Abstract
SerpinB2 (plasminogen activator inhibitor type 2) has been called the “undecided serpin” with no clear consensus on its physiological role, although it is well described as an inhibitor of urokinase plasminogen activator (uPA). In macrophages, pro-inflammatory stimuli usually induce SerpinB2; however, expression is constitutive in Gata6+ large peritoneal macrophages (LPM). Interrogation of expression data from human macrophages treated with a range of stimuli using a new bioinformatics tool, CEMiTool, suggested that SerpinB2 is most tightly co- and counter-regulated with genes associated with cell movement. Using LPM from SerpinB2−/− and SerpinB2R380A (active site mutant) mice, we show that migration on Matrigel was faster than for their wild-type controls. Confocal microscopy illustrated that SerpinB2 and F-actin staining overlapped in focal adhesions and lamellipodia. Genes associated with migration and extracellular matrix interactions were also identified by RNA-Seq analysis of migrating RPM from wild-type and SerpinB2R380A mice. Subsequent gene set enrichment analyses (GSEA) suggested SerpinB2 counter-regulates many Gata6-regulated genes associated with migration. These data argue that the role of SerpinB2 in macrophages is inhibition of uPA-mediated plasmin generation during cell migration. GSEA also suggested that SerpinB2 expression (likely via ensuing modulation of uPA-receptor/integrin signaling) promotes the adoption of a resolution phase signature.
Similar content being viewed by others
Introduction
SerpinB2 (aka plasminogen activator inhibitor type 2 or PAI-2) is a member of the clade B or ovalbumin-like serine protease inhibitor (ov-serpin) subgroup of the serpin superfamily. SerpinB2 is typically described as an inhibitor of the protease urokinase plasminogen activator (uPA) and (to a lesser extent) tissue plasminogen activator (tPA)1,2,3,4,5,6. uPA and tPA convert plasminogen to the active protease, plasmin, which has a wide range of physiological and pathological activities7,8,9. For instance, plasmin is well known for its involvement in fibrinolysis and thrombolysis, with both plasminogen activator inhibitor type 1 (PAI-1) (also known as SerpinE1) and SerpinB2 traditionally regarded as key inhibitors in these processes3. However, although PAI-1−/− mice have clear fibrinolysis/thrombolysis defects10,11,12, evidence that SerpinB2 inhibits fibrinolysis/thrombolysis in vivo is actually quite limited12 and not without controversy13.
Despite over 1100 publications on PAI-2/SerpinB2, no clear consensus on the physiological function of SerpinB2 has emerged, leading to labels such as “the undecided Serpin”14 or the “enigmatic serpin”4,13. Like other ov-serpins, SerpinB2 lacks a classical secretory signal peptide and is usually found in the cytoplasm15, with its presumed primary target, uPA, generally localized outside the cell. How SerpinB2 might reach the extracellular milieu has been controversial16,17; however, SerpinB2 externalization via microparticle formation has recently been illustrated for macrophages, cancer cells and syncytiotrophoblasts18,19. Nevertheless, a bewildering array of intracellular and extracellular binding partners and a diversity of functions and activities have been attributed to SerpinB220. For example, SerpinB2 has been reported (i) to modulate Th1/Th2 immunity20,21,22,23,24,25, (ii) to inhibit apoptosis in certain settings20,26,27,28, (iii) to inhibit cancer metastasis and migration18,29,30,31, (iv) to regulate differentiation and proliferation32,33,34,35,36 and (v) to inhibit IL-1β processing37. Although a range of uPA-independent functions for SerpinB2 have been reported37,38,39,40,41,42, uPA inhibition has been implicated in the majority of SerpinB2’s activities3,12,18,31,43,44,45.
SerpinB2 can be expressed by a number of different cell types including monocyte/macrophages, where SerpinB2 expression is usually inducible under inflammatory conditions3,18,20,46 and can reach up to 0.27% of total cellular protein47. However, the wide plasticity of macrophage differentiation is now well recognized, with a range of specialized macrophage populations identified in specific tissues48,49,50. Amongst these populations, constitutive expression of high levels of SerpinB2 mRNA appears to be a unique feature of large peritoneal macrophages (LPM)51,52,53. Resident peritoneal macrophages (RPM) are known to express readily detectable levels of SerpinB2 protein constitutively25, with RPM comprising of two subpopulations, LPM that are CD11bhi and F4/80hi52 and small peritoneal macrophages (SPM) that are F4/80lo and express slightly lower levels of CD11b52,53. Gata6 is believed to be a key transcription factor for differentiation of LPM, with two studies52,53 (but not a third54) suggesting that Gata6 induces SerpinB2 expression. An important function for F4/80hi, Gata6+ LPM was recently identified; rapid non-vascular migration from the peritoneum into sites of tissue injury, involving migration via lamellipodia extensions and ultimately the adoption of an alternatively activated phenotype to promote tissue repair55.
Herein we use a new bioinformatic tool, CEMiTool56, to identify genes in human macrophages that are usually co- and counter-regulated with respect to SerpinB2 to gain insights into SerpinB2’s function. RPM/LPM from both SerpinB2−/− mice (conventional knockout) and new SerpinB2R380A mice (active site mutant generated using CRISPR technology) and their respective controls were then used to further characterize the role of SerpinB2 in migration and differentiation using IncuCyte live cell analysis, RNA-Seq and gene set enrichment analyses (GSEA).
Results
Insights into serpinB2 function from CEMiTool analysis
To obtain an insight into the physiological function of SerpinB2 in human macrophages, the recently released CEMiTool56 was used to interrogate publicly available microarray data (GSE46903) on human peripheral blood-derived monocytes and macrophages that were stimulated in vitro with a range of agents, alone or in combination (n = 65 different treatments)49. The CEMiTool analysis identified 129 transcripts (Table S1a) that (across the 65 different treatments) were either (i) co-regulated with SerpinB2 (i.e. usually up-regulated when SerpinB2 was up-regulated) or (ii) counter-regulated with SerpinB2 (i.e. usually down-regulated when SerpinB2 was up-regulated) (Fig. 1a).
CEMiTool and IPA analyses of mRNA microarray data. (a) Microarray data sets from human monocytes/macrophages cultured under 65 different conditions were analyzed using CEMiTool. CEMiTool identified 129 genes (Table S1a) that were either (i) co-regulated with SerpinB2 (i.e. genes usually up-regulated when SerpinB2 was up-regulated) or (ii) counter-regulated with SerpinB2 (i.e. genes usually down-regulated when SerpinB2 was up-regulated). Expression z scores for SerpinB2 and the 129 genes after the 65 different treatments are shown. (b) Co-regulated genes were given a nominal expression value of 2 and counter-regulated genes were give a nominal expression value of −2, and the gene list analyzed using IPA (Direct only); 117 of the 129 genes were recognized by IPA. IPA identified Cellular Movement as the top scoring Molecular and Cellular Function; 51/117 genes appeared in 1 or more of the 96 Categories/Diseases or Functions Annotations that comprise Cellular Movement (Table S1b). The box plots are provided by IPA for the indicated function; each of the many (e.g. 96) internal boxes represents one annotation, with the box size indicating log10 p value and the color indicating the z-score.
We undertook two processes to validate this CEMiTool analysis for mouse RPM. IL-6 (an important product of macrophages50 and important for macrophage differentiation57) was identified by CEMITool as usually being co-regulated with SerpinB2 (Table S1a). In agreement with this result, RPM from SerpinB2+/+ mice showed significantly higher IL-6 secretion than RPM from SerpinB2−/− mice (Fig. S1a). Secondly, the 129 transcripts identified by CEMiTool were uploaded into Ingenuity Pathway Analysis (IPA), with 117 of these identified as genes by IPA. Co-regulated genes were given a nominal fold change of 2 and counter-regulated genes were given a nominal fold change of −2. Prominent in the top Up-Stream Regulators (USRs) identified by IPA where USRs associated with NF-κb (Fig. S1b). Consistent with these findings, significantly higher levels of DNA binding by NF-κb family members was seen in nuclear extracts from SerpinB2+/+ RPM than in nuclear extracts from SerpinB2−/− RPM (Fig. S1c). Overall these NF-κb results are consistent with the previously published up-regulation of SerpinB2 by NF-кB in human58 and mouse macrophages26. Also identified by the IPA USR (Fig. S1b) was (i) HMGB1, the prototypical endogenous danger molecule59, with a positive z-score, consistent with up-regulation of SerpinB2 by pathogens and endogenous danger signals20,60 and (ii) the glucocorticoid receptor (NR3C1) with a negative z-score, consistent with down-regulation of SerpinB2 by glucocorticoids61.
The top Regulator Effect Network identified by IPA analysis of the CEMiTool genes was “RELA Recruitment of cells” (Fig. S1d). The top scoring Molecular and Cellular Function returned by IPA was Cellular Movement (Fig. 1b). Cellular Movement was returned as the top scoring Molecular and Cellular Function irrespective of the IPA settings (i.e. direct only or direct and indirect, with or without nominal fold change). Cellular Movement comprised 96 Diseases or Functions Annotations (Table S1b), providing the p-value range (Fig. 1b). Of the 117 genes identified by CEMiTool, a series of co-regulated chemokines (Table S1a) played a dominant role in IPA’s Diseases or Functions Annotations (Table S1b, Molecules), with chemokines generally associated with promotion of leucocyte migration. Cell Death and Survival was ranked second and Cell-to-Cell Signaling and Interaction third (within Molecular and Cellular Functions) in this analysis (Fig. 1b).
CEMiTool56 thus provided a unique insight into the function of SerpinB2 in human macrophages by identifying genes that were consistently co- and counter-regulated with SerpinB2 under a large range of conditions. Subsequent IPA analysis of these genes indicated that they are primarily involved in cell movement/migration, suggesting SerpinB2 is part of a transcriptional program that regulates macrophage migration.
