in the database (MK213321 or MK552378), as the sequencing results from Td-HR and TdT-HR strains were exactly the same. Phylogenetic analyses were conducted based on ITS-2 or COI gene to evaluate the backgrounds of Td-HR and TdT-HR strains. Cytb gene was not used because of lacking enough data in GenBank. For either of the two genes, a data matrix was constructed, including all T. dendrolimi reference sequences deposited in GenBank to represent the diversity, 17 other Trichogramma species (COI and ITS-2 sequences from the same individuals)45 serving as outgroups to evaluate the interspecific distance, and our data. These sequences were aligned by Clustal X version 2.0 (www.clustal.org)46 and analyzed using both maximum parsimony (MP) and neighbor joining (NJ) methods with MEGA version 5.047,48, to verify the consistency of the phylogenetic results. Bootstrap analysis was done with 1000 replications to estimate the robustness of the nodes. Evolutionary distances were calculated based on a maximum composite likelihood model. The reference sequences were listed in Supplementary Tables 1 and 2.
Endosymbionts detection, infection frequency and phylogeny
The Cardinium, Rickettsia and Wolbachia infection were investigated in both Td-HR and TdT-HR strains. Briefly, the DNA of either Td-HR or TdT-HR strain were extracted and then checked for the presence of endosymbionts through PCR with three primer pairs respectively for Cardinium (CLO-f1: 5′-GGAACCTTACCTGGGCTAGAATGTATT-3′, CLO-r1: 5′-GCCACTGTCTTCAAGCTCTACCAAC-3′), Rickettsia (R1: 5′-GCTCTTGCAACTTCTATGTT-3′, R2: 5′-CATTGTTCGTCAGGTTGGCG-3′) and Wolbachia (81F: 5′-TGGTCCAATAAGTGATGAAGAAAC-3′, 691R: 5′-AAAAATTAAACGCTACTCCA-3′)49. PCR amplifications were performed under the conditions described previously and the products were purified, cloned and sequenced as mentioned earlier49,50. Fifty individuals were randomly checked each strain. However, only Wolbachia were found and only thelytokous individuals were infected. The infection frequency of Wolbachia in the original population was then investigated by random detection in 200 individuals stored in the ethanol (see above). Sequence of the Wolbachia surface protein gene wsp was submitted to NCBI GenBank database and was assigned accession number MG914000. The phylogenetic analysis of wsp was carried out as described above. In this analysis, 27 wsp gene sequences were used, including our data and 26 reference sequences from NCBI (Supplementary Table 3). To fortify the phylogenetic reconstruction reliability, a MLST typing system was also adopted. PCR amplification was performed for 5 MLST alleles (gatB, coxA, hcpA, ftsZ, fbpA) following standard protocols and universal primers (https://pubmlst.org/wolbachia/info/protocols.shtml). Nucleotide sequencing data were subsequently deposited in PubMLST database (https://pubmlst.org/wolbachia/). The ST number 486 and id number 1851 were assigned. A matrix comprising our and other 38 unique concatenated MLST sequences retrieved from PubMLST database was used in Maximum Likelihood (ML) analysis to develop the phylogenetic relationship. GTR + G + I nucleotide substitution model was determined as the best fitting choice. ML tree was reconstructed with Mega 5.0 using the following parameters: “No. of Bootstrap Replication” - 1000, “Gaps/Missing Data Treatment” - Use all sites, “No. of Discrete Gamma Categories” - 5. The reference MLST profiles were listed in Supplementary Table 4.
Antibiotic treatment
To decide whether thelytoky was non-revertible, the females of TdT-HR strain were treated with tetracycline (mixed with 10% honey-water). Five concentrations were used: 0 (control), 0.0001, 0.001, 0.01 and 0.1 mg/ml (C0-C4). Newly emerged females were allowed to feed on tetracycline for 36 h. Then 15 of them were randomly selected from each concentration and supplied individually with approximate 150 (surplus) C. cephalonica eggs to parasite for 36 h. These females were gently removed after that and the eggs were maintained under 25 ± 1 °C, 70 ± 5% RH condition with a 16: 8 h light: dark photoperiod. The brood size, number of female, male or intersex individulas were counted afterwards. Identification of intersex was attributed to Tulgetske and Stouthamer16. Same tetracyline treatments were administrated to mated Td-HR females to serve as a control.
Introgression experiment
To test if individuals with a thelytokous genome are able to reproduce thelytokously, alleles of the TdT-HR strain were introgressed into the genomes of Td-HR strain (Fig. 1A). Firstly, 90 females from Td-HR strain were crossed with 90 males from TdT-HR strain (produced by antibiotic treatment) respectively. Then each of these females (F0) was placed in a 4 ml plastic tube with 10% honey-water and supplied with a fresh egg card containing approximately 150 (surplus) C. cephalonica eggs to oviposit for 24 h. Parasitized eggs blackened within 4 ~ 5 days when wasps pupated. These black eggs were isolated to ensure the offspring (F1) being unmated. Hybrid F1 females were mated again with TdT-HR strain males. This backcross was repeated for 5 generations and the resulting F5 individuals should possess 96.9% of the genome stemming from TdT-HR strain according to the relationship 1 – (0.5)n 17. In our study, the proportion of hybrid females that produced at least one daughter decreased gradually for the possible decayed sexual traits in thelytokous strain or the diploid males which are usually stertile or sire inviable or sterile triploid females51,52,53,54. Consequently, the number of hybrid lines used for reproduction mode analysis decreased from 90 to 33 in F1 generation, to 17 in F2, to 8 in F3, to 7 in F4 and 4 in F5. The reproduction mode of hybrid females was tested every generation (averaging 12 virgins selected for each hybrid line) by supplying them with C. cephalonica eggs. All female offspring indicated the mode is thelytoky, whereas all male offspring indicated a arrhenotokyous reproduction mode. The control was conducted by performing a continuous cross between bisexual Td-HR females and males.