Peritoneal macrophage populations in serpinB2−/− and serpinB2+/+ mice
SerpinB2 protein is constitutively expressed by RPM, making them ideal for analyzing the function of SerpinB2 without the requirement to up-regulate SerpinB2 expression with pro-inflammatory agents20,25. RPM can be readily isolated from peritoneal lavage cells by adherence (see Material and Methods). Peritoneal lavage cells from C57BL/6 mice contain a large population of CD11bhi and F4/80hi LPM and a smaller population of CD11bmed and F4/80low SPM cells62. Peritoneal lavage cells from SerpinB2−/− and SerpinB2+/+ mice show similar proportions of LPM and SPM (Fig. 2a, red and green gates, respectively). This result was confirmed for LPM over 7 independent experiments (Fig. S2a). The number of LPM (from peritoneal lavage cells) per mouse from SerpinB2−/− and SerpinB2+/+ mice was also not significantly different over 4 independent experiments (Fig. S2b). RPM populations from both SerpinB2−/− and SerpinB2+/+ mice comprised ≈80% F4/80hi cells (corresponding to LPM), as determined in 3–4 independent experiments (Fig. S2c). Following the convention on macrophage nomenclature63, we make the distinction hereafter between RPM (isolated by adherence) and LPM (identified/isolated by FACs sorting), with RPM comprising ≈80% LPM.
LPM in SerpinB2−/− and SerpinB2+/+ mice. (a) Peritoneal lavage cells from SerpinB2−/− and SerpinB2+/+ mice were stained with CD11b and F4/80 and analyzed by FACS. Numbers (±SD) show the percentage of SPM and LPM (gates indicated) as a proportion of all peritoneal lavage cells (n = 3 mice per group). The F4/80hi lines indicate the cut-off used to FACS sort LPM. (b) Immunofluorescent antibody staining of FACS-sorted LPM using anti-murine SerpinB2 antibody and FITC-phalloidin (F-actin) imaged using confocal microscopy. All three images for SerpinB2+/+ LPM and SerpinB2−/− LPM are of the same field. (c) Immunoblotting of LPM from SerpinB2+/+ and SerpinB2−/− mice using anti-murine SerpinB2 and anti-GAPDH antibodies. Full length gels are shown in Fig. S15.
LPM express serpinB2 protein
SerpinB2 protein was readily detectable in nearly all FACS-sorted LPM by confocal microscopy (Fig. 2b) using a polyclonal anti-murine SerpinB2 antibody raised against a CD interhelical loop region of murine SerpinB243. No SerpinB2 protein was detected in SerpinB2−/− LPM (Fig. 2b). Immunoblotting further illustrated the high level of specificity for SerpinB2 of this antibody, recognizing the expected 47 kD band in SerpinB2+/+ LPM lysates and showing no detectable reactivity against any protein in lysates of SerpinB2−/− LPM (Fig. 2c). These data clearly illustrate SerpinB2 protein expression in LPM, consistent with constitutive SerpinB2 mRNA expression in these cells52. In contrast, bone marrow derived macrophages express SerpinB2 mRNA, but little if any SerpinB2 protein25.
FACS sorted-SPM were found not to express detectable levels of SerpinB2 protein, consistent with low levels of SerpinB2 mRNA expressed by these cells (data posted on Immgen by Gautier et al.)64. (SPM from both SerpinB2−/− and SerpinB2+/+ mice are thus SerpinB2 negative and any differences between RPM from SerpinB2−/− and SerpinB2+/+ mice thus likely resides within the LPM populations).
Increased migration of serpinB2−/− RPM on matrigel
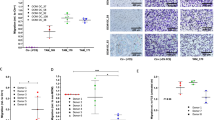
Given the CEMiTool results and the reported role of SerpinB2 in suppressing cancer cell metastasis18,65, the migration of RPM from SerpinB2−/− and SerpinB2+/+ mice was compared using the standard IncuCyte™ scratch wound cell migration assays. RPM from SerpinB2−/− mice migrated significantly faster than RPM from SerpinB2+/+ mice, when cells were plated onto Matrigel (Fig. 3a; a repeat experiment is shown in Fig. S3a). This effect (Fig. 3a) was not due to any cell viability differences, with SerpinB2−/− and SerpinB2+/+ RPM after seeding onto Matrigel showing similar survival over the period of the experiment (Fig. S3b).
RPM migration and length of membrane protrusions. (a) Migration into “scratch wounds” of ex vivo SerpinB2−/− and SerpinB2+/+ RPM seeded onto Matrigel-coated wells analyzed using the standard IncuCyte scratch wound assay. Data was generated (for both SerpinB2−/− and SerpinB2+/+ RPM) from 3 pools of RPM, each derived from 3 mice, with each pool seeded into 12 wells (i.e. n = 9 SerpinB2−/− and n = 9 SerpinB2+/+ mice). The mean of the 3 pools is shown (n = 3). Statistics by repeat measures ANOVA. (b) As for (a) but using ex vivo RPM from SerpinB2R380A and C57BL/6J mice, one pool of 3 mice (for each strain) and 23 Matrigel coated wells. (c) Western blot of RPM from the indicated mice strains, stained with the murine anti-SerpinB2 antibody. (d) Length of cellular protrusions of RPM within the scratch areas for mice described in (a). The number of cells interrogated is provided within each bar (n), with the longest protrusion from each cell used (i.e. one value for each cell). (e) As for (d) for mice described in (b). (f,g) Representative phase images of the RPM described in (d) at 25 hours post seeding. See also Fig. S3d,e.
uPA plays a key role in migration of monocytes and macrophages via plasmin-mediated degradation of the extracellular matrix (ECM)66,67. These data thus support the view that SerpinB2 inhibits uPA-dependent cell migration in macrophages, with SerpinB2 externalized via microparticle formation during migration68 and thereby able to inhibit uPA18. We (like others20) have been unable to detect SerpinB2/uPA complexes in in vivo or in ex vivo settings. Nevertheless, RPM can be shown to secrete proteolytically active uPA, with such secretion unaffected by SerpinB2 deficiency (Fig. S3c).
Increased migration of serpinB2R380A RPM on matrigel
SerpinB2−/− mice may potentially not be ideal for studying SerpinB2 deficiency for a number of reasons. The SerpinB10 gene neighbors the SerpinB2 gene on mouse chromosome 1, and a role for this gene in food allergy was recently reported69. C57BL/6 mice have a premature stop codon in SerpinB1070, whereas the original SerpinB2−/− mice71 do not. It is highly unlikely that backcrossing onto C57BL/625 would separate these two adjacent Serpin genes, so SerpinB2+/+ littermate mice are SerpinB10−/−, and SerpinB2−/− mice are SerpinB10+/+. In addition, neighboring gene perturbation affects have previously been identified in Serpin knock-out mice72, with such affects potentially changing expression of other genes in the Serpin cluster. To address these potential issues, a homozygous SerpinB2 active-site mutant mouse line (SerpinB2R380A) on a C57BL/6 background was generated using CRISPR technology45, with C57BL/6 mice used as controls. As SerpinB2 has been reported to have functions that are independent of its protease inhibition activity20,39,40,73, the SerpinB2R380A mouse would also assess the role of SerpinB2’s protease inhibition activity.
RPM from SerpinB2R380A mice migrated significantly faster than RPM from C57BL/6J mice in the scratch test (Fig. 3b), clearly confirming that SerpinB2 inhibits migration on Matrigel and that SerpinB2’s protease inhibition activity is required. Immunoblotting with anti-SerpinB2 antibody showed good expression of SerpinB2R380A protein in RPM from SerpinB2R380A mice (Fig. 3c), illustrating that this mutation does not compromise protein expression or stability. To our knowledge, this is the first time a phenotype for a SerpinB2 reactive site mutant has been shown in primary cells. The data is consistent with uPA being the target of macrophage SerpinB2, with uPA and plasmin described as key players in macrophage migration67,74,75,76,77.
SerpinB2 reduced the length of membrane protrusions in RPM
SerpinB2 expression in cancer cells has been shown to reduced migration and metastasis18,65 and to reduce the length of migration-associated invadopodia-like structures18,29. Macrophages also generate migration-associated membrane protrusions, primarily filopodia and lamellipodia. RPM membrane protrusions were measured after plating onto Matrigel and were significantly longer in SerpinB2−/− RPM (Fig. 3d) and SerpinB2R380A RPM (Fig. 3e) than in their respective wild-type controls. Phase images further illustrate the differences (Fig. 3f,g); additional phase images are provided in Figs. S3d,e. These data argue that SerpinB2 has a similar role in inhibiting migration in both cancer cells and macrophages.
SerpinB2 can be externalized in cancer cells and macrophages via formation of microparticles18. To provide evidence that extracellular SerpinB2 can indeed result in shortening of cellular protrusions, small quantities of recombinant SerpinB2 and SerpinB2R380A (generated using transfected HEK239T cells) were incubated with SerpinB2−/− RPM and B16 melanoma seeded onto Matrigel. Recombinant SerpinB2, but not recombinant SerpinB2R380A was able (i) to form the expected covalent complex with uPA and (ii) to reduce the length of cellular protrusions in SerpinB2−/− RPM and B16 cells (Fig. S4). These data support the view that extracellular SerpinB2 interacting with extracellular uPA leads to shortening of migration-associated protrusions and inhibition of migration.