Experiment Design. (A) Diagram for Introgression experiment. Black symbols, thelytokous genotypes; Grey symbols, bisexual genotypes. (B) Diagram for the detection of suppressor genes or PSR Chromosome. S, dominant suppressor allele; s, recessive suppressor allele; n, non-suppressor allele; C, cured individual.
Factors restraining expansion of thelytoky
From the results of above experiments, it can be concluded that Wolbachia induce the thelytoky of T. dendrolimi (see Results and Discussion for further details). And also, because the infection only existed in thelytokous wasps (see Endosymbionts detection, infection frequency and phylogeny), our fourth question, i.e., what restrains the expansion of thelytoky, was thereby simplified to what keeps the frequency of Wolbachia low? Stouthamer et al.55 proposed three factors that could serve to the restricted spread of Wolbachia, including: (1) inefficient transmission of Wolbachia which causes some offspring of infected females to lose infection. (2) suppressor genes based on the conflict between nuclear and cytoplasmic genes. Since Wolbachia are cytoplasmic inherited, they cannot benefit from the male fitness and favor a female biased population. In the meantime, males would try to suppress the transmission of Wolbachia. Several instances have been reported where suppressors act in two ways (i.e., by killing the bacteria or by negating the effects from them) against cytoplasmic sex-ratio distorters56,57,58. (3) presence of paternal sex ratio (PSR) chromosome which acts by destroying the paternal set of chromosomes after fertilization, with the exception of itself. Therefore the resulting sons only carry maternal and PSR chromosomes55,59,60. With regard to the first factor, if Wolbachia do transfer inefficiently, the expression level of thelytokous phenotype in hosts will decrease gradually with increasing generations, especially under adverse environments. Apparently, this is exactly what we need to determine in the second question, i.e., is the thelytoky stable? To answer this question, a successive observation on sex ratio was carried out for thelytokous strain at four temperatures: 17, 21, 25 and 29 °C. The 33 °C was not performed because it caused a substantial number of deaths. Thirty five TdT-HR wasps were individually maintained at each rearing temperature in a growth chamber (Panasonic, MLR-352H-PC, Osaka, Japan) for four generations. For each generation, superfluous C. cephalonica eggs were supplied. The number of emerged female, male or intersex wasps was counted, then average and total (i.e., compared to total emerged offspring) rates of male or intersex were calculated. Another experiment for differentiating the latter two factors was designed according to Stouthamer et al.55 (see also Fig. 1B). Sixty cross lines (F0) between bisexual Td-HR males and thelytokous TdT-HR females were established, 15 (if possible) resulting virgin daughters (F1) from each cross line were selected randomly to produce F2 offspring (granddaughters). The virgin production was continued one more generation by selecting 30 granddaughters (2 granddaughters × 15 daughter lines) each cross line. Virgin females were obtained as described in Introgression section. Suppose that suppressor genes do present, a male carrying a suppressor allele will produce cured daughters (dominant allele or both male and female carrying a recessive allele), or cured granddaughters (when male with a recessive allele)55. In light of this, males would occur in F2 or F3 generation. Under PSR chromosome model, however, males would occur in F1 and usually with a high proportion. Additionally, if such high male proportion occurred, the males from high male biased lines would be crossed with thelytokous females three more generations.
Statistical analysis
The possible effects of tetracycline treatment on brood size, sex ratio (males / total emerged wasps) were evaluated by using generalized linear model (GLM), with tetracycline treatment as the explanatory variable, while brood size or sex ratio was modeled as response variable based on a Poisson distribution or a binomial logit distribution. To identify if number of TdT-HR male or intersex offspring changed with different antibiotic concentrations, generalized linear mixed model (GLMM) was perfromed based on Poisson distribution. Tetracycline concentration was used as explanatory variable, male or intersex offspring number as the response variable, and brood size as the random factor.
All statistical analyses were performed in R software (version 3.4.3)61.
Results
Genetic backgrounds of Td-HR and TdT-HR strains
ITS-2 gene showed a relative high variation. The distance based on ITS-2 gene between TdT-HR and Td-HR was 0.015 (1.55% sequence divergence), higher than that between TdT-HR and most other T. dendrolimi strains or isolates (0.003–0.012). Not a single base difference was found in COI or Cytb sequences between Td-HR and TdT-HR strains. Mean intraspecific and interspecific distances were 0.043 and 0.276 for ITS-2, whereas were 0.006 and 0.065 for COI. The reconstructed phylogenetic relationship using ITS-2 or COI sequences is presented as a phylogram in Figs. 2 or 3. As the trees inferred from neighbor joining and maximum parsimony methods were similar, only the former one is displayed (same for Wolbachia phylogeny). ITS-2 tree indicates that both Td-HR and TdT-HR strains belong to a widely unresolved monophyletic polytomy (94% bootstrap support) of highly similar T. dendrolimi isolates and strains from various origins, including those from Germany, Japan and different regions of China (Supplementary Table 1). The clade appearing as highly homogenous T. dendrolimi genetic pool is clearly divergent from all the other Trichogramma species used in this study (Fig. 2). A similar phylogenetic COI-based tree further indicates that our strains and isolate TdJS are sister taxa (95% bootstrap support). Interestingly, the isolate TdJS is from Jilin province, adjacent to our sampling location on the east by city Ji’an and north by city Tonghua, implying that these two populations may have same geographic origin.
Phylogenetic Neighbor Joining tree of Trichogramma species based on ITS-2 sequences. Accession number follows the taxon name. Designations in bold are the sequences of bisexual and thelytokous T. dendrolimi strains in this study. Values above the branches are the percentages of bootstrap support estimated from 1000 replicates.
Phylogenetic Neighbor Joining tree of Trichogramma species based on COI sequences. Accession number follows the taxon name. Designations in bold are the sequences of bisexual and thelytokous T. dendrolimi strains in this study. Values above the branches are the percentages of bootstrap support estimated from 1000 replicates.
Wolbachia infection frequency and phylogeny
The detection of endosymbionts was performed for both Td-HR and TdT-HR strains, but only Wolbachia was found and only thelytokous wasps were infected. Among 200 examined wasps, two females (1%) were infected by Wolbachia. The phylogenetic wsp-based tree (Fig. 4) is obviously divided into two supergroups (A and B) which is consistent with previous studies50,62. According to the grouping criterion (2.5% divergence) by Zhou et al.50, wsp sequences of supergroup B used in this study were subdivided into eight groups. Our Wolbachia strain showed 0.6 to 2.1% wsp sequence differences from other Wolbachia strains in Sib group and thus fell within this group. Similarly, ML phylogeny of Wolbachia based on 5 concatenated MLST alleles (2,079 bp after aligned and trimmed) from our data and different supergroups (A, B, D, F and H) from PubMLST reveals an incidence of Wolbachia in TdT-HR wasps belonging to supergroup B and a monophyletic higher relatedness to T. deion (Fig. 5).
Phylogenetic Maximum Likelihood tree of Wolbachia based on 5 MLST alleles (gatB, coxA, hcpA, ftsZ, fbpA). ST number and supergroup follow the taxon name. Designation in bold is the Wolbachia strain from our thelytokous T. dendrolimi. Values above the branches are the percentages of bootstrap support estimated from 1000 replicates.
Antibiotic treatment
To investigate whether thelytoky is unrevertible, five concentrations of tetracycline (C0-C4) were administrated to the females of TdT-HR strain. Statistical analyses showed that both male and intersex offspring gradually increased with higher tetracycline concentrations, ranging from 0 to 91 (11.73%) for male (GLMM, χ2 = 76.09, d.f. = 1, P < 0.001) and to 35 (4.51%) for intersex (GLMM, χ2 = 46.904, d.f. = 1, P < 0.001) (Fig. 6 and Supplementary Fig. 2). Same tetracycline treatment was performed on Td-HR strain to exclude the possible effects of tetracycline. But no intersex individuals were found and no significant differences were observed in sex ratio (GLM, χ2 = 1.8361, d.f. = 4, P = 0.7659) or brood size (GLM, χ2 = 1.4848, d.f. = 4, P = 0.8293) among tetracycline-treated and untreated groups.
Introgression
If the thelytoky is genetically determined, the bisexual Td-HR wasps should gain thelytokous reproduction function after alleles of thelytokous TdT-HR wasps are introgressed into their genomes. A continuous introgression carried out between TdT-HR males and Td-HR females showed that the hybrid virgin females in every generation remained arrhenotokous (Table 1), proving that the genome is not responsible for the thelytoky of T. dendrolimi.
Factors restraining expansion of thelytoky
Of three possible factors that may restrict the spread of Wolbachia, inefficient transmission results in a high proportion of abnormal (male or intersex) offspring. But this was not found in TdT-HR strain. As shown in Table 2, males and intersex individuals were rare or absent in each generation under low (17 and 21 °C), moderate (25 °C) and high (29 °C) temperatures, demonstrating the stable transmission of Wolbachia. Assuming that suppressor genes are present, the offspring of cured virgin daughters (F1) or granddaughters (F2) should be all male (See Fig. 1B). However, no such daughters or granddaughters were found (Supplementary Table 5). Instead, our results accorded well with the model of PSR chromosome. Among 54 hybrid lines established successfully (i.e., the F0 female produced at least one offspring), nine (16.67%) showed high male proportion in F1 generation (Supplementary Table 5). Males from these male biased lines were crossed with TdT-HR females for three consecutive generations. All resulting hybrid lines produced mostly male offspring (Supplementary Table 6).
Discussion
Trichogramma dendrolimi is one of the most important biocontrol agents widely used in agriculture and forestry production for its high parasitism rate, adaption to multiple lepidopteran pests, wide distribution, and ability to be mass reared on big or artificial eggs18,32,33,34,35,36,37. But as mentioned earlier, an acceptable parasitism effect requires an inundative condition which makes a high cost. A good solution to this is the practice of releasing its thelytokous counterparts. We here report the first thelytokous T. dendrolimi strain in China which co-occurs with its bisexual form in a natural population.
We first analyzed the phylogenetic backgrounds of the two strains. ITS-2 gene has widely proven to be a powerful tool in distinguishing among closely related Trichogramma species42,63,64,65,66. Its usefulness for species identification in Trichogramma is because: (1) smaller divergence within species than between species; (2) morphologically distinct cryptic species can be discriminated by sequence differences42. The conservative sequence variation is related to concerted evolution which results in homogenization of variable repeats and produces a mostly uniform sequence within individuals of a population and hence among panmictic populations within species67,68,69. Phylogenetic analysis based on ITS-2 gene apparently supported that our two strains are part of the highly homogeneous T. dendrolimi genetic pool and can be assumed to have similar genetic backgrounds, as suggested by Kishani et al.44,70. Such relationship was further substantiated by similar COI-based reconstruction and by undifferentiated COI and Cytb sequences between two strains. These two mitochondrial genes have also been proved to provide abundant phylogenetic information in Trichogramma45,71,72. Worthy of note is that: 1) ITS-2 gene in T. dendrolimi appears to represent a higher intraspecific variation and a faster evolution (i.e., longer interspecific distances) than COI; 2) Td-HR is not phylogenetically closest to TdT-HR based on ITS-2 but COI. The higher intraspecific variation of ITS-2 seems unusual since COI generally has a greater nucleotide diversity within species41,73,74,75. Several alternative hypotheses could be applied to the polymorphous ITS-2. Firstly, it could be simply interpreted as the result of mutation accumulation (e.g., Taraxacum offcinale)76. Secondly, the gene flow between races, ecotypes or subspecies of a species that have distinct histories could serve to the mixing of differentiated ITS-2 sequences, postponing the homogenization73. The final hypothesis is the lack of gene flow causing retention of different ITS-2 morphisms77.