SerpinB2 localizes to focal adhesions and lamellipodia
Microparticle formation has been reported to be intimately associated with formation of membrane protrusions during migration68. Using confocal microscopy of SerpinB2+/+ RPM, SerpinB2 was consistently found adjacent to, or close to, actin in focal adhesions and lamellipodia (Fig. 4). Enlargements of the four focal adhesions in the merged image in Fig. 4a (right hand image) are shown in Fig. S5. Quantitation of green (actin), red (SerpinB2) and yellow (merged) pixels are provided, with yellow pixels comprising about 10–25% of all green/red/yellow pixels (Fig. S5). The close proximity of actin and SerpinB2 could also be in seen in the leading edge of lamellipodia (Fig. 4b, Merged, white arrowheads). Just behind the leading edge, the close proximity of actin and SerpinB2 could again be seen in focal adhesions (Fig. 4b, Merged, white arrows), with enlarged images of these focal adhesions again showing significant levels of overlapping staining (Fig. 4c). Both uPA, the uPA receptor and plasmin are found associated with focal adhesions and lamellipodia66,78,79, with plasmin generation required for degradation of the ECM and de-adhesion67,78,80,81. Thus SerpinB2 localizes to sites associated with microparticle formation, uPA activity and migration, consistent with the view that SerpinB2 inhibits migration by inhibiting uPA18,65,82.
Confocal immunofluorescence microscopy. Adherent LPM from wild-type mice were dual labeled with FITC-phalloidin and anti-SerpinB2 antibody. (a) Actin; FITC-phalloidin (F-actin) staining showing actin concentrations at focal adhesions (green). SerpinB2; SerpinB2 staining localized around and in the focal adhesions (red). Merge; overlapping staining (yellow); see Fig. S5 for enlargements and quantitation of overlap. (b) Actin; FITC-phalloidin staining showing F-actin concentration along the leading edge of a lamellipodia (bottom left, green). SerpinB2; SerpinB2 staining (red) also staining the leading edge of a lamellipodia (bottom left, red). Merge; overlapping staining at the leading edge of a lamellipodia (yellow, arrowheads). (c) The two clusters of focal adhesions indicated in b (arrows) are enlarged to show overlapping localization of actin and SerpinB2 staining. The images shown are representative of 2 independent experiments where 5 images were examined containing ≈10 cells per image with ≈50 cells examined per experiment. Approximately 30% of cells examined showed overlapping staining at focal adhesions and/or lamellipodia.
The generalized uPA-dependent peri-cellular proteolysis reported previously for macrophages67, was not increased in SerpinB2−/− RPM (Fig. S6). This observation is consistent with the lack of an effect of SerpinB2 expression on extracellular levels of active uPA (Fig. S3c). uPA-dependent peri-cellular proteolysis is not ordinarily associated with microparticle release, arguing that SerpinB2-mediated inhibition of uPA is restricted to settings where microparticle formation allows SerpinB2 externalization, such as during the formation and retraction of membrane protrusion that is associated with migration68.
RNA-Seq of migrating RPM from SerpinB2R380A and C57BL/6 mice
RPM from SerpinB2R380A and C57BL/6 mice migrating on Matrigel (as in Fig. 3b) for 24 hours were analyzed by RNA-Seq. Library size graphs and MDS plots are shown in Fig. S7a,b. A complete list of gene counts are provided in Table S1c, from which a list of differentially expressed genes (DEGs) was generated (q < 0.01, n = 1481) (Table S1d). Validation by qRT PCR of differentially expression of 4 genes is shown in Fig. S7c–f.
The aforementioned 1481 DEGs were analyzed using Enrichr, with the Mouse Gene Atlas and ARCHS4 Tissues data bases returning Macrophage as the top term (Figs 5a, S8a); consistent with the FACS data showing that these RPM cultures comprise >80% F4/80hi macrophages (Fig. S2c). The fibrinolysis pathway was also returned as the top term by BioCarta 2013 (Fig. S8a, blue arrow). When the DEG list was analyzed by IPA the most significant Molecular and Cellular Function was Cellular Movement and the most significant Physiological System Development and Function was Immune Cell Trafficking (Figs 5a, S8b); consistent with the results in Figs 1b and 3b.
Summary of RNA-Seq data and pre-ranked GSEA for resolution phase macrophages. (a) RPM from SerpinB2R380A and C57BL/6 mice were seeded onto Matrigel for 24 h and were then analyzed by RNA-Seq; 1481 DEGs were identified (using a filter of q < 0.01); 624 DEGs were up-regulated, and 857 were down-regulated in SerpinB2R380A RPM. These DEGs were analyzed by Enrichr and IPA. (b) Up-regulated DEGs from resolution phase macrophages (Table S1g) were analyzed by GSEA against the complete RNA-Seq gene list (Table S1c), with genes pre-ranked by p-value. The NES score (normalized enrichment score) and false discovery rate (FDR) q values are indicated. (c) Heat map of core enriched genes from b, illustrating that genes up-regulated in wild-type (WT) RPM (relative to SerpinB2R380A RPM) are also up-regulated in resolution phage macrophages (relative to naïve and inflammatory macrophages). The core enriched gene list is also provided in Table S1g.
When the DEG list (n = 1481; Table S1d) was further analyzed using Enrichr, multiple data bases returned top ranking terms associated with focal adhesions, integrin signaling and ECM interactions; consistent with Fig. 4. These terms emerged to be largely present in the down-regulated genes (n = 857) (Figs 5a, S9); an observation consistent with reduced matrix interactions for faster migrating SerpinB2R380A RPM (Fig. 3b). Panther 2016 also returned Plasminogen activating cascade (Fig. S9, blue arrow).
SerpinB2 and M1/M2 polarization
When the DEG list (n = 1481; Table S1d) was analyzed using IPA upstream regulator function (USR, direct only), a series of M1-associated USRs (see below) were identified (Fig. 5a, Table S1e) that were largely retained when only the up-regulated genes (n = 624) were used (Table S1f). Amongst the USRs was RELA (Table S1e), consistent with Fig. S1d and the increased migration seen for SerpinB2R380A RPM (Fig. 3b). Particularly prominent (high activation z-scores) amongst the USRs that were more active in SerpinB2R380A RPM, were a series of transcription factors associated with M1-polarization83,84; specifically, interferon response factors (IRF7, IRF1 and IRF5), STAT1 and NF-кb family members (REL, NFKB2, RELA, NFKB1) (Fig. 5a, Table S1e). STAT6, an inhibitor of M1 polarization84, was also identified with a negative activation z-score (Table S1e). These results are arguably consistent with multiple studies in SerpinB2−/− mice reporting that in certain settings SerpinB2 can regulate Th1/Th2 immunity20,21,22,23,24,25, with Th1 associated with M1 and Th2 associated with M2 macrophage polarization85. However, IRF3 (generally associated with M284) is also predicted to be more active in SerpinB2R380A RPM, and IRF8 and IRF2 (that usually promote M184) were identified with negative z-scores (Fig. 5a, Table S1e). These results suggest that SerpinB2’s effects on macrophage polarization are not a straightforward fit with the classical M1/M2 model50. This notion is supported by qRT-PCR analysis showing no significant differences in the mRNA levels of the classical M2 marker, Arg-1, in SerpinB2−/− and SerpinB2+/+ RPM (Fig. S10a). Macrophage polarization has been associated with changes in phagocytic activity86,87,88 and phagocytosis of carboxylate-modified polystyrene beads88 was also unaffected in SerpinB2−/− mice (Fig. S10b).
SerpinB2 expression promotes adoption of a resolution phase signature
Gata6+ LPM ultimately adopt an alternatively activated phenotype as part of the tissue repair process55. To test whether loss of SerpinB2 function might influence this process, a list of 224 genes up-regulated in mouse resolution phase peritoneal macrophages (relative to both naive peritoneal macrophages and inflammatory peritoneal macrophages) (Table S1g) was generated from publicly available microarray data, using data and methods described in Stables et al.89. The 224 DEGs were then used in a GSEA against a pre-ranked gene list of all genes from the RNA-Seq analysis of C57BL/6 versus SerpinB2R380A RPM. A significant correlation emerged (Fig. 5b), suggesting SerpinB2 expression promotes up-regulation of a number of genes associated with the resolution phase signature in peritoneal macrophages (Fig. 5c). Core enriched genes (n = 31) (Fig. 5c) analyzed by IPA (direct and indirect) returns IL-27 (p = 3.9 × 10e8, activation z-score = 2.4), IL-6 (p = 4.4 × 10e8, activation z-score = 1.5) and STAT3 (p = 1.1 × 10e6, activation z-score = 0.58) as top USRs by p value. IL-27, IL-6 and STAT3 are associated with alternative activation and/or anti-inflammatory activity57,90. This bioinformatics analysis is consistent with the significantly elevated levels of IL-6 secreted by cultured SerpinB2+/+ compared to SerpinB2−/− RPM (Fig. S1a). Taken together these results argue that SerpinB2 expression in LPM contributes to the alternatively activated tissue repair program adopted by LPM at the site of injury55.