The mutation accumulation, if does exist, one would then expect random mutations of ITS-2 gene. However, no differences were found in sequences of cloned ITS-2 gene from either bisexual or thelytokous wasps. Gene flow requires different clusters with distinct historic backgrounds. This overtly contradicts the undifferentiated COI and Cytb sequences between Td-HR and TdT-HR strains. The lack of gene flow between thelytokous and bisexual wasps is more plausible. If the polymorphism in ITS-2 sequences is present in ancestral population, the subsequent Wolbachia infection causing thelytoky (see below), male mating preference for bisexual females78,79, and PSR chromosome eliminating paternal genome after inseminating thelytokous wasps (see below) would reduce or prevent the gene flow, and hence sharpen the different patterns of ITS-2 between bisexual and thelytokous wasps. It is also possible that the polymorphism happens after Wolbachia infection. In this case, Wolbachia is responsible for nonrandom mutation of ITS-2 in thelytokous strain. Either way, Wolbachia seem to play a role in driving the variation of ITS-2. In addition, the phylogenetic COI-based reconstruction revealed that our population represents a sister taxon to the one from Jilin province that is adjacent to our sampling location. This may imply a wider distribution of Wolbachia-infected thelytokous T. dendrolimi.
Of three endosymbionts causing thelytoky of arthropods, the wasps of TdT-HR strain were only infected by Wolbachia. Phylogenetic analyses indicated that this Wolbachia strain belongs to Sib group (in supergroup B) which has proven to be able to induce thelytoky in Trichogramma. To date, 16 Wolbachia supergroups (A–F and H–Q) have been established80. Nevertheless, the thelytoky-inducing strains (with firm demonstrations) are reported only in two supergroups: A and B11,81. In supergroup A, two groups (i.e., Dro and Uni) cause thelytoky of four species, including Telenomus nawai, Muscidifurax uniraptor, and two Aphytis species62,82,83,84. Meanwhile, three such groups (i.e., Dei, Div and Sib) are found in supergroup B: Dei and Sib are responsible for the thelytoky of 9 Trichogramma species, including T. brassicae, T. chilonis, T. cordubensis, T. deion, T. dendrolimi, T. embryophagum, T. kaykai, T. oleae and T. pretiosum, while Div only induces thelytoky in Apoanagyrus diversicornis10,28,62,85,86,87,88,89,90. Since for many host species the Wolbachia group information was not supplied (e.g., Bryobia praetiosa)91, the thelytokous groups listed here could be incomplete. It is interesting to note that the group Dei-induced thelytokous T. dendrolimi was demonstrated in our previous study by transferring Wolbachia from T. embryophagum10. This suggests that the reproduction of a host can be manipulated by different Wolbachia.
Both antibiotic treatment and introgression experiment proved that current thelytoky is a result of microorganism infection instead of a genetically determined mechanism. Furthermore, the expression of thelytokous phenotype significantly weakened when tetracycline concentration was increased. It may suggest that the microorganism(s) inside the wasps was responding to the change of antibiotic when the effects of tetracycline on T. dendrolimi were already ruled out. Previous studies have reported a positive correlation between thelytokous phenotype and Wolbachia titer: Wang et al.92 showed a decreased female offspring proportion of thelytokous Encarsia Formosa with lower Wolbachia titer; Ma et al.93 pointed out that the males of thelytokous Asobara japonica increased with the decrease of Wolbachia titer caused by Rifampicin. Indeed, through quantitative PCR, we were able to prove that Wolbachia titer significantly decreased with higher tetracycline concentrations (or with weakened thelytokous phenotype) (see Supplementary Fig. 3).
The similar backgrounds, endosymbiont-induced thelytoky, Wolbachia infection, thelytoky-inducing group Sib, and positive correlation between Wolbachia titer and thelytokous phenotype allowed us to reasonably reach a conclusion that Wolbachia do induce the thelytoky of TdT-HR wasps. And we could thereby proceed to the next question: why the Wolbachia infection rate was so low in this population? Generally, a fixed Wolbachia infection is expected, i.e., all individuals (or nearly 100%) are female94,95. But the low infection level appears to be not rare: the rate was 0.9%, 4.5% and 4–26% for T. evanescens, T. turkestanica and T. kaykai Pinto & Stouthamer, respectively31,55. Among the factors that serve to the restricted spread of Wolbachia, the inefficient transmission was firstly examined. We failed to find a high, but an extremely low rate of abnormal individuals in TdT-HR strain at low, moderate or high temperature. This suggested a stable transmission of Wolbachia, which is the prerequisite for us to employ this strain in biocontrol practice. In Northeast China, ACB generally has two generations per year corresponding to two different environmental temperatures18. In first generation when it is around 25 °C20, ABC larvae emerge from eggs on whorl-stage corn and cause seriously direct damages. But a much greater indirect damage occurs in the second generation when corn reaches silking or pollen-shedding stage at much higher temperatures96, because the feeding activity from larvae can cause ear and kernel rot which substantially raise the risk of grains contamination by mycotoxins19. Using TdT-HR strain to control ABC might be practicable on account of its stable thelytokous phenotype under different temperatures. It also seems likely that this strain displays a good control of Dendrolimus punctatus for being a native parasitoid of this pest species.
Based on models attributed to Stouthamer et al.55, we were able to detect and distinguish between suppressor genes and PSR chromosome. Our results evidently supported the latter. Thus far, the selfish chromosome PSR has only been described in two wasps, Nasonia vitripennis and Trichograma kaykai Pinto & Stouthamer97. We here provide T. dendrolimi as the third case. Additionally, it is important to note the extremely low Wolbachia infection caused by PSR chromosome. As in such a case the corresponding thelytokous wasps would be too rare to be discriminated from normal bisexual wasps. Consequently, the frequency of Wolbachia-induced thelytoky in nature would be misestimated, or worse, we cannot give an objective assessment of the interaction between Wolbachia and their hosts. In current study, a description of how we “accidentally” found the thelytokous strain was provided, that is, by setting up isofemale lines. We herein also recommend that researchers firstly establish the isofemale lines of parasitoid wasps (or other haplo-diploid species) from fields before detecting Wolbachia infection for the fact of rare thelytoky.