SerpinB2 and Gata6
Two studies52,53 (but not a third54) suggested that Gata6 induces SerpinB2 expression. The USRs described above also include Gata6 (Table S1e, z score = 2.3, p = 9.8 × 10−9), suggesting loss of SerpinB2 activity increased the activity of Gata6. GSEAs were thus undertaken comparing DEGs from SerpinB2R380A RPM (Table S1d) with the published microarray analyses from studies on Gata6−/− LPM. For GSE4704953, up-regulated DEGs from SerpinB2R380A RPM showed significant correlation with genes down-regulated in Gata6−/− LPM (Fig. 6a). In addition, down-regulated DEGs from SerpinB2R380A RPM showed significant correlation with genes up-regulated in Gata6−/− LPM (Fig. 6a). The same patterns emerged for GSE5668452 (Fig. 6b). The third study GSE3744854, which did not identify SerpinB2 as a Gata6-regulated gene, showed significant co-regulation (rather than counter-regulation) for up-regulated genes (Fig. 6c, Up DEGs). For down-regulated genes, core enriched genes again showing clear counter-regulation (with considerable overlap with the other studies), although this did not reach significance (Fig. 6c, Down DEGs). A very similar picture merged when the same GSEAs were undertaken using DEGs (listed in Table S1i) obtained from RNA microarray analyses of ex vivo FACS-sorted F4/80hi cells (gate is shown in Fig. 2a) from SerpinB2−/− and SerpinB2+/+ mice (Fig. S11). Thus transcriptome analysis of migrating SerpinB2R380A RPM and ex vivo SerpinB2−/− F4/80hi cells both suggest that a significant number of genes regulated by Gata6 are counter-regulated by SerpinB2 expression.
GSEA using microarray data sets from Gata6−/− LPM. The 624 up-regulated and the 857 down-regulated DEGs in SerpinB2R380A RPM were compared by GSEA with 3 microarray data sets that compared LPM from Gata6+/+ and Gata6−/− mice; (a) GSE47049, (b) GSE56684 and (c) GSE37448. GSEAs showing significant counter-regulation (i.e. up in SerpinB2R380A and down in Gata6−/− or down in SerpinB2R380A and up in Gata6−/−) are indicated with red p values. Heat maps of core enriched genes showing counter-regulation are shown for (a,b) and for Down DEGs in (c), which showed counter-regulation that did not reach significance (q = 0.094). Up DEGs in (c) showed co-regulation (positive NES score) indicating no significant counter-regulation. The core enriched genes illustrated by the heat maps are listed in Table S1h.
The core enriched genes from GSEAs using GSE47049 and GSE56684 (Table S1h) for DEGs up-regulated in SerpinB2R380A RPM (Fig. 6a,b, Up DEGs) were analyzed by IPA (direct only). The top scoring Molecular and Cellular Functions was Cellular Movement (p-value range 2.49e-03 - 4.03e-09). When the core enriched genes (Table S1h) from all 3 studies for DEGs down-regulated in SerpinB2R380A RPM (Fig. 6a–c, Down DEGs) were analyzed by IPA, the top Physiological System Development and Function was Immune Cell Trafficking (1.50e-03 - 1.02e-08). In broad terms these results are consistent with a role for Gata6 in promoting migration91,92,93,94,95 and SerpinB2-expression inhibiting migration. We speculate about a potential mechanism that might underpin this counter-regulation in Fig. S12 (see Discussion).
SerpinB2 and RPM survival
uPA and plasmin have been linked to apoptosis in a variety of cell types96,97 including monocytes/macrophages98,99,100 and cancer cells27,101,102, with the latter studies illustrating that uPA/plasmin can both promote and suppress apoptosis depending on the setting. SerpinB2 has similarly been reported to both inhibit20,26,41,42 and promote apoptosis103,104, with several studies also showing no effect on apoptosis18,43,105. We were unable to see any differences in cell death of SerpinB2−/− and SerpinB2+/+ RPM induced by LPS, IFNγ, ATP or L-Leucyl-L-Leucine methyl ester (Fig. S13). Viability on Matrigel was also unaffected by SerpinB2 deficiency (Fig. S3b).
IL-1β induction and secretion was unaffected in SerpinB2−/− RPM
SerpinB2 has been reported to inhibit caspase 142 and IL-1β processing37. However, we were unable to see any significant increases in pro-IL-1β expression or IL-1β secretion in SerpinB2−/− RPM (Fig. S14). We also previously saw no role for keratinocyte SerpinB2 in IL-1β expression or processing43.
Discussion
Herein we provide multiple lines of evidence that the function of macrophage SerpinB2 is inhibition of migration. The data was derived from bioinformatics analyses and investigation of ex vivo murine LPM/RPM that constitutively express SerpinB2. LPM from two SerpinB2-deficient mouse strains (and their respective wild-type controls) were used, (i) conventional knock-out SerpinB2−/− mice and (ii) SerpinB2R380A mice generated using CRISPR technology. Macrophage SerpinB2 thus appears to have a similar function to SerpinB2 expression in cancer cells, where in a range of settings SerpinB2 expression has been shown to inhibit metastasis/invasion18,29,31,44,65,82.
A large body of evidence supports a role for uPA and plasmin in promoting migration66,67,74,75,76,77,106. That SerpinB2 can inhibit uPA in vitro is not in dispute3 (Fig. S4b). That SerpinB2 inhibits uPA in vivo has been less easy to demonstrate20, with SerpinB2-uPA complexes (to the best of our knowledge) - demonstrated in ex vivo samples in only one45 (perhaps two107) publications. We have also been unable unequivocally to demonstrate such complexes from primary macrophages, perhaps because most such complexes are not cell associated, are present at low concentrations and/or because they are rapidly degraded108. The Enrichr analyses of the RNA-Seq data did provide some evidence for a role for uPA/plasmin, with 2 data bases, BioCarta 2013 (Fig. S7A, blue arrow) and Panther 2016 (Fig. S8, blue arrow), identifying fibrinolysis/plasminogen activation, although neither reached significance after multiple comparisons adjustment of the p-value. However, the general (and perhaps inappropriate13) assumption that SerpinB2 (like PAI-1) is associated only with fibrinolysis, may have resulted in a paucity of appropriate data and data-base annotation. The evidence that SerpinB2 inhibits uPA in vivo thus remains largely indirect. The increased migration of SerpinB2R380A RPM provides compelling evidence that the protease inhibition function of SerpinB2 is involved; an observation consistent with uPA inhibition. SerpinB2 and actin staining overlapped in focal adhesions and lamellipodia, structures that are also associated with uPA, the uPA receptor and plasmin66,78,79. SerpinB2 can be externalized via microparticle formation18, a process intimately associated with formation of membrane protrusions during migration68. In addition, rSerpinB2 (but not rSerpinB2R380A) formed complexes with uPA and reduced the length of cellular protrusions (Fig. S4), with SerpinB2 (but not SerpinB2R380A) expression similarly reducing the length of migration-associated cellular protrusions (Fig. 3d,e).
There is a widespread view that SerpinB2 inhibits apoptosis20,26,41,42, although this activity appears to be context dependent18,43,103,104,105. The evidence presented herein may provide an explanation, given that migration and ECM attachment can influence susceptibility to apoptosis109,110, with uPA and plasmin also linked to apoptosis in a variety of cell types96,97 including monocytes/macrophages98,99,100 and cancer cells27,101,102. Increased SerpinB2 may simply reduce plasmin-mediated ECM degradation and cell detachment, thereby promoting cell survival111. For instance, SerpinB2 may inhibit cell death by reducing signaling that promotes anoikis (ECM detachment-induced apoptosis)112.
We have been unable to find any evidence in keratinocytes43 or in RPM (Fig. S14) that SerpinB2 regulates IL-1 processing37,113. However, IL-1α and IL-1β were identified by CEMiTool as being consistently co-regulated with SerpinB2 (Table S1a). Both SerpinB2 and IL-1 are externalized (secreted) under pro-inflammatory conditions by non-conventional processes involving secretory vesicles or via loss of the plasma membrane barrier18,43,114,115, although the role of gasdermin D pores (that facilitate IL-1β secretion114) in SerpinB2 secretion from macrophages remains to be determined. Thus, although SerpinB2 does not appear to regulate IL-1, SerpinB2 and IL-1 often appear to be co-regulated and may be secreted by similar mechanisms.
Counter-regulation of some Gata6-regulated genes associated with cell migration by SerpinB2 expression is a novel finding, although the observation is consistent with (i) promotion of cell migration via elevated levels of Gata6 in a number of settings91,92,93,94,95, perhaps mediated in part by direct up-regulation of uPA116,117 and (ii) inhibition of migration by SerpinB2 via reduced uPA-mediated plasmin formation, with plasmin associated with promotion of migration106. The mechanistic basis of this counter-regulation remains to be characterized; however, one might speculate that ERK is involved. SerpinB2+/+ RPM express lower levels of phospho-ERK1/2 than RPM from SerpinB2−/−mice (Fig. S12a). Less phospho-ERK1/2 activity would reduce phosphorylation and activation of FOXO1118, with a SerpinB2-associated reduction in FOXO1 activity supported by IPA analysis of the core enriched genes from Fig. 6 (Fig. S12b). FOXO1 has recently been shown positively to regulate Gata6 expression in peritoneal resident macrophages119, so reduced FOXO1 would lead to reduced Gata6 expression (Fig. S12c). Less GATA6 activity in SerpinB2+/+ RPM is also predicted by IPA analysis of the RNA-Seq data (see Table S1e; showing more GATA6 activity in SerpinB2R380A RPM).
That changes in migration might contribute to altered macrophage differentiation is perhaps not surprising120,121,122. Simple inhibition of plasmin production would appear unlikely to provide a straightforward mechanism, as plasmin has been reported to promote both M1/pro-inflammatory responses123,124,125 and M2-associated macrophage polarization126,127. Modulation of integrin and uPAR signaling events are more likely involved44,128,129,130,131,132,133. For instance, uPAR/integrin signaling often involves ERK activation44,129,131,134. Phospho-ERK-1/2 levels were higher in SerpinB2−/− RPM (Fig. S11a), consistent with observations in cancer cells135. ERK activation is often associated with M1 polarization136,137,138,139 and migration140,141. SerpinB2 has been associated with suppression of some M1-associated activities in certain settings20,21,22,23,24,25; however, the overall fit with the M1/M2 paradigm appeared to be poor (Fig. 5a). The key observation made herein is that SerpinB2-associated modulation of macrophage differentiation perhaps has less to do with M1/M2; instead, SerpinB2 expression appears to inhibit migration and promote adoption of a resolution phase signature (Fig. 5b), consistent with the role of LPM in tissue repair55.