For a long time, thelytokous Trichogramma have been considered to enhance the efficacy of biocontrol. But their application is prevented by the fact of being scarce in nature. Efforts have been made to artificially create thelytokous Trichogramma wasps27, and the common way is to transfer Wolbachia from native into foreign hosts. So far, two methods have now been mainly adopted: microinjection and host sharing. The former is more general, whereas the latter has only been used in parasitoid wasps10,27,28,98,99,100. Successful Wolbachia transinfection are reported notwithstanding, compared to native hosts, Wolbachia in the foreign ones universally induce a weaker or undetectable phenotype and usually cannot be maintained stably, even if the transinfection is between conspecifics. The transmission rate of thelytoky-inducing Wolbachia decreased to 0% at seventh generation in the new host Drosophila simulans (Muscidifurax uniraptor as the donor)98, and similar results were found in the recipient hosts T. kaykai, T. deion and T. atopovirilia (Intra- or interspecific Trichogramma as the donors)28; a low vertical transmission rate of Wolbachia causing cytoplasmic incompatibility was shown in the recipient host Ostrinia scapulalis (Ephestia kuehniella as the donor)100. In contrast, the Wolbachia infection and induced phenotype in natural strains are maintained stably in general29,30, possibly for the long-term co-adaptation between Wolbachia and their native hosts.
Finally, we are able to answer the questions addressed in Introduction: yes, thelytokous and bisexual wasps are phylogenetically related; and yes, the thelytoky is stable; Wolbachia is responsible for current thelytoky and PSR chromosome restrains their expansion. However, we still lack the comprehensive studies and integrate evaluation to determine the biocontrol potential of our thelytokous strain before it can be used in practice. And our further studies will focus on this aspect by evaluating indicators like body size, vagility101, decision-making20, and adaptive capacity to the long-term storage90.
Data availability
All data included in this study are available upon reasonable request by contact with the corresponding author.
References
van Lenteren, J. C. The state of commercial augmentative biological control: plenty of natural enemies, but a frustrating lack of uptake. BioControl 57, 1–20 (2012).
van Lenteren, J. C. & Bueno, V. H. P. Augmentative biological control of arthropods in Latin America. BioControl 48, 123–139 (2003).
Bale, J., van Lenteren, J. C. & Bigler, F. Biological control and sustainable food production. Philos. T. R. Soc. B. 363, 761–776 (2008).
Cock, M. J. et al. Do new access and benefit sharing procedures under the convention on biological diversity threaten the future of biological control? BioControl 55, 199–218 (2010).
Zhang, J. J. et al. Advantages of diapause in Trichogramma dendrolimi mass production on eggs of the Chinese silkworm, Antheraea pernyi. Pest Manag. Sci. 74(4), 959–965 (2018).
Tabone, T., Bardon, C., Desneux, N. & Wajnberg, E. Comparative assessment of parasitism of different Trichogramma spp. on Plutella xylostella L. on greenhouse cauliflower. J. Pest Sci. 83, 251–256 (2010).
Desneux, N. et al. Biological invasion of European tomato crops by Tuta absoluta: ecology, history of invasion and prospects for biological control. J. Pest Sci. 83, 197–215 (2010).
Cascone, P. et al. Improving the efficiency of Trichogramma achaeae to control Tuta absoluta. BioControl 60, 761–771 (2015).
Vavre, F., de Jong, J. H. & Stouthamer, R. Cytogenetic mechanism and genetic consequences of thelytoky in the wasp Trichogramma cacoeciae. Heredity 93, 592–596 (2004).
Zhang, H. Y. Wolbachia transfection and Biological Control Capability of Trichogramma spp. PhD Dissertation. Shenyang Agricultural University (2009).
Ma, W. J. & Schwander, T. Patterns and mechanisms in instances of endosymbionts-induced parthenogenesis. J. Evolution. Biol. 30, 868 (2017).
Hilgenboecker, K., Hammerstein, P., Schlattmann, P., Telschow, A. & Werren, J. H. How many species are infected with Wolbachia?-a statistical analysis of current data. FEMS Microbiol. Lett. 281, 215–220 (2008).
Werren, J. H., Baldo, L. & Clark, M. E. Wolbachia: master manipulators of invertebrate biology. Nat. Rev. Microbiol. 6, 741–751 (2008).
Zug, R. & Hammerstein, P. Still a host of hosts for Wolbachia: analysis of recent data suggests that 40% of terrestrial arthropod species are infected. PLoS One 7, e38544 (2012).
Stouthamer, R., Pinto, J. D., Platner, G. R. & Luck, R. F. Taxonomic status of thelytokous forms of Trichogramma (Hymenoptera: Trichogrammatidae). Ann. Entomol. Soc. Am. 83, 475–481 (1990).
Tulgetske, G. M. & Stouthamer, R. Characterization of intersex production in Trichogramma kaykai infected with parthenogenesis-inducing Wolbachia. Naturwissenschaften 99, 143–152 (2012).
Stouthamer, R., Luck, R. F. & Hamilton, W. D. Antibiotics cause parthenogenetic Trichogramma (Hymenoptera: Trichogrammatidae) to revert to sex. P. Natl. Acad. Sci. USA 87, 2424–2427 (1990).
Wang, Z. Y., He, K. L., Zhang, F., Lu., X. & Babendreier, D. Mass rearing and release of Trichogramma for biological control of insect pests of corn in China. BioControl 68, 136–144 (2014).
Wang, Z. Y., He, K. L. & Yan, S. Large-scale Augmentative Biological Control of Asian Corn Borer Using Trichogramma in China: A Success Story in Proceedings of the Second International Symposium on Biological Control of Arthropods (ed. Mark, S. H.) 487–494 (Davos, Switzerland, 2005).
Liu, Q. Q. et al. Decision-making in a bisexual line and a thelytokous Wolbachia-infected line of Trichogramma dendrolimi Matsumura (Hymenoptera: Trichogrammatidae) regarding behavior toward their hosts. Pest Manag. Sci. 74, 1720–1727, https://doi.org/10.1002/ps.4867 (2018).
Hassan, S. A. A simple method to select effective Trichogramma strains for use in biological control in Trichogramma and other egg parasitoids (eds. Wajnberg, E. & Vinson, S. B.) 201–205 (Les Colloques de l’INRA, 1990).