Materials and Methods
CEMiTool analysis
CEMiTool56 was used to analyze microarray data (GSE46903) on human peripheral blood-derived monocytes and macrophages stimulated in vitro with 65 different treatments49. The raw microarray gene expression data was downloaded from the Gene Expression Omnibus website (GEO, www.ncbi.nlm.nih.gov/geo/). The arrayQualityMetrics Bioconductor package142 was used to remove samples that failed at least 3 of the 5 tests. The expression normalization was performed by the affy package’s RMA function (Bioconductor)143. CEMiTool was used with default parameters to identify co-expression modules using normalized expression data56; specifically to identify genes that were co-regulated or counter-regulated with SerpinB2.
IPA analyses
Data were analyzed through the use of Ingenuity Pathway Analysis (IPA)144 (QIAGEN Inc., https://www.qiagenbioinformatics.com/products/ingenuity- pathway-analysis). Ingenuity Pathway Analysis was undertaken using default parameters (direct only or direct and indirect, as stipulated for each analysis) using Build version 478438 M, Content version 44691306 (Release Date 2018-06-15).
Ethics statement
Mouse work was approved by the QIMR Berghofer Medical Research Institute Animal Ethics Committee. All mouse work was conducted in accordance with the “Australian code for the care and use of animals for scientific purposes” as defined by the National Health and Medical Research Council of Australia. Mice were euthanized using carbon dioxide.
Mice
SerpinB2−/− and SerpinB2+/+ mice backcrossed 12 times onto C57BL/6 J mice have been described previously25 and were bred in-house at QIMR Berghofer. Heterozygous mice with a SerpinB2R380A active site mutation were generated at the Australian Genome Research Facility Ltd. (Melbourne, Australia). The active site (P1) Arg38020 (codon AGA) of SerpinB2 was changed to Ala380 (codon GCA); i.e. nucleotides 1222 and 1223 (with reference to accession NM_011111.4) were changed from AG to GC. Two silent mutations were also introduced GCAACTGGACATGGTGGCCCACAGTTTGTC to prevent cutting of the oligonucleotide during genome editing. A homozygous SerpinB2R380A line was generated in-house. The genotype was confirmed by tail tipping, extraction of DNA (Extract-N-Amp Tissue PCR Kit, Sigma), PCR (primers Forward tctgaggtgttccatcaag, Reverse ctaccaacaaatagtatcgtgtg) and sequencing of the PCR products45.
Isolation of peritoneal lavage cells
Peritoneal lavage cells were isolated essentially as described145. Female mice were used to avoid any potential issues arising from fighting and injury occasionally seen in male mice. RPM were isolated by adherence as described25; see also Fig. S2.
FACS analysis
FACS analysis of peritoneal lavage cells used the LSR Fortessa 4 (BD Biosciences) and FACS sorting the MOFLO XDP (Beckman Coulter, Inc.), with analysis undertaken using FLowJo vX.0.7 software. Antibodies used were APC anti-mouse CD11b (Cat 101212, clone M1-70, IgG2b, Biolegend), isotype control APC Rat IgG (Cat 400612, clone RTK4530, IgG2b, Biolegend), FITC anti-mouse F4/80 (Cat MCA497FB, clone CI:A3-1, IgG2b, AbD Serotec), and isotype control FITC Rat IgG (Cat 400505, clone RTK2758, Biolegend).
IncuCyte scratch wound cell migration assay
Peritoneal lavage cells were seeded onto wells of 96 well plates (ImageLock, Essen Bioscience Inc, MI, USA) coated with Corning Matrigel Matrix (Corning Incorporated, Tewksbury, MA, USA) (overnight 100 μg/ml). Cells were incubated for 3 hours; medium RPMI1640 supplemented with 10% endotoxin free (tested using RAW264 reporter cells146) fetal calf serum (Gibco), glutamine (Gibco) and penicillin/streptomycin (Thermo Fisher Scientific). Wells were washed 3 times in PBS. The plates were analyzed at the indicate times via the IncuCyte ZOOM System (Essen Bioscience) and the 2016A live cell analysis software after application of a scratch. The Relative Wound Density (%) over time was determined with the cell density in the scratch wound area expressed relative to the cell density outside of the scratch wound area.
The lengths of cellular protrusions from RPM within the scratch wound areas were measured using the images provided by IncuCyte (and uploaded into ImageJ), with the longest protrusion from each cell used (i.e. one value for each cell). Round cells (protrusion length equal to zero) were not included as these cells were deemed to be non-migrating.
Anti-mouse serpinB2 antibody
An affinity purified rabbit polyclonal, anti-murine SerpinB2 antibody was supplied by Peptide Specialty Labs GmbH (Heidelberg, Germany). A coupled CD loop region peptide EIGSYGITTRNPENFSGC was used as the immunogen43. Immunoblotting was performed as described43.
Confocal florescent microscopy
Confocal microscopy was undertaken using Leica TCS confocal microscope (Leica Microsystems, North Ryde, NSW, Australia), with images shown using 2–3 adjacent 500 nm Z stacks. Staining with the anti-mouse SerpinB2 antibody and phalloidin-rhodamine (for actin) has been described previously18. Briefly cells were cultured on glass coverslips, fixed in 2% paraformaldehyde and permeabilized with 0.1% Triton X-100 in PBS.
RNA-Seq
Peritoneal lavage cells from SerpinBR380A and C57BL/6 mice were seeded onto Matrigel in 6 well plates; 107.cells from 3 mice per well (3 wells per mouse strain as biological replicates). After 3 hours plates were extensively washed with PBS. After 24 hours culture, RPM were extracted using RNeasy Plus Mini kit (including a RNase free DNase I step) according to manufacturer’s instructions. Samples were sent to the Australian Genome Research Facility (Melbourne, Australia), cDNA libraries were prepared and sequenced (100 bp single end reads) using the Illumina HiSeq2500 Sequencer (Illumina Inc.) with reads mapped to the mouse genome (Mus_musculus.GRCm38) as described147. The read counts were used to determine gene expression and identify differentially expressed genes (DEGs) using R packages (R version 3.2.0) ‘edgeR’ (v3.18.1) and ‘limma’ (3.32.7). (https://bioconductor.org/packages/release/bioc/html/edgeR.html). The default TMM normalization method of edgeR was used to normalize the counts. The GLM model was used to perform differential expression comparison between the groups. Genes that had >1 CPM in at least 3 samples were used for further analysis. The differentially expressed gene list was generated by applying a Benjamini-Hochberg corrected p-value (i.e. FDR or q value) filter of <0.01.
Gene set enrichment analysis (GSEA)
GSEAs148 were conducted using the software GSEA Desktop v3.0 (Broad Institute Inc., MIT, USA). A pre-ranked GSEA was performed to determine whether the up-regulated genes from resolution phase macrophages89 were enriched in the SerpinB2R380A RPM expression dataset (Fig. 5b). Resolution phase macrophages in the Stables et al. publication89 were obtained by purification of macrophages from peritoneal lavage cells by B cell depletion and adherence >48 hours after intraperitoneal treatment with 0.1 mg zymozan. They were distinguished from both naïve macrophages (no treatment) and pro-inflammatory macrophages (intraperitoneal treatment with 10 mg zymozan). The RNA-Seq expression ranked file contained genes with >1 CPM in at least 3 samples. The gene rank values were calculated by -log10(p-value) from DEG analysis (limma package, Bioconductor), where the up-regulated genes (in WT) were given a positive p-value and down-regulated genes given a negative p-value. The resolution phase gene set (Table S1g) was generated using the microarray data (Gene Express; E-MEXP-3189) and methods described previously89. The parameters used in the pre-ranked GSEA were the classic enrichment statistic, tTest metric, with 1,000 gene set permutations. GSEA was also used to determine whether up or down-regulated genes in WT versus SerpinB2R380A RPM were enriched in 3 expression sets from Gata6−/− mice (Fig. 6), with the GSEAs using weighted p2 enrichment statistic. The raw microarray gene expression data was downloaded from the GEOwebsite and process as described above for CEMiTool.
Statistics
Statistical analysis of experimental data was performed using IBM SPSS Statistics for Windows, Version 19.0. The t test was used when the difference in variances was <4, skewness was ≥2 and kurtosis was ≤2. When differences in variances were >4 the non-parametric Kolmogorov–Smirnov test was used. The repeat measure ANOVA was used for IncuCyte data (Fig. 3a,b) given cells were repeatedly imaged over time.
Data Availability
The Illumina RNA-Seq data generated for this study are available from NCBI SRA accession: SRP158653.
References
Ritchie, H., Robbie, L. A., Kinghorn, S., Exley, R. & Booth, N. A. Monocyte plasminogen activator inhibitor 2 (PAI-2) inhibits u-PA-mediated fibrin clot lysis and is cross-linked to fibrin. Thromb Haemost 81, 96–103 (1999).
Corsetti, J. P. et al. Influences on plasminogen activator inhibitor-2 polymorphism-associated recurrent cardiovascular disease risk in patients with high HDL cholesterol and inflammation. Atherosclerosis 250, 1–8 (2016).
Kruithof, E. K., Baker, M. S. & Bunn, C. L. Biological and clinical aspects of plasminogen activator inhibitor type 2. Blood 86, 4007–4024 (1995).
Medcalf, R. L. Plasminogen activator inhibitor type 2: still an enigmatic serpin but a model for gene regulation. Methods Enzymol 499, 105–134 (2011).