Stouthamer, R. The use of sexual versus asexual wasps in biological control. BioControl 38, 3–6 (1993).
Neto, L. Interactions génétiques entre les trichogrammes (Hym. Trichogrammatidae) et leurs hôtes: Rôle d’un symbiote. PhD Dissertation. INSA (1996).
Silva, I. M. M. S. et al. Biological control potential of Wolbachia-infected versus uninfected wasps: laboratory and greenhouse evaluation of Trichogramma cordubensis and T. deion strain. Biocontrol Sci. Techn. 10, 223–228 (2000).
Ramirez-Romero, R., Sivinski, J., Copeland, C. S. & Aluja, M. Are individuals from thelytokous and arrhenotokous populations equally adept as biocontrol agents? Orientation and host searching behavior of a fruit fly parasitoid. BioControl 57, 427–440 (2012).
Prabhulinga, T., Jalali, S. K., Kumar, K. P. & Doddabasappa, B. Biological characteristics of arrhenotokous and thelytokous Trichogramma pretiosum Riley in Arthropod Diversity and Conservation in the Tropics and Sub-tropics (eds. Chakravarthy, A. & Sridhara, S.) 327–333 (Springer, Singapore, 2016).
Grenier, S. et al. Successful horizontal transfer of Wolbachia symbionts between Trichogramma wasps. Philos. T. R. Soc. B. 265, 1441–1445 (1998).
Huigens, M. E., De Almeida, R. P., Boons, P. A. H., Luck, R. F. & Stouthamer, R. Natural interspecific and intraspecific horizontal transfer of parthenogenesis-inducing Wolbachia in Trichogramma wasps. Philos. T. R. Soc. B. 271, 509–515 (2004).
Dedeine, F. et al. Removing symbiotic Wolbachia bacteria specifically inhibits oogenesis in a parasitic wasp. P. Natl. Acad. Sci. USA 98, 6247–6252 (2001).
Gottlieb, Y. & Zchori-Fein, E. Irreversible thelytokous reproduction in Muscidifurax uniraptor. Entomol. Exp. Appl. 100, 271–278 (2001).
Gonçalves, C. I. et al. Natural occurrence of Wolbachia-infected and uninfected Trichogramma species in tomato fields in Portugal. Biol. Control 37, 375–381 (2006).
Zhang, S. F. et al. Sensory and immune genes identification and analysis in a widely used parasitoid wasp Trichogramma dendrolimi (Hymenoptera: Trichogrammatidae). Insect Sci. 23, 417–429 (2016).
Yuan, X. H. et al. Performance of four Chinese Trichogramma species as biocontrol agents of the rice striped stem borer, Chilo suppressalis, under various temperature and humidity regimes. J. Pest Sci. 85(4), 497–504 (2012).
Li, J., Zhao, L., Li, Y., Zhao, Z. & Ma, R. Inoculative releases of Trichogramma dendrolimi for suppressing the oriental fruit moth (Grapholita molesta) in peach orchard in China. Fruits 71, 123–128 (2016).
Göttig, S. & Herz, A. Are egg parasitoids of the genus Trichogramma (Hymenoptera: Trichogrammatidae) promising biological control agents for regulating the invasive Box tree pyralid, Cydalima perspectalis (Lepidoptera: Crambidae)? Biocontrol Sci. Techn. 26, 1471–1488 (2016).
Hou, Y. Y. et al. Effect of oriental armyworm Mythimna separata egg age on the parasitism and host suitability for five Trichogramma species. J. Pest Sci. 91, 1–9 (2018).
Li, T. H. et al. Multiparasitism with Trichogramma dendrolimi on egg of Chinese oak silkworm, Antheraea pernyi, enhances emergence of Trichogramma ostriniae. J. Pest Sci. 92(2), 707–713 (2019).
Yang, C. C., Wang, J. L. & Zhang, J. Screening the diet prescription for rice moth rearing. J. Shenyang Agric. Univ. 21, 48–52 (1990).
Sumer, F. et al. A molecular key to the common species of Trichogramma of the Mediterranean region. BioControl 54, 617–624 (2009).
Yang, C. H., Chang, H. W., Ho, C. H., Chou, Y. C. & Chuang, L. Y. Conserved PCR primer set designing for closely-related species to complete mitochondrial genome sequencing using a sliding window-based PSO algorithm. PLoS One 6(3), e17729 (2011).
Wagener, B., Reineke, A., Löhr, B. & Zebitz, C. P. Phylogenetic study of Diadegma species (Hymenoptera: Ichneumonidae) inferred from analysis of mitochondrial and nuclear DNA sequences. Biol. Control 37, 131–140 (2006).
Stouthamer, R., Hu, J., van Kan, F. J., Platner, G. R. & Pinto, J. D. The utility of internally transcribed spacer 2 DNA sequences of the nuclear ribosomal gene for distinguishing sibling species of Trichogramma. BioControl 43, 421–440 (1999).
Simon, C. et al. Evolution, weighting, and phylogenetic utility of mitochondrial gene sequences and a compilation of conserved polymerase chain reaction primers. Ann. Entomol. Soc. Am. 87, 651–701 (1994).
Kishani Farahani, H. et al. Does Wolbachia infection affect decision-making in a parasitic wasp? Entomol. Exp. Appl. 155, 102–116 (2015).
Venkatesan, T. et al. Differentiation of some indigenous and exotic trichogrammatids (Hymenoptera: Trichogrammatidae) from India based on Internal transcribed spacer-2 and cytochrome oxidase-I markers and their phylogenetic relationship. Biol. Control 101, 130–137 (2016).
Larkin, M. A. et al. Clustal W and Clustal X version 2.0. Bioinformatics 23, 2947–2948 (2007).
Saitou, N. & Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 4, 406–425 (1987).
Tamura, K. et al. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739 (2011).
Duron, O. et al. The diversity of reproductive parasites among arthropods: Wolbachia do not walk alone. BMC Biol. 6, 27 (2008).