Al-Horani, R. A. Serpin regulation of fibrinolytic system: implications for therapeutic applications in cardiovascular diseases. Cardiovasc Hematol Agents Med Chem 12, 91–125 (2014).
Booth, N. A. Fibrinolysis and thrombosis. Baillieres Best Pract Res Clin Haematol 12, 423–433 (1999).
Aisina, R. B. & Mukhametova, L. I. [Structure and functions of plasminogen/plasmin system]. Bioorg Khim 40, 642–657 (2014).
Foley, J. H. Plasmin(ogen) at the Nexus of Fibrinolysis, Inflammation, and Complement. Semin Thromb Hemost 43, 135–142 (2017).
Draxler, D. F., Sashindranath, M. & Medcalf, R. L. Plasmin: A Modulator of Immune Function. Semin Thromb Hemost 43, 143–153 (2017).
Farrehi, P. M., Ozaki, C. K., Carmeliet, P. & Fay, W. P. Regulation of arterial thrombolysis by plasminogen activator inhibitor-1 in mice. Circulation 97, 1002–1008 (1998).
Carmeliet, P. et al. Plasminogen activator inhibitor-1 gene-deficient mice. II. Effects on hemostasis, thrombosis, and thrombolysis. J Clin Invest 92, 2756–2760 (1993).
Siefert, S. A. et al. Enhanced venous thrombus resolution in plasminogen activator inhibitor type-2 deficient mice. J Thromb Haemost 12, 1706–1716 (2014).
Gardiner, E. E. & Medcalf, R. L. Is PAI-2 really a PAI after all? J Thromb Haemost 12, 1703–1705 (2014).
Medcalf, R. L. & Stasinopoulos, S. J. The undecided serpin. The ins and outs of plasminogen activator inhibitor type 2. FEBS J 272, 4858–4867 (2005).
Bird, C. H. et al. Nucleocytoplasmic distribution of the ovalbumin serpin PI-9 requires a nonconventional nuclear import pathway and the export factor Crm1. Mol Cell Biol 21, 5396–5407 (2001).
von Heijne, G., Liljestrom, P., Mikus, P., Andersson, H. & Ny, T. The efficiency of the uncleaved secretion signal in the plasminogen activator inhibitor type 2 protein can be enhanced by point mutations that increase its hydrophobicity. J Biol Chem 266, 15240–15243 (1991).
Ye, R. D., Wun, T. C. & Sadler, J. E. Mammalian protein secretion without signal peptide removal. Biosynthesis of plasminogen activator inhibitor-2 in U-937 cells. J Biol Chem 263, 4869–4875 (1988).
Schroder, W. A. et al. Tumor cell-expressed SerpinB2 is present on microparticles and inhibits metastasis. Cancer Med 3, 500–513 (2014).
Guller, S. et al. Protein composition of microparticles shed from human placenta during placental perfusion: Potential role in angiogenesis and fibrinolysis in preeclampsia. Placenta 32, 63–69 (2011).
Schroder, W. A., Major, L. & Suhrbier, A. The role of SerpinB2 in immunity. Crit Rev Immunol 31, 15–30 (2011).
Shea-Donohue, T., Zhao, A. & Antalis, T. M. SerpinB2 mediated regulation of macrophage function during enteric infection. Gut Microbes 5, 254–258 (2014).
Zhao, A. et al. SerpinB2 is critical to Th2 immunity against enteric nematode infection. J Immunol 190, 5779–5787 (2013).
Schroder, W. A. et al. SerpinB2 deficiency modulates Th1Th2 responses after schistosome infection. Parasite Immunol 32, 764–768 (2010).
Major, L. D. et al. Induction of SerpinB2 and Th1/Th2 modulation by SerpinB2 during lentiviral infections in vivo. PLoS One 8, e57343 (2013).
Schroder, W. A. et al. A physiological function of inflammation-associated SerpinB2 is regulation of adaptive immunity. J Immunol 184, 2663–2670 (2010).
Delhase, M. et al. TANK-binding kinase 1 (TBK1) controls cell survival through PAI-2/serpinB2 and transglutaminase 2. Proc Natl Acad Sci USA 109, E177–186 (2012).
Valiente, M. et al. Serpins promote cancer cell survival and vascular co-option in brain metastasis. Cell 156, 1002–1016 (2014).
Ramnefjell, M., Aamelfot, C., Helgeland, L. & Akslen, L. A. Low expression of SerpinB2 is associated with reduced survival in lung adenocarcinomas. Oncotarget 8, 90706–90718 (2017).
Bae, S. Y., Park, H. J., Hong, J. Y., Lee, H. J. & Lee, S. K. Down-regulation of SerpinB2 is associated with gefitinib resistance in non-small cell lung cancer and enhances invadopodia-like structure protrusions. Sci Rep 6, 32258 (2016).
Longhin, E., Camatini, M., Bersaas, A., Mantecca, P. & Mollerup, S. The role of SerpinB2 in human bronchial epithelial cells responses to particulate matter exposure. Arch Toxicol 92, 2923–2933 (2018).
Harris, N. L. E. et al. SerpinB2 regulates stromal remodelling and local invasion in pancreatic cancer. Oncogene 36, 4288–4298 (2017).
Antalis, T. M. & Dickinson, J. L. Control of plasminogen-activator inhibitor type 2 gene expression in the differentiation of monocytic cells. Eur J Biochem 205, 203–209 (1992).
Yu, H., Maurer, F. & Medcalf, R. L. Plasminogen activator inhibitor type 2: a regulator of monocyte proliferation and differentiation. Blood 99, 2810–2818 (2002).
Zhang, S. et al. The sGC activator inhibits the proliferation and migration, promotes the apoptosis of human pulmonary arterial smooth muscle cells via the up regulation of plasminogen activator inhibitor-2. Exp Cell Res 332, 278–287 (2015).
Elsafadi, M. et al. SERPINB2 is a novel TGFbeta-responsive lineage fate determinant of human bone marrow stromal cells. Sci Rep 7, 10797 (2017).
Lee, N. H. et al. SERPINB2 is a novel indicator of stem cell toxicity. Cell Death Dis 9, 724 (2018).
Chuang, S. Y. et al. TLR-induced PAI-2 expression suppresses IL-1beta processing via increasing autophagy and NLRP3 degradation. Proc Natl Acad Sci USA 110, 16079–16084 (2013).
Hsieh, H. H., Chen, Y. C., Jhan, J. R. & Lin, J. J. The serine protease inhibitor serpinB2 binds and stabilizes p21 in senescent cells. J Cell Sci 130, 3272-3281 (2017).
Lee, J. A. et al. SerpinB2 (PAI-2) Modulates Proteostasis via Binding Misfolded Proteins and Promotion of Cytoprotective Inclusion Formation. PLoS One 10, e0130136 (2015).
Fan, J. et al. Interaction of plasminogen activator inhibitor-2 and proteasome subunit, beta type 1. Acta Biochim Biophys Sin (Shanghai) 36, 42–46 (2004).
Tonnetti, L. et al. SerpinB2 protection of retinoblastoma protein from calpain enhances tumor cell survival. Cancer Res 68, 5648–5657 (2008).
Park, J. M. et al. Signaling pathways and genes that inhibit pathogen-induced macrophage apoptosis–CREB and NF-kappaB as key regulators. Immunity 23, 319–329 (2005).
Schroder, W. A. et al. SerpinB2 deficiency results in a stratum corneum defect and increased sensitivity to topically applied inflammatory agents. Am J Pathol 186, 1511–1523 (2016).
Croucher, D. R., Saunders, D. N., Lobov, S. & Ranson, M. Revisiting the biological roles of PAI2 (SERPINB2) in cancer. Nat Rev Cancer 8, 535–545 (2008).
Schroder, W. A. et al. SerpinB2 deficiency in mice reduces bleeding times via dysregulated platelet activation. Platelets 30, 658–663 (2019).
Major, L., Schroder, W. A., Gardner, J., Fish, R. J. & Suhrbier, A. Human papilloma virus transformed CaSki cells constitutively express high levels of functional SerpinB2. Exp Cell Res 317, 338–347 (2011).
Maurer, F. & Medcalf, R. L. Plasminogen activator inhibitor type 2 gene induction by tumor necrosis factor and phorbol ester involves transcriptional and post-transcriptional events. Identification of a functional nonameric AU-rich motif in the 3′-untranslated region. J Biol Chem 271, 26074–26080 (1996).
Hume, D. A., Summers, K. M. & Rehli, M. Transcriptional Regulation and Macrophage. Differentiation. Microbiol Spectr 4, 1–19 (2016).
Xue, J. et al. Transcriptome-based network analysis reveals a spectrum model of human macrophage activation. Immunity 40, 274–288 (2014).
Shapouri-Moghaddam, A. et al. Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol 233, 6425–6440 (2018).
Gautier, E. L. et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat Immunol 13, 1118–1128 (2012).
Okabe, Y. & Medzhitov, R. Tissue-specific signals control reversible program of localization and functional polarization of macrophages. Cell 157, 832–844 (2014).
Rosas, M. et al. The transcription factor Gata6 links tissue macrophage phenotype and proliferative renewal. Science 344, 645–648 (2014).
Gautier, E. L. et al. Gata6 regulates aspartoacylase expression in resident peritoneal macrophages and controls their survival. J Exp Med 211, 1525–1531 (2014).
Wang, J. & Kubes, P. A Reservoir of Mature Cavity Macrophages that Can Rapidly Invade Visceral Organs to Affect Tissue Repair. Cell 165, 668–678 (2016).
Russo, P. S. T. et al. CEMiTool: a Bioconductor package for performing comprehensive modular co-expression analyses. BMC Bioinformatics 19, 56 (2018).