Zhou, W., Rousset, F. & O’Neill, S. Phylogeny and PCR-based classification of Wolbachia strains using wsp gene sequences. Philos. T. R. Soc. B. 265, 509–515 (1998).
Whiting, P. Multiple alleles in complementary sex determination in Habrobracon. Genetics 28, 365–382 (1943).
Liebert, A., Sumana, A. & Starks, P. Diploid males and their triploid offspring in the paper wasp Polistes dominulus. Biol. Letters 1, 200–203 (2005).
de Boer, J. G., Ode, P. J., Vet, L. E. M., Whitfield, J. B. & Heimpel, G. E. Diploid males sire triploid daughters and sons in the parasitoid wasp Cotesia vestalis. Heredity 99, 288–294 (2007).
Ma, W. J., Pannebakker, B. A., Beukeboom, L. W., Schwander, T. & van de Zande, L. Genetics of decayed sexual traits in a parasitoid wasp with endosymbiont-induced asexuality. Heredity 113, 424–431 (2014).
Stouthamer, R., van Tilborg, M., de Jong, J. H., Nunney, L. & Luck, R. F. Selfish element maintains sex in natural populations of a parasitoid wasp. Philos. T. R. Soc. B. 268, 617–622 (2001).
Cavalcanti, A. L., Falcao, D. N. & Castro, L. E. “Sex-Ratio” in Drosophila prosaltans-A character due to interaction between nuclear genes and cytoplasmic factors. Am. Nat. 91, 327–329 (1957).
Cosmides, L. M. & Tooby, J. Cytoplasmic inheritance and intragenomic conflict. J. Theor. Biol. 89, 83–129 (1981).
Rigaud, T. & Juchault, P. Conflict between feminizing sex ratio distorters and an autosomal masculinizing gene in the terrestrial isopod Armadillidium vulgare Latr. Genetics 133, 247–252 (1993).
Werren, J. H. The paternal-sex-ratio chromosome of Nasonia. Am. Nat. 137, 392–402 (1991).
van Vugt, J. F. A., Salverda, M., de Jong, J. H. & Stouthamer, R. The paternal sex ratio chromosome in the parasitic wasp Trichogramma kaykai condenses the paternal chromosomes into a dense chromatine mass. Genome 46, 580–587 (2003).
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, https://www.R-project.org (2017).
van Meer, M. M. M., Witteveldt, J. & Stouthamer, R. Phylogeny of the arthropod endosymbiont Wolbachia based on the wsp gene. Insect Mol. Biol. 8, 399–408 (1999).
Ercan, S. F., Oztemiz, S., Tuncbilek, S. A. & Stouthamer, R. Sequence analysis of the ribosomal DNA ITS2 region in two Trichogramma species (Hymenoptera: Trichogrammatidae). Arch. Biol. Sci. 63, 949–954 (2011).
Karimi, J., Darsouei, R., Hosseini, M. & Stouthamer, R. Molecular characterization of Iranian Trichogrammatids (Hymenoptera: Trichogrammatidae) and their Wolbachia endosymbiont. J. Asia-Pac. Entomol. 15, 73–77 (2012).
Almeida, R. P. & Stouthamer, R. ITS-2 sequences-based identification of Trichogramma species in South America. Braz. J. Biol. 75, 974–982 (2015).
Liu, Y., Hou, M. & Song, K. Molecular identification of Trichogramma species from South and South East Asia and natural Wolbachia infection. Entomol. Fennica 28, 57–66 (2017).
Zimmer, E. A., Jupe, E. R. & Walbot, V. Ribosomal gene structure, variation and inheritance in maize and its ancestors. Genetics 120, 1125–1136 (1988).
Hillis, D. M., Moritz, C., Porter, C. A. & Baker, R. J. Evidence for biased gene conversion in concerted evolution of ribosomal DNA. Science 251, 308–310 (1991).
Vogler, A. P. & DeSalle, R. Evolution and phylogenetic information content of the ITS-1 region in the tiger beetle Cicindela dorsalis. Mol. Biol. Evol. 11, 393–405 (1994).
Kishani Farahani, H. et al. Decrease of memory retention in a parasitic wasp: an effect of host manipulation by Wolbachia? Insect Sci. 24, 569–583 (2017).
Hua, H. Q. et al. Inter-and intra-specific differentiation of Trichogramma (Hymenoptera: Trichogrammatidae) species using PCR-RFLP targeting. COI. J. Econ. Entomol. 11, 1860–1867 (2018).
Fu, H. B. Molecular identification and population differentiation of Trichogramma spp. PhD Dissertation, Shenyang Agricultural University (2006).
Navajas, M., Lagnel, J., Gutierrez, J. & Boursot, P. Species-wide homogeneity of nuclear ribosomal ITS2 sequences in the spider mite Tetranychus urticae contrasts with extensive mitochondrial COI polymorphism. Heredity 80, 742–752 (1998).
Navajas, M. & Boursot, P. Nuclear ribosomal DNA monophyly versus mitochondrial DNA polyphyly in two closely related mite species: the influence of life history and molecular drive. Philos. T. R. Soc. B. 270, S124–S127 (2003).
Li, Y. et al. COI and ITS2 sequences delimit species, reveal cryptic taxa and host specificity of fig-associated Sycophila (Hymenoptera: Eurytomidae). Mol. Ecol. Resour. 10, 31–40 (2010).
King, L. M. & Schaal, B. A. Genotypic variation within asexual lineages of Taraxacum officinale. P. Natl. A. Sci. 87, 998–1002 (1990).
Campbell, C. S., Wojciechowski, M. F., Baldwin, B. G., Alice, L. A. & Donoghue, M. J. Persistent nuclear ribosomal DNA sequence polymorphism in the Amelanchier agamic complex (Rosaceae). Mol. Biol. Evol. 14, 81–90 (1997).
Kremer, N. et al. A new case of Wolbachia dependence in the genus Asobara: evidence for parthenogenesis induction in Asobara japonica. Heredity 103, 248 (2009).