Mauer, J. et al. Signaling by IL-6 promotes alternative activation of macrophages to limit endotoxemia and obesity-associated resistance to insulin. Nat Immunol 15, 423–430 (2014).
Mahony, D., Kalionis, B. & Antalis, T. M. Plasminogen activator inhibitor type-2 (PAI-2) gene transcription requires a novel NF-kappaB-like transcriptional regulatory motif. Eur J Biochem 263, 765–772 (1999).
Yang, H., Wang, H., Chavan, S. S. & Andersson, U. High Mobility Group Box Protein 1 (HMGB1): The Prototypical Endogenous Danger Molecule. Mol Med 21, S6–S12 (2015).
Orozco, L. D. et al. Unraveling inflammatory responses using systems genetics and gene-environment interactions in macrophages. Cell 151, 658–670 (2012).
Hamilton, J. A. et al. Interleukin-4 suppresses plasminogen activator inhibitor-2 formation in stimulated human monocytes. Blood 80, 121–125 (1992).
Ghosn, E. E. et al. Two physically, functionally, and developmentally distinct peritoneal macrophage subsets. Proc Natl Acad Sci USA 107, 2568–2573 (2010).
Murray, P. J. et al. Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41, 14–20 (2014).
Shay, T. & Kang, J. Immunological Genome Project and systems immunology. Trends Immunol 34, 602–609 (2013).
Praus, M., Wauterickx, K., Collen, D. & Gerard, R. D. Reduction of tumor cell migration and metastasis by adenoviral gene transfer of plasminogen activator inhibitors. Gene Ther 6, 227–236 (1999).
Estreicher, A., Muhlhauser, J., Carpentier, J. L., Orci, L. & Vassalli, J. D. The receptor for urokinase type plasminogen activator polarizes expression of the protease to the leading edge of migrating monocytes and promotes degradation of enzyme inhibitor complexes. J Cell Biol 111, 783–792 (1990).
Fleetwood, A. J. et al. Urokinase plasminogen activator is a central regulator of macrophage three-dimensional invasion, matrix degradation, and adhesion. J Immunol 192, 3540–3547 (2014).
Leong, H. S. et al. Invadopodia are required for cancer cell extravasation and are a therapeutic target for metastasis. Cell Rep 8, 1558–1570 (2014).
Marenholz, I. et al. Genome-wide association study identifies the SERPINB gene cluster as a susceptibility locus for food allergy. Nat Commun 8, 1056 (2017).
Puente, X. S. & Lopez-Otin, C. A genomic analysis of rat proteases and protease inhibitors. Genome Res 14, 609–622 (2004).
Dougherty, K. M. et al. The plasminogen activator inhibitor-2 gene is not required for normal murine development or survival. Proc Natl Acad Sci USA 96, 686–691 (1999).
Teoh, S. S. et al. Maspin is not required for embryonic development or tumour suppression. Nat Commun 5, 3164 (2014).
Jensen, P. H. et al. The exon 3 encoded sequence of the intracellular serine proteinase inhibitor plasminogen activator inhibitor 2 is a protein binding domain. J Biol Chem 271, 26892–26899 (1996).
Miles, L. A. et al. New insights into the role of Plg-RKT in macrophage recruitment. Int Rev Cell Mol Biol 309, 259–302 (2014).
Miles, L. A. et al. Deficiency of plasminogen receptor, Plg-RKT, causes defects in plasminogen binding and inflammatory macrophage recruitment in vivo. J Thromb Haemost 15, 155–162 (2017).
O’Connell, P. A., Surette, A. P., Liwski, R. S., Svenningsson, P. & Waisman, D. M. S100A10 regulates plasminogen-dependent macrophage invasion. Blood 116, 1136–1146 (2010).
Genua, M. et al. Treatment with a Urokinase Receptor-derived Cyclized Peptide Improves Experimental Colitis by Preventing Monocyte Recruitment and Macrophage Polarization. Inflamm Bowel Dis 22, 2390–2401 (2016).
Wiesner, C., Le-Cabec, V., El Azzouzi, K., Maridonneau-Parini, I. & Linder, S. Podosomes in space: macrophage migration and matrix degradation in 2D and 3D settings. Cell Adh Migr 8, 179–191 (2014).
Pollanen, J., Hedman, K., Nielsen, L. S., Dano, K. & Vaheri, A. Ultrastructural localization of plasma membrane-associated urokinase-type plasminogen activator at focal contacts. J Cell Biol 106, 87–95 (1988).
Reichel, C. A., Kanse, S. M. & Krombach, F. At the interface of fibrinolysis and inflammation: the role of urokinase-type plasminogen activator in the leukocyte extravasation cascade. Trends Cardiovasc Med 22, 192–196 (2012).
Kindzelskii, A. L. et al. Pericellular proteolysis by leukocytes and tumor cells on substrates: focal activation and the role of urokinase-type plasminogen activator. Histochem Cell Biol 121, 299–310 (2004).
Laug, W. E., Cao, X. R., Yu, Y. B., Shimada, H. & Kruithof, E. K. Inhibition of invasion of HT1080 sarcoma cells expressing recombinant plasminogen activator inhibitor 2. Cancer Res 53, 6051–6057 (1993).
Roy, S. et al. Redefining the transcriptional regulatory dynamics of classically and alternatively activated macrophages by deepCAGE transcriptomics. Nucleic Acids Res 43, 6969–6982 (2015).
Chistiakov, D. A., Myasoedova, V. A., Revin, V. V., Orekhov, A. N. & Bobryshev, Y. V. The impact of interferon-regulatory factors to macrophage differentiation and polarization into M1 and M2. Immunobiology 223, 101–111 (2018).
Mills, C. D. & Ley, K. M1 and M2 Macrophages: The Chicken and the Egg of Immunity. J Innate Immun 6, 716–726 (2014).
Genua, M. et al. The urokinase plasminogen activator receptor (uPAR) controls macrophage phagocytosis in intestinal inflammation. Gut 64, 589–600 (2015).
Varin, A., Mukhopadhyay, S., Herbein, G. & Gordon, S. Alternative activation of macrophages by IL-4 impairs phagocytosis of pathogens but potentiates microbial-induced signalling and cytokine secretion. Blood 115, 353–362 (2010).
Peng, J. et al. Modulatory effects of adiponectin on the polarization of tumor-associated macrophages. Int J Cancer 137, 848–858 (2015).
Stables, M. J. et al. Transcriptomic analyses of murine resolution-phase macrophages. Blood 118, e192–208 (2011).
Bosmann, M. & Ward, P. A. Modulation of inflammation by interleukin-27. J Leukoc Biol 94, 1159–1165 (2013).
Shen, F. et al. GATA6 predicts prognosis and hepatic metastasis of colorectal cancer. Oncol Rep 30, 1355–1361 (2013).
Tian, F. et al. Aberrant expression of GATA binding protein 6 correlates with poor prognosis and promotes metastasis in cholangiocarcinoma. Eur J Cancer 49, 1771–1780 (2013).
Deng, L. & Liu, H. MicroRNA-506 suppresses growth and metastasis of oral squamous cell carcinoma via targeting GATA6. Int J Clin Exp Med 8, 1862–1870 (2015).
Beuling, E. et al. GATA6 is required for proliferation, migration, secretory cell maturation, and gene expression in the mature mouse colon. Mol Cell Biol 32, 3392–3402 (2012).
Song, Y. et al. GATA6 is overexpressed in breast cancer and promotes breast cancer cell epithelial-mesenchymal transition by upregulating slug expression. Exp Mol Pathol 99, 617–627 (2015).
Doeuvre, L., Plawinski, L., Goux, D., Vivien, D. & Angles-Cano, E. Plasmin on adherent cells: from microvesiculation to apoptosis. Biochem J 432, 365–373 (2010).
Hildenbrand, R. et al. The urokinase-system–role of cell proliferation and apoptosis. Histol Histopathol 23, 227–236 (2008).
Mitchell, J. W., Baik, N., Castellino, F. J. & Miles, L. A. Plasminogen inhibits TNFalpha-induced apoptosis in monocytes. Blood 107, 4383–4390 (2006).
Paland, N., Aharoni, S. & Fuhrman, B. Urokinase-type plasminogen activator (uPA) modulates monocyte-to-macrophage differentiation and prevents Ox-LDL-induced macrophage apoptosis. Atherosclerosis 231, 29–38 (2013).
Zeng, P. et al. Apigenin Attenuates Atherogenesis through Inducing Macrophage Apoptosis via Inhibition of AKT Ser473 Phosphorylation and Downregulation of Plasminogen Activator Inhibitor-2. Oxid Med Cell Longev 2015, 379538 (2015).
Pavet, V. et al. Plasminogen activator urokinase expression reveals TRAIL responsiveness and supports fractional survival of cancer cells. Cell Death Dis 5, e1043 (2014).
Besch, R., Berking, C., Kammerbauer, C. & Degitz, K. Inhibition of urokinase-type plasminogen activator receptor induces apoptosis in melanoma cells by activation of p53. Cell Death Differ 14, 818–829 (2007).
Gan, H. et al. Mycobacterium tuberculosis blocks crosslinking of annexin-1 and apoptotic envelope formation on infected macrophages to maintain virulence. Nat Immunol 9, 1189–1197 (2008).
Suwa, D., Konno, H., Tanaka, T. & Urano, T. Intraperitoneal infusion of recombinant plasminogen activator inhibitor type 2 induced apoptosis in implanted human colon cancer and inhibited its growth and liver metastasis. Anticancer Res 28, 693–698 (2008).
Fish, R. J. & Kruithof, E. K. Evidence for serpinB2-independent protection from TNF-alpha-induced apoptosis. Exp Cell Res 312, 350–361 (2006).