Liu, Q. Q. et al. Effects of PI-Wolbachia on Mating Behavior and Biological Characteristics in Trichogramma dendrolimi Matsumura. J. Shenyang Agric. Univ. 49, 393–401 (2018).
Glowska, E., Dragun-Damian, A., Dabert, M. & Gerth, M. New Wolbachia supergroups detected in quill mites (Acari: Syringophilidae). Infect. Genet. Evol. 30, 140–146 (2015).
Werren, J. H. Biology of Wolbachia. Annu. Rev. Entomol. 42, 587–609 (1997).
Zchori-Fein, E. et al. Parthenogenesis-inducing microorganisms in Aphytis (Hymenoptera: Aphelinidae). Insect Mol. Biol. 4, 173–178 (1995).
Gottlieb, Y., Zchori-Fein, E., Faktor, O. & Rosen, D. Phylogenetic analysis of parthenogenesis-inducing Wolbachia in the genus Aphytis (Hymenoptera: Aphelinidae). Insect Mol. Biol. 7, 393–396 (1998).
Arakaki, N., Noda, H. & Yamagishi, K. Wolbachia-induced parthenogenesis in the egg parasitoid Telenomus nawai. Entomol. Exp. Appl. 96, 177–184 (2000).
Stouthamer, R. & Kazmer, D. J. Cytogenetics of microbe-associated parthenogenesis and its consequences for gene flow in Trichogramma wasps. Heredity 73, 317–327 (1994).
Pijls, J. W. A. M., Steenbergen, H. J. V. & Alphen, J. J. V. Asexuality cured: the relations and differences between sexual and asexual Apoanagyrus diversicornis. Heredity 76, 506–513 (1996).
Schilthuizen, M. O. & Stouthamer, R. Horizontal transmission of parthenogenesis-inducing microbes in Trichogramma wasps. Philos. T. R. Soc. B. 264, 361–366 (1997).
Pintureau, B., Chaudier, S., Lassablière, F., Charles, H. & Grenier, S. Addition of wsp sequences to the Wolbachia phylogenetic tree and stability of the classification. J. Mol. Evol. 51, 374 (2000).
Pintureau, B., Grenier, S., Heddi, A. & Charles, H. Biodiversity of Wolbachia and of their effects in Trichogramma (Hymenoptera: Trichogrammatidae). Ann. Soc. Entomol. Fr. 38, 333–338 (2002).
Nazeri, M., Ashouri, A. & Hosseini, M. Can Wolbachia infection improve qualitative characteristics of Trichogramma brassicae reared on cold stored eggs of the host? Int. J. Pest Manage. 61, 243–249 (2015).
Weeks, A. R. & Breeuwer, J. A. J. Wolbachia-induced parthenogenesis in a genus of phytophagous mites. Philos. T. R. Soc. B. 268, 2245–2251 (2001).
Wang, X. X., Qi, L. D., Jiang, R., Du, Y. Z. & Li, Y. X. Incomplete removal of Wolbachia with tetracycline has two-edged reproductive effects in the thelytokous wasp Encarsia formosa (Hymenoptera: Aphelinidae). Sci. Rep-UK. 7, 44014 (2017).
Ma, W. J. et al. Diploid males support a two-step mechanism of endosymbiont-induced thelytoky in a parasitoid wasp. BMC Evol. Biol. 15, 84 (2015).
Stouthamer, R. Wolbachia-induced parthenogenesis in Influential Passengers: Inherited Microorganisms and Arthropod Reproduction (eds. O’Neill, S. L., Werren, J. H. & Hoffmann, A. A) 102–124 (Oxford University Press, Oxford, 1997).
Huigens, M. E. & Stouthamer, R. Parthenogenesis associated with Wolbachia in Insect Symbiosis (eds. Bourtzis, K. & Miller, T. A.) 247–266 (CRC Press, Boca Raton, Florida, 2003).
Dong, H., Liu, Q. Q., Xie, L. N., Cong, B. & Wang, H. Functional response of Wolbachia-infected and uninfected Trichogramma dendrolimi Matsumura (Hymenoptera: Trichogrammatidae) to Asian corn borer, Ostrinia furnacalis Guenée (Lepidoptera: Pyralidae) eggs. J. Asia-Pac. Entomol. 20, 787–793 (2017).
Werren, J. H. & Stouthamer, R. PSR (paternal sex ratio) chromosomes: the ultimate selfish genetic elements. Genetica 117, 85–101 (2003).
van Meer, M. M. M. & Stouthamer, R. Cross-order transfer of Wolbachia from Muscidifurax uniraptor (Hymenoptera: Pteromalidae) to Drosophila simulans (Diptera: drosophilidae). Heredity 82, 163–169 (1999).
Kawai, S., Matsumoto, Y., Gotoh, T. & Noda, H. Transinfection of Wolbachia in planthoppers: nymphal injection of cultured Wolbachia and infection dynamics. Environ. Entomol. 38, 1626–1633 (2009).
Sakamoto, H. et al. Transinfection reveals the crucial importance of Wolbachia genotypes in determining the type of reproductive alteration in the host. Genet. Res. 85, 205–210 (2005).
Boivin, G. Phenotypic plasticity and fitness in egg parasitoids. Neotrop. Entomol. 39, 457–463 (2010).
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2017YFD0201000) and the Natural Science Foundation of Liaoning Province (2015020768).
Author information
Authors and Affiliations
Contributions
Quan-quan Liu and Jin-cheng Zhou performed the majority of the work, wrote the main manuscript text and prepared all figures. Hui Dong supervised this work and assisted with writing of the manuscript. Chen Zhang performed the offspring observation in Introgression experiment and Factors restraining expansion of thelytoky. Su-fang Ning, Li-jia Duan assisted with statistical analyses. All authors commented on and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, Qq., Zhou, Jc., Zhang, C. et al. Co-occurrence of thelytokous and bisexual Trichogramma dendrolimi Matsumura (Hymenoptera: Trichogrammatidae) in a natural population. Sci Rep 9, 17480 (2019). https://doi.org/10.1038/s41598-019-53992-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-53992-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.