Mahmood, N., Mihalcioiu, C. & Rabbani, S. A. Multifaceted Role of the Urokinase-Type Plasminogen Activator (uPA) and Its Receptor (uPAR): Diagnostic, Prognostic, and Therapeutic Applications. Front Oncol 8, 24 (2018).
Kinnby, B., Lecander, I. & Martinsson, G. & B, A. Tissue Plasminogen Activator and Placental Plasminogen Activator Inhibitor in Human Gingival Fluid. Fibrinolysis 5, 239–242 (1991).
Al-Ejeh, F., Croucher, D. & Ranson, M. Kinetic analysis of plasminogen activator inhibitor type-2: urokinase complex formation and subsequent internalisation by carcinoma cell lines. Exp Cell Res 297, 259–271 (2004).
Keller, N., Ozmadenci, D., Ichim, G. & Stupack, D. Caspase-8 function, and phosphorylation, in cell migration. Semin Cell Dev Biol 82, 105–117 (2018).
Lv, P. C., Jiang, A. Q., Zhang, W. M. & Zhu, H. L. FAK inhibitors in Cancer, a patent review. Expert Opin Ther Pat 28, 139–145 (2018).
Buchheit, C. L., Weigel, K. J. & Schafer, Z. T. Cancer cell survival during detachment from the ECM: multiple barriers to tumour progression. Nat Rev Cancer 14, 632–641 (2014).
Dickinson, J. L., Bates, E. J., Ferrante, A. & Antalis, T. M. Plasminogen activator inhibitor type 2 inhibits tumor necrosis factor alpha-induced apoptosis. Evidence for an alternate biological function. J Biol Chem 270, 27894–27904 (1995).
Greten, F. R. et al. NF-kappaB is a negative regulator of IL-1beta secretion as revealed by genetic and pharmacological inhibition of IKKbeta. Cell 130, 918–931 (2007).
Evavold, C. L. et al. The Pore-Forming Protein Gasdermin D Regulates Interleukin-1 Secretion from Living Macrophages. Immunity 48, 35–44 e36 (2018).
Daniels, M. J. & Brough, D. Unconventional Pathways of Secretion Contribute to Inflammation. Int J Mol Sci 18, E102 (2017).
Belaguli, N. S. et al. GATA6 promotes colon cancer cell invasion by regulating urokinase plasminogen activator gene expression. Neoplasia 12, 856–865 (2010).
Carriero, M. V. & Stoppelli, M. P. The urokinase-type plasminogen activator and the generation of inhibitors of urokinase activity and signaling. Curr Pharm Des 17, 1944–1961 (2011).
Cabrera-Ortega, A. A., Feinberg, D., Liang, Y., Rossa, C. Jr. & Graves, D. T. The Role of Forkhead Box 1 (FOXO1) in the Immune System: Dendritic Cells, T Cells, B Cells, and Hematopoietic Stem Cells. Crit Rev Immunol 37, 1–13 (2017).
Oh, M. H. et al. mTORC2 Signaling Selectively Regulates the Generation and Function of Tissue-Resident Peritoneal Macrophages. Cell Rep 20, 2439–2454 (2017).
Meznarich, J. et al. Urokinase plasminogen activator induces pro-fibrotic/m2 phenotype in murine cardiac macrophages. PLoS One 8, e57837 (2013).
Santibanez, J. F. Transforming growth factor-Beta and urokinase-type plasminogen activator: dangerous partners in tumorigenesis-implications in skin cancer. ISRN Dermatol 2013, 597927 (2013).
Deryugina, E. I. & Quigley, J. P. Cell surface remodeling by plasmin: a new function for an old enzyme. J Biomed Biotechnol 2012, 564259 (2012).
Li, Q. et al. Plasmin plays an essential role in amplification of psoriasiform skin inflammation in mice. PLoS One 6, e16483 (2011).
Syrovets, T., Lunov, O. & Simmet, T. Plasmin as a proinflammatory cell activator. J Leukoc Biol 92, 509–519 (2012).
Munakata, S. et al. Inhibition of plasmin protects against colitis in mice by suppressing matrix metalloproteinase 9-mediated cytokine release from myeloid cells. Gastroenterology 148, 565–578 e564 (2015).
Carlson, S. et al. Cardiac macrophages adopt profibrotic/M2 phenotype in infarcted hearts: Role of urokinase plasminogen activator. J Mol Cell Cardiol 108, 42–49 (2017).
Sugimoto, M. A. et al. Plasmin and plasminogen induce macrophage reprogramming and regulate key steps of inflammation resolution via annexin A1. Blood 129, 2896–2907 (2017).
Cha, B. H. et al. Integrin-Mediated Interactions Control Macrophage Polarization in 3D Hydrogels. Adv Healthc Mater 6, 1700289 (2017).
D’Alessio, S. & Blasi, F. The urokinase receptor as an entertainer of signal transduction. Front Biosci (Landmark Ed) 14, 4575–4587 (2009).
Smith, H. W. & Marshall, C. J. Regulation of cell signalling by uPAR. Nat Rev Mol Cell Biol 11, 23–36 (2010).
Wolff, C. et al. Signalling networks associated with urokinase-type plasminogen activator (uPA) and its inhibitor PAI-1 in breast cancer tissues: new insights from protein microarray analysis. J Pathol 223, 54–63 (2011).
Zhang, Y. L. et al. SPON2 Promotes M1-like Macrophage Recruitment and Inhibits Hepatocellular Carcinoma Metastasis by Distinct Integrin-Rho GTPase-Hippo Pathways. Cancer Res 78, 2305–2317 (2018).
Nguyen, D. H. et al. Urokinase-type plasminogen activator stimulates the Ras/Extracellular signal-regulated kinase (ERK) signaling pathway and MCF-7 cell migration by a mechanism that requires focal adhesion kinase, Src, and Shc. Rapid dissociation of GRB2/Sps-Shc complex is associated with the transient phosphorylation of ERK in urokinase-treated cells. J Biol Chem 275, 19382–19388 (2000).
Juliano, R. L. et al. Integrin regulation of cell signalling and motility. Biochem Soc Trans 32, 443–446 (2004).
Cochran, B. J., Croucher, D. R., Lobov, S., Saunders, D. N. & Ranson, M. Dependence on endocytic receptor binding via a minimal binding motif underlies the differential prognostic profiles of SerpinE1 and SerpinB2 in cancer. J Biol Chem 286, 24467–24475 (2011).
Zhu, L. et al. TSC1 controls macrophage polarization to prevent inflammatory disease. Nat Commun 5, 4696 (2014).
Lu, H. et al. ANG II facilitated CD11(+) Ly6C(hi) cells reprogramming into M1-like macrophage through Erk1/2 or p38-Stat3 pathway and involved in EAM. J Leukoc Biol 103, 719–730 (2018).
Tian, L. et al. Cannabinoid Receptor 1 Participates in Liver Inflammation by Promoting M1 Macrophage Polarization via RhoA/NF-kappaB p65 and ERK1/2 Pathways, Respectively, in Mouse Liver Fibrogenesis. Front Immunol 8, 1214 (2017).
Blom, K. et al. The anticancer effect of mebendazole may be due to M1 monocyte/macrophage activation via ERK1/2 and TLR8-dependent inflammasome activation. Immunopharmacol Immunotoxicol 39, 199–210 (2017).
Eblen, S. T. Extracellular-Regulated Kinases: Signaling From Ras to ERK Substrates to Control Biological Outcomes. Adv Cancer Res 138, 99–142 (2018).
Kan, X. H. et al. Kv1.3 potassium channel mediates macrophage migration in atherosclerosis by regulating ERK activity. Arch Biochem Biophys 591, 150–156 (2016).
Kauffmann, A., Gentleman, R. & Huber, W. arrayQualityMetrics–a bioconductor package for quality assessment of microarray data. Bioinformatics 25, 415–416 (2009).
Gautier, L., Cope, L., Bolstad, B. M. & Irizarry, R. A. affy–analysis of Affymetrix GeneChip data at the probe level. Bioinformatics 20, 307–315 (2004).
Kramer, A., Green, J., Pollard, J. Jr. & Tugendreich, S. Causal analysis approaches in Ingenuity Pathway Analysis. Bioinformatics 30, 523–530 (2014).
Ray, A. & Dittel, B. N. Isolation of mouse peritoneal cavity cells. J Vis Exp 35, 1488 (2010).
Johnson, B. J. et al. Heat shock protein 10 inhibits lipopolysaccharide-induced inflammatory mediator production. J Biol Chem 280, 4037–4047 (2005).
Prow, N. A. et al. Lower temperatures reduce type I interferon activity and promote alphaviral arthritis. PLoS Pathog 13, e1006788 (2017).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 102, 15545–15550 (2005).
Acknowledgements
The work was supported by a Project grant from the National Health and Medical Research Council (NHMRC) of Australia, and São Paulo Research Foundation FAPESP Grant No. 2014/24162-2 and 2017/17345-1. Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/PROEX) – Finance Code 001 provided a PhD scholarship for T.D.H. A.S. holds a Principle Research Fellowship from the NHMRC. The authors would like to thank the QIMRB animal house and IT staff for their assistance.
Author information
Authors and Affiliations
Contributions
W.A.S. and A.S. conceived the project. W.A.S., T.T.L., J.G. E.N., D.P. and G.M.B. undertook the experiments. T.D.H., H.I.N. and J.E. undertook the bioinformatics analyses. A.S. wrote the manuscript with input from all the authors.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schroder, W.A., Hirata, T.D., Le, T.T. et al. SerpinB2 inhibits migration and promotes a resolution phase signature in large peritoneal macrophages. Sci Rep 9, 12421 (2019). https://doi.org/10.1038/s41598-019-48741-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-48741-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